JAK1/2 inhibitor therapy has potential to improve the pre-HCT performance status by effective control of MF-related symptom burden
Several transplant studies have shown the relationship with, or better performance status with, improved survival after transplant
If stopped suddenly prior to HCT, can cause rapid return of MF-related symptoms. Therefore, a strategy of gentle tapering over 4–5 days prior to HCT is recommended
Reduced spleen size may help in faster hematological recovery
Myelosuppressive properties of JAK inhibitors may have impact of hematological recovery after HCT, and may also limit the use of JAK inhibitor therapy in the post-HCT setting
JAK1/2 inhibitor therapy has recently been shown to be effective in reducing the risk of GVHD in a mouse model by inhibiting donor T-cell expansion and inflammatory cytokine production
Beneficial effect also demonstrated on 6 patients with steroid-refractory GVHD
There is proven increased risk of urinary tract infections and zoster infection with JAK1/2 inhibitor therapy in comparison to other available treatments
In addition, anecdotal case reports of opportunistic infections such as reactivation of hepatitis, mycobacterial infections, and invasive fungal infections have raised concerns of opportunistic infections with JAK inhibitor therapy
Impaired function of NK cells and dendritic cells
Due to CYP3A4 inhibition, potential of significant drug interactions with routine transplant medications such as calcineurin inhibitors and antifungal medications
Cases of tumor lysis syndrome reported in a recent prospective study; not clear whether ruxolitinib may cause chemosensitization to drugs used in conditioning therapy
HCT, hematopoietic cell transplantation; MF, myelofibrosis; GVHD, graft-versus-host disease; GVL, graft-versus-leukemia; NK, natural killer.
Timing of HCT in patients responding to JAK inhibitors
As highlighted in cases 2 and 3, the decision making can be very complex in patients who are responding to JAK1/2 inhibitors. In such scenarios, there are three potential models with regard to timing of HCT as shown in Figure 11.1.
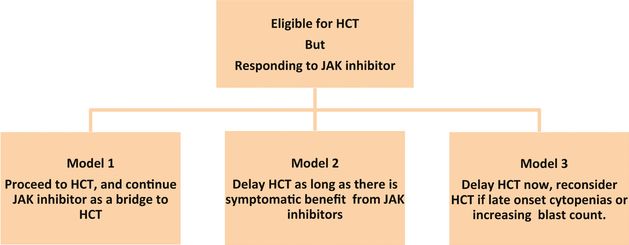
Timing of hematopoietic cell transplantation (HCT) in myelofibrosis. Source: Shanavas M, Gupta V. Controversies and dilemmas in allogeneic transplantation for myelofibrosis.
At present, there are no comparative data on outcomes of HCT vs. non-transplant treatment options. Given the rarity of MF, such comparative studies are logistically extremely difficult, if not impossible. We believe that in such scenarios patients should be provided a balanced discussion about the pros and cons of each approach. A careful consideration of patient, disease, and transplant related characteristics should be done before selecting a patient for transplantation (Figure 11.2).
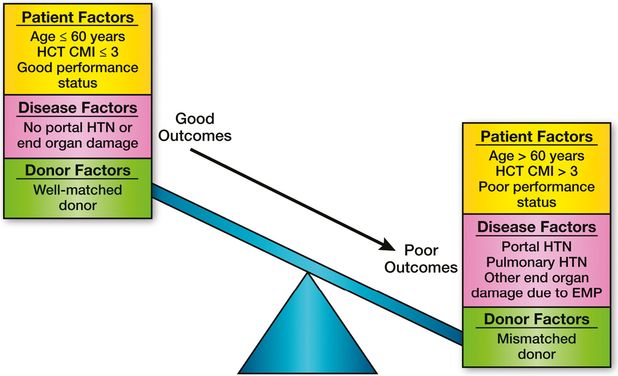
Factors influencing outcome in hematopoietic cell transplantation (HCT) for myelofibrosis. CMI, comorbidity index; HTN, hypertension; EMP, extramedullary hematopoiesis.
Early HCT may lead to significant TRM and loss of quality of life in some patients, who could have years of reasonable quality of life with other treatment options. On the other hand, delay in HCT may result in worse outcome due to increasing age, higher disease-specific score, poor performance status, worsening splenomegaly, transfusion-associated iron overload, and the risk of LT. A recent case series highlighted the issue of LT in patients, showing symptom benefit on ruxolitinib.27
Another important factor to consider here is the discontinuation rate of JAK1/2 inhibitors in MF, unlike tyrosine kinase inhibitors in chronic myeloid leukemia. A recent update on multicenter COMFORT-1 trial reported discontinuation rates of 21%, 35%, and 51% at 1, 2, and 3 years, respectively.28
Another important concern is whether failure of JAK inhibitor therapy has any adverse effect on HCT outcomes. A small study from Germany showed that the outcomes of transplantation were better in patients who were responding to JAK inhibitor, with regard to spleen size, compared to those who have failed or lost response.24 However, at present there are not enough data to arrive at a conclusion.
There are no data from comparative studies to guide the decision regarding need and timing of HCT in MF, and there are obvious logistical difficulties in conducting such trials. Hence, Markov decision models comparing transplant vs. non-transplant strategies may be a reasonable approach to resolve this dilemma, as has been successfully used in myelodysplastic syndrome.29,30 The Chronic Leukemia Working Committee of the CIBMTR has planned such an analysis, and the results are eagerly awaited. At present we recommend an individualized approach after careful consideration of patient, disease, and transplant-related factors (Figure 11.2).
We suggest that the decision of transplantation should be individualized in each patient. Both transplant and non-transplant options, along with their pros and cons, should be discussed, and a treatment plan should be made after taking patient wishes and goals of therapy in to consideration. Even in patients responding to JAK inhibitors, certain factors, such as younger age and good performance status, may tilt the balance towards transplantation.
Is there a role for splenectomy in the era of JAK inhibitors?
Splenomegaly is a frequent finding in patients with MF, and often is associated with debilitating symptoms and complications, including abdominal pain, difficulty in bending and walking, early satiety which leads to weight loss, cytopenias, and finally, in some cases, portal hypertension and splenic infarction. Splenectomy is an effective therapeutic option and may relieve the symptoms, but it is not without its complications. Splenectomy carries the risk of postoperative thrombocytosis, thrombohemorrhagic complications, and infectious complications, resulting in significant postoperative morbidity and mortality.31
The role of splenectomy before transplant has been extensively debated, and has remained a controversial issue (Table 11.2). Earlier studies did not show that splenectomy before HCT enhanced survival. Several recent studies suggested that splenectomy may enhance survival in selected HCT patients. In the French study, men who had not under gone splenectomy had a higher mortality (hazard ratio 3.45) even when adjusted for the Lille score that was still highly statistically significant.32 In the CIBMTR study published by Ballen et al., there was a trend towards increased disease-free survival in splenectomized patients (relative risk 0.77).33 A recent large series of patients who underwent splenectomy before HCT was reported by the Seattle group. They found in their study that patients who underwent splenectomy before HCT had a lower mortality than those patients with intact spleen.34 However, one of the major issues in these studies is lack of information on the patients who underwent splenectomy prior to HCT, and never proceeded to HCT due to an adverse event related to splenectomy.
| Center | Transplant | Outcome | Reference |
|---|---|---|---|
| Fred Hutchinson Cancer Research Center (FHCRC) | MA | No difference in overall survival | Li et al.43 |
| European Group for Blood and Marrow Transplantation (EBMT) | RIC | Increase in relapses in splenectomized | Kroeger et al.44 |
| Ospedale San Martino in Genova | RIC | Increase relapse deaths in non-splenectomized (spleen >22 cm) No impact on overall survival | Bacigalupo et al.45 |
| Gruppo Italiano Trapianto di Midollo Osseo (GITMO) | MA (48%) RIC (52%) | No impact on treatment-related morbidity, overall survival | Patriarca et al.46 |
| French (Société Française de Greffe de Moelle et de Therapie Cellulaire: SFGM-TC) | MA (31%) RIC (69%) | Non-splenectomized males had an increase in relapses and non-relapse mortality | Shanavas and Gupta47 |
| Center for International Blood and Marrow Transplant Research (CIBMTR) | MA (83%) RIC (17%) | No impact on survival | Ballen et al.33 |
| FHCRC | MA (89%) RIC (11%) | Decreased post-HCT mortality | Scott et al.34 |
HCT, hematopoietic cell transplantation; MA, myeloablative; RIC, reduced-intensity conditioning.
The availability of JAK inhibitors by and large will replace the need for surgery, and consequences of surgical procedure. Some patients after JAK inhibitor therapy may still have a large spleen, as demonstrated by case 2. The role of splenectomy in such patients is controversial. In the JAK inhibitor era, splenectomy may be considered on a case-by-case basis in those who may have lost their benefit to JAK inhibition or never showed a response.
In our opinion, splenectomy prior to HCT is not required for those patients responding to JAK inhibitor therapy. Decision regarding splenectomy in patients losing clinical benefit or refractory to JAK inhibitor therapy should be considered on a individual basis by careful assessment of the risk–benefit ratio.
Alternative donor transplantation in myelofibrosis
Due to improvements in human leukocyte antigen (HLA) matching and supportive care, the differences in outcomes between fully matched related donors and unrelated donors are generally getting smaller, but still exist in MF. In a prospective study of RIC in MF from EBMT, there were no differences in outcomes between related donors and fully matched unrelated donors.35 In contrast, a recently published study from MPD-RC17 as well as CIBMTR11 reported a higher non-relapse mortality in matched unrelated donors when compared to matched sibling donors. However, both the EBMT as well as the CIBMTR study reported clearly inferior outcomes with mismatched unrelated donors with a hazard ratio of mortality almost more than double compared to matched sibling or well-matched unrelated donor transplantation.
There are minimal data on use of umbilical cord blood (UCB) or haplo-identical transplants in patients with MF. A retrospective review of Japanese registry included 11 cases of unrelated UCB, and these patients had a lower probability of hematopoietic recovery when compared to related donor BM transplantation. The difference in overall survival was not statistically significant, but definite conclusions cannot be made due to small sample size.36 Another study from Eurocord evaluated the role of UCB transplantation in 35 patients with MF.37 There were 7 patients with secondary acute myeloid leukemia from previous MF in this study. Two-year overall survival and progression-free survival were 44% and 30%, respectively. While these studies have demonstrated the feasibility of UCB transplantation in patients with MF, results appear significantly inferior in comparison to sibling donor or well-matched unrelated donor transplantation.
Poor outcomes of mismatched unrelated donor and UCB transplantation may influence the decision of timing of transplantation in patients who have good response to JAK inhibitor therapy.
In our opinion, in patients who are responding to JAK-1/2 inhibitor therapy, and do not have matched sibling or well-matched unrelated donor, the option of mismatched unrelated donor or UCB transplantation should be considered at the failure of JAK inhibitor therapy or in those patients at high risk of LT or unable to tolerate JAK inhibitor therapy due to severe cytopenias.
Graft failure, graft rejection, and relapse after HCT – diagnosis and management
Graft failure/graft rejection after HCT is a serious complication with a high mortality. Established risk factors are BM graft, matched unrelated, HLA mismatch, low cell count in the graft, and T-cell depletion of the graft.38 The distinction between graft failure/graft rejection and relapse is important to make appropriate therapeutic decisions. In the presence of full donor chimerism but poor hematological recovery, the cause may be drugs or infectious issues such as cytomegalovirus. In this situation, treating the underlying cause, such as discontinuing the medication or treating the infectious issue, is important. However, if there is no improvement after adequate treatment, one may consider the option of stem cell boost or second transplantation.
In rejection, chimerism status in the CD3/CD33 is mixed with the presence of both donor and recipient cells. In such situations, differentiation between graft failure and relapse could be difficult. Relapsed disease as defined by the International Working Group consensus criteria is a loss of complete response (CR), partial response (PR), or clinical improvement (CI). In other words, patients are considered to have relapsed if they no longer fulfill the criteria for CR, PR, or CI.39 However, monitoring of remission according to these criteria and further relapse can be misleading, as documentation of CR requires resolution of disease-related clinical signs and symptoms as well as cytopenias. Persistence of cytopenias post-HCT may be due to several other causes, such as viral reactivation or medication-related. Fibrosis may persist for some time after transplant. Monitoring minimal residual disease posttransplant in MF for patients who have specific disease markers may be helpful in diagnosing relapse. The availability of CALR mutation will make it feasible that an informative driver mutation marker such as JAK2 V617F, CALR, MPL, and exon 12 can monitored in >90% with MF.
Alchalby et al. reported the results in 90 patients with MF who underwent HCT. They utilized a highly sensitive polymerase chain reaction method to follow specific molecular markers of malignant cells post-HCT as a pre-emptive strategy to detect early relapse, so that application of donor lymphocyte infusions (DLIs) at the stage of molecular relapse as opposed to full-blown relapse could be implemented.40
Therapeutic modalities used to treat graft failure (primary or secondary) or relapse include growth factor and transfusion support, DLI, stem cell boost without conditioning (unselected CD34 vs. CD34 selected), or second transplant. The decision is based not only on chimerism analysis, which helps differentiate between graft rejection and relapse, but also on evidence of markers of driver mutations, and patients’ overall performance status.
For graft rejection, transplantation of additional stem cells could be performed as regrafting after administration of a conditioning regimen or infusion of stem cells without prior conditioning from the same donor. Success with regrafting after conditioning can be seen with 66–100%. However, non-relapse-related mortality has been reported in up to 50–60%, with an overall survival of 20–25% due to GVHD and infectious complications. Therefore, Klyuchnikov et al. used the approach of CD34 selected stem cell boost in 32 patients with graft failure. Hematological improvement was seen in 81% of patients at a median of 30 days. The cumulative incidence of acute GVHD and chronic GVHD was 17% and 26%, respectively. The overall survival at 2 years was 45%, suggesting that CD34 selected stem cell boost may be an effective approach in some patients.41
Depending on the risk of the MF, relapse can occur in 10–30% of patients who undergo HCT within 3 years. There seems to be no standard treatment approach for these patients. Early withdrawal of immunosuppression, use of DLI, and second HCT using RIC are possible therapeutic options. Klyuchnikov et al. studied 30 patients with MF who had relapsed disease and/or graft rejection and who underwent a DLI and/or second HCT using RIC. Thirteen of the patients received DLI in dose escalation fashion; in those patients, acute and chronic GVHD occurred in 12% and 36% of cases. Seventeen patients underwent a second allogeneic stem cell transplantation (13 who failed a DLI and 4 patients who did not receive DLI). Acute GVHD and chronic GVHD were seen in 47% and 46%. Overall response rates after second HCT was 80% (CR in 9 patients and PR in 3). The cumulative incidence of relapse was 24% and 2-year overall survival and progression-free survival were 70% and 67% for all patients for a median follow-up of 27 months. These results indicate that DLI and/or second HCT may be effective in selected patients.42 In patients with poor performance status after relapsed disease, one may consider the option of JAK1/2 inhibitor therapy to control MF-related symptoms.
In our opinion, the distinction between graft rejection, graft failure, and relapse needs to be made so that early intervention is possible. Options include either CD34 selected or unselected boost with or without prior conditioning, DLI, or second transplant.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree