html xmlns=”http://www.w3.org/1999/xhtml” xmlns:mml=”http://www.w3.org/1998/Math/MathML” xmlns:epub=”http://www.idpf.org/2007/ops”>
Case 1
A 62-year-old man is referred to hematology clinic for a new diagnosis of polycythemia vera (PV). His older brother was diagnosed with PV 6 months ago after presenting to an emergency room with a submassive pulmonary embolism and the patient wants to know what his risk is for developing thrombosis and how he can reduce it. His past medical history is notable for hyperlipidemia and hypertension. He has no personal history of thrombosis and is otherwise asymptomatic. His only medications are simvastatin and hydrochlorothiazide. Initial evaluation is notable for a hematocrit of 52% and a platelet count of 620,000/μL.
Epidemiology of thrombosis in myeloproliferative neoplasms
Thrombosis rates are roughly similar among PV, essential thrombocythemia (ET) and primary myelofibrosis (PMF) patients, averaging 4.9% per year in PV, 1.4% per year in ET, and 1.75% per year in PMF (see Table 18.1 for arterial and venous rates).1–3 The remainder of myeloproliferative neoplasms (MPNs) are less well studied (chronic eosinophilic leukemia/hypereosinophilic syndrome, systemic mastocytosis) or known to have a distinct molecular biology (chronic myeloid leukemia). In this chapter, we will focus primarily on thrombosis in ET and PV as the two most common and pathophysiologically similar MPNs.
| Condition | % with history of VTE | % with history of arterial TE | Annual VTE rate (%) | Annual arterial TE rate (%) |
|---|---|---|---|---|
| PVa | 13.7% | 28.7% | 2.1% | 2.7% |
| ETb | 4.1% | 14.5% | 0.6% | 0.8% |
| PMFc | 4.5% | 4.4% | 1.1% | 0.7% |
VTE, venous thromboembolism; TE, thromboembolism; PV, polycythemia vera; ET, essential thrombocythemia; PMF, primary myelofibrosis.
a All patients treated toward goal hematocrit ≤45%, half on aspirin.
b Most patients treated with antiplatelet agents, >75% with cytoreductive agents.
c Patients received a variety of therapies, ranging from watch and wait to cytoreductive agents to anticoagulants.
Risk stratification
Individual thrombosis risk stratification often directs treatment plan in patients with MPNs, including initiation of cytoreductive agents. Current risk stratification is largely based on age and history of thrombosis, as the combination of age >60 years and previous history of thrombosis confers a hazard ratio (HR) of 17.3 for future thrombosis. Patients with neither are considered at low risk, and all others are considered at high risk. A number of other potential risk factors have also been studied in these patients.
Cardiovascular risk factors
Traditional cardiovascular risk factors, including hypercholesterolemia,4 male gender, and smoking,5 are all associated with increased rates of thrombosis, particularly arterial thrombosis, in patients with MPNs.
Thrombocytosis
While thrombocytosis is a hallmark of ET and is often noted in PV, the degree of platelet elevation does not clearly correlate with thrombosis risk. Further implications and management of thrombocytosis will be addressed later in this chapter.
Leukocytosis
Several studies have demonstrated that leukocytosis, both at baseline and during cytoreductive therapy, is a marker of increased risk of primary and recurrent thrombosis in patients with MPNs. Most evidence points to an increased risk of arterial events over venous; the association seems strongest for acute coronary syndromes (ACS).6 The predictive value of leukocytosis appears to be greatest in young, traditionally low-risk patients,7 but these data must be interpreted with caution as the mechanism and clinical significance of this association have yet to be fully established.
Mutations
A number of mutations have been identified in patients with MPNs, including JAK2 V617F in >95% of patients with PV,8 MPL in 5–10% of patients with ET,9 and the more recently identified CALR mutation, which is present in 50–70% of ET patients with wild-type (WT) JAK2 and MPL genes.10 For unclear reasons, the JAK2 V617F mutation appears to be associated with endothelial dysfunction and patients with this mutation have higher levels of circulating immature platelets compared to WT, providing multiple potential mechanisms for increased thrombotic risk.11,12
The majority of studies demonstrate an association between JAK2 V617F and increased thrombosis risk, particularly among ET patients with a high allele burden.13–15 A 2009 systematic review reported a twofold increase in thrombosis risk for JAK2 V617F-positive ET patients vs. WT, but there was significant heterogeneity among the included studies, and the treatment implications of this risk difference are uncertain.16
Compared to persons with either the JAK2 V617F or MPL mutation, patients with CALR mutation appear to have1–3 a lower risk of thrombosis (10-year incidence of thrombosis 5.2% vs. 14.5% in JAK2-positive patients or 19.5% in MPL-positive patients).10
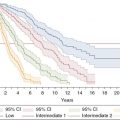
IPSET-Thrombosis score
In light of the increasing number of studies attempting to establish clear risk factors for thrombosis, one group has developed a new risk stratification tool incorporating cardiovascular risk factors and JAK2 V617F mutation status to define a third, intermediate-risk, category among patients with ET.17 Patients in this retrospective analysis were stratified according to a point system, assigning one point each for age >60 years and presence of cardiovascular risk factors and two points each for history of thrombosis and JAK2 V617F mutation. Cardiovascular risk factors were defined as hypertension, diabetes, and active tobacco use. Patients with 0–1 points were classified as low-risk, those with two points were considered intermediate-risk, and those with greater than three points were classified as high-risk.
Of note, most patients who were considered high-risk were treated with cytoreductive therapy during this study (76%), whereas only 37% of patients who were classified as low-risk at the beginning of the study were on cytoreductive therapy by the end. Sixty-eight percent of patients were treated with low-dose aspirin; rates of aspirin use within the different risk categories were not reported. Rates of thrombosis among each group are included in Table 18.2.
| Low risk | Intermediate risk | High risk | |
|---|---|---|---|
| Patients (% total) | 474 (39%) | 471 (39%) | 275 (23%) |
| Thrombosis risk | 1.03% patients/year | 2.35% patients/year | 3.56% patients/year |
While this tool was internally and externally validated prior to publication, it is the product of a retrospective analysis and has not yet undergone rigorous testing by other groups and its role in clinical decision-making has not been defined.
Primary prevention of thromboembolism
Preventing a first clot in a patient with MPN is important not only because both venous and arterial thrombosis are associated with morbidity and mortality, but also because long-term anticoagulation therapy and other interventions that may follow even a relatively minor thrombosis carry risks as well as burdens. For patients with PV who have never had thrombosis, phlebotomy remains the backbone of treatment, but a number of other options exist for PV patients who are likely to benefit from additional risk reduction as well as for ET patients, for whom phlebotomy is not used.
Phlebotomy
Phlebotomy is widely accepted as appropriate and beneficial first-line management in PV patients, particularly as the risk of thrombosis has been clearly correlated with hematocrit. There is now high-quality evidence that targeting a hematocrit less than 45% reduces cardiovascular mortality and major thrombosis risk more effectively than higher hematocrit targets.18
Aspirin
Low-dose aspirin has also been a mainstay of primary prevention therapy since the European Collaboration on Low-dose Aspirin in Polycythemia Vera (ECLAP) trial demonstrated a 58% relative risk reduction in the incidence of thrombosis with the addition of low-dose aspirin to routine treatment of PV.19 This benefit is assumed to extend to most patients with ET, although the evidence that ET patients will benefit from aspirin is less robust.20
Specifically, daily aspirin has been demonstrated to decrease arterial thrombosis rates in low-risk ET patients (JAK2 V617F-negative patients with no history of thrombosis, age <60 years) who have traditional cardiovascular risk factors (smoking, hypertension, hyperlipidemia, or diabetes). Patients in the same study without cardiovascular risk factors did not experience a benefit from aspirin. Among patients with the JAK2 V617F mutation, however, antiplatelet agents appear to lower venous thromboembolism (VTE) rates as compared to no treatment.21
Currently, daily low-dose aspirin is recommended for all PV patients and all ET patients with microvascular symptoms such as erythromelalgia, but the benefit of aspirin for ET patients who are JAK2-negative and have no history of thrombosis, no microvascular symptoms, and no cardiovascular risk factors remains uncertain.
Cytoreductive agents
In general, therapy with cytoreductive agents is reserved for high-risk patients and is used for primary prevention only in patients >60 years or in PV patients who have failed phlebotomy. The use of cytoreductive agents for secondary prevention will be discussed later.
In PV patients, cytoreductive treatment is titrated toward goal hematocrit. However, for both PV and ET, the ideal platelet count is less clear and there is little or no high-quality evidence to support any particular platelet count. Indeed, higher platelet counts (>1,000,000/μL) are more closely associated with hemorrhage than thrombosis and one study actually demonstrated greater thrombosis-free survival in ET patients with higher platelet counts, up to a maximum count of 1,500,000/μL, above which VTE rates increased again.3 There is no high-quality evidence showing that platelet targets lower than 600,000/μL (the target used in the Cortelazzo et al. trial of hydroxyurea vs. placebo22) will further reduce the risk of thrombosis. However, at least one set of expert recommendations suggests that, for cytoreductive therapies being studied in a clinical trial, a platelet count below 400,000/μL should be used, along with other criteria, to define a complete remission.23
If hematocrit can be appropriately lowered with phlebotomy in patients such as the one described here, there is no evidence that additional benefit would be derived from lowering platelet count.
A number of agents, with differing side effect profiles, are currently in use and have been shown to be safe and effective in the MPN patients, including the very elderly. Patients may transition between agents either for intolerance or inefficacy.24
Hydroxyurea
Hydroxyurea is an antimetabolite that leads to panmyelosuppression via prevention of DNA synthesis. The first trial to demonstrate benefit of hydroxyurea was a prospective study of 114 high-risk patients with ET randomized to hydroxyurea titrated to platelet count <600 versus no myelosuppressive therapy. The authors noted a statistically significant reduction in occurrence of thrombosis (24% vs. 3.6%, p = 0.003) in the hydroxyurea group over a 27-month treatment period; the majority of events in the control arm were transient ischemic attack (TIA) or digital microvascular ischemia.22
While concern over possible leukemogenicity persists, no high-quality evidence that long-term hydroxyurea use adds to the inherent risk that a patient with PV or ET will develop myelodysplastic syndrome (MDS), acute myeloid leukemia (AML), or another stem cell neoplasm has ever been published.
Anagrelide
Anagrelide is a phosphodiesterase 3 inhibitor which also reduces platelet production by disrupting the maturation phase of megakaryocytes.
In 2005 a large, randomized controlled trial comparing hydroxyurea plus aspirin to anagrelide plus aspirin in patients with ET demonstrated several important findings. At a median follow-up of 39 months, the primary endpoint (a composite of arterial thrombosis, venous thrombosis, serious hemorrhage, or death from thrombotic or hemorrhagic causes) favored hydroxyurea (odds ratio, 1.57; 95% confidence interval, 1.04–2.37; p = 0.03). However, several secondary endpoints are noteworthy. First, anagrelide was superior to hydroxyurea with regard to prevention of venous thrombosis (3 vs. 14 events, p = 0.006). This VTE benefit was offset by a higher rate of arterial events in the anagrelide group (37 vs. 17, p = 0.004), but this latter difference is based almost entirely on an imbalance in TIAs (14 vs. 1), a poorly standardized outcome that is especially problematic in an open-label study.25
Although evidence more favorable to anagrelide has recently been published, it remains a second-line agent for most patients primarily because, when combined with aspirin, anagrelide confers an unacceptably high rate of serious bleeding, possibly because anagrelide reduces not only platelet number but also platelet function.26 Although an earlier comparison25 to hydroxyurea suggested that anagrelide-treated patients may have a higher risk of progression to myelofibrosis (odds ratio, 2.92; 95% confidence interval, 1.24–6.86; p = 0.01), a more recent study, powered for non-inferiority, suggests that more evidence about the risk of progression to myelofibrosis associated with anagrelide is needed.26
Interferon
Interferon achieves successful cytoreduction by direct suppression of hematologic progenitors, but use is often limited by its well-known side effect profile, including fatigue, flu-like symptoms, anorexia, and depression. Pegylated interferon alfa-2A has demonstrated hematologic response as well as a decrease in JAK2 V617F-positive malignant clones in both ET and PV patients, but very little is known about the impact of interferon on thrombosis risk.27
Interferon is most often used in patients for whom hydroxyurea and anagrelide are contraindicated. Interferon-alpha has been used successfully in some high-risk patients who become pregnant, as it does not have the established teratogenicity of alternative agents and is not known to cross the placenta.28
Alternative cytoreductive agents
Pipobroman and busulfan have been used historically and in a handful of clinical trials, but are not currently in wide use. Pipobroman, used historically in the USA and Europe, had been shown to be effective in reducing hematocrit, but has fallen out of favor since a large prospective trial demonstrated it to be leukemogenic in comparison to hydroxyurea.29 Busulfan appears to effectively lower platelet counts and have low toxicity in elderly patients, but it is likely leukemogenic and efficacy for reducing risk of thrombosis has yet to be confirmed in a trial.
A number of other therapeutic alternatives, including small-molecule JAK2 inhibitors, are under investigation, but no high-quality evidence relevant to thrombosis risk is currently available. The JAK2 inhibitor ruxolitinib, in particular, has been shown to reduce symptoms, including splenomegaly, in patients who are refractory or intolerant to other cytoreductive agents, but has no established role for thrombosis prevention at this time.30
Additional risk factor modification
As in other patients with history of VTE, attention should be paid to modification of lifestyle and careful avoidance of other risk factors, such as smoking and exogenous estrogen sources, including oral contraceptives and hormone replacement therapy.
Case 2
A 57-year-old woman with no known medical problems presents to the emergency room with dyspnea and pleuritic chest pain of several hours’ duration. She has no recent history of travel or injury. She takes no medications and has no personal or family history of thrombophilia. Recent age-appropriate cancer screening, including mammography and cervical cancer screening, is negative. Her vital signs are notable for mild tachycardia and a room-air oxygen saturation of 92%. Physical exam is significant for a painful, erythematous, edematous right calf. Chest computed tomography with angiography reveals large bilateral pulmonary emboli and initial labs demonstrate a platelet count of 900,000/µL with normal hemoglobin. Anticoagulation with low-molecular-weight heparin (LMWH) bridge to warfarin is started. Three months later she returns to her primary care provider to discuss discontinuing anticoagulation. Platelet count at this visit is 830,000/µL and she is found to have the JAK2 V617F mutation.
Prevalence of MPNs in patients who present with thrombosis
Up to one-third of patients with PV and 10–29% of patients with ET will have thrombosis at the time of diagnosis, making it a relatively common presentation of MPNs.31 In one cohort, those diagnosed with ET within 24 months of first thrombosis had longer thrombosis-free survival than those who were diagnosed >24 months after first thrombosis. This highlights the importance of considering MPN as a possible cause of otherwise unexplained thrombosis.3
Mechanisms of thrombosis in MPNs
The pathogenesis of thrombosis in MPNs is multifactorial, incompletely understood, and likely involves multiple mechanisms. Historically, hyperviscosity, stasis, and shear have been accepted as major contributors to the hypercoagulable state but recent data also implicate chronic systemic inflammation.32 On a cellular level, increased activation of all blood cell lines appears to play a role.
Platelets
Platelets, elevated by definition in patients with ET and often those with PV, are found circulating in immature and activated forms in these patients; both are associated with increased risk of thrombosis.11,32 Ex vivo studies also suggest that both ADP-induced platelet aggregation as well as thrombin generation are increased in PV and ET patients as compared to controls.33
Erythrocytes
Increased hematocrit is known to contribute to hyperviscosity and alter shear principles in patients with PV and recent data also suggest alterations in red cell adhesion to the subendothelium mediated by a JAK2 V617F pathway.34
Leukocytes
The mechanism of involvement of leukocytes is less clear, but some studies suggest that elevated leukocyte count (>10,000/μL and especially >15,000/μL) is associated with increased risk of thrombosis.6,7,35 This association highlights the possible importance of inflammation as a possible underlying etiology for thrombosis in MPNs.
Endothelial cells
Endothelial cell activation and/or dysfunction is also suspected of playing a role, at least in part due to altered shear forces resulting from hyperviscosity. Data suggest patients with MPNs have elevated levels of both circulating endothelial cells and endothelial activation markers and that the JAK2 V617F mutation may play a role in the dysfunction of endothelial cells.12,36 Some studies also note increased circulating vascular endothelial growth factor, supporting a theory of increased angiogenesis in ET patients.36
Molecular changes
On a molecular level, several studies have identified inflammatory cytokines, acute-phase reactants, procoagulant microparticles, and expressed adhesion molecules in elevated levels in patients with MPNs and thrombosis.8,32 Specifically, increased rates of thrombin generation and an acquired resistance to protein C appear to play major roles.37
Acute management of VTE
Early management of acute VTE in MPNs does not differ significantly from treatment of non-MPN-associated VTE and generally involves initiation with low-molecular-weight or unfractionated heparins or fondaparinux with subsequent transition to warfarin. The target-specific oral anticoagulants are highly effective for the treatment and secondary prevention of VTE, but there is little or no published experience with these agents in MPN-associated or cancer-associated VTE.
Among patients with arterial events, there appears to be no benefit to treating with anticoagulants prior to starting secondary prophylaxis with antiplatelet agents (31.3% vs. 31.4% rate of re-thrombosis).38 This will be discussed in more detail later in this chapter.
Secondary prevention of VTE in MPNs
After first thrombosis, the cumulative probability of recurrence remains high in all-comers regardless of treatment. Recurrences occur more commonly in the same vascular bed as initial events.38
One large observational study examined rates of recurrent thrombosis in 494 patients with PV or ET and thrombosis followed by an Italian hematology group for a total of 2,952 patient-years. Patients received a variety of treatments, including antiplatelet agents (73%), phlebotomy (31%), pharmacologic cytoreduction (71%), and long-term anticoagulation (18%); the observational (non-controlled) nature of this study means there is significant potential for confounding in the results. That notwithstanding, the combination of cytoreductive therapy plus an antiplatelet agent was associated with a lower risk of thrombosis than either cytoreductive therapy alone (HR = 0.56) or antiplatelet therapy alone (HR = 0.67). A significant decrease in thrombosis risk was associated with combination of a cytoreductive agent (hydroxyurea, pipobroman, busulfan, interferon, or anagrelide) and oral anticoagulation (HR = 0.37 compared to cytoreduction alone; HR = 0.33 compared to oral anticoagulation alone). However, the combination of cytoreductive therapy plus anticoagulation was accompanied by a 2.8% annual risk of major bleeding.38
Since Cortelazzo et al. showed that high-risk patients (more than one-third of whom had prior thrombosis) have a lower rate of thrombosis with hydroxyurea compared to no therapy,22 many patients, like the one in the case we have presented, receive hydroxyurea. That being said, the specific evidence that hydroxyurea reduces the risk of recurrent MPN-associated venous thrombosis is relatively weak.
Pending further evidence, clinical decisions about how to prevent recurrent thrombosis in patients with MPN should probably incorporate general principles that apply to treating thrombosis in the general population (e.g., antiplatelet therapy for arterial thrombosis, extended anticoagulation for unprovoked venous clot).
Case 3
A 68-year-old man with a known history of PV presents with ACS. His medical history also includes chronic obstructive pulmonary disease, hypertension, and diabetes. He has a 50-pack-year smoking history. Current medications include aspirin, metformin, and lisinopril and his PV is treated with phlebotomy, goal hematocrit <45%. Cardiac catheterization demonstrates a 95% occlusion of the right coronary artery and a drug-eluting stent is placed with resolution of symptoms.
Prevalence of arterial events in MPNs
Arterial events, including cerebrovascular and coronary artery events, are even more prevalent than venous thrombosis among patients with MPN. Of the 40% of ECLAP patients who experienced thrombosis prior to enrollment, approximately three-quarters had arterial thrombosis. Over the course of follow-up (median 2.7 years), an additional 5.9% experienced arterial thrombosis.1 Rates in ET appear to be lower, at approximately 1%/year in one large study following 1,144 patients over a median of 5.4 years.3
Stroke/TIA has been shown to be the most common arterial event among patients with ET or PV in most studies to date,1,3,39 and ischemic stroke is responsible for >8% of deaths in PV patients regardless of mutation status. Cardiovascular disease, including myocardial infarction and heart failure, is responsible for another 7–15% of deaths in patients with PV.1,40 As with other thrombotic events, risk does not correlate with platelet count.
One theory for the increased number of arterial events is that atherosclerosis is accelerated by the chronic inflammatory state of patients with MPNs, as evidenced by increased incidence of major thrombotic events in ET and PV patients with elevated inflammatory biomarkers, including white blood cell count, C-reactive protein, and long pentraxin 3.32
Risk factors favoring arterial events over venous in patients with ET include history of thrombosis (HR 2.07), cardiovascular risk factors (smoking, diabetes, and/or hypertension) (HR 1.91) and JAK2 V617F positivity (HR 2.57). Age >60 years, white blood cell count >11, and hemoglobin <12 g/dL appear to favor arterial events to a lesser degree.39
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree