8
Pain from skeletal metastases is the most common source of intractable pain in cancer patients. It is the presenting system in nearly 80% of patients with bone metastases.1 In an ambulatory patient group, only 45% described adequate control of pain from medications, and 23% stated that analgesics had no effect at all. This group was otherwise highly functional: 27% still worked, and only 16% considered themselves disabled. The long-term effects of poorly controlled pain from skeletal metastases are undeniable. Over time, mobility and quality of life are diminished, resulting in anxiety and depression. Traditional oncologic therapies provide benefit to many, but not all, patients. Additionally, these historic treatments have a delayed onset of pain relief. Thermal ablation has emerged as a viable option to treat patients with pain from skeletal metastases based on published outcomes, the limited number of treatment cycles required, and the relatively rapid onset of relief. In this chapter, the current status of ablation is compared with outcomes with traditional therapies. Given the emergence of vertebroplasty and cementoplasty, as well as the combined use of these techniques with ablation, the relative role of these options is reviewed as well. Technical tips are provided, as well as guidance regarding patient evaluation and management.
♦ Current Treatment Options
Traditional oncologic therapies have been divided into three major categories: surgical, radiation therapy, and medical. The selected treatment option for a given patient varies depending on the type of malignancy, the number of tumors, and their location.
Surgery
Absolute indications for surgery are a point of discussion, although there is agreement that repair or extirpation of painful skeletal metastases is palliative. A variable indication is the expected time of survival, with some practitioners favoring surgery in patients with a potential survival of 4 weeks or greater, whereas others recommend surgery only if minimal expected survival is 3 months.2,3 Accepted historic indications include radiation-resistant disease, spinal instability, spinal cord/nerve compression, acute neurologic deterioration, and either an impending or present pathologic fracture.3,4 A pathologic fracture is defined as a fracture occurring in a bone under the setting of normal physiologic stress.4 Most skeletal metastases develop in long bones. In a series of surgically treated patients, almost 60% of metastases requiring surgery were in the femur alone.3 In this study, the humerus was involved next most commonly (18.8%), followed by the spine (10.2%) and pelvis (5.9%). Skeletal metastases are rarely solitary and are isolated in only 5 to 15% of patients.3
When needed, it is preferable for surgical repair to be performed prior to the development of a pathologic fracture. In long bones, indications include pain as well as osteolytic foci >2.5 cm in diameter or involvement of >50% of the circumference of the bone. Greater than 50% involvement of the cortical circumference is associated with a 50% risk of pathologic fracture.3 Spine surgery is more complex, with most indications surrounding avoiding or treating devastating neurologic complications. Primary objectives include nerve root decompression, spinal stabilization, and reconstruction of the spinal column.4 The approach to spinal reconstruction depends on the involved portion of the column. Tumors isolated to the posterior elements or the anterior three quarters of the vertebral bodies are approached posteriorly or anteriorly, respectively. Tumors involving the posterior quarter of the vertebral body require both anterior and posterior access, a more complex and challenging intervention.4
Most surgical series are retrospective and consist of treatment of metastases from a variety of tumor types in different bones. One series described surgery on 76 pathologic fractures and 41 impending fractures.3 Only 15% of these patients had a solitary metastasis, and 80.4% of the treated areas were in the long bones of the upper and lower extremities. The majority of the remaining patients (16.1%) had tumors in the spine and pelvis. The surgeries were associated with high morbidity. Patients required three to eight units of packed red blood cells, and the average postoperative admission time was 17.8 days. Several patients were not able to bear weight after surgery, a finding that has been described in other studies following hemipelvectomy.5 Over half the patients maintained the same need for narcotic medications following surgery that they had prior to reconstruction.
Katzer et al3 did not categorize complications as major or minor, but instead described “wound” and “systemic” complications, which occurred in 24.8% and 14.5% of patients, respectively. Significantly, 7.9% of patients died in the hospital following surgery. When surgery is performed in the pelvis, spine, and long bones, a wide resection margin is favored.5 If this is not possible, supplemental radiation is typically added to the treatment regimen to decrease the risk of local disease progression. When fixation screws are placed, radiation therapy is often added to the treatment regimen to prevent loosening secondary to progression of disease. One study found a decrease in loosening of spinal hardware from 15% to 3% with the addition of postsurgical radiation. Optimally, surgery is performed first to maximize healing.6
Radiation
Radiation therapy is the most commonly employed therapy for patients with painful metastases. Interestingly, given the common utility of this technique, standard protocols are not established and remain a point of debate without optimally defined delivery methods. External beam radiotherapy (EBRT) is commonly delivered with a target dose reached over several sessions/fractions. The most commonly described schedule is a total of 30 Gy achieved over 10 fractions.7 This fractionation schedule is based on a Radiation Therapy Oncology Group study of 1016 patients in which five different regimens were compared.8 In this study 54% of patients achieved complete relief of pain, and 83% had at least partial relief. The median duration of complete and partial pain relief was 12 and 20 weeks, respectively. A later metanalysis compared various treatment regimens with outcomes. Included in the comparison were single versus multiple fraction treatment plans. Additionally, different target doses in multifraction treatment plans were compared.9 No difference was found when treatment was given in a single fraction versus multiple fractions when partial and complete pain relief were evaluated. Most of the single fraction therapies prescribed a lower total dose. Not surprisingly, patients treated with a single fraction were also more likely to have recurrent pain and require re-treatment than were those treated with multiple fractions.
Prospective studies have since been performed evaluating the benefits of EBRT given in a single versus multiple fractions. A Dutch multicenter trial compared 24 Gy over six fractions to 8 Gy in one fraction. Achieved palliation was equal between groups: 73% of multiple fraction patients achieved pain relief versus 71% of the single fraction group.10 Again, 24% of the single fraction patients who achieved benefit required reirradiation versus 6% of the multiple fraction patients. A separate randomized trial followed patients for a median of 12 months and found no difference in time to first improvement in pain, time to complete pain relief, or time to increase in pain or toxicity with any of three treatment regimens. These regimens included 30 Gy in ten fractions, 20 Gy in five fractions, and a single fraction of 8 Gy.11 Consistent with previous studies, the single fraction group required re-treatment more commonly than patients undergoing the other regimens.
External beam radiotherapy is helpful for the majority of treated patients. However, the maximum benefit of relief is often not manifested until up to 4 weeks after treatment.7 Only 50% of patients have any relief of pain at 2 weeks.12 Not all tumors are equally radiosensitive. EBRT is the most successful in treating lymphoma, myeloma, seminoma, and neuroblastoma, with more moderate success when treating breast and prostate metastases.4 EBRT also has several toxicities. Common side effects include fatigue, skin erythema, and nausea. Depending on the location of the field, diarrhea or esophagitis is common. Larger fields, particularly over the pelvis and spine are associated with myelosuppression. This risk is likely elevated in patients pretreated with chemotherapy. Pathologic fractures are a potential risk in 4 to 18% of patients following EBRT.8 The risk is likely greater in long bones than in the spine.7
Bisphosphonates
Bisphosphonates are analogs of pyrophosphate that inhibit osteoclast-mediated bone resorption. A variety of agents have been developed and are principally used in patients with breast cancer and, less commonly, multiple myeloma.13 Breast cancer has been the most commonly studied tumor for several reasons: bone metastases develop in up to 70% of this patient group, and survival of breast cancer patients with skeletal metastases is 25 months, much longer than prostate (6 months) or lung (3 months) cancer.14 Given the high incidence of skeletal metastases in patients with breast cancer and the relatively prolonged survival, 10 to 20% of these patients develop pathologic fractures. Once initiated, bisphosphonates are given for life.
Typically, outcomes with bisphosphonates have been measured by a decrease in skeletal-related events (SREs), which include pathologic fracture, requirement of EBRT for intractable pain, surgery for spinal cord compression, and hyper-calcemia of malignancy. The newest bisphosphonate on the market, zoledronic acid (Zometa, Novartis, East Hanover, NJ) is 10,000 times more powerful than first-generation agents and is the first drug in this class to be approved for treatment of skeletal metastases from other primary tumors. In preclinical testing, zoledronic acid had a 41% reduction in SREs compared with placebo.13
Although outcomes with zoledronic acid are promising, bisphosphonate use is not without controversy. Critics point out that evaluation of SREs does not include measuring or evaluating for changes in the level of pain.14 Other points of contention include that a decrease in SREs with bisphosphonates is not noticeable until 3 to 6 months after the initiation of therapy, and one study found that the cost of the drug being evaluated exceeded the cost savings from the SREs that were prevented. This time to benefit is one reason why this group of drugs was not used for metastasis outside of breast cancer in the past. The most common toxicity with bisphosphonates is osteonecrosis of the jaw.13 When using zoledronic acid, the risk is 1% at 1 year with an increase to 21% at 3 years.
Radionuclide Therapy
Radionuclides are used in the setting of multiple metastases, particularly when EBRT has failed. They are not appropriate for lytic metastases, patients with an impending or pathologic fracture, spinal cord compression, renal insufficiency, or compromised bone marrow function.7 Two agents are most commonly used: strontium-89 and samarium-153.
Strontium-89
Strontium-89 (89Sr) decays by beta-emission with a maximum energy of 1.46 MeV, an average soft tissue penetration of 2.4 mm, and a half-life of 50.6 days. 89Sr is taken into the mineral matrix of bone and concentrates at areas of active resorption of skeletal metastases. The retention is proportional to the tumor burden at a given site, and given the long half-life, up to 80% of the administered dose may be taken into the bone by 100 days.15 Patients can experience a pain flare shortly after treatment. When 89Sr was studied versus placebo, 20% and 50% of patients treated with 89Sr reported complete and partial pain relief, respectively.16 The typical administered dose is 1.5 MBq/kg.17 In a study of 83 patients, this dose resulted in some pain relief in 75% of patients and complete pain relief in 22%.17 In this trial, the onset of pain relief was typically 10 to 20 days after injection, the benefit maximum was at 6 weeks, and the duration of benefit was approximately 6 months.
Samarium-153
Samarium-153 (153Sm) emits beta-energy at 0.81 MeV, with a half-life of 46.3 hours. Additionally, this isotope has 28% emission of 103 keV gamma rays.7 Compared with 89Sr, 153Sm has a shorter half-life and a higher dose rate, resulting in shorter dose delivery times: 75% of 153Sm is delivered in 4 days compared with 101 days for 89Sr.7 153Sm is chelated to ethylenediamine-tetramethylene phosphonate (EDTMP, which results in preferential localization to osteoblastic areas of bone. Given the natural production of gamma rays, 153Sm can also be used for diagnostic purposes as well. In a randomized trial versus placebo, a 1.0-mCi/kg dose resulted in pain relief in 72% of 32 patients.18 In this study 31% of patients reported complete/marked pain relief, and 43% of patients reporting improvement in pain had persistent benefit at 16 weeks.
Outcomes with Thermal Ablation
Radiofrequency Ablation
Initial retrospective reports on radiofrequency ablation (RFA) described excellent clinical outcomes. Gronemeyer and colleagues19 used RFA to treat 10 patients with 21 painful spinal metastases. Four patients were treated with supplemental vertebroplasty 3 to 7 days following ablation. Nine of the 10 patients reported a decrease in pain at the time the review was written, with a 74% average pain reduction. A case report by Ohhigashi et al20 described successful thermal ablation of recurrent rectal carcinoma involving the sacrum.
A larger retrospective trial reviewed 34 treatments in 30 patients.21 Using a 10-point pain scale, the authors evaluated the patients’ worst pain, average pain, and mean pain interference with daily life prior to the treatment, and 1 day, 1 week, and 8 weeks following the procedure. They found that 19 of the 30 patients had reduction in pain within 24 hours. At 1 week and 8 weeks, all treated patients had significantly reduced pain and decreased interference with daily life from pain.
In the largest reported multicenter prospective trial, Goetz et al22 reported outcomes in 43 patients. Patients were followed for 24 weeks after treatment. Prior to ablation, the mean worst pain of the day was 7.9/10. At 4, 12, and 24 weeks after treatment, the worst pain of the day decreased to 4.5/10, 3.0/10, and 1.4/10, respectively. All decreases were statistically significantly improved from the baseline score; 41 of 43 patients experienced significant reductions in pain. Three major complications occurred, all of which should be noted by practitioners performing thermal ablation of skeletal metastases:
- A second-degree skin burn at a grounding pad site. Operators performing RFA need to be meticulous about grounding pad placement, ensuring that positioning is appropriate and that there are no gaps in contact with the skin.
- Transient bowel and bladder dysfunction following treatment of a sacral metastasis. Treatment in the sacrum can be associated with motor defects (typically at S1) or, more inferiorly (S2–4), foot drop or bowel and bladder dysfunction. It is critical to discuss these risks with patients and to obtain their signed consent in the interventional radiology (IR) clinic.
- A fracture of the acetabulum following treatment of a supraacetabular lesion. Weight-bearing bones are at risk of pathologic fracture when treated with thermal ablation. This potential complication is discussed in greater detail below.
Cryoablation
Callstrom et al23 reported a series of patients with painful skeletal metastases that were treated with cryoablation. Fourteen patients were treated and followed for up to 6 months. Tumors up to 11 cm in diameter were treated. The mean worst pain score prior to ablation was 6.7/10. This rating dropped to 3.8/10 four weeks after treatment. There was also a significant decrease in pain interference with activities of normal living. All patients who required narcotics prior to treatment had a reduced requirement for medication following cryoablation. No major complications occurred. Patients with tumors in weight-bearing areas such as in the prospective multicenter RFA trial were excluded from this study.
Ablation Combined with Cementoplasty/Vertebroplasty
Hoffmann and colleagues24 reported outcomes in 22 patients who were treated with both RFA and cementoplasty/vertebroplasty over a 5-year period. Treated tumors were in the thoracic and lumbar spine, sacrum, pelvis, acetabulum, femur, and tibia. All treated patients had improvement in pain within 24 hours, with a mean worst pain score decrease from 8.5/10 to 5.5/10, a statistically significant reduction. By 3 months, the worst pain of the day had decreased to 3.5/10. The rapid onset of pain relief with combined therapy is notable. Over a mean follow-up of 7.7 months, 15 of the patients had a reduced narcotic requirement. Two patients had an increased narcotic requirement secondary to tumor progression outside the treated area. No major complications were observed.
A study by Munk et al25 reviewed 25 combined treatments in 19 patients. Tumors in the lumbar and thoracic spine, acetabulum, sacrum, pubis, and humerus were treated. Pain was assessed prior to treatment and at 6 weeks after therapy. The mean score changed from 7.9/10 before treatment to 4.2/10 afterward. There were several asymptomatic leaks of cement, and one transient thermal nerve injury when postprocedural CT demonstrated extravasated cement in contact with the sciatic nerve. The patient developed a mixed sensory loss and weakness following the procedure, which resolved almost entirely at 48 hours after treatment.
As treatment options for patients with painful metastases continue to evolve, combination therapy is an intriguing option. Treatment of spinal tumors with thermal ablation is not without risk, as is discussed below (see Anatomic Considerations). Given that skeletal metastases have a propensity to form within the axial skeleton, combined therapy is an appropriate option for some patients to avoid postablation pathologic fracture.26
♦ Patient Selection, Tumor Selection, and Anatomic Considerations
Patient Selection
Patients undergoing thermal ablation of skeletal metastases should meet the following inclusion criteria:
- A worst pain score >4/10 per day using the Brief Pain Inventory-Short Form in the 24-hour period prior to treatment27
- No more than two dominant sites of painful skeletal metastases with pain as described above
- Life expectancy of >2 months
- The ability to provide informed consent
- Platelet count >75,000/microliter
- International normalized ratio <1.5
Exclusion criteria include the following:
- Widespread painful metastases
- Impending fracture of the targeted bone
- Osteoblastic tumors
- Tumors within 1 cm of the spinal cord, brain, aorta, inferior vena cava, bowel, or bladder
- Not meeting inclusion criteria listed above
Tumor Selection
As noted above, blastic tumors are generally considered inappropriate for thermal ablation and have been considered an exclusion criterion for most ablation trials.22,23,28 In limited situations, these tumors can be treated with some success. Primary tumors of bone, including osteosarcoma, multiple myeloma, lymphoma, and leukemic infiltration, are primarily best treated systemically as an initial measure.
Anatomic Considerations
Several patients who are considered poor surgical candidates with painful metastatic disease have tumors either in the spine or supraacetabular pelvis. Both these areas are weight bearing, and it should be kept in mind that tumors treated by thermal ablation alone do not support weight any better than untreated tumors.29 A pathologic fracture occurring following thermal ablation of a supraacetabular metastasis has been reported.22 Treatment of weight-bearing bones of the lower extremity such as the femur and tibia with osteoplasty has been described. When treating weight-bearing bones, supplemental osteoplasty with polymethylmethacrylate should be done when possible to prevent a pathologic fracture.
♦ Bone and Relevant Musculoskeletal Anatomy
The “brick oven” effect during thermal ablation of hepatocellular carcinoma describes the treatment of a relatively spongy tumor surrounded by fibrotic liver that insulates the generated heat within the tumor during ablation.30 In several patients, cortical bone can have a similar insulating effect.25 This relative protection is important because the spinal cord and peripheral/motor nerves are frequently adjacent to metastatic lesions. With radiofrequency (RF) energy, temperatures at or exceeding 45°C have been associated with both spinal cord and peripheral nerve damage.31,32 Dupuy et al33 placed RF electrodes in the vertebral bodies of pigs using a transpedicular approach.33 Thermistors were placed from 5 to 15 mm from the active electrode tip including one in the anterior epidural space in all animals. Control ablations were also performed in the paraspinal soft tissues with similar temperature measurements. Maximal temperatures at 5, 10, and 15 mm from the thermal electrode in the vertebral body were 48°, 41°, and 39°C. In contrast, maximal temperatures at the same distances following ablation in the paraspinal muscles were 84°, 62°, and 58°C. These findings suggest that intact cortical bone provides an insulation effect during thermal ablation. Further evidence of the insulating effect of bone was provided by the ablation parameters. The authors used a system that temporarily switches off in the setting of increased impedance. In the skeletal ablations, the maximum achievable current was 220 ± 35 mA, whereas in the soft tissue treatments it was 326 ± 42 mA.
It is important to note that the above study was performed in a healthy animal model. An operator should not assume that tumor-infiltrated bone provides thermal insulation, particularly in the thecal sac. In a study combining thermal ablation with cementoplasty, Nakatsuka et al34 treated 17 patients with spinal tumors. All patients were treated with sedation, and ablation was halted immediately if radicular pain was described by the patient. Four of these patients (24%) had significant neural injury. In all patients, the ablation was terminated immediately upon the patient’s communicating the sensation of radiating pain. However, irreversible damage was already present in these patients. Of note, all four patients had tumor involving the posterior cortex of the vertebral body or the pedicle used for placement of the RF electrode. The three patients with posterior cortical involvement all developed incomplete hemiplegia.
♦ Technique: How I Do It
Patient Selection
Both thermal ablation and cement injection can benefit several patients with painful metastases. The author’s preference is to use cement in painful metastases involving weight-bearing areas, including the vertebral bodies and supraacetabular region. Cement injection can also be used when thermal ablation outcomes are suboptimal. If there is concern that neurologic structures are at risk, ablation is deferred in favor of vertebroplasty/cementoplasty alone. In other locations, thermal ablation is used as a primary therapy. For supraacetabular lesions or large pelvic tumors, ablation and cementoplasty can be combined in a single session.
Preprocedure Evaluation
All patients are seen in the IR clinic for formal consultation.35 A physical examination is performed, and a history is taken. At a minimum, the worst and average pain of the day is documented using the 1 to 10 Visual Analogue Scale. Narcotic and other pain medication requirements are noted. Relevant imaging is reviewed. If multiple sites are potentially involved or if the dominant source of pain is uncertain, repeat CT scan is performed with a radiopaque marker on the pain source. Alternatively, examination under fluoroscopy may be done for spinal masses. Examination under fluoroscopy is often helpful if a patient has partial collapse of several vertebral levels. Direct palpation frequently reveals that a discrete few levels may be the source of all the pain a patient is experiencing. By determining which levels are directly involved in patient discomfort, the treating interventional radiologist can avoid unnecessary treatment of asymptomatic levels, shorten procedure times, and limit radiation exposure to the patient and the operator.
Step-by-Step Radiofrequency Ablation of a Painful Metastasis
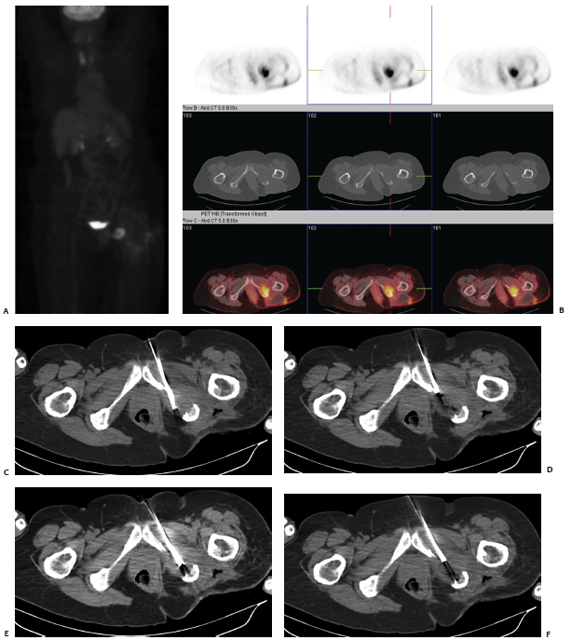
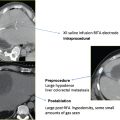
A patient was referred with a painful ischial metastasis from breast carcinoma, which had previously been radiated (Fig. 8.1). Positron emission tomography (PET)/CT demonstrated hypermetabolic activity in the left ischial bone. Upon presentation to the IR clinic, the patient had excruciating pain, which worsened during the day and prevented her from sitting unless she leaned to her right side, taking the pressure of the site. The ablation procedure was performed with general anesthesia. Many operators use general anesthesia to control pain during RFA.
The ablation was performed with CT guidance. Routine use of antibiotics prior to thermal ablation is not necessary. A cluster Cool-Tip electrode with 2.5-cm active tips (Covidien, Boulder, CO) was used for this procedure. Under CT fluoroscopic guidance, the electrodes were placed in several different positions. One of the principal theories regarding the benefits of thermal ablation for painful metastases is that pain is generated by stretching and infiltration of the well-innervated periosteum. The margin of a targeted tumor involving the periosteum should be included in the treatment field. Ablation was performed as per the manufacturer’s guideline, and the temperature was measured following each delivery of RF energy (60°C is the target with this device). The electrode was then repositioned in areas of the mass that had not reached tumoricidal temperatures. After three deployments, final imaging was performed and the electrode and grounding pads were removed. Inspection of the skin demonstrated no evidence of a grounding pad burn. The patient was admitted overnight. Patients are routinely given a patient-controlled analgesia (PCA) pump because pain can worsen in the immediate postprocedure period. The following morning, the patient was tolerating a normal diet, and her pain was controlled with oral analgesics alone. She was discharged without incident.
Step-by-Step Cryoablation of a Painful Metastasis
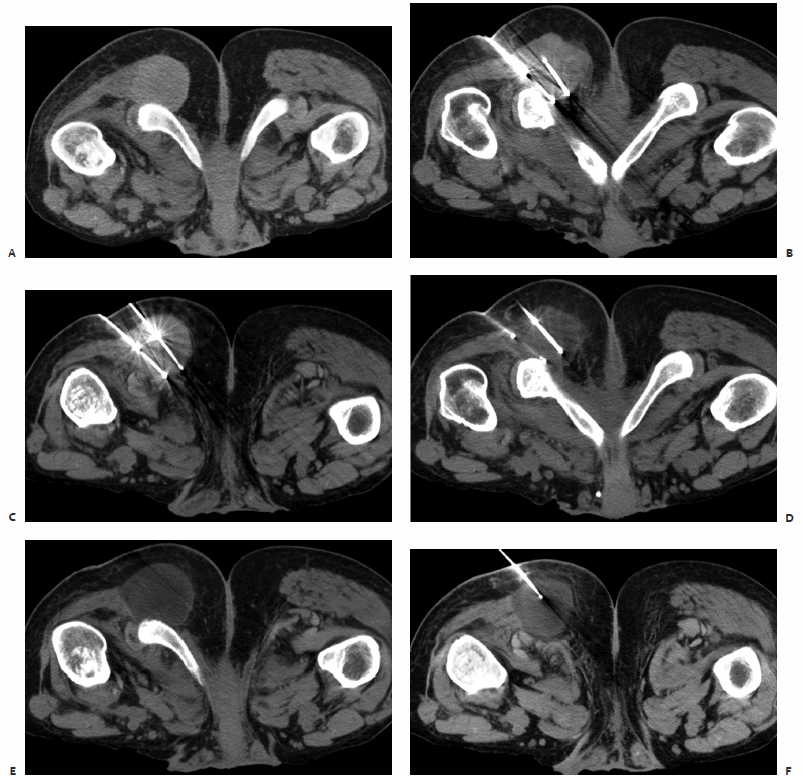
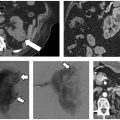
A patient was referred to the IR clinic with a painful colorectal cancer metastasis extending to his right ischium (Fig. 8.2). He had previously undergone radiation therapy, which improved his pain for several months before it acutely worsened. His pretreatment images were obtained with the patient in the prone position because his pain was so severe that he could not tolerate lying on his back or side, including during his visit to the IR clinic. Given the dimensions of this tumor, the decision was made to treat him with cryoablation.
Fig. 8.1 (A,B) Positron emission tomography/computed tomography (PET/CT) demonstrates a hypermetabolic focus in the left ischium. (C) A cluster electrode is advanced into the medial aspect of the lytic mass. Care was taken to ablate along the periosteal margin without extending too close to the rectum. (D) After 4 minutes of ablation, the electrode was withdrawn until the measured temperature was less than 60°C. Note the gas/cavitation defect within the tumor. (E) The electrode was then advanced into a more lateral position and ablation repeated. (F) After the second ablation, the electrode was again withdrawn, until a temperature of less than 60°C was noted. It was redirected laterally to ensure coverage of the lateral periosteal margin.
Fig. 8.2 (A) CT scan obtained at the time of interventional radiology (IR) consultation demonstrates a sizable soft tissue mass extending to the ischium. The patient’s pain was so severe that he was living in the prone position. (B,C) Multiple cryoprobes were positioned within the mass, covering up to the interface with the ischium. The needles are separated by approximately 2 cm in all cases. (D) Image obtained at the end of the first 10-minute freeze cycle. (E,F) Final images obtained following the second 10-minute freeze cycle. The tumor plus a margin is encompassed by the ice ball.
Step-by-Step Treatment of a Supraacetabular Metastasis using Radiofrequency Ablation and Cementoplasty
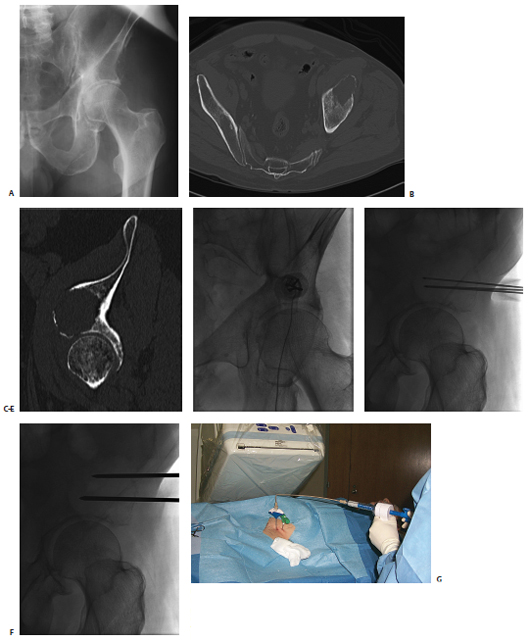
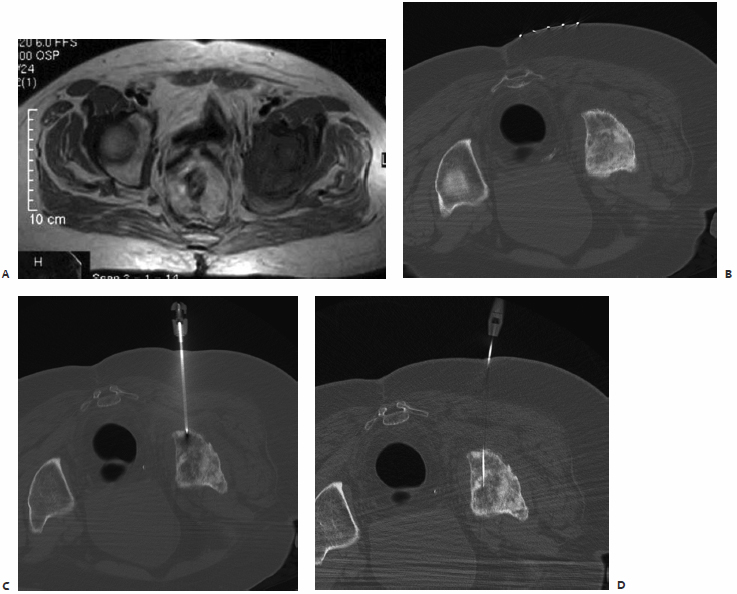
A patient had known metastatic renal cell carcinoma with a previously radiated metastasis to the ilium (Fig. 8.3). Thin-cut CT demonstrated that the acetabular roof was discontinuous. Given the location of the tumor, osteoplasty was clearly indicated as part of the therapy. However, there was concern about the potential for cement leakage into the joint space. Given the size of the tumor, we decided to combine osteoplasty with thermal ablation. In the IR clinic, the patient was wheelchair bound from a combination of pain and concern for a pathologic fracture, as described by his referring oncologist. The potential risk of cement leakage into the joint space was discussed with the patient.
The procedure was performed in one visit using general anesthesia support. Prophylactic antibiotics are given prior to cement delivery (typically IV cefazolin), as is done for vertebroplasty. Antibiotics are appropriate given the permanent implantation of polymethylmethacrylate. Fluoroscopic guidance was used for the entire procedure. This option was selected over CT because the tumor could be easily defined with fluoroscopy, and our biggest concern was cement leakage. Fluoroscopy allowed us to deliver the cement with realtime monitoring, which we thought would limit the risk or volume of spillage into the joint space.
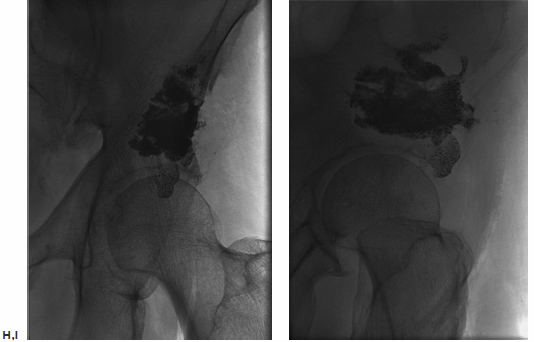
The ablation was performed first using a cluster electrode targeted to the deepest inferior portion of the tumor. After 4 minutes of ablation, the electrode was pulled back along the same plane into the lateral inferior aspect, and ablation repeated when temperatures in the thermistor registered less than 60°C. The superior aspect was then similarly treated. Two 11-gauge needles were then placed in the inferior and superior aspects of the tumor. Cement was injected in the inferior needle, first with the stylet removed from the superior needle in an attempt to “vent” the cement toward the second needle and away from the joint space. After injecting approximately 9 cc of cement via the inferior needle, 6 cc was injected via the superior needle. Final imaging, including C-arm CT reconstruction, was performed. There was no leakage into the joint space. The patient was observed overnight and discharged the following morning with minimal residual pain. He began physical therapy, and at 1-month follow-up in the IR clinic he was able to ambulate with a walker.
Step-by-Step Treatment of a Painful Metastasis with Cementoplasty
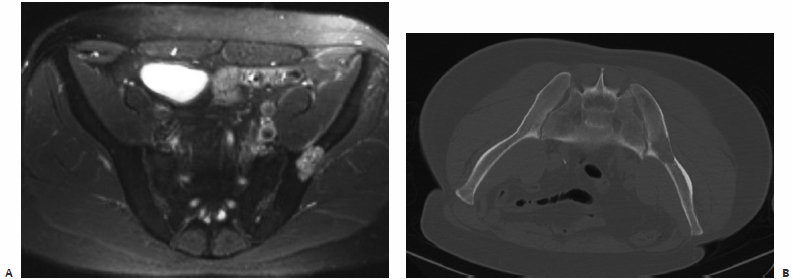
A patient was admitted with intractable pain from a pubic bone metastasis secondary to uterine carcinoma (Fig. 8.4). It had been previously treated with radiation therapy. An inpatient IR consultation was performed and recent plain films and CT were reviewed. Based on the proximity of the femoral triad to the tumor, the author and colleagues were concerned about the possibility of thermal injury to the femoral nerve. Additionally, we thought that the supplemental support provided by cementoplasty might prevent future pathologic fracture.
The procedure was performed under fluoroscopic guidance with conscious sedation. A Foley catheter was placed immediately prior to the procedure to decompress the bladder. The patient’s pannus was taped superiorly and laterally in an attempt to displace the femoral vessels laterally. The location of the femoral artery was tracked by palpation and marked prior to selecting an insertion site for the needle. Using a medial-to-lateral approach, an 11-gauge needle was advanced into the tumor under fluoroscopic guidance. Approximately 6 cc of cement was slowly injected under fluoroscopic guidance. No spillage into the pelvis or hip joint was identified. The Foley catheter was removed immediately following the procedure. The next morning, the patient’s pain was significantly improved. Her narcotics were quickly tapered, and she was discharged with minimal residual pain.
Vertebroplasty of a Painful Metastasis
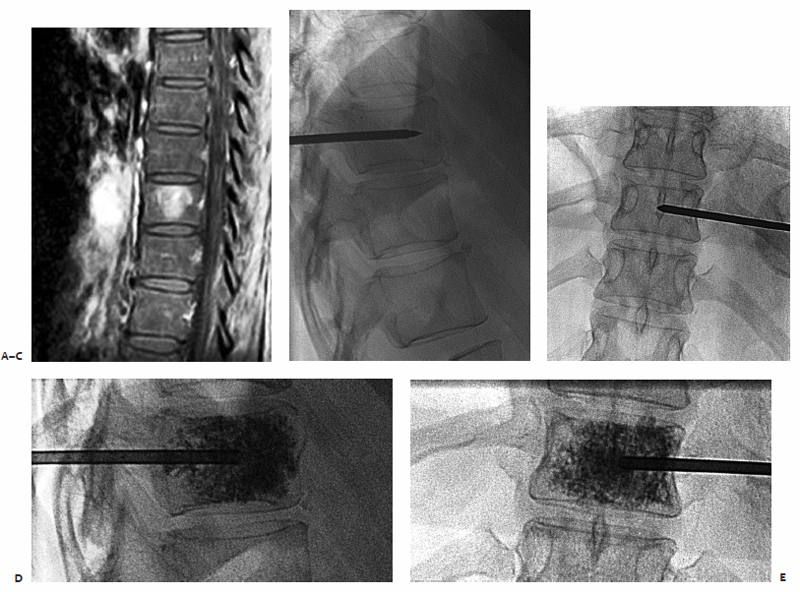
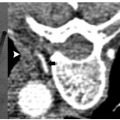
A patient had metastatic uveal melanoma and had been treated with immunoembolization of multiple hepatic metastases. She developed nonradiating back pain, and magnetic resonance imaging (MRI) confirmed metastatic infiltration of T10 (Fig. 8.5). During an IR clinic visit, examination under fluoroscopy confirmed this level as the source of the patient’s pain, which was 8/10 at its worst during the day.
The day of the procedure, the patient was given prophylactic antibiotics and placed in the prone position. An 11-gauge needle was advanced via the right pedicle to the posterior aspect of the vertebral body. Importantly, the medial border of the pedicle was not crossed. As the needle was advanced through the vertebral body, the hub was directed laterally to allow the tip to reach midline. The needle tip was advanced to the anterior quarter of the vertebral body using the lateral projection for guidance. Imaging in the anterior projection confirmed that the needle was midline. From this position, 5 cc of cement was injected in the lateral projection. Cement was injected in the lateral projection to ensure that posterior leakage into and around the spinal canal did not occur. Final imaging in the anterior projection confirmed that filling extending between the pedicles and nearly from one end plate to the other, which is the targeted outcome. The patient was observed for 2 hours and discharged. Her pain was better at discharge, and 2 days later it had decreased to 1/10 in severity.
Fig. 8.3 (A) Conventional radiograph of the pelvis demonstrates the lytic metastasis to the iliac bone immediately above the acetabulum. (B,C) Axial CT image and sagittal reconstruction delineate the destructive lytic process with discontinuity of the acetabulum. (D) The cluster ablation electrode is lined up over the metastasis. (E) The electrode was advanced into the most medial aspect of the tumor with fluoroscopic guidance. (F) After several overlapping ablations, two needles were placed for cement injection. (G) Both stylets were removed from the needles and cement injected into the inferior needle, allowing the polymethylmethacrylate to “vent” to the superior needle and avoid spillage into the joint space. (H,I) Frontal and oblique spot films after injection of 15 cc of cement, confirm filling of the majority of the tumor without spill into the joint space.
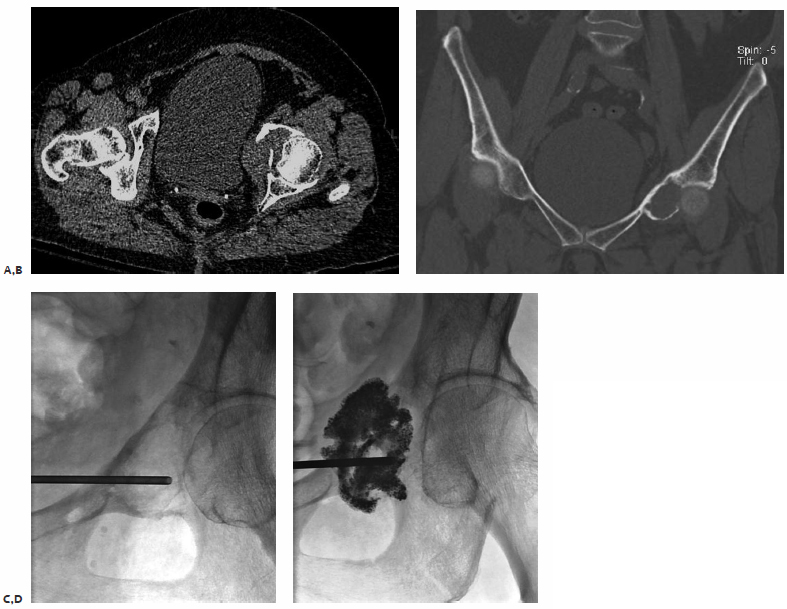
Fig. 8.4 (A,B) Axial CT and coronal thin-cut reconstruction demonstrate a lytic metastasis deep in the pelvis. Given the immediate proximity of the femoral neurovascular structures, there was concern about neurologic complications from thermal ablation. The author elected to perform cementoplasty. (C) A Foley catheter was placed into the patient’s bladder to decompress it and allow access to the tumor from a medial approach. We wanted to access the tumor from the most lytic portion. (D) Final image after cement injection demonstrates no spill into the joint space. There is filling of the tumor as well as some of the soft tissue component.
Fig. 8.5 (A) Fat-suppressed contrast-enhanced MRI demonstrates an enhancing metastasis in T10. (B) Via a transpedicular approach, an 11-gauge needle was advanced into the anterior portion of the vertebral body. The medial border of the pedicle should not be crossed during needle advancement. (C) Anterior projection with the needle at the midline. After ensuring entry to the vertebral body, the needle tip can be directed medially. Over 90% of the time, vertebroplasty of a given level can be performed with a single needle. (D) Lateral view following cement injection. Filling of the anterior two thirds of the vertebral body should be achieved. (E) Filling from pedicle to pedicle is present following polymethylmethacrylate injection.
Patient Follow-Up
Ablation patients are admitted to the IR service for overnight observation and given a PCA pump for pain control. Basal rates are not prescribed, so true narcotic requirements can be estimated by patient demand dose utilization. Although some operators perform ablation as an outpatient procedure, many patients have a temporary increase in pain after having a large needle placed into a painful tumor. By the following morning, virtually all patients have significantly improved and can easily be discharged.
Vertebroplasty/cementoplasty patients are discharged 2 to 3 hours following treatment to allow sedation to wear off and perform initial pain assessment. The IR service contacts all treated patients 2 to 3 days following therapy to ensure that pain is adequately controlled. Patients are seen 1 month after treatment in the IR clinic for formal pain assessment. If the patient’s pain is well controlled, physical examination is satisfactory without additional imaging. Virtually all patients will be getting restaged every 2 to 3 months with imaging, so our service typically waits until the next routine CT/MRI/PET in those patients with well-controlled pain following therapy.
In patients with poorly controlled pain following treatment, repeat imaging is ordered to evaluate whether the patient has developed a pathologic fracture or if there is a new tumor. More than 10% of patients treated with vertebroplasty develop adjacent fractures following therapy.36 Patients who call in with new pain after treatment are seen rapidly in the IR clinic with new imaging in hand to facilitate triage and procedure scheduling.
♦ Tricks of the Trade
Preprocedure
Patient selection is the cornerstone to a successful practice. Treatment of tumors in weight-bearing bones using ablation alone without supplemental stabilization can predispose patients to devastating pathologic fractures. Clinic visits require frank discussion of potential procedural risks. Several tumors localize to the sacrum, and the possibility of motor nerve injury or foot drop from treatment may be present. By providing informed consent, the patient participates in decision making regarding whether pain is severe enough to tolerate associated risks. Although virtually all patients treated with ablation in reported studies benefited from therapy, total elimination of pain does not occur in all patients. Managing patient expectations is a key component of the preprocedure consent process.
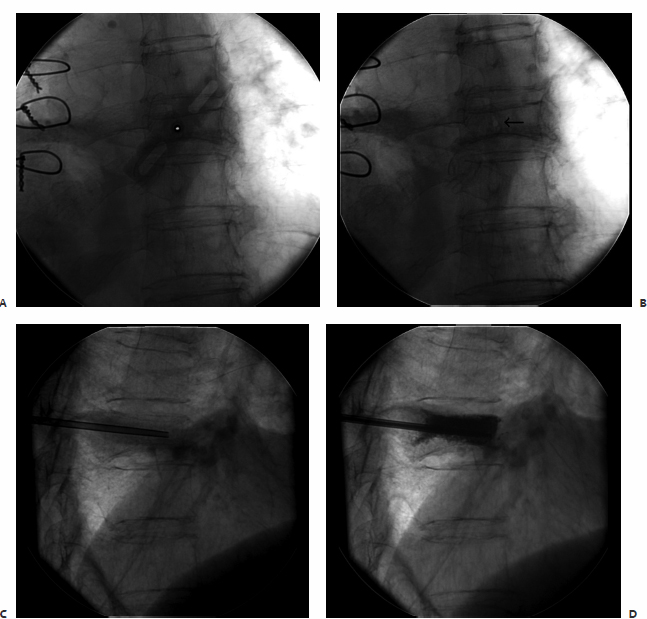
Fig. 8.6 (A) The image intensifier is lined up directly over the barrel of the needle. (B) After the needle is removed, a radiolucent defect remains visible at the entry site (arrow). (C) The needle is easily readvanced into the vertebral body using the defect as a target. (D) Vertebroplasty is performed.
Intraprocedural
Protecting the Skin from Thermal Injury
Skeletal ablation can involve treating tumors that are relatively close to the skin surface. If the operator is not careful, cutaneous injury to the patient can result.37 When using RFA, this risk can be lowered by placing ice in a sterile glove, which is then tied off and left on the skin as a thermal protectant. For cryoablation, warm saline can be poured onto sterile gauze and placed on the skin to prevent freezing.
Bull’s-Eye Technique
Given the viscosity of cement, large needles are used for vertebroplasty. Despite their size, these needles can occasionally become clogged, necessitating needle exchange. Additionally, many operators perform biopsy and vertebroplasty in the same setting. Some needles are designed to be completely removed at biopsy to maximize the size of the retained specimen. To expedite needle replacement, the “bull’s-eye” technique is helpful.38
To perform a needle exchange, the image intensifier is lined up directly over the needle with the stylet removed (Fig. 8.6). The lumen of the needle stands out in stark contrast to the radiodense barrel. After removing the needle, the tract is visible as a radiolucent defect. The needle can be replaced by mimicking the angle of the image intensifier. When the tract is accessed, the needle generally can be advanced to its previous position without resistance.
Postprocedure
One of the principal benefits of ablation and cementoplasty is the rapid onset of pain relief that patients experience. Often, these patients have limited their activities of daily living to accommodate their incapacitating pain. When the pain improves, there is a potential for deconditioned patients to overexert themselves. A report in the literature discussed two patients who had normal neurologic examinations following RFA who developed neuropathy several days after the procedure from overuse.39 There is a normal inflammatory response to ablation that resolves over time. Both patients in the review above responded completely to a short course of steroids.
♦ Difficult Cases
Blastic Metastasis
Generally, percutaneous therapies are most beneficial for patients with lytic disease. It can be difficult, if not impossible, to advance needles safely into the appropriate position for treatment. In this case, the patient was hospitalized with intractable pain from a pelvic metastasis from lung cancer (Fig. 8.7). The tumor had been previously radiated. The IR service was consulted, and treatment was scheduled based on the findings on a recent MRI.
At the time of the procedure, the initial CT images demonstrated that the tumor was largely blastic with one mixed lytic-blastic component. A nonexpandable electrode was chosen given the density of the tumor. However, the needle could not be advanced through the fibrotic cortex. With some difficulty, an 11-gauge needle was advanced into the bone. However, advancing the ablation electrode coaxially through the needle was not an option because of concerns that the current would track along the 11-gauge needle and cause a skin burn. After advancing the vertebroplasty needle close to the far cortex, it was removed. Mimicking the “bull’s-eye” technique described above, the ablation electrode was advanced through the tract, with palpation and ablation performed without incident. Interestingly, despite the relative small portion of the tumor that was treated, the patient had a significant reduction in his pain following the procedure. This case does confirm that patients who are to be treated with ablation should have a CT scan ahead of therapy to evaluate the relative osteoblastic nature of a metastasis.
Cementoplasty to Supplement Suboptimal Ablation
A patient had intractable pain from an iliac wing metastasis from renal cell carcinoma (Fig. 8.8). An angle of approach was selected for ablation that theoretically would have allowed the active tips of the cluster electrode to span the length of the tumor. However, during advancement the tips converged. After two cycles of ablation and attempts at repositioning, the maximal postablation temperature that could be obtained was 51°C. Therefore, an 11-gauge needle was advanced under CT guidance into the tumor. Using CT fluoroscopic guidance, 4 cc of cement was slowly injected, with imaging obtained every half cc. The tumor filled adequately and the patient had resolution of his pain by the following morning.
If this patient’s tumor was near a joint space or in the spine, we would have terminated the procedure following the ablation, with the intention of performing cementoplasty separately under fluoroscopic guidance. Given the distance of the targeted tumor from vital or at-risk structures, we carefully injected the cement in this specific instance with CT guidance.
Fig. 8.7 (A) T1-weighted MRI outlining the extensive metastasis in the left hemipelvis. (B) Prone CT scan prior to ablation reveals the metastasis to have blastic component. Shown is the only lytic portion of this previously radiated metastasis. (C) After failing to advance an ablation electrode, a vertebroplasty needle was advanced into the metastasis. (D) To avoid conduction along the vertebroplasty needle, it was removed, and the ablation electrode was advanced using a modification of the “bull’s-eye” technique.
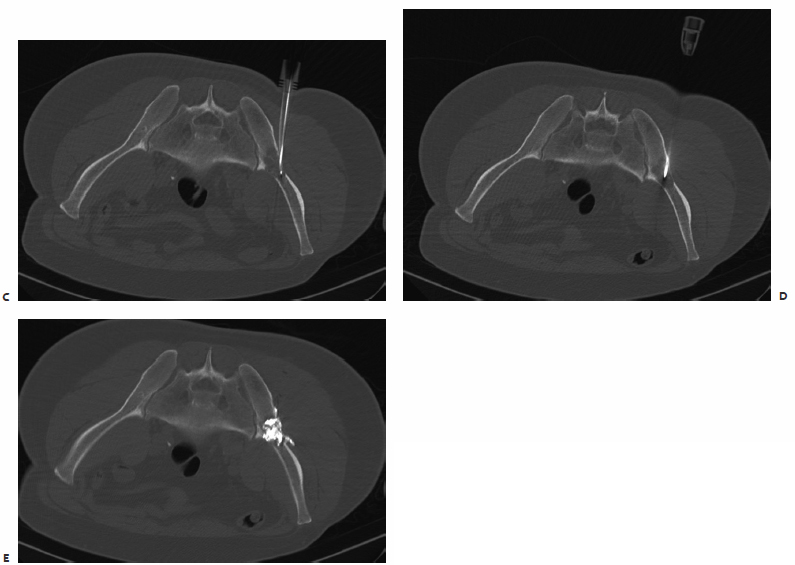
Fig. 8.8 (A) A short-time inversion recovery MRI image demonstrates a metastatic focus from renal cell carcinoma in the left iliac wing. (B) CT scan of the patient in the prone position with the long axis of the malignancy along the length of the bone. Our intention was to try and approximate the long axis of the tumor using a direct posterior approach. (C) Despite multiple repositionings, the ablation electrode cluster grouped together at the anterior aspect of the tumor. Suboptimal temperatures were obtained following multiple rounds of ablation. (D) An 11-gauge needle was advanced into the tumor. (E) Cement injection filled the posterior/untreated component of the tumor as well as the suboptimally treated anterior portion.
- Radiation therapy is the most commonly employed therapy for patients with painful metastases, but the maximum benefit of relief can be delayed up to 4 weeks, and up 50% of patients have no relief of pain at 2 weeks. Radiation may require multiple visits.
- Percutaneous thermal treatments include RFA, cryoablation, and combining thermal ablation with cementoplasty.
- Osteolytic mass-forming bone metastatic lesions are the best candidates for thermal ablation; in general, osteoblastic metastases are not good candidates for ablation.
- The use of cement in painful metastases involving weight-bearing areas, including the vertebral bodies and supraacetabular region, can be synergistic and protect from fractures.
- Use an objective measure of pain to quantify postablation outcomes, such as the 1 to 10 Visual Analogue Scale. Patient selection is the cornerstone to a successful bone pain practice.
- Clinical examination including palpation is important to select only symptomatic sites/levels to avoid treatment of unnecessary sites.
- Managing patient expectations is a key component of the preprocedure consent process, including possible side effects and loss of neurologic function.
- After thermal ablation, pain may improve, with the possible risk that patients will then overexert themselves; patients should be counseled not to do so.
- During cementoplasty, clogging of the access needle may occur. Needle exchange is possible with the “bull’s-eye” technique.