Introduction
Endogenous Cushing’s syndrome (Cs) is rare with an incidence of 1,8–3,2 cases per million per year [ ]. Pregnancy is uncommon in women with Cs with fewer than 250 cases reported in the literature. Hypercortisolemia and hyperandrogenism suppress the hypothalamus–pituitary gland–ovary axis and impair follicular development and ovulation, which leads to menstrual irregularities and infertility [ ]. Diagnosis is challenging because pregnancy and Cs may experience similar clinical features and changes such as fatigue, weight gain, hypertension, and hypercortisolemia. However, because Cs leads to serious fetal and maternal complications, early and accurate diagnosis and treatment are crucial.
Hormonal changes to the hypothalamic–pituitary–adrenal axis during pregnancy
Pregnancy is a normal state of hypercortisolism due to hormonal changes to the hypothalamic–pituitary–adrenal axis. It is characterized by a rise in corticotrophin-releasing hormone (Crh), adrenocorticotrophic hormone (Acth), and cortisol levels [ , ].
In nonpregnant women, Crh is mainly produced by the hypothalamus but has also been detected in ovaries [ ]. During pregnancy placenta secretes also Crh more intensively during the second and third trimesters [ , ]. Furthermore, a rise in Crh-binding protein is observed [ ]. Placental Crh, molecularly identical to the hypothalamic form, stimulates maternal pituitary and adrenal glands to produce Acth and cortisol, respectively [ , ]. Plasma Crh rises 1000-fold as gestation progresses with a peak of 4000 pg/mL at 40 weeks of gestation and normalization to nonpregnancy levels 24 hours after delivery [ , ].
Plasma Acth concentrations rise progressively during pregnancy with a peak during labor and delivery [ ]. Causes of Acth higher levels during pregnancy include placental synthesis, higher pituitary response to Crh, or desensitization to cortisol negative feedback [ ].
High levels of estrogen in pregnancy stimulate the production of corticosteroid-binding globulin by the liver at least till the 12th postpartum day, leading to an overestimation of serum total cortisol levels. Both urinary and serum cortisol levels rise during gestation to levels similar to Cs. Total circulating cortisol levels start to increase from the 11th week of gestation with a peak in the first (1.6-fold) and second (2.4-fold) trimesters and a plateau in the third (3-fold) trimester [ , ]. Free cortisol both in urine and serum presents a 3-fold and 1.8-fold increment, respectively [ , ]. Moreover, night time salivary cortisol increases with a peak of 2-fold in the third trimester in comparison to nonpregnancy [ ]. Although cortisol rises (urinary, serum, and salivary), it maintains its circadian rhythm [ ]. Despite the rise of maternal cortisol, the fetus is protected through the placental 11β hydroxysteroid dehydrogenase type 2, which converts active cortisol to inactive cortisone [ ].
Response of Acth and cortisol to exogenous Crh changes during pregnancy. Schulte et al. reported no significant rise in Acth and cortisol levels after an intravenous dosage of 1 μg/kg Crh administration, whereas a dosage of 2 μg/kg Crh in the third trimester produces Acth and cortisol levels similar to nonpregnancy [ , ].
Fetal Crh is mainly produced by the placenta and Acth is detectable at 12 weeks of gestation [ ]. Crh levels progressively increase especially in the late trimester, playing an important role in labor’s onset and fetal lung maturation [ ]. Fetal cortisol is mainly of maternal origin. Fetal adrenal glands, which are larger compared with those of adults, produce mainly dehydroepiandrosterone sulfate and convert progesterone to cortisol [ ]. Another source of fetal cortisol is the amniotic fluid through the conversion of cortisone to cortisol.
Maternal plasma Crh, Acth, and cortisol levels rise rapidly at the onset of labor and delivery. The peak of Crh seems to be 48 hours before delivery. Subsequently, the placenta’s removal leads to decreased Crh and Acth levels 2 hours after birth, whereas a drop of cortisol follows [ ]. On the fourth postpartum day Crh, Acth, and cortisol levels return to prepregnancy levels, whereas the impaired response of Crh to dexamethasone may persist until the fifth postpartum week [ ]. Free and total cortisol levels normalize almost 1 week and two to 3 months postpartum, respectively [ ].
Cushing syndrome in pregnancy
Epidemiology
Cs in pregnancy is rare, with fewer than 250 cases in the literature, because hypercortisolism impairs follicular development and ovulation, leading to amenorrhea [ ].
Primary adrenal pathology (adenomas, bilateral macronodular hyperplasia, or carcinoma) is the responsible cause for more than half of Cs cases (54%) during pregnancy, in contrast to nonpregnant women where Cushing disease (CD) is the main etiology (75%) [ , ]. Other causes are Cd (28%), ectopic ACTH secretion (4%), and iatrogenic (0.5%). Among the adrenal causes, adenomas account for 44%, followed by carcinoma (9.5%) and bilateral hyperplasia (1%) [ ]. Although it is not fully understood, women with Cd are more hyperandrogenic and subsequently less ovulatory compared with those with adrenal pathology [ ]. Furthermore, patients with ectopic Acth present severe hypercortisolism and menstrual disturbances. The aforementioned may explain their low prevalence in pregnancy [ ].
There are also a few cases, known as pregnancy induced Cs, where clinical and biochemical features of Cs are present during pregnancy but remit after delivery [ ]. Cs may be induced either because of adrenal stimulation by placental Acth, high estrogen pregnancy levels or abnormal luteinizing hormone receptors in the adrenal cortex, where the high hormone chorionic gonadotropin levels of pregnancy lead to increased levels of cortisol [ ].
Diagnosis of CS during pregnancy
Clinical features
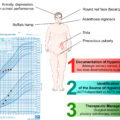
Diagnosis of Cs during pregnancy is difficult because of the physiologic hypercortisolism normally present during pregnancy. Moreover, symptoms and signs of Cs present similarities with those of normal pregnancy. Weight gain, striae, mood changes, hypertension, and diabetes mellitus are commonly present during normal pregnancy and have low discriminative value. However, the report of fetal loss, hypertension, or type 2 diabetes before pregnancy and deep purple striae (especially outside the abdomen), muscular weakness, proximal myopathy, or osteoporosis during pregnancy may imply Cs [ ]. Given the challenging diagnosis, laboratory and imaging tests are necessary.
Screening tests
Laboratory tests in the general population include: 1) measurement of 24 hours urinary free cortisol (Ufc) levels 2) low dose dexamethasone suppression test and 3) midnight serum cortisol or late night salivary cortisol. However, the normal hormonal changes during pregnancy alter the aforementioned, complicating the laboratory diagnosis.
Ufc measurement reflects the circulating free cortisol. In nonpregnant women, a 4-fold increment in Ufc levels is diagnostic for Cs. However, Ufc excretion during pregnancy is normal in the first trimester and rises to 3 times the upper normal value during the second and third trimesters. Thus, only Ufc values greater than 3-fold the upper limit of normal are considered indicative of Cs [ ].
Low dose dexamethasone suppression test is not recommended in pregnancy because of the blunted suppression of cortisol, which can lead to false positive rates [ ].
Late night salivary cortisol is normally elevated 2-fold in pregnancy [ ]. Pregnant women preserve the circadian rhythm, presenting a diurnal cortisol variation, whereas pregnant women with Cs lose this ability. This is a distinguishing difference, indicating Cs.
Determining the etiology of CS
The next step is determining the etiology of Cs by measuring Acth levels, performing dynamic tests (Crh or desmopressin test, high dose dexamethasone suppression test), bilateral and inferior petrosal sinus sampling, or imaging studies [ ].
High levels of cortisol inhibit Acth secretion by the pituitary. In nonpregnant women with autonomous adrenal pathology Acth is reduced (<10 pg/mL), whereas Acth levels are normal or increased in Cd or ectopic Acth secretion [ ]. However, in pregnant women with adrenal adenomas, Acth may not be suppressed because of placental Acth secretion or pituitary Acth stimulation by placental Crh [ ]. Thus, Acth levels are not diagnostic for adrenal Cs during pregnancy, but they are for Cd when they are in the upper of normal reference or even higher [ , , ].
Concerning dynamic tests, the dosage of 100 μg intravenous Crh administration has not largely been used in pregnancy because ovine Crh is a category C drug, used only when is clinically indicated [ ]. The principle of this test is that adrenal tumors or ectopic Acth secretion do not respond to Crh, but pituitary tumors do [ ]. Studies have shown that Crh did not manage to sufficiently increase Acth in pregnancy [ , ]. An alternative to the Crh test is the administration of 10 μg intravenous desmopressin, which is cheaper and more available than Crh but with poorer rates of sensitivity and specificity (84% vs. 77%, 83% vs. 73%) [ ]. Two case reports during pregnancy demonstrated an increment of 70% in Acth 15 minutes after desmopressin administration and a higher response compared with Crh, respectively [ , ].
The basis of the high dose dexamethasone test is the fact that pituitary tumors have a relative responsiveness to glucocorticoid feedback whereas adrenal tumors and ectopic Acth do not. Reports showed that cortisol has decreased by more than 80%, distinguishing Cd from adrenal sources during pregnancy [ , ].
Bilateral inferior petrosal sinus sampling is not recommended during pregnancy, due to the risk of thromboembolic events and radiation [ ].
In cases of hypercortisolism and normal or low Acth levels, an adrenal Mri without contrast or ultrasound could be performed in pregnancy [ ]. A pituitary Mri without contrast should be performed if surgery is planned before birth. It can be diagnostic when pituitary tumor >6 mm are identified [ ]. However, the fact that the pituitary gland is normally enlarged 2-fold by the third trimester can conceal small tumors [ ]. For ectopic Acth cases, a chest or abdominal Mri can be performed.
Maternal and fetal complications
Cs during pregnancy is associated with major maternal and fetal complications. Maternal complications include hypertension (40%–68%), impaired glucose tolerance or diabetes (25%–37%), preeclampsia (14%–27%), and miscarriages (24%) [ , , ]. Less common are osteoporosis and fractures (5%), psychiatric complications (4%), cardiac failure(3%), poor wound healing (2%), and maternal death (2%) [ , ]. Fetal morbidity include prematurity (43%–66%), intrauterine growth retardation or death (5%–24%), stillbirth (6%), and hypocortisolism (2%). Later in life, it seems that female children tend to present a high body mass index [ ].
Treatment
Untreated Cs is associated with adverse maternal and fetal outcomes. Due to the rarity of Cs during pregnancy, treatment is not well-established. Treatment requires a multidisciplinary approach and should be individualized according to gestational age, symptoms severity, patient’s wish, etiology, maternal comorbidities, and potential maternal or fetal risks [ ]. Based on the aforementioned, it can be conservative, medical, or surgical.
Conservative management
Conservative treatment during pregnancy is preferable for cases of mild hypercortisolism without complications or cases diagnosed late in pregnancy. It includes close monitoring of glucose and blood pressure, use of low molecular weight heparin and supportive care when comorbidities are present [ ].
Surgical treatment
First line treatment during pregnancy is surgery. Transsphenoidal pituitary surgery and bilateral adrenalectomy are performed according to etiology. Pituitary surgery is performed in the second trimester and seems to be safe with a remission rate of 77% [ , , ]. Complications include diabetes insipidus, cerebrospinal fluid leak, and inappropriate diuretic hormone secretion. Adrenalectomy has been performed in the second trimester with favorable outcomes [ ].
Medical treatment
Medical treatment, most commonly used in second and third trimesters as second line therapy, includes metyrapone, ketoconazole, cabergoline, mitotane, aminoglutethimide, and cyproheptadine.
Metyrapone, an 11β hydroxylase inhibitor, is the most common medicine used in pregnancy, with a quick onset of action and UFC normalization in a few weeks [ ]. Adverse effects include hypokalemia, secondary to 11β deoxycorticosterone hypertension, preeclampsia, and edema [ ]. It seems that it crosses the placenta in animal studies and recently in a human, thus it may influence fetal adrenal steroidogenesis [ ]. However, no fetal abnormalities have been reported [ , ].
Ketoconazole is a steroidogenesis inhibitor, rarely used in pregnancy [ ]. It crosses the placental membrane with an increased risk of teratogenicity and abortion in rats [ ]. Although ketoconazole has been used in the first trimester, due to its potential antiandrogenic effect it is preferable to be used in the second and third trimesters [ ]. However, recent case reports treated with ketoconazole have reported good outcomes [ , ].
Treatment with cabergoline in pregnancy is not well established. It has been used only in four cases alone or in combination with other medical treatments with good results [ , ]. One drawback is that lactation is disrupted due to its direct effect on the pituitary. Other treatments such as mitotane or aminoglutethimide are not recommended during pregnancy because they can induce teratogenicity and fetal masculinization, respectively [ ]. Cyproheptadine is not recommended due to lack of efficacy [ ].
Postpartum management
Reassessment of the hypothalamic–pituitary–adrenal axis is recommended at 2 to 3 months after delivery and a new pituitary Mri for untreated Cd at 3 to 6 months [ , ]. In the postpartum period, women with Cs should be advised for contraception and those with active disease should be addressed before a new pregnancy [ ]. Due to the risk of thrombotic events, estrogen and progestin oral contraceptives should be avoided [ , ].
Conclusion
Cs in pregnancy is rare probably because of the anovulatory effect of hypercortisolism and hyperandrogenism. Moreover, it is difficult to diagnose due to the overlapping of clinical and biochemical features between normal pregnancy and pregnancy complicated with Cs.
Diagnosis is based on the greater than threefold upper normal limit of Ufc and latenight salivary cortisol levels. For localizing the source of Cs, Acth and pituitary or adrenal Mri imaging without contrast are performed.
Compared with treated Cs, untreated Cs has an increased risk for maternofetal complications. Hypertension, preeclampsia, diabetes mellitus, prematurity and intrauterine growth retardation are some of these. Treatment is necessary for the prevention of these complications and should be individualized. Management by a tertiary center expertized in high risk pregnancies is recommended. First line treatment for cases of moderate to severe hypercortisolism in the second trimester is surgery, whereas medical treatment is preferable for the first and third trimesters. For cases with mild hypercortisolism medical treatment or conservative management are considered.
In postpartum period, the hypothalamic–pituitary–adrenal axis can be reassessed at 2 to 3 months. Contraception should be provided in these patients, especially when active disease is present.
References
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree