Disclosure Statement
CT received research funding on treatment of abnormal growth hormone secretion by Pfizer. CF has nothing to disclose.
Introduction
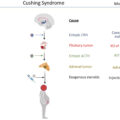
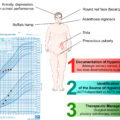
Cushing’s syndrome (CS) refers to exposure of the body to supraphysiologic levels of glucocorticoids [ ]. Exogenous CS (often referred to as iatrogenic) caused by administration of pharmacologic doses of adrenocorticotropic hormone (ACTH) or most commonly glucocorticoids is not uncommon due to the widespread use of these medications for the management of various disorders (autoimmune, malignant, and other) [ ]. However, endogenous CS is a rare endocrine condition with an estimated incidence of 3–50 cases per million people per year depending on the population studied, and it remains an uncommon diagnosis for the clinical endocrinologist [ ]. We herein present the etiologies and genetic causes of CS, which can guide the diagnostic evaluation of patients and the identification of other associated findings based on the pathogenetic mechanism.
Causes of Cushing’s syndrome
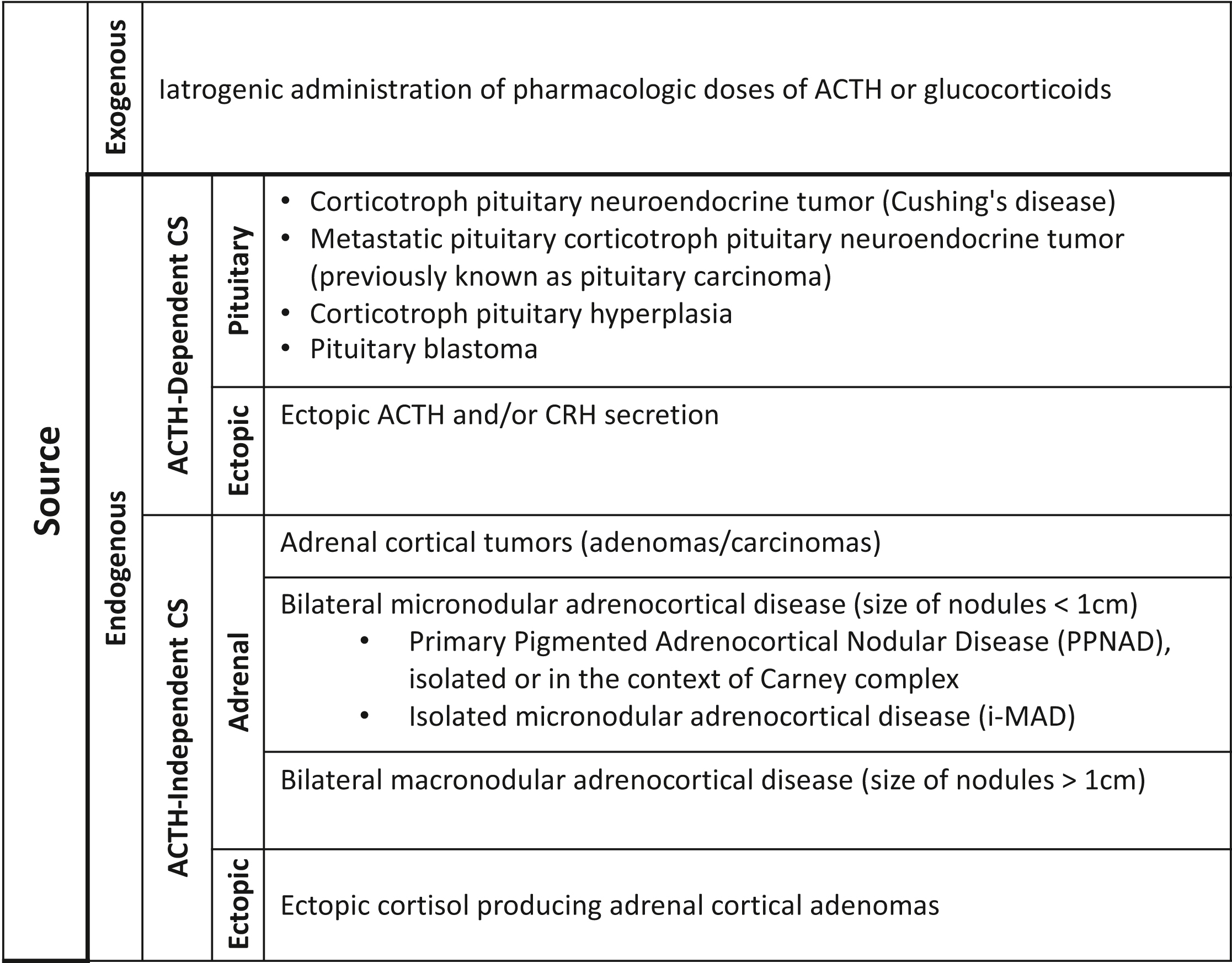
The main causes of CS are shown in Table 2.1 . We often classify causes of CS based on ACTH levels, which represent the involvement of ACTH in the pathogenesis of the disease. ACTH-independent CS is associated with adrenal disorders, which lead to autonomous cortisol secretion. On the other hand, ACTH-dependent CS is caused by either pituitary or ectopic causes of ACTH or rarely corticotropin-releasing hormone (CRH) excess. In macronodular adrenocortical disease, paracrine effects of locally secreted ACTH have been reported, and thus the exact mechanism of hypercortisolemia may be the combination of multiple factors. Since the measured plasma concentration of ACTH remains low, this condition is presented under ACTH-independent CS [ ].
Table 2.1
Causes of Cushing’s syndrome.
|
The frequency of each etiology differs based on the age group of the patient. Overall, corticotroph pituitary neuroendocrine tumors (PitNETs), also known as Cushing’s disease (CD) or corticotroph pituitary adenomas, are the most common cause of CS in children older than 5–7 years of age and adults [ , ]. ACTH-independent CS is the most common cause of CS in children less than 5 years of age, while ectopic CS is the most rare cause, mostly encountered in adult patients [ , ].
ACTH-independent Cushing’s syndrome—adrenal disorders
The most common cause of ACTH-independent CS are cortisol-producing adrenal cortical adenomas (ACAs) [ ]. These unilateral tumors usually involve isolated cortisol production. Less commonly adrenal cortical carcinomas (ACCs) may present with cortisol excess often cosecreted with adrenal androgens [ ]. Imaging findings, such as the size and the precontract attenuation of the lesion in computed tomography (CT), and biochemical findings, such as the urine steroid profile, may help in presurgical differentiation of benign from malignant adrenal lesions [ ]. The genetic landscape of these lesions also differs, suggesting different pathways of pathogenesis and variable prognosis [ ].
Less commonly, bilateral adrenocortical disorders may lead to ACTH-independent CS. These represent <2% of all cases of CS [ ]. In the 2022 World Health Organization (WHO) classification of adrenal cortical tumors, the nomenclature of bilateral adrenocortical disorders has been clarified and followed herein [ ]. The two main categories are based on the size of the identified nodules as macro- or micronodular.
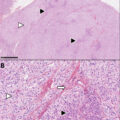
Bilateral micronodular adrenocortical disease is characterized by multiple nodules most of which are <1 cm [ ]. This category is further divided based on the presence of black pigment on gross pathology sections of the adrenals (lipofuscin pigment deposition) and internodular atrophy, as seen in primary pigmented adrenocortical disease (PPNAD), or in the absence of these findings, as seen in isolated micronodular adrenocortical disease (i-MAD) [ ]. PPNAD is commonly diagnosed in the context of Carney complex (CNC), but in rare cases, it may present as isolated PPNAD (i-PPNAD) [ ]. Notably, CS may be atypical or cyclical, but most patients with PPNAD will have a paradoxical response to dexamethasone during Liddle’s test when performed during active CS, which is pathognomonic for PPNAD [ ].
Bilateral macronodular adrenocortical disease is characterized by multiple bilateral nodules most of which are >1 cm [ ]. This is more common in adults and rarely seen in pediatric patients [ ]. Many of these lesions present with aberrant expression of G protein–coupled receptors (GPCRs) such as glucose-dependent insulinotropic peptide (GIP), vasopressin, b-adrenergic receptors, luteinizing hormone/choriogonadotropin (LH/hCG), and others, which may have implications for their presentation, diagnosis, and treatment [ , ].
Although ectopic adrenal cortical tissue has been reported, especially in the gonads, cases of ACTH-independent CS due to ectopic (extraadrenal) autonomous cortisol production are extremely rare [ ]. These ectopic ACAs are found usually in the pararenal region [ ].
ACTH-dependent Cushing’s syndrome—pituitary
Corticotroph PitNETs are the most common cause of ACTH-dependent CS [ ]. PitNETs represent monoclonal lesions originating from clonal expansion of corticotroph cells [ ]. Most of these lesions are slowly progressing and maintain characteristics of the normal corticotroph cell, such as the expression of the CRH receptor, which help in the diagnostic evaluation of patients [ ]. Metastatic corticotroph PitNETs is the term currently favored to replace the previously used term pituitary carcinoma [ ]. Metastatic corticotroph PitNETs are extremely rare and characterized by distant metastasis outside the pituitary gland commonly in the central nervous system (CNS), liver, and other organ [ ]. Certain histologic subtypes, such as Crooke cell adenomas or silent corticotroph tumors, predispose to more aggressive behavior [ ].
Corticotroph pituitary hyperplasia is observed in rare cases of ectopic CRH-secreting NETs and presents with expanded but intact reticulin staining and pituitary enlargement without distinct lesions [ ]. In rare cases, primary corticotroph pituitary hyperplasia has been reported as the cause of ACTH-dependent CS [ ]. This has also been hypothesized in rare germline genetic defects [ ].
Finally, in infantile onset of ACTH-dependent CS, pituitary blastomas should be considered. Pituitary blastomas are pituitary lesions with characteristic histologic findings involving primitive undifferentiated Rathke-type epithelium cells, along with secreting cells most prominently corticotrophs [ ].
ACTH-dependent Cushing’s syndrome—ectopic
Ectopic secretion of ACTH and/or CRH is a rare cause of CS accounting for up to 10% of all cases [ , ]. Most common sources of ectopic CS in adolescents and adults include bronchial carcinoids, pancreatic NETs, thymic carcinoids, medullary thyroid carcinomas, and other rare tumors [ ]. In young children less than 5–10 years old, ectopic CS is often associated with primitive cell–derived tumors such as Wilms tumors, Ewing sarcomas, neuroblastomas, and others [ , ].
Genetics of Cushing’s syndrome
ACTH-independent Cushing’s syndrome
Adrenal cortical tumors are commonly associated with germline or somatic pathogenic variants in the tumor suppressor gene TP53 found in approximately 20% of all cases of ACCs [ , ]. In the pediatric population, TP53 pathogenic variants are found in up to 70% of adrenocortical tumors and a germline founder mutation (p.R337H) is considered one of the leading causes of adrenocortical tumors in the Brazilian pediatric population [ , ]. Germline pathogenic variants in TP53 are also associated with Li Fraumeni syndrome (LFS). LFS is a cancer predisposition syndrome in which patients have increased risk for ACC, breast cancer, central nervous system tumors, osteosarcomas, and soft tissue sarcomas [ ].
Additional multiple neoplasia/tumor syndromes are associated with ACTH-independent CS. Lynch syndrome is a cancer predisposition genetic syndrome associated with increased risk for ACCs, colorectal, endometrial, and other cancers. Lynch syndrome is due to germline variants in mismatch repair genes, including MSH2 and MSH6 [ ]. Cortisol-secreting adrenal cortical adenomas and bilateral macronodular adrenocortical disease have also been described in familial adenomatous polyposis (FAP), caused by pathogenic variants in APC [ ]. Further, Multiple endocrine neoplasia type 1 (MEN1), due to pathogenic variants in MEN1 gene may present with unilateral or bilateral adrenal tumors, including isolated nodules and carcinomas [ , ]. McCune–Albright syndrome (MAS) is due to postzygotic somatic activating variants in GNAS gene, which codes for the alpha subunit (G s a protein) that mediates the signaling of GPCRs, leading to increased cyclic AMP (cAMP) concentration and constitutive activation of the downstream protein kinase A (PKA) pathway. MAS is also associated with bilateral macronodular adrenocortical disease often presenting in the neonatal period and always within the first year of life [ , ]. Additionally, somatic GNAS pathogenic variants are identified in sporadic cortisol-secreting ACAs [ ]. Last, the syndrome of hereditary leiomyomatosis and renal cell carcinoma, caused by inactivating pathogenic variants in FH gene, coding for fumarate hydratase, may present with bilateral adrenocortical tumors and CS [ ].
Chromosome 11p15 is a locus involving a cluster of imprinted genes, including IGF2 , H19 , and CDKN1C . Both germline and somatic variants in chromosome 11p15, as well as epigenetic alterations at this locus, are associated with cortisol-producing adrenocortical tumors [ , ].
Furthermore, activation of the Wnt signaling pathway, often associated with somatic activating CTNNB1 gene pathogenic variants, has been described in both benign and malignant adrenocortical tumors [ , ].
Somatic variants resulting in activation of the cAMP/PKA pathway, including PRKACA, PRKAR1A , and PRKACB, are also associated with cortisol-secreting ACAs [ ]. In 2014, two groups reported that 35%–70% of cortisol-secreting ACAs harbored the recurrent somatic p.L206R pathogenic variant in PRKACA gene [ , ]. PRKACA codes for the catalytic subunit of PKA, and the p.L206R variant leads to loss of binding with the inhibitory regulatory subunit, resulting in activation of the PKA activity [ ]. Patients with PRKACA -related ACAs are often younger in age and have higher cortisol levels compared with patient without PRKACA -related tumors [ ]. PRKACB copy number gains have also been later reported in a small number of patients with cortisol-secreting ACAs [ ].
Bilateral micronodular adrenocortical disease due to PPNAD is often presenting in the context of CNC. CNC is most commonly caused by pathogenic variants or less often by partial or whole gene deletions of the PRKAR1A gene [ , ]. A second locus on chromosome 2p16, referred to as CNC2 locus, has been linked with CNC patients without PRKAR1A gene defects [ ]. CNC presents with the constellation of myxomas (cutaneous, cardiac), spotty skin pigmentation, and endocrine overactivity, affecting adrenal, thyroid, pituitary, and gonadal function [ , ]. PPNAD occurs in about 60% of patients with CNC, with median age of diagnosis at 34 years [ ]. i-PPNAD has also been associated with sporadic pathogenic variants in PRKAR1A gene and variants at the chromosome 2p16 locus, as well as variants in PDE11A [ ].
Bilateral micronodular adrenocortical disease due to i-MAD is a very rare disorder. Germline inactivating pathogenic variants in PDE11A and PDE8B genes have been described in few families [ , ]. PDE11A and PDE8B are dual-specificity phosphodiesterases (PDE) involved in cAMP hydrolysis, and inactivating mutations of the genes lead to increased cAMP signaling and activation of cAMP-responsive elements [ , ]. Germline copy number gains (duplication or triplication) of the PRKACA gene have been reported in few patients with i-MAD and macronodular adrenocortical disease [ ]. Recently, a mosaic PRKACA duplication has been reported in a patient with i-MAD and acral cutaneous mucinosis, suggesting a potential new association [ ].
Bilateral macronodular adrenocortical disease is most commonly associated with inactivating variants in ARMC5 , which are present in up to 55% of cases [ ]. Bilateral macronodular adrenocortical disease due to ARMC5 variants has been described in both familial and sporadic cases [ ].
Bilateral macronodular adrenocortical disease may often involve aberrant GPCR expression [ ]. GIP-dependent CS, also known as food-dependent CS, is an interesting subtype, often presenting with low fasting cortisol and paradoxical GIP-stimulated postprandial hypercortisolemia with large adrenal adenomas and/or macronodules [ , ]. Biallelic germline and somatic pathogenic variants in KDM1A gene have recently been identified in patients with GIP-dependent CS [ ].
ACTH-dependent Cushing’s syndrome—Pituitary causes
Corticotroph PitNETs may be associated with germline or somatic gene defects. Germline gene defects are found infrequently in approximately 10% of patients with CD, while somatic gene defects are found in 40%–60% of patients depending on the population studied [ , ].
Multiple endocrine neoplasia syndrome type 1 (MEN1) is an autosomal dominant syndrome caused by germline pathogenic variants of MEN1 gene [ ]. MEN1 has high penetrance over lifetime, but manifestations are variable [ ]. Patients with MEN1 present commonly with pituitary tumors in 40%–60% of all cases, most of them prolactin-secreting or nonfunctioning [ , ]. Corticotroph PitNETs involve a small number of these pituitary tumors (0%–4% of all PitNETs detected) at an average age of 34–45 years old, with the youngest patient presenting at 5 years of age [ ]. Other manifestations of MEN1 include primary hyperparathyroidism, gastroenteropancreatic, and other NETs, carcinoids of various organs and other [ ]. Obtaining a comprehensive personal and family history for findings potentially related with MEN1 features is important for screening for this syndrome.
Multiple endocrine neoplasia type 4 (MEN4) is a rare autosomal dominant syndrome considered as the MEN1 phenocopy because patient present with similar findings as MEN1 without a MEN1 gene defect [ , ]. Germline pathogenic variants in CDKN1B gene are identified as the cause of this syndrome [ , , ]. CDKN1B codes for p27 kip1 , a cyclin-dependent kinase inhibitor that regulates cell cycle and acts as a tumor suppressor gene [ ]. CD is diagnosed in approximately 7%–10% of all patients with MEN4 and may present in pediatric age [ ]. A study by Sekiya et al. showed an increased frequency of polymorphism rs2066827 V109G in sporadic corticotropinomas and noted a change of corticotroph cell proliferation in the presence of this variant, further supporting the involvement of this gene in pituitary tumorigenesis [ ].
On the contrary, MEN type 2 (MEN2) is rarely associated with pituitary tumors, and only single case reports of patients with germline RET gene pathogenic variants and corticotropinomas have been published [ , ]. Somatic RET gene variants are also not common in sporadic pituitary tumors suggesting that there is limited role of this gene in corticotroph regulation [ ].
Over the past decade, much information has been reported on DICER1 involvement in ACTH-dependent CS. DICER1 gene codes for a small RNA endoribonuclease involved in microRNA processing. Patients with germline DICER1 pathogenic variants present with DICER1 syndrome involving multiple tumors in lungs, kidneys, thyroid gland, and other tissues [ ]. In 2014, de Koch et al. reported that germline DICER1 pathogenic variants were identified in most patients with ACTH-secreting pituitary blastomas and a second somatic hit was also common in these tumors [ ]. All patients were young with age at diagnosis ranging from 7 to 24 months and had macroadenomas that were managed with either surgery alone or surgery and chemotherapy [ ]. Recently, DICER1 variants have also been reported in patients with sporadic CD without histologic findings of blastoma [ ]. The reported variants were not associated with the RNaseIIb domain as those reported in pituitary blastomas, nor with somatic loss of the normal allele [ ]. All patients were pediatric cases with median age of 9 years (range: 7.8–16.3) and median tumor diameter of 7 mm [ ].
Familial isolated pituitary adenoma (FIPA) syndrome refers to familial presentation of pituitary tumors in at least two family members without additional associated manifestations. CD is one of the less commonly encountered tumors in this syndrome, representing <10% of FIPA families [ ]. Of families with FIPA, 15%–20% carry a pathogenic variant in AIP gene [ , ]. AIP gene pathogenic variants may also be identified in apparent nonfamilial pituitary tumor cases often referred to as “simplex” cases especially in young-onset [ , ]. Most of these families and simplex cases represent noncorticotroph PitNETs. CD in association with germline AIP pathogenic variants has been reported in few studies involving only apparently “simplex” cases [ , ]. Diagnosis of an AIP pathogenic variant is important because patients present earlier in age and have larger and more aggressive pituitary tumors [ ].
As mentioned before, CNC is an autosomal dominant multiple neoplasia syndrome often caused by inactivating pathogenic variant in PRKAR1A gene leading to constitutive activation of PKA activity [ , ]. Although patients with CNC present often with pituitary tumors, these are mostly associated with growth hormone secretion; CD in the context of CNC has only been reported in few case reports [ , ].
The association of pituitary adenoma with paraganglioma/pheochromocytoma (3 PAs) has been described initially in patients with SDHx pathogenic variants and later expanded to additional gene defects associated with pheochromocytoma/paraganglioma such as MAX , VHL, and other [ ]. Patients with familiar 3 PA association involve mostly growth hormone, prolactin, or nonfunctioning tumors, and CD is an infrequent presentation [ ]. Due to this association, screening of sporadic corticotroph tumors has identified variants in these genes, but causative association has not yet been completed [ ].
Loss of Cables1 function has been shown in vitro to stimulate corticotroph proliferation and correlate with p27 kip1 expression [ ]. Germline CABLES1 gene variants were reported in four patients with CD (2.7% of the cohort) in one study and thus represent a rare cause of CD [ ].
A rare association of PitNETs has been reported with tuberous sclerosis complex (TSC). TSC is a neurocutaneous disorder presenting with hamartomas of various tissues (brain, lung, kidneys, and others), characteristic skin findings, seizures, and other features [ ]. TSC is caused by pathogenic variants in TSC1 or TSC2 genes coding for hamartin and tubulin, which lead to activation of the mTOR pathway and cell proliferation [ ]. At least two patients with TSC and CD have been reported in the literature, a pediatric patient 13.5 years old and a young adult of 33 years old [ ]. The mechanism of this association has not been further investigated yet [ ].
Somatic gene defects are more commonly identified as the cause of CD. In 2015, two research groups reported the presence of USP8 gene defects in 40%–60% of patients with CD [ , ]. USP8 codes for a deubiquitinase. Pathogenic variants in a 14-3-3 binding motif hot spot of USP8 gene lead to enhanced cleavage of USP8, which further leads to increased catalytic activity. One of the substrates of USP8 is the epidermal growth factor receptor (EGFR); increased catalytic activity of USP8 leads to increased EGF signaling which results in increased POMC gene expression and ACTH secretion [ ].
One patient with a germline USP8 hotspot pathogenic variant has been reported in the literature [ ]. Her presentation included early-onset severe recurrent CD along with other nonpituitary manifestations potentially associated with increased EGF signaling [ ].
In USP8 -negative tumors, somatic defects have been reported in other genes. Somatic USP48 pathogenic variants have been reported in 10%–23% of tumors without a USP8 pathogenic variant [ , ]. Furthermore, TP53 or more rarely BRAF (V600E) pathogenic variants have been reported in these tumors [ , ].
Aggressive corticotroph tumors and carcinomas may be associated with ATRX somatic gene defects. These tumors present with decreased ATRX tumor expression in up to 19% of cases, and all of these tumors harbored an ATRX somatic pathogenic variant [ ]. Most of these tumors had additional somatic gene defects in known tumor suppressor genes, such as TP53 , PTEN , and others [ ].
Finally, copy number variations in large genomic areas have also been involved in PitNETs. In a pediatric CD cohort, genomic alterations across all chromosomes were reported in almost 20% of the cases and were associated with larger size and higher chance of cavernous sinus invasion [ ]. Further studies suggested that these disrupted corticotroph PitNETs are not associated with USP8 somatic pathogenic variant but often harbor TP53 pathogenic variants and are associated with more aggressive behavior [ ].
ACTH-dependent Cushing’s syndrome—Ectopic tumors
Genetic causes of ectopic ACTH secretion NETs are not yet clear and differ based on the tissue of presentation.
MEN1 syndrome may present with ectopic CS in the context almost exclusively of thymic carcinoids [ ]. Ectopic CS is the least common cause of CS in MEN1 and presents in 0%–8% of all cases reported [ ]. Medullary thyroid carcinomas with or without RET gene pathogenic variants may present with ectopic CS. In a recent literature review of 96 patients with MTC and ectopic CS, the median age at diagnosis was 44 years with a range from 10 to 84 years old, but the genetic background of these tumors was not reported [ ]. Recently, gene fusions involving EWSR1 :: BEND2 , KMT2A :: BCOR , and TFG :: ADGRG7 have been reported in all pancreatic NETs associated with ectopic CS, but the mechanism of POMC activation has not been investigated [ ]. Finally, rare case reports of ectopic CS with TP53 , VHL and other gene pathogenic variants have been reported.
Conclusion
Endogenous CS is caused by a variety of disorders involving the pituitary gland, the adrenals, and ectopic sources. Several germline and somatic genetic defects have been reported in the pathogenesis of endogenous CS. The clinical presentation and histologic findings may guide the appropriate genetic testing of patients which may reveal additional findings or provide evidence for their prognosis and management.
References
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree