Introduction
Adrenal gland disorders, which include adenomas, carcinomas, and bilateral adrenal disease, account for approximately 20%–25% of endogenous Cushing’s syndrome (CS) cases. Bilateral adrenal disease is classified as either macronodular or micronodular, based on nodule size being greater than 1 cm or less than 1 cm in diameter, respectively [ , ]. Although the specific incidence of overt adrenal CS is infrequently investigated, it is speculated to be around 1.5 cases per million annually, given that adrenal tumors are relatively common, affecting 3%–10% of the general population [ ]. Women are more likely to exhibit CS associated with a benign or malignant adrenal tumor, but the pathophysiological explanation for this difference remains unclear [ ]. Radiological studies show that approximately 3% of adults over 50 years old and up to 10% of patients over 80 years old harbor at least one adrenocortical nodule.
Among adrenal incidentalomas, less than 5% are primary malignant tumors (adrenocortical carcinoma, or ACC). The remaining 85% are adrenal adenomas and hyperplasia, with 20%–50% having mild autonomous cortisol secretion (MACS) and 1%–4% being responsible for overt Cushing’s syndrome [ ]. Steroid assay using mass-spectrometry techniques revealed this last decade that most tumors previously considered clinically inactive are, in fact, producing some steroids [ , ].
In benign adrenal tumors that produce cortisol, the main altered signaling pathway is the protein kinase A (PKA) pathway, which affects both unilateral adenomas and bilateral adrenal disease [ , ]. Adrenocortical carcinoma (ACC) rarely causes adrenal CS, with an estimated annual incidence of 0.5–2 cases per million in adults [ ]. ACC can secrete various steroids, and pure cortisol secretion is not its most characteristic feature. This chapter will exclusively discuss benign adrenal tumors causing cortisol excess.
Morbidity associated with adrenal Cushing’s is high and is characterized primarily by the occurrence of diabetes, hypertension, thrombosis, and osteoporosis. Mortality rates for malignant adrenocortical cancer are high, but this is mainly explained by tumor growth and metastasis [ ]. Even when excluding deaths due to ACC, the mortality rate from untreated adrenal Cushing’s syndrome is up to twice that of the general population, primarily due to cardiovascular and cerebrovascular events and infections. Furthermore, a recent meta-analysis showed that patients with MACS have a higher mortality rate compared with those with nonfunctioning adrenal tumors, along with a higher relative risk of developing diabetes, dyslipidemia, and hypertension [ ].
Genetics and molecular findings
Cortisol producing unilateral adenomas
Genetic alterations leading to PKA activation, by affecting the PKA holoenzyme, are the most frequent alterations identified in cortisol-producing adenomas. In 2014, four independent teams identified activating somatic pathogenic variants of the PRKACA gene, which encodes the catalytic subunit α of PKA, in CPA [ , ] ( Table 8.1 , Fig. 8.1 ).
Table 8.1
Genetic alterations in adrenal Cushing.
| CPA | PPNAD | PBMAH | |
|---|---|---|---|
| Somatic alterations | Germline alterations | Postzygotic alterations | |
| PRKAR1A ¤ | PRKAR1A ¤ | MEN1 ¤ | GNAS1 |
| PRKACA ¤ | PRKACA° | APC ¤ | |
| GNAS1 ∗ | (PDE11A) ¤ | FH ¤ | |
| CTNNB1 ∗ | ARMC5 ¤ | ||
| KDM1A ¤ | |||
| DOT1L ¤ | |||
| HDAC9 ¤ | |||
| EDNRA ¤ | |||
| MC2R∗ | |||
| (PDE11A) ¤ | |||
Type of genetic alterations: ¤ loss of function; ∗gain of function; °duplication. Gene function: Italic black = certain causal gene; Italic grey = causal gene to be confirmed; (Italic brackets black) = modifier gene.
Genetic alterations and signalling pathways in adrenal Cushing.
a. Activating pathogenic variants of MC2R (PBMAH). b. Activating pathogenic variants of GNAS1 in McCune Albright syndrome (PBMAH), apart from McCune Albright syndrome (CPA). c. Illegitimate G-protein-coupled receptors expression (PBMAH). d. KDM1A inactivation (germline pathogenic variant and somatic second hit = loss of heterozygosity) (PBMAH). e. APC germline pathogenic variants (PBMAH). f. ARMC5 inactivation (germline pathogenic variant and somatic second hit = pathogenic variant or loss of heterozygosity) (PBMAH). g. FH germline pathogenic variants (PBMAH). h. MEN1 germline pathogenic variants (PBMAH). i. PRKAR1A inactivation (germline pathogenic variant +/_ somatic second hit) (PPNAD) or (point somatic pathogenic variant or LOH) (CPA). j. PRKACA duplication (PPNAD). k. Activating pathogenic variant of PRKACA (CPA).
Somatic PRKACA pathogenic variants are highly specific to patients with overt CS caused by a unilateral adenoma, being absent or very rare in other adrenocortical lesions [ ]. Patients carrying these variants have a more severe endocrine clinical phenotype compared with those with CPA who have no genetic alteration. Approximately 40% of CPAs harbor PRKACA pathogenic variants [ ]. These variants are mainly located in a hotspot region of the PRKACA gene, with the most common being a missense variant affecting amino acid 206 (p.L206R). Previous studies demonstrated that p.L206R alters the interaction between the catalytic and regulatory subunits, which destabilizes the PKA holoenzyme. This destabilization leads to the continuous activation of the mutant catalytic subunit and degradation of the regulatory subunit RIIβ (PRKAR2B) protein of PKA [ ]. Other missense variants of PRKACA have been identified in CPA, with all but one (p.E32V) located in the hotspot region and interfering with the regulatory subunit [ ]. Overexpression of the PRKACA variants p.199_200insW, p.200_201_insV, and p.S213R+insIILR also partially abolishes the interaction with both regulatory subunits RIα and RIIβ.
In addition to PRKACA pathogenic variants, a somatic pathogenic variant in the PRKACB gene, which encodes the catalytic subunit β of PKA, was described in one CPA patient who had severe hypercorticism [ ]. This missense variant, p.S54L, increases the sensitivity of the tetramer to cAMP, thereby elevating PKA activity.
Somatic alterations of the PRKAR1A gene, which encodes the regulatory subunit 1A of PKA, were also described in CPA. These alterations include loss of heterozygosity (LOH) at the PRKAR1A locus 17q, which was found in 7 of the 29 adenomas studied. Inactivating pathogenic variants of PRKAR1A have been identified by Sanger sequencing [ ] and in a whole-exome sequencing (WES) study [ , ]. In rare cases of adrenocortical adenomas, somatic activating pathogenic variants of the GNAS1 gene, which encodes the Gs protein α-subunit, have been reported [ , ]. GNAS1 alterations inactivate GTPase activity, which leads to the constitutive activation of PKA signaling through adenylate cyclase activation [ ]. Activation of the cAMP/PKA signaling pathway causes different pathway alterations in CPA. CPA with GNAS1 pathogenic variants showed an overexpression of extracellular matrix receptor interaction and focal adhesion pathways. In contrast, CPA with PRKAR1A pathogenic variants overexpressed genes related to the Wnt/β-catenin signaling pathway [ ].
Activation of the Wnt/β-catenin pathway was also reported in approximately 40% of adrenocortical adenomas that lacked any somatic PRKAR1A mutation, most of which harbored somatic pathogenic variants of the CTNNB1 gene, which encodes β-catenin [ ] ( Table 8.1 ). The β-catenin protein accumulates in the cytoplasm and nucleus of activated cells. These pathogenic variants lead to abnormal β-catenin activation and induce adrenocortical tumorigenesis [ ]. This constitutive β-catenin activation due to CTNNB1 pathogenic variants is the most frequent molecular alteration identified in both benign adrenocortical tumors (nonsecreting adenomas, CPA, or aldosterone-producing adenomas) and ACC [ ]. Moreover, pathogenic variants of other genes involved in this pathway, such as APC or ZNRF3 , have also been identified in adrenocortical tumors [ ].
Bilateral cortisol producing nodular disease
Beyond CPA, adrenal CS can be caused by bilateral benign tumors, which are divided into two main groups based on nodule size [ ]:
-
•
Micronodular adrenal hyperplasia (MiAH) is defined by bilateral nodules measuring less than 1 cm in diameter. The most common form of MiAH is Primary Pigmented Nodular Adrenal Disease (PPNAD).
-
•
Macronodular hyperplasia is defined by nodules over 1 cm in diameter, the most common form being the Primary Bilateral Macronodular Adrenal Hyperplasia (PBMAH).
The bilateral nature of these tumors suggests a genetic predisposition. Similar to CPA, genetic alterations leading to constitutive activation of the cAMP/PKA pathway have been identified in patients with bilateral disease. Thus, pathogenic variants of the PDE11A and PDE8B genes, which encode for phosphodiesterases (PDEs), have been described in both PMAH and PPNAD [ , , ]. Interestingly, PDEs alterations have been identified in association with other genetic defects, supporting the hypothesis that PDEs may be a secondary event capable of amplifying the activation of cAMP/PKA signaling, resulting in hypercortisolism [ ].
Primary pigmented nodular adrenal disease (PPNAD)
Pathogenic variants of the PRKAR1A gene, located in 17q22-24, explain the majority of PPNAD cases (around 80% of cases) ( Table 8.1 , Fig. 8.1 ). The variants are reported in 37% of sporadic Carney Complex (CNC) cases and more than 70% of familial CNC cases, with almost complete penetrance [ , , ].
Frameshift pathogenic variants of PRKAR1A , which lead to a premature stop codon, were initially described in association with LOH and subsequent loss of the wild-type allele in these tumors [ ]. No hotspot location has been identified, as the PRKAR1A pathogenic variants (more than 100 described to date) are distributed all along the coding sequence of the gene and even in the intronic sequence (affecting splicing). However, a higher prevalence was found for c.709-7del6 and c.491-492delTG pathogenic variants [ ]. These specific variants lead to distinct phenotypes: isolated PPNAD or PPNAD associated with cardiac myxoma, lentigines, and thyroid tumors, respectively. Most PRKAR1A variants (80%) were subject to mRNA nonsense-mediated decay (NMD), resulting in mRNA not being translated into protein. The pathogenic variants escaping NMD (20%) led to the expression of an alternative protein and were responsible for a more aggressive form of CNC. Despite the presence of three other regulatory subunits of PKA (PRKAR1B, PRKAR2A, PRKAR2B), the haploinsufficiency of the regulatory subunit R1A may be sufficient to increase the cAMP/PKA pathway in adrenocortical cells.
In addition, alterations in the regulation of the catalytic subunits of PKA were reported. Triplication of the 1p31.1 chromosome region, including the PRKACB gene, was observed in a patient with CNC who had abnormal skin pigmentation, myxomas, and acromegaly, but no PPNAD [ ]. Copy number gain of the 19p region, including the PRKACA gene, was identified in patients with bilateral adrenal disease and CS, with pathological features similar to those observed in patients with PRKAR1A haploinsufficiency, compatible with PPNAD [ , ]. Functional studies of PRKACA duplication showed a gain of function, resulting in higher protein levels and activation of cAMP/PKA signaling.
PDE11A pathogenic variants have been identified in patients with isolated PPNAD. A causative role was suspected when three germline pathogenic variants of PDE11A were identified in patients with PPNAD who lacked PRKAR1A alteration [ ]. Single nucleotide variants (SNV) of PDE11A , which showed decreased enzymatic activity in vitro [ ], were also found more often in CNC patients with PRKAR1A pathogenic variants who developed PPNAD (25.3%) compared with those who did not develop PPNAD (6.8%), suggesting that PDE11A could also act as a modifier gene [ ]. In addition to the cAMP/PKA pathway, the involvement of the Wnt/ beta-catenin pathway was also reported in patients with PPNAD, with some somatic CTNNB1 pathogenic variants and Beta-catenin accumulation noted in some tumors [ , ].
Linkage studies suspected a second potential locus in 2p16 responsible for CNC, where LOH and gains in the number of copies were found, suggesting the potential presence of an oncogene that has not yet been determined [ , ]. This region includes Proopiomelanocortin ( POMC ) gene and DNA-mismatch repair gene MSH2 , but those genes were further excluded [ ].
Primary bilateral macronodular adrenal hyperplasia (PBMAH)
Primary bilateral macronodular adrenal hyperplasia (PBMAH) is the most common etiology of CS due to bilateral adrenal involvement. It is usually considered to represent less than 1% of endogenous cases [ , , ]. However, the diagnosis of PBMAH is becoming more frequent following the investigation of patients with bilateral adrenal incidentalomas associated with mild autonomous cortisol secretion (MACS), suggesting a higher prevalence than previously described [ ].
Several different names have been used for this disease: adrenocorticotropic hormone (ACTH)-independent massive bilateral adrenal disease (AIMBAD) [ ], massive macronodular adrenal hyperplasia or disease (MMAD) [ ], ACTH-independent macronodular adrenal hyperplasia (AIMAH) [ ], macronodular adrenal hyperplasia (MAH) [ ], bilateral macronodular adrenal hyperplasia (BMAH) [ ], primary macronodular adrenal hyperplasia (PMAH) [ ]. The term “ACTH-independent” was abandoned after it was shown that, in some forms of the disease, intraadrenal ACTH synthesis was responsible for the local stimulation of cortisol production [ ]. Due to the discovery of several genetic alteration causing the disease, some articles introduced the term bilateral macronodular adrenocortical disease (BMAD) [ ]. The new term was endorsed by the 2022 World Health Organization (WHO) pathological classification of adrenal tumors [ ].
PBMAH can be part of genetic tumors predisposition syndromes, due to pathogenic variants of MEN 1 in type 1 multiple endocrine neoplasia (NEM1), of APC in familial adenomatous polyposis and (APC) or FH in hereditary leiomyomatosis- kidney cancer syndrome [ ] ( Table 8.1 , Fig. 8.1 ). These syndromic forms are rare, and isolated PBMAH is the most common form of the disease [ ].
Given the description of familial cases and the bilateral adrenal involvement in isolated PBMAH, specific genetic alterations leading to this disease have been suspected. In one study of leukocyte and tumor DNA from 33 patients with isolated PBMAH who underwent adrenalectomy, LOH was detected at the 16p locus by SNP array in 24% of the cases [ ]. Consequently, ARMC5 gene alterations, which map to 16p11.2, were identified as the first gene responsible for isolated PBMAH ( Table 8.1 , Fig. 8.1 ).
ARMC5 acts as a tumour suppressor gene: a first event (mostly frameshift or nonsense variants) is present in the DNA of all cells, and a second hit (point pathogenic variant or LOH) is specific to each adrenal nodule. Other studies subsequently confirmed the involvement of ARMC5 in the pathophysiology of PBMAH in cohorts with sporadic or family cases [ , , , ]. Pathogenic variants of ARMC5 are found in around 21%–26% of PBMAH cases [ , ].
The precise function of ARMC5 is not yet fully understand. It is a cytosolic protein, containing seven armadillo domains, as beta-catenin, and one BTB domain [ ]. Functional studies showed that inactivation of ARMC5 in human adrenocortical cancer H295R led to a decrease in the expression of steroidogenesis enzymes and less efficient cortisol synthesis. However, ARMC5 missense mutations or deletions led to reduced apoptosis [ , ]. Thus, despite a decreased steroidogenic capacity of each cell, the increased number of adrenocortical cells due to the impaired apoptosis leads to a global excess in cortisol production [ ]. Moreover, ARMC5 interacts with the E3-ubiquitin ligase cullin3 through its BTB domain, leading to ARMC5 ubiquitination and further degradation by the proteasome and maybe cell cycle (dys)regulation [ ]. Recently, genomic studies identified a common pattern of gene expression in patients with ARMC5 pathogenic variants, characterized by a gonadal transcriptomic signature, a specific miRNome clustering and a global hypermethylation in CpG islands [ , ].
The aberrant expression of G Protein–Coupled Hormone Receptors (GPCRs) in PBMAH, which leads to increased secretion of cortisol in response to various hormonal stimuli, has been largely studied. Several stimulating ligands, binding to GPCRs, have been described: Glucose-dependent insulinotropic peptide (GIP) responsible for food-dependent Cushing syndrome [ ], LH/HCG responsible for Cushing syndrome during pregnancy and after menopause [ ], β-adrenergic receptors (β-AR), vasopressin (V2–V3-vasopressin receptor), serotonin (5-HT7 receptor) and glucagon [ ] ( Fig. 8.1 ). The prevalence of illegitimate membrane receptors in PBMAH is high, varying from 77% to 87% among studies [ , ].
The prevalence of illegitimate membrane receptors in PBMAH is high, varying from 77% to 87% among studies. The prevalence of cortisol response to illegitimate β-AR, V2–V3-vasopressin receptor, and 5-HT7 receptor is similar in patients with ARMC5 pathogenic variants and wild-type patients [ , ]. Conversely, ARMC5 pathogenic variants have never been reported in GIP-dependent PBMAH related to ectopic GIPR expression, which is consistent with the recent identification of KDM1A inactivation in food-dependent Cushing and the mutual exclusion of KDM1A and ARMC5 pathogenic variants ( Table 8.1 , Fig. 8.1 ).
KDM1A , also called LSD1 , encodes the lysine demethylase type 1A [ ], and acts in PBMAH as a tumor suppressor gene, with germline pathogenic variant associated with somatic LOH. In affected adrenal nodules from affected patients, KDM1A mRNA and protein levels were when compared with PBMAH without food-dependent Cushing. The precise mechanism of regulation of GIPR expression by KDM1A is not fully understood, but it could be involved in epigenetic regulation of gene expression and regulation of other cellular pathways by several mechanisms including protein-protein interactions, protein stability, regulation of subcellular localization, or promoter binding [ ].
Potentially inactivating variants of ACTH receptor ( MC2R ), which could cause a loss of ligand binding and responsiveness and subsequent autonomous cortisol secretion have been reported in some rare cases of PBMAH [ ] ( Table 8.1 , Fig. 8.1 ). Moreover, postzygotic activating mutations of GNAS1 , results in constitutive activation of cAMP/PKA pathway and autonomous cortisol secretion in the context of Mc Cune Albright syndrome [ , ] ( Table 8.1 , Fig. 8.1 ). Finally, WES studies in patients with PBMAH reported the involvement of other potential causal genes as DOT1L (coding for a histone H3 lysine methyl-transferase), HDAC9 (coding for a histone deacetylase) and Endothelin Receptor type A ( EDNRA ) gene [ ].
Diagnosis
Clinical presentation
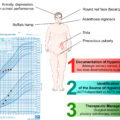
Regardless of the etiology, patients with cortisol-producing adrenal tumors can present with Cushing syndrome (CS) of varying severity. In adults, specific catabolic signs of CS include proximal muscle amyotrophy, wide purplish striae, bruising without trauma, and thinning of the skin. Less specific signs include facial plethora, buffalo neck, supraclavicular fat pads, and abdominal obesity [ ]. CS can also lead to various complications, such as diabetes, dyslipidemia, hypertension, osteopenia or osteoporosis, obstructive sleep apnea, hypogonadotropic hypogonadism, and immunosuppression. Menstrual irregularities or amenorrhea are common in women with CS due to the suppression of gonadotropin-releasing hormone secretion by high cortisol levels, which leads to low luteinizing hormone and follicle-stimulating hormone levels [ ].
The number and severity of CS symptoms are variable, depending on the degree and duration of hypercortisolism and likely individual factors. Adrenocortical adenomas are often responsible for mild autonomous cortisol secretion (MACS) with few or no specific signs of CS, but they are usually associated with glucose intolerance, obesity, and hypertension[ ]. In women with adrenocortical carcinoma or ACTH-stimulated hyperandrogenism, signs of androgen excess—such as hirsutism, acne, oily facial skin, and increased libido—can be present [ ].
PPNAD and PBMAH may have syndromic presentations, making it essential to look for associated manifestations. PPNAD is the most frequent endocrine manifestation of CNC, a tumor predisposition syndrome, reported in 26%–60% of patients [ , ]. The clinical presentation is variable, with hypercortisolism that is more or less marked, often diagnosed in the first 2 decades, and can have a sudden, insidious, or cyclic onset [ , ]. Manifestations of CNC that can be associated with PPNAD are cardiac or cutaneous myxomas, lentiginous spots, thyroid tumors, acromegaly, testicular tumors, schwannoma, and osteochondromas [ ]. Most PBMAH patients present with CS in their fifth and 6th decades, unless it is part of McCune-Albright syndrome. There is a slight increased prevalence in females, around 60% of cases [ , , ]. Patients can present with manifestations of associated genetic syndromes, such as multiple endocrine neoplasia type 1 syndrome (mainly hyperparathyroidism and pituitary adenoma), familial adenomatous polyposis, and hereditary leiomyomatosis and renal cell cancer disorder. Moreover, in PBMAH patients presenting with KDM1A pathogenic variants, multiple myeloma or monoclonal gammopathy of unknown significance (MGUS) might occur, as this gene predisposes to familial multiple myeloma [ , , ]. Meningioma has also been reported in PBMAH patients with an ARMC5 pathogenic variant.
Hormonal evaluation
The diagnosis of adrenal CS implies a 2 steps approach: first the diagnosis of the hypercortisolism and second the diagnosis of its adrenal cause. First it is essential to exclude an exogenous glucocorticoid intake. The diagnosis of CS is confirmed when at least two different first-line screening tests are abnormal [ ]. These include: abnormal overnight 1-mg Dexamethasone Suppression Test (DST) with morning cortisol cutoffs <1.8 μg/dL or 50 nmol/L, increased urinary free 24-hour cortisol (UFC), and increased late-night salivary cortisol levels. Other hormonal investigations, such as midnight serum cortisol or loss of cortisol circadian rhythm, have been described for diagnosing CS but are less commonly used. Moreover, in some situations, the Dexamethasone-Corticotropin Releasing Hormone (CRH) test or the Desmopressin test should be considered to rule out nonneoplastic hypercorticism. The 1-mg Dexamethasone suppression test is considered highly sensitive for adrenal Cushing. When this test is the only alteration, and the cortisol level is between 50 and 138 nmol/L, the patient is considered to have MACS [ ]. Some rare patients have cyclic CS, characterized by excess cortisol episodes alternating, regularly or irregularly, with periods of normal cortisol secretion [ ]. For these patients, UFC or late-night salivary cortisol has been suggested to be a better diagnostic tool than the DST. If initial testing is normal but clinical suspicion is high, repeated testing is suggested.
Once the diagnosis of CS is established, measurement of plasma ACTH is essential to confirm adrenal CS. Plasma ACTH concentrations are low in most patients with adrenal CS, but up to 25% may present values within the normal range. This can be due to assay methodology, incomplete suppression of pituitary ACTH secretion, or adrenal tumor ACTH production [ , ]. Conversely, around 10% of patients with Cushing’s disease present with low ACTH levels (below 10 pg/mL or 2 pmol/L), blurring the boundaries between the etiologies. In these cases, CRH stimulation test can be useful to differentiate patients with pituitary-dependent hypercortisolism, who respond with a raise in ACTH levels, from patients with adrenal Cushing, who do not [ ].
More recently, steroid measurement by liquid chromatography-tandem mass spectrometry (LC-MS/MS) has been shown to be efficient in discriminating patients with adrenal CS from ACTH-dependent CS, and even benign from malignant adrenocortical tumors [ , ]. Even if LC-MS/MS steroid profiling is not yet commonly used in routine clinical practice, this is a very promising approach that should improve the diagnosis and follow-up of adrenal tumors.
Some specific hormonal work-up can be useful in identifying the causes of adrenal Cushing. In patients with PPNAD, a paradoxical increase in cortisol secretion has been shown during the Liddle’s test, which consists of administering 2 mg per day of Dexamethasone for 2 days, followed by 8 mg/day for 2 days [ ]. An increase of more than 50% of the basal level in UFC on the second day of high-dose dexamethasone administration supports the diagnosis of PPNAD. Even if this paradoxical cortisol response is used as a biological criterion for the diagnosis of the disease, it is not pathognomonic [ ]. It has been observed in 69% of patients with PPNAD, but also reported in patients with cortisol-producing adrenocortical adenomas, attributed to the occurrence of somatic PRKAR1A pathogenic variants [ ].
Patients with GIP-dependent and overt CS often present a specific phenotype, characterized by low fasting plasma cortisol levels that increases after meal intake. GIP-dependent CS has been reported both in patients with PBMAH, occurring mainly in women [ , ], and in patients with unilateral adenomas [ , ]. Ectopic GIPR can be diagnosed by an increase over 50% of serum cortisol 2 hours after an oral 75-g glucose load or a mixed meal, compared with basal cortisol level.
To demonstrate the presence of other aberrant GPCR expression, which is frequently observed in cortisol secretion dysregulation in PBMAH, the response to different stimuli could be assessed. Different protocols have been described to identify aberrant receptors in adrenal CS [ , ]. They consist of modulating the plasma levels of diverse hormone ligands, either directly or indirectly. All tests are performed following an overnight fast and with the patient in a supine position for at least 1 hour. The full research protocol lasts 3 days and sequentially includes: a posture test to screen for receptors to angiotensin II, vasopressin, or catecholamines; a standard mixed meal to assess the presence of GIP or other gastrointestinal hormone receptors; and a 250 μg intravenous (IV) ACTH 1–24 test as a reference. Administration of GnRH 100 μg IV evaluates responses to GnRH, LH, and follicle-stimulating hormone; IV TRH 200 μg assesses modulation by TRH, thyrotropin, or prolactin. On the third day, sequential intramuscular (IM) administration of glucagon 1 mg, vasopressin 10 UI IM, and 10 mg orally metoclopramide (HTR4 agonist) are performed. Measurements of ACTH, cortisol, and other steroids are performed at 30- to 60-minute intervals for 2–3 hours following the intervention. A raise from baseline steroid levels of 25%–49% defines a partial response, and a raise over 50% defines a complete response. Further tests may be performed in case of a positive response to better define the hormone and the specific receptor involved. These tests have been very useful for research, but today their practical implications for clinical care are limited.
Imaging
Several imaging techniques may be used for the identification and diagnosis of adrenal tumors. Computed tomography (CT) and Magnetic resonance imaging (MRI) are mainly used to identify benign lesions [ , ]. Conversely, [18F]2-deoxy-D-glucose (FDG)-positron emission tomography/computed tomography (PET/CT) is primarily used for the detection of malignant disease [ ] (not treated in this chapter). CT has high spatial and quantitative contrast resolution, which allows for the assessment of tissue density by measuring the X-ray absorption of tissues. Noncontrast (or “unenhanced”) CT is currently the most widely used imaging method for adrenal tumor investigation. MRI is a second-line imaging modality for assessing adrenal lesions, particularly useful in children, pregnant women, and patients with claustrophobia or allergy to iodine contrast reagents. Similar to CT, MRI is reliable for evaluating the fat content of an adrenal mass: the so-called chemical shift observed in lipid-rich lesions is defined by a drop of signal on the T1-weighted out-of-phase images compared with the T1-weighted in-phase images. In cortisol-secreting adrenal masses, 131-iodine-norcholesterol scintigraphy or NP-59 scintigraphy can also be performed. This can be valuable to ascertain atypical cases of bilateral lesions or assess modest cortisol secretion in adrenal incidentalomas, but it is performed only in a few specialized centers [ , ].
Unilateral cortisol producing adenomas
Adrenal adenomas are usually regular shaped, homogeneous, lipid-rich masses, less than 4 cm in diameter on CT imaging [ ]. Thus, the current European guidelines on the management of adrenal incidentalomas, consider that all homogenous adrenal masses with a spontaneous density ≤10 Hounsfield units (HU) or HU between 11 and 20 and a tumor size <4 cm are adenomas [ ]. Even if, unenhanced attenuation value has a poor sensitivity to diagnose an adrenal adenoma in case of cortisol oversecretion due to poor lipid content, the accuracy of washout analysis is preserved [ ]. On MRI adrenal adenomas are characterized by low adrenal-liver and adrenal-spleen ratios and loss of signal intensity on out-of-phase images ( Fig. 8.2 ).
Abdominal-pelvic unenhanced computed tomography scan (axial section) showing a left cortisol producing adenoma of 22 mm of diameter and spontaneous density of 24 Hounsfield Units and right adrenal atrophy without mass syndrome.
PPNAD
Normal adrenal glands on CT scan or MRI were reported in about 20% of the patients with PPNAD according to the literature [ , ]. Adrenal glands may also appear as moderately enlarged and high definition CT scans (slice thickness less than 5 mm) may visualize small round hypodense nodules, usually less than 10 mm ( Fig. 8.3A ). Moreover, in some adult patients have also been described adrenal macronodules associated with micronodules [ ]. NP-59 scintigraphy shows in patients with PPNAD a bilateral adrenal uptake, rarely asymmetrical [ ] ( Fig. 8.3B ).
Adrenal imaging of a primary pigmented nodular adrenal disease.
(A) Computed tomography scan showing slight adrenal enlargement due to subcentimetric nodules (B) NP-59 scintigraphy showing bilateral asymmetrical adrenal uptake.
PBMAH
The most common radiological presentation of PBMAH is a bilateral enlargement of the two adrenal glands, harboring at least one macronodule, more than 1 cm in diameter, with internodular hyperplasia (or less frequently hypoplastic internodular tissue) [ , ] ( Fig. 8.4A ). In some cases, there is no clearly visible nodule and just a diffuse hyperplasia. On noncontrast CT scan attenuation values of PBMAH can range from less than 10 HU up to 25 HU [ , ]. According to different series, patients with ARMC5 pathogenic variants have significantly larger adrenals and more numerous nodules than wild-type patients, correlated with the level of cortisol excess, and some patients with KDM1A pathogenic variants present with myelolipoma. Regarding MRI imaging, PBMAH nodules were described as isointense relative to muscle with T1-weighted sequences and hyperintense relative to liver with T2-weighted sequences in previous studies. NP-59 scintigraphy shows bilateral adrenal uptake, but when asymmetric can guide the indication of unilateral adrenalectomy ( Fig. 8.4B ).
Adrenal imaging of a primary bilateral macronodular adrenal hyperplasia.
(A) Computed tomography scan showing bilateral adrenal enlargement due to multiples nodules over 1 cm diameter (B) NP-59 scintigraphy showing bilateral symmetrical adrenal uptake.
Pathology
Cortisol producing unilateral adenomas
Macroscopy
At gross examination, CPA are usually single, round, or oval shaped nodules measuring less than 5 cm. They are well-delineated, intraadrenal and golden-yellow or light brown in color [ , ]. Most cases are homogeneous, but some CPA can present with hemorrhage or cystic remodeling [ ]. Due to autonomous cortisol production, the adrenal cortex is atrophic (less than 2 mm thick in adults) [ ] ( Fig. 8.5 ).
Macrophotograph of two sections of a typical cortisol producing adenoma (white arrowhead). The adjacent adrenal gland is atrophic (black arrowhead).
Microscopy
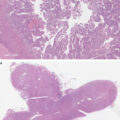
Microscopically, CPA are composed of nest or cords surrounded by capillaries and small fibrous trabeculae highlighted by Gordon Sweet histochemistry [ ] ( Fig. 8.6A ). They are made of clear cells with abundant cytoplasm rich in lipidic microvacuoles similar to zona fasciculata. The nucleus is round with regular contour and can be nucleolated. There is a variable proportion of compact eosinophilic cells similar to the zona reticularis [ ] ( Fig. 8.6B ). Most cases present with no sign of aggressiveness. The mitotic index is low (<5 mitosis/10 mm 2 ) with no atypic mitosis. Necrosis area and signs of invasion (capsular, venous, or sinusoidal) are generally not present [ ]. The diagnosis is based on Weiss score [ ]. The WHO 2022 classification of adrenal cortical tumors also endorses the reticulin algorithm, based on Gordon Sweet staining [ ] and the Helsinki scoring system based on mitotic index, tumoral necrosis and Ki67 immunohistochemistry [ ].
Microphotographs of a cortisol producing adenoma.
(A) the adenoma (white arrowhead) is round and well distinguished from the adjacent atrophic adrenal gland (black arrowhead), HES staining, magnification ×25. (B) The adenoma is composed by a majority of clear cells nests surrounded by capillaries and fibrous trabeculae (black arrowhead) and by nests of compact eosinophilic cells (white arrowhead), HES staining, magnification ×200.
PPNAD
Macroscopy
In PPNAD, the adrenal glands are of normal or reduced size and weight [ ]. On section, both adrenal glands contain multiple round, dark-brown nodules less than 5 mm in diameter [ , ]. These nodules are separated by areas of atrophic adrenal cortex [ ] ( Fig. 8.7 ).
Macrophotograph of 3 sections of a primary pigmented nodular adrenal disease with multiple round pigmented nodules separated by atrophic adrenal cortex (white arrowhead).