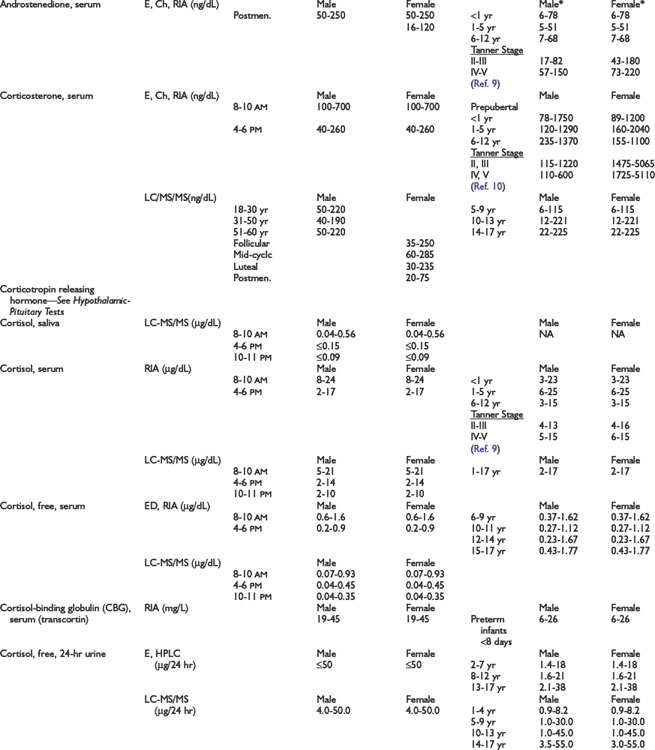
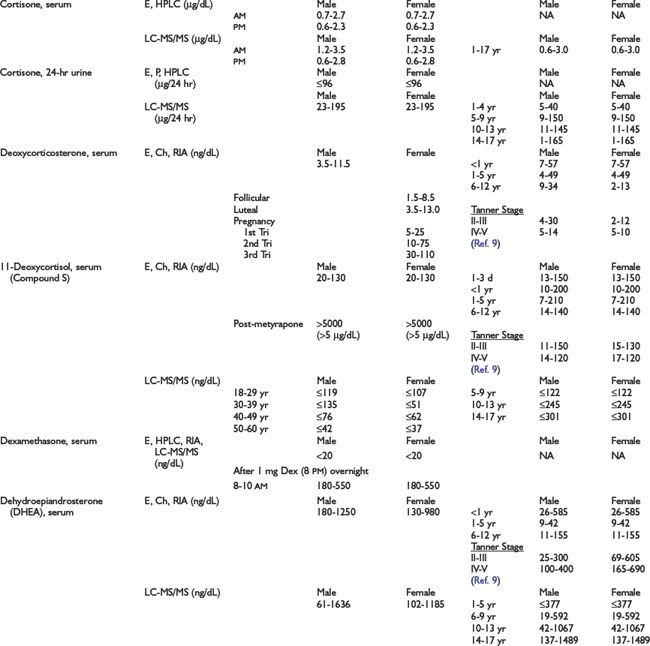
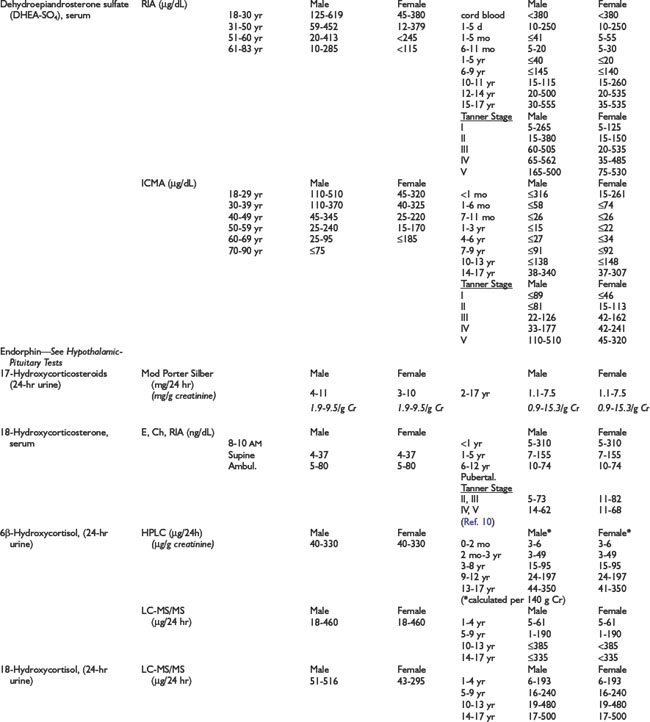
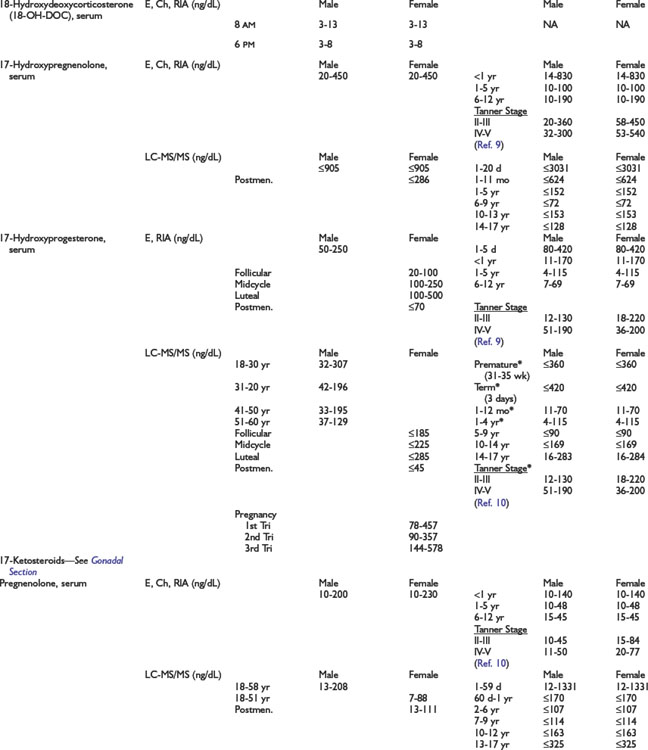
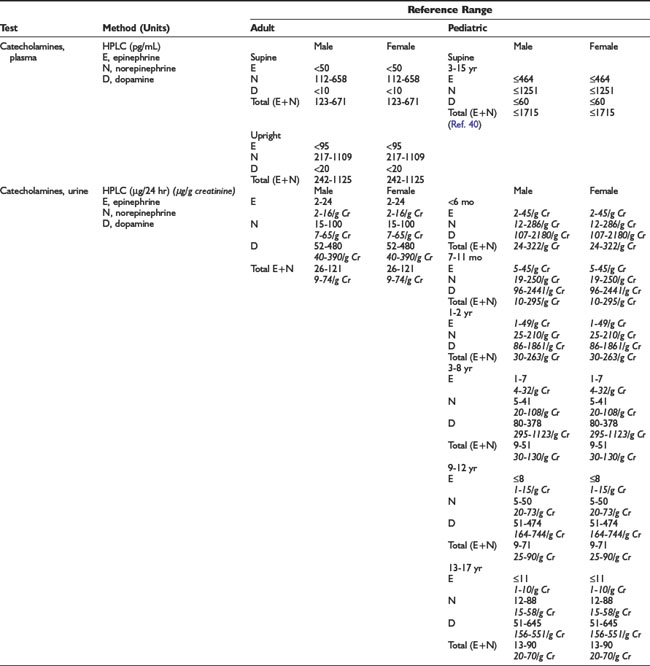
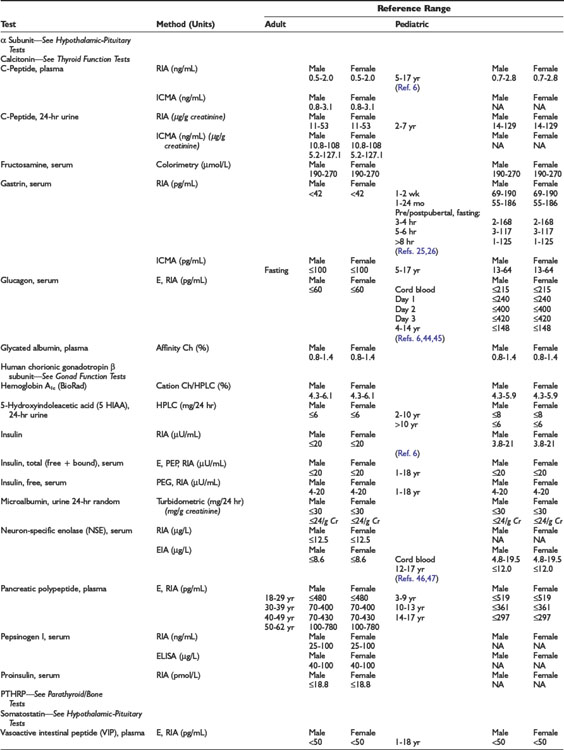
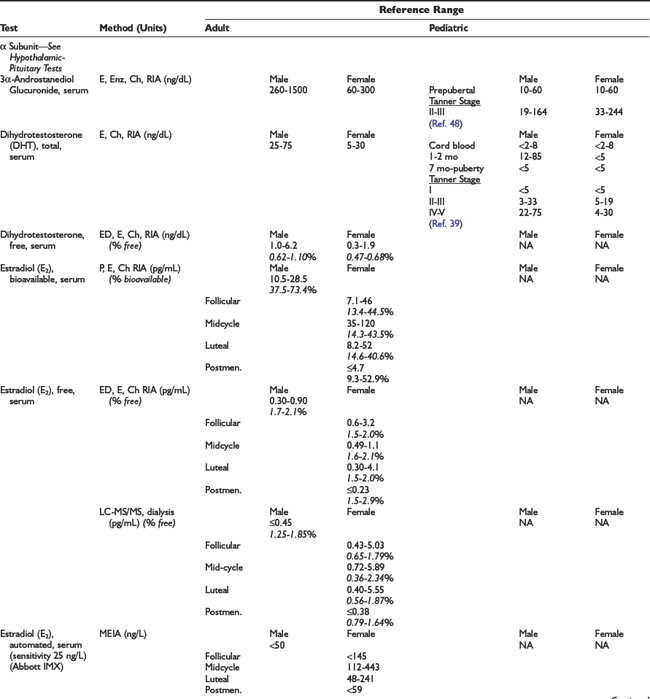
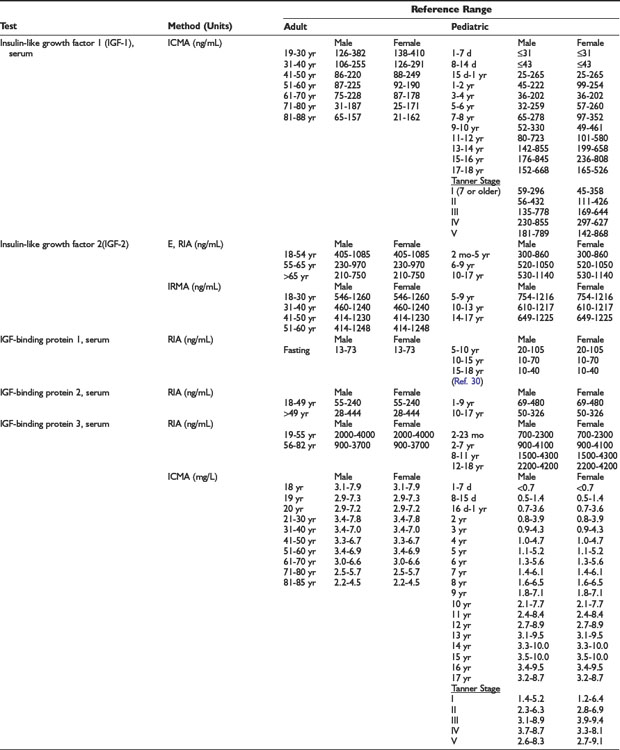
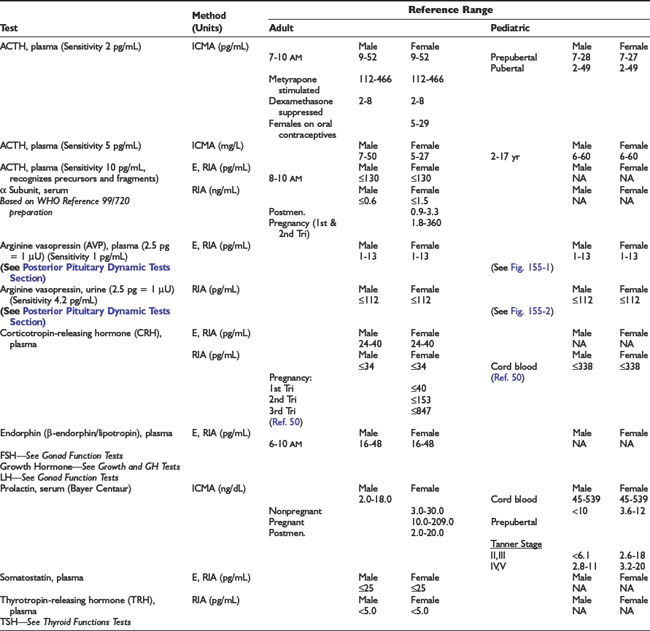
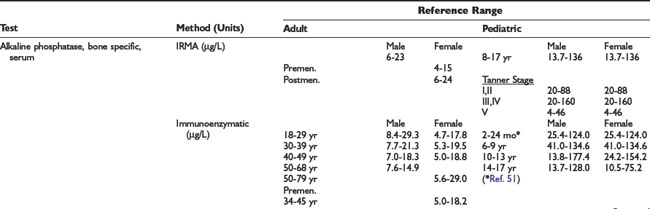
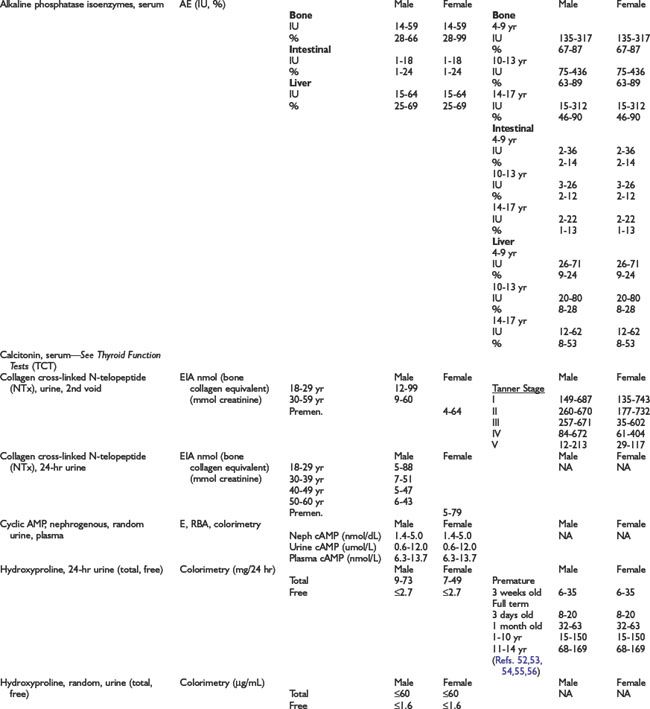
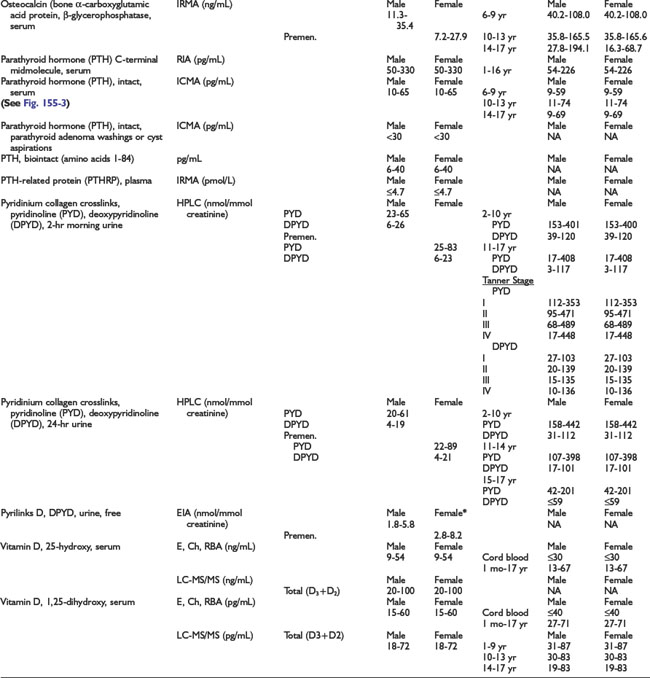
Adrenal Dynamic Tests
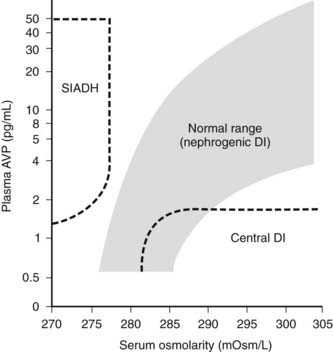
FIGURE 155-1. Relationship of serum osmolarity and plasma arginine vasopressin (AVP) levels in normal subjects and patients with central and nephrogenic diabetes insipidus (DI) and the syndrome of inappropriate antidiuretic hormone secretion (SIADH). The nephrogenic DI range and the normal range are similar (Ref. 1).
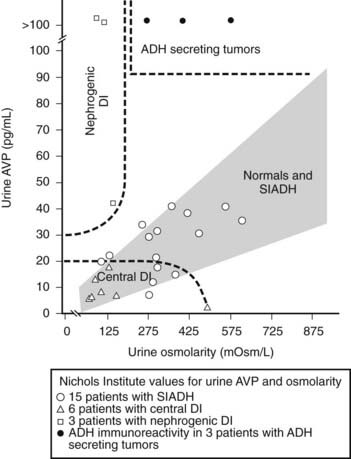
FIGURE 155-2. Relationship of urine osmolarity and urine arginine vasopressin (AVP) levels in normal subjects and in patients with central and nephrogenic diabetes insipidus (DI) and antidiuretic hormone (ADH)-secreting tumors (Ref. 1). SIADH, Syndrome of inappropriate antidiuretic hormone secretion.
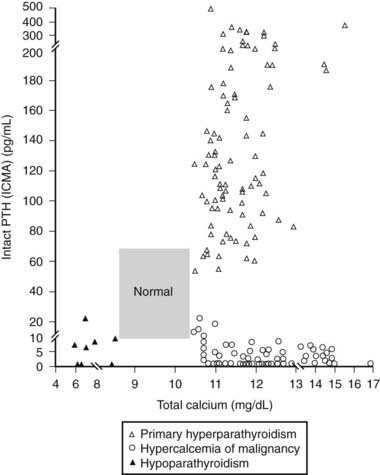
FIGURE 155-3. Relationship of serum (total) calcium and intact parathyroid hormone (PTH) concentrations in patients with primary hyperparathyroidism, hypoparathyroidism, and hypercalcemia of malignancy (Ref. 1). Patients with hypercalcemia of malignancy usually also have elevated levels of parathyroid-related protein (PTHRP). ICMA, Immunochemiluminometric assay.
(Data from Clinical Correlations Department Quest Diagnostics Nichols Institute.)
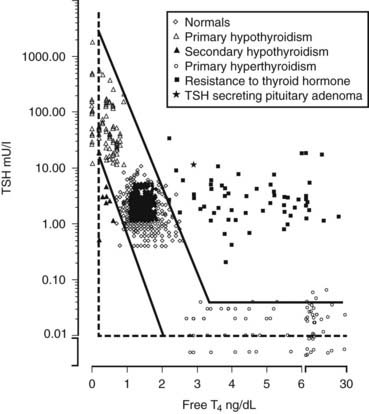
FIGURE 155-4. Relationship of serum free thyroxine (T4) (direct dialysis) and thyroid-stimulating hormone (TSH) concentrations in normal subjects and patients with thyroid disorders (Ref. 1). The log linear relationship is shown, whereby a linear change in serum free T4 produces a logarithmic change in serum TSH concentration. Normal values are shown for 589 normal subjects (289 children, 5 months to 20 years of age and 300 adults 21 to 54 years of age), 84 hypothyroid, and 116 hyperthyroid patients obtained through the Nichols Institute Clinical Correlations division. The samples from patients with thyroid hormone resistance were provided by Dr. Samuel Refetoff of The University of Chicago. Samples from patients with central hypothyroidism were provided by Dr. Peter Singer of the University of Southern California, Dr. Mary Samuels of Oregon Health Sciences University, Dr. Susan Clark of Loma Linda University, and Dr. Arlan Rosenbloom of the University of Florida. All patients were sampled prior to treatment, thus representing the native disease state.
Table 155-1. Adrenocorticotropic Hormone Stimulation Test, Standard56,57
Table 155-2. Rapid Adrenocorticotropic Hormone Stimulation Results in Normal Adults: (250 µg Intravenous Bolus 1–24 ACTH) Glucocorticoid Pathway*
| Steroid | Blood Level (ng/dL) Except Cortisol* | |
|---|---|---|
| 0 | 60 min | |
| 17-Hydroxypregnenolone | 20–450 | 290–910 |
| 17-Hydroxyprogesterone | 17–104 | 42–250 |
| Dehydroepiandrosterone | 230–955 | 545–1845 |
| Androstenedione | 56–135 | 72–290 |
| 11-Deoxycortisol | 21–130 | 82–290 |
| Cortisol (µg/dL) | 6–19 | 14–41 |
* Quest Diagnostics Nichols Institute: 10 females, 10 males, healthy young adults; results are observed ranges (Ref. 1).
Table 155-3. Mineralocorticoid Pathway*
| Steroid | Blood Level (ng/dL) | |
|---|---|---|
| 0 | 60 min | |
| Progesterone | 5–50 | 21–44 |
| Deoxycorticosterone | 3–10 | 14–33 |
| 18-Hydroxycorticosterone | 11–46 | 54–161 |
| Aldosterone | 2–9 | 5–20 |
* Quest Diagnostics Nichols Institute: 10 females, 10 males, healthy young adults; results are observed ranges (Ref. 1).
Table 155-4. Rapid Adrenocorticotropic Hormone Stimulation Results in Normal Children and Adolescents (250 µg Intravenous Bolus 1–24 ACTH) Glucocorticoid Pathway*
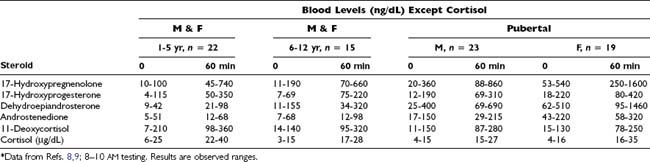
Table 155-6. Adrenocorticotropic Hormone Stimulation Test, Prolonged56
Table 155-7. Adrenocorticotropic Hormone Stimulation Test, Low-Dose56,57
Table 155-8. Low-Dose Short Synacthen Testing (LDSST)11
| Indications: |
| Testing: |
| Expected Results: |
| Comment: |
• There is a high correlation between the ITT and LDSST peak cortisol responses (r = 0.89; p < 0.0001) |
Table 155-9. Aldosterone Suppression Test56,58
Table 155-10. Corticotropin-Releasing Hormone Stimulation Test56
Table 155-11. Corticotropin-Releasing Hormone Stimulation Test, Adrenal Venous Sampling59
Table 155-12. CRH Stimulation Test, Petrosal Venous Sinus Sampling56,60
Indication: Differentiation between ectopic and pituitary causes of Cushing’s disease. This procedure can better distinguish between ectopic and pituitary ACTH hypersecretion than dexamethasone suppression, but it is invasive, may be associated with serious complications, and therefore should be performed by an experienced radiologist. This procedure appears to be less useful for lateralization of the tumor in cases in which the ACTH secretion appears to be of pituitary origin. Sampling: Catheters are inserted via the jugular or femoral veins into both inferior petrosal veins. Blood is obtained from a peripheral vein via a third catheter or from a port in the iliac vein. Blood samples are drawn simultaneously from both inferior petrosal sinuses and the peripheral vein for plasma ACTH. At least 4 sets of 3 samples each are obtained: 2 sets are drawn immediately before injection of CRH as a baseline and 2 more are drawn between 2 and 3 and between 5 and 6 minutes after peripheral intravenous injection of CRH.
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|