Challenging thrombosis scenarios. MPN, myeloproliferative neoplasm; APC, activated protein C resistance. As reviewed in: Falanga A, Marchetti M. Thrombosis in myeloproliferative neoplasms. Semin Thromb Hemost. 2014;40(3):348–58. Adapted from: McMahon B, Stein B L. Thrombotic and bleeding complications in classical myeloproliferative neoplasms. Semin Thromb Hemost. 2013;39:101–11.
| Query | Comment |
|---|---|
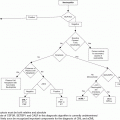
| When should MPN molecular markers be ordered in a patient presenting with thrombosis? | Testing for JAK2 V617F is useful in those with BCS or PVT, even when myeloproliferation is absent; there is less utility in testing for JAK2 exon 12 and MPL mutations, and the prevalence of CALR mutations in AVT is unknown Testing for MPN molecular markers is less productive in CVT, DVT, PE, and arterial thrombosis in the absence of myeloproliferation |
| What is the role for inherited thrombophilia testing in MPN patients? | There is no consistent correlation between MPN, inherited thrombophilia, and thrombosis, and routine testing is not recommended |
| What is the ideal treatment for microvascular disturbances? | Aspirin or other antiplatelet drugs are the initial treatments. The role of twice-daily aspirin, dual antiplatelet therapy, and alternative antiplatelet therapies is not yet clear Cytoreductives may be required in refractory cases |
| What is the ideal anticoagulant to treat MPN-associated thrombosis? What is the optimal duration of anticoagulation? | These questions remain unanswered In AVT, expert opinion suggests indefinite anticoagulation JAK inhibition and pegylated interferon are being evaluated in those with a history of AVT |
BCS, Budd–Chiari syndrome; PVT, portal vein thrombosis; AVT, abdominal venous thrombosis; CVT, cerebral vein thrombosis, DVT, deep-vein thrombosis; PE, pulmonary embolism.
Case 1: part 1
A 70-year-old woman with hypertension and hyperlipidemia experienced redness, swelling, and pain of the right fifth digit. Initially, her symptoms responded to non-steroidal anti-inflammatory drugs, but over the next week, swelling and pain involved all fingers, and her hand had an “angry” red appearance. She was told of possible Raynaud’s phenomenon, and referred to a rheumatologist. The rheumatologist felt that the appearance was inconsistent with Raynaud’s, and recommended a hematology referral after noting thrombocytosis dating back for 2 years. The hematologist did not report splenomegaly, but noted a leukocyte count of 16.4 × 109/L, hemoglobin of 16.6 g/dL, and platelet count of 1500 × 109/L. A reduced erythropoietin level and positive JAK2 V617F mutation test were discovered, confirming the diagnosis of PV. Subsequently, she developed pain in her right leg due to superficial thrombophlebitis, which resolved with supportive care.
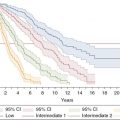
In this case, the focus is on the recognition and management of microvascular disease associated with MPN, which can include erythromelalgia, headaches, transient visual disturbances, transient ischemic attacks (TIA), atypical chest discomfort, dizziness, and numbness. These symptoms have been characterized as “vasomotor” in nature, and have been reported in 28.5% of patients from a large retrospective study of contemporary PV patients.4 In a study of 402 MPN patients, headache, dizziness, and numbness were observed in patients with ET (47.1%, 56.1%, and 58.8%, respectively), PV (52.2%, 52.1%, and 66.2%) and MF patients (44.6%, 58.1%, and 58.1%).5 Erythromelalgia, considered the hallmark microvascular disturbance, is characterized by symptoms of asymmetric redness and burning pain, typically involving the skin of the upper or lower extremities, as well as palms and fingers, or sole of the foot and toes.6 Erythromelalgia is often preceded by pruritus or parasthesias, provoked by exercise or warmth, and relieved by cold or elevation. The condition can mimic superficial thrombophlebitis, and can progress to acrocyanosis, necrosis, and gangrene if untreated.
Erythromelalgia, and other microvascular disturbances such as headache, TIA, and visual disturbances, may share a similar pathophysiology, and accordingly, treatment strategy.6 In ET and PV patients with thrombocytosis, it has been reported that erythromelalgia is a consequence of spontaneous intravascular activation and aggregation of hypersensitive platelets with transient digital arterial occlusion. Supportive evidence includes the demonstration of von Willebrand factor rather than fibrin in stained specimens from erythromelalgic skin areas, implicating a platelet-mediated process.6 In addition, increased urinary thromboxane B2 excretion has been noted, consistent with increased platelet aggregation. Further, in comparison with controls, patients with erythromelalgia had higher levels of thrombomodulin, reflecting endothelial damage, and platelet factor 4 and plasma β-thromboglobulin levels, indicating platelet activation.6 These levels normalized with aspirin, and clinically, microvascular symptoms were promptly relieved. Decrease in the platelet count with cytoreductives (busulfan in this report) also relieved symptoms, whereas coumadin was ineffectual, supporting the concept that platelets mediated the microvascular disease. The investigators cited similar findings of aspirin sensitivity in ET patients who had cerebral, ocular, and coronary microvascular circulatory disturbances.6
Case 1: part 2
Shortly after the diagnosis of PV was established, the patient was started on a treatment program of phlebotomy (goal hematocrit <45%), aspirin 81 mg, and hydroxyurea, titrated to a normal leukocyte count and platelet count. The hydroxyurea was initiated as she was considered to be at higher risk for thrombosis on the basis of her age. Aspirin promptly relieved her erythromelalgia, and she has not had any recurrent microvascular disturbances. During 30 months of follow-up, she has not had a thrombotic event or complication from aspirin or hydroxyurea.
This case highlights the nature and prevalence of microvascular disease in the MPN, including erythromelalgia, which can have a dramatic presentation. It also emphasizes the association between qualitative and often quantitative platelet abnormalities and microvascular disturbances. These disorders stem from hypersensitive platelets which appear to spontaneously aggregate and activate, and are often controlled by antiplatelet agents, such as aspirin.6 Cytoreductive therapy may be necessary to control microcirculatory disease resistant to aspirin. Interestingly, in vitro study suggests that low-dose aspirin, when administered twice daily, may exert a more pronounced inhibition of platelet aggregation by decreasing thromboxane production.7 Neither this strategy, nor the use of dual antiplatelet therapy or alternative antiplatelet agents (clopidrogel), has been validated by clinical trials.
Case 2: part 1
A 27-year-old woman presented with right upper-quadrant (RUQ) abdominal pain. Six years previously, leukocytosis and thrombocytosis were noted during a routine evaluation. She delivered her first child 6 months prior to presentation, and was subsequently placed on oral contraceptives. Three weeks before admission, she had a laparoscopic cholecystectomy because of RUQ discomfort and cholelithiasis. She had a history of smoking 3–4 cigarettes per day. She was evaluated because of persistent RUQ symptoms despite surgery. Her examination was notable for abdominal distension, RUQ tenderness, and obesity. Imaging confirmed ascites, hepatosplenomegaly, and chronic thrombosis of the hepatic vein. She was diagnosed with chronic Budd–Chiari syndrome (BCS); an evaluation for inherited thrombophilia was negative. Her leukocyte count was 10.1 × 109/L, hemoglobin 9 g/dL, and platelets 200 × 109/L; on the same day as this complete blood count result, a bone marrow biopsy revealed a normocellular bone marrow, with only a slight increase in megakaryocytes, and no increase in reticulin fibrosis. Overall, the bone marrow findings were felt atypical for an MPN. However, a subsequent workup confirmed the presence of a JAK2 V617F mutation.
MPN-associated thrombosis can involve atypical sites, resulting in abdominal venous thrombosis (AVT, also known as splanchnic vein thrombosis), which includes hepatic, portal (PVT), mesenteric, and splenic vein thrombosis. Interestingly, MPN can be occult in this context, as myeloproliferation can be masked by splenomegaly, hemodilution, bleeding, and iron deficiency.8 As in the case presentation, AVT can represent the first manifestation of an MPN. Affected patients encountered in this way often fall outside the typical demographic, as these patients are often younger women.9 The mechanism for AVT in those with JAK2 V617F mutations is not clearly established, though it has been shown that endothelial cells of the splanchnic veins harbor the JAK2 V617F mutation, and may interact with activated platelets or leukocytes to increase thrombotic risk.10,11
Recently, a meta-analysis of 1,062 and 855 patients with BCS and PVT, respectively, identified an MPN in nearly 41% of patients.8 The JAK2 V617F mutation was positive in 80.3% and 17% did not have typical MPN features. When classifying the specific MPN subtype, PV and ET were most common (53% and 23%), followed by MPN-unclassified (17%) and MF (7%). In those with PVT, MPN was identified in 31.5%, and 86% had JAK2 V617F mutations; 15% without typical MPN features had a JAK2 V617F mutation. Similarly, PV (27.5%) and ET (26.2%) were the most common MPN subtypes, followed by MPN-unclassified (17.7%) and MF (12.8%). JAK2 exon 12 mutations were not identified, and MPL mutations were found in <1% of cases. This meta-analysis confirms a considerable prevalence of MPN in those with AVT, and validates JAK2 V617F as an appropriate part of the diagnostic evaluation, even when MPN features are absent.8 Regarding prognosis, among 241 patients with AVT, those with BCS and an MPN or JAK2 V617F mutations had more severe liver dysfunction at diagnosis and shorter event-free survival. However, these factors did not influence overall survival, perhaps due to prompt hepatic decompression.12 JAK2 status or the presence of MPN did not impact prognosis among those with PVT.
Treatment of BCS and PVT requires a multidisciplinary approach, with collaboration between the hepatologist and the hematologist. The hepatologist may advise on the need for transjugular intrahepatic portosystemic shunting (TIPS), angioplasty with or without stenting, or thrombolysis, and when hepatic dysfunction is severe, transplantation.13,14 The hematologist may guide anticoagulation, which is often recommended indefinitely in the absence of contraindication. Those with AVT are at higher risk for bleeding when given anticoagulants, because hepatic dysfunction and portal hypertension increase the risk for esophageal varices and portal gastropathy; additionally, gastric varices can complicate splenic varices. In addition to anticoagulation, cytoreduction of thrombocytosis is recommended.13
Case 2: part 2
Initially, the patient was treated with paracentesis to relieve symptomatic ascites, followed by hepatic decompression with a TIPS procedure. She was initiated on heparin anticoagulation, and transitioned to therapeutic warfarin. Unfortunately, follow-up imaging revealed TIPS occlusion, requiring local tissue plasminogen activator, thrombectomy, and angioplasty. Her platelet count at the time was less than 400 × 109/L, but nevertheless, antiplatelet therapy (aspirin 81 mg daily) was added to warfarin. After 2 years of follow-up, she has remained free from thrombotic complications or symptoms from her underlying MPN.
Many unanswered questions remain regarding MPN-associated AVT, and are reviewed by Sekhar et al.14 First, it is not known whether all patients should receive indefinite anticoagulation; in addition, the ideal anticoagulant (low-molecular-weight heparin, warfarin, or target-specific anticoagulants) is uncertain. Second, though recommended, it is not clear if cytoreductive therapy further reduces the risk of recurrence in those affected patients already receiving anticoagulants. Third, the ideal cytoreductive agent has not yet been established in this patient population.14 Fourth, while JAK2 exon 12 and MPL mutations appear to be infrequent, it is not known if CALR mutations are prevalent in those with JAK2-negative AVT. As reviewed by De Stefano et al., CALR mutations appear to be infrequently identified in patients with MPN-AVT.15 Regarding future therapies, a phase II study of ruxolitinib has been initiated in AVT patients, with the hypothesis that this JAK inhibitor would not only address symptomatic splenomegaly, but also decrease pressure in the splanchnic circulation, reducing the risk for variceal formation.16 Finally, an international study will address the safety and efficacy of pegylated interferon in those with a history of AVT (clinical trials identifier NCT0125981).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree