Introduction
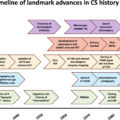
Medical therapy for Cushing’s syndrome (CS) has historically played a minor role in the disease management; however, it has recently acquired an emerging role, with the availability of several drugs with different therapeutic targets, efficacy, and safety profiles .
Particularly, in the most recent consensus, medical therapy has been advocated before surgery, as preoperative treatment, especially in patients with severe disease, and/or when a rapid hypercortisolism control is required; or after pituitary or ectopic surgery, as adjuvant treatment, in patients with persistent or recurrent CS; before or after pituitary radiotherapy in Cushing’s disease (CD), as bridging treatment; or, lastly, as primary alternative treatment in case of refusal or contraindication to surgery, such as invisible tumors or tumors with unfavorable location [ , , ].
The spectrum of available drugs includes three main categories of compounds: the pituitary-directed agents, the adrenal-blocking agents, and the glucocorticoid receptor (GR) antagonist.
When choosing a medical therapy, clinicians should consider different characteristics of drugs and patients: the drug’s rapidity of action, manageability, safety and tolerability, local availability, approval, costs, as well as patients’ sex-related factors, specific characteristics, mainly including the presence or not of tumor in case of CD [ ].
This chapter provides an overview of the different drugs for the treatment of CS, illustrating efficacy, safety, and highlighting key practical clinical considerations and recommendations for the use of each drug ( Table 15.1 ) .
Table 15.1
Clinical improvements, AEs, AEs of special interest, and management of AEs of special interest of the medical therapies for Cushing’s syndrome.
| Drug | Clinical improvements | AEs | AEs of special interest | Management of AEs of special interes |
|---|---|---|---|---|
| Pasireotide sc | Clinical signs of CD: facial rubor, bruising, supraclavicular and dorsal fat pads; body weight; waist circumference; blood pressure; lipid profile; depressive status; QoL | Hyperglycemia-related events (72.8%); diarrhea (58.0%); nausea (51.9%); cholelithiasis (30.2%); mild transient elevations in liver enzyme levels (29.0%); headache (28.4%); abdominal pain (24.1%); fatigue (19.1%); asthenia (11.1%); hypocortisolism-related events (8.0%); prolongation of the QTc interval >480 msec (1.9%) | Impairment of glucose metabolism; liver dysfunctions; hypocortisolism-related AE; QT prolongation |
|
| Pasireotide LAR | Clinical signs of CD: facial rubor, bruising, striae, supraclavicular and dorsal fat pads; body weight; waist circumference; blood pressure; lipid profile; QoL | Hyperglycemia-related events (76.6%); diarrhea (39.3%); cholelithiasis (32.6%); nausea (20.6%); headache (18.6%); nasopharyngitis (18.6%); fatigue (17.3%); abdominal pain (14.6%) | ||
| Cabergoline | Clinical signs of CD: facial mooning, plethora; waist circumference; muscle status; blood pressure; glucose metabolism | Dizziness and/or nausea (0%–35%), asthenia (0%–30%), hypotension (0%–10%), vertigo (3.2%), constipation (2.4%), diarrhea, edema, nasal congestion, personality change and visual hallucination (0.8%) | Impairment of behavior (particularly impulse control disorders); hypotension-related AEs; cardiac valve damage |
|
| Metyrapone | Body weight; blood pressure; glucose metabolism; lipid profile; muscle status; psychiatric symptoms | Hirsutism and/or acne in women (4.5%–71.4%); hypertension (6%–48.4%); dizziness (9.7%–44.4%); nausea (5.3%–33.3%); fatigue (13%-26%); headache (22%); edema (6%–20%); arthralgia (8.7%-18.2%); hypokalemia (6%–13.6%) | Hypocortisolism-related AEs; adrenal hormone precursor accumulation related AEs |
|
| Ketoconazole | Body weight; muscle and bone status; blood pressure; glucose metabolism; potassium levels; psychiatric symptoms; hirsutism and menstrual cycles in women | Hepatotoxicity (10.7%–18.7%), particularly increase in liver enzymes (2.6%–18.4%); gastrointestinal disturbances (3.7%–18.7%); adrenal insufficiency (5.3%–18.5%); gynecomastia (16.7%); skin rash (3.6%–6.2%) | Liver dysfunctions; hypocortisolism-related AEs; QT prolongation; drug-drug interactions |
|
| Levoketoconazole | Body weight; glucose metabolism; lipid profile; peripheral edema; QoL; depressive status; hirsutism and acne in women. | Increase in liver enzymes (11.7%–44.6%); nausea (29%–31.9%); headache (23%–27.6%); hypokalemia (10.6%–26%); hypertension (17%–24%). | Liver dysfunctions; hypocortisolism-related AEs; QT prolongation; drug-drug interactions. |
|
| Osilodrostat | Body weight; blood pressure; glucose metabolism; lipid profile; QoL; depressive status | Fatigue (28.5%–58.3%); hypocortisolism-related AEs (27.4%-51.1%); adrenal hormone precursors accumulation-related AEs (42.3%-61.6%), mainly increased blood testosterone (24.7%) and hypertension (12.4%-21.9%); nausea (31.6%–41.7%); headache (25%–33.6%); diarrhea (25%–31.6%); QT prolongation (3.6%) | Hypocortisolism-related AEs; adrenal hormone precursor accumulation related AEs; QT prolongation; pituitary tumor enlargement |
|
| Mifepristone | Body weight, waist circumference; body fat; increase in insulin sensitivity | Nausea and fatigue (48%); headache and hypokalemia (44%); endometrial thickening (28.6%); peripheral edema (26%); hypertension (24%); abnormal uterine bleeding (14.5%); adrenal insufficiency (4%) | Hypocortisolism-related AEs; hypokalemia; endometrial thickening and vaginal bleeding; drug-drug interactions |
|
AE , Adverse event; ALP , alkaline phosphatase; AST , aspartate aminotransferase; ALT , alanine aminotransferase; CD , Cushing’s disease; DPP-4 , dipeptidyl peptidase-4; ECG , electrocardiography; gamma-GT , gamma-glutamyltransferase; GLP-1 , glucagon-like peptide 1; LAR , long-acring release; MRI , magnetic resonance imaging; QoL , quality of life; sc , subcutaneous; ULN , upper limit of normal; pts, patients; US , ultrasound.
Pituitary-directed agents
The pituitary-directed drugs, the somatostatin analog pasireotide and the dopamine agonist cabergoline, act at pituitary level, inhibiting adrenocorticotropic hormone (ACTH) and secondarily cortisol secretion, and targeting the cause of CD, the pituitary tumor.
Pasireotide
Pasireotide acts by binding to somatostatin receptors (SSTR) type 1,2,3, and 5, with a high affinity for SSTR5, which is highly expressed in ACTH-secreting pituitary tumors.
Pasireotide is the first medical therapy officially approved by European Medicine Agency (EMA) and United States Food and Drug Administration (FDA) for the treatment of CD in patients for whom surgery is not an option or for whom surgery has failed or has not been curative [ , ].
It is available in two different formulations, the first historical subcutaneous (sc) formulation, administered in a twice daily regimen, and the new, recently developed, long-acting release (LAR) pasireotide formulation, administered in a once monthly regimen, with the potential advantage to improve patient’s compliance.
Pasireotide sc is administered twice daily at a dosage of 600–1800 μg/day: it should be started at 1200 μg/day or at lower doses of 600 μg/day in patients with moderately impaired hepatic function. After the first 2 months of therapy, an intermediate efficacy evaluation should be performed. In case of urinary free cortisol (UFC) levels reduction without normalization, treatment should be continued until UFC normalization or an up-titration to 1800 μg/day should be considered, if the previous dose has been well tolerated. In case of no response, pasireotide sc discontinuation should be considered. A down-titration to 600 μg/day, temporary interruption or discontinuation should be considered on the basis of safety profile.
The pivotal phase III study showed that pasireotide sc induced UFC normalization in 14.6%–26.3% of patients at 6 months, without dose up-titration. Regardless of dose increase, UFC normalization was obtained in 15.9%–28.8% of patients at 6 months, and in 13.4%–25% of patients at 12 months [ ].
These percentages appeared higher in a real-world evidence study, up to 70% after 6 months of treatment, probably due to the enrollment of patients with very mild to moderate disease, compared with those with moderate to very severe disease enrolled in the pivotal study [ ].
Pasireotide sc efficacy was also evaluated in long-term evidence studies, with UFC normalization reported in 34.5% of patients at 24 months [ ], 68.8% of patients at 60 months [ ], and 50% of patients at 120 months [ ].
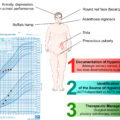
The pasireotide sc treatment was accompanied by improvement in clinical signs of CD, such as facial rubor, bruising, supraclavicular and dorsal fat pads, as well as in body weight, waist circumference, blood pressure, and lipid profile, depressive status, and quality of life (QoL) [ , , , ].
Notably, the treatment appeared also associated with a decrease in tumor volume ≥20% in 16%–57% and 36%–78% of patients, at 6 and 12 months, respectively [ ].
The most frequently reported AEs in the pivotal study were hyperglycemia-related events (72.8%), diarrhea (58%), nausea (51.9%), cholelithiasis (30.2%), mild transient elevations in liver enzyme levels (29%), headache (28.4%), abdominal pain (24.1%), fatigue (19.1%), asthenia (11.1%), hypocortisolism-related events (8%), and prolongation of the QTc interval >480 msec (1.9%) [ ].
Pasireotide LAR is intramuscularly administered every 4 weeks at a dosage of 10–40 mg: it should be started at 10 mg every 4 weeks. After the first 4 months of therapy, an intermediate efficacy evaluation should be performed. In case of UFC levels reduction without normalization, an up-titration up to 40 mg every 4 weeks should be considered, if the previous dose has been well tolerated. In case of no response, pasireotide LAR discontinuation should be considered. A down-titration to the previous tolerated dose, temporary interruption or discontinuation should be considered on the basis of safety profile [ ].
The pivotal phase III study showed that pasireotide LAR induced UFC normalization in 40.8%–41.9% of patients at 7 months, regardless of dose up-titration [ ].
Pasireotide LAR efficacy was also evaluated in a long-term evidence study, with UFC normalization reported in 46.9% of patients at 24 months and 72.2% of patients at 36 months [ ].
The pasireotide LAR treatment was accompanied by improvement in clinical signs of CD, such as facial rubor, bruising, striae, supraclavicular, and dorsal fat pads, as well as in body weight, waist circumference, blood pressure, lipid profile, and QoL [ , ].
Notably, the treatment appeared also associated with a decrease in tumor volume ≥20% in 42.9%–47.4% of patients at 12 months [ ].
The most frequently reported AEs in the pivotal study were hyperglycemia-related events (76.6%), diarrhea (39.3%), cholelithiasis (32.6%), nausea (20.6%), headache (18.6%), nasopharyngitis (18.6%), fatigue (17.3%), and abdominal pain (14.6%) [ ].
Some specific safety issues related to pasireotide treatment should be considered in clinical practice, mainly impairment of glucose metabolism, liver dysfunction, hypocortisolism-related AE, and QT prolongation.
Considering that impairment of glucose metabolism is a common complication of CS, glycemic status should be assessed prior to starting pasireotide treatment, and in patients with poorly controlled diabetes mellitus, intensive antidiabetic therapy should be initiated before starting treatment [ ].
Moreover, during treatment, patients need to be carefully monitored, weekly for the first 3 months and periodically thereafter, as clinically indicated, as well as over the first 4 to 6 weeks after any dose increase.
If hyperglycemia develops, the initiation or adjustment of antidiabetic therapy is recommended. Experts recommended that after lifestyle changes and medical treatment with metformin, if the control of glucose metabolism is not achieved or maintained (HbA1c levels of >7%–7.5%), combination therapy with agents targeting the incretin pathway is recommended. First, therapy with metformin and a dipeptidyl peptidase-4 (DPP-4) inhibitor may be established. If glycemic target values are not reached, the DPP-4 inhibitor may be replaced by a glucagon-like peptide-1 receptor agonist, with the advantages of higher HbA1c levels lowering effect, without increasing the risk of hypoglycemia, and with the potential ability to reduce body weight. If hyperglycemia remains uncontrolled by these combinations, establishing insulin therapy together with maintaining metformin treatment may be necessary. In these cases, initial combination therapy of metformin with long-acting basal insulin, targeting fasting plasma glucose, may be the first option. If the individual HbA1c target levels are not met or the postprandial glucose levels are high with basal insulin, short-acting prandial insulin therapy has to be finally established. If uncontrol of glucose metabolism persists, despite appropriate medical management, pasireotide dose reduction or discontinuation should be considered, and also after pasireotide discontinuation, glucose monitoring should be performed according to clinical practice [ ].
Liver dysfunctions, and particularly mild transient elevations in aminotransferases, are commonly observed in patients treated with pasireotide, therefore, a careful monitoring of liver and gallbladder functions is recommended, before treatment with pasireotide, after the first 2-3 weeks, then monthly for 3 months on treatment, and thereafter periodically as clinically indicated. If transaminase levels increase, a more frequent monitoring is recommended, until values return to pre-treatment levels. If the patient develops jaundice or other signs suggestive of clinically significant liver dysfunction, in the event of a sustained increase in aspartate aminotransferase (AST) or alanine aminotransferase (ALT) > 5xULN, or if AST or ALT elevations > 3xULN occur concurrently with bilirubin elevations > 2xULN, pasireotide should be discontinued. Pasireotide should not be restarted if the liver function abnormalities are suspected to be related to pasireotide [ , , ].
Hypocortisolism-related AEs may occur in patients treated with pasireotide, therefore, caution should be used in advising, monitoring, and instructing them on how to manage signs and symptoms of hypocortisolism as weakness, fatigue, anorexia, nausea, vomiting, weight-loss, and hypotension [ , , ]. If necessary, pasireotide dose reduction or interruption and/or a temporary exogenous steroid replacement therapy should be evaluated [ , , ].
QT prolongation or bradycardia are less frequently observed in patients treated with pasireotide, however, caution should be used especially in patients who have congestive heart failure (NYHA Class III or IV), unstable angina, sustained ventricular tachycardia, clinically significant bradycardia, advanced heart block, history of acute myocardial infarction or in patients with risk factors for torsade de pointes as patients with hypokalemia, congenital long QT prolongation or family history of long QT syndrome, or in patients treated with concomitant medications known to prolong QT interval [ , ]. An electrocardiography is recommended before, 1 week after the beginning of the treatment, and thereafter periodically as clinically indicated. Concomitantly, hypokalemia and/or hypomagnesemia must be corrected and monitored. Increase of QTcF > 480 ms should trigger a referral to a cardiologist. If QTcF is >500 ms pasireotide should be discontinued [ , ].
Cabergoline
Cabergoline acts on the entire cohort of dopamine receptors, including the stimulatory D1 and D5 receptor subtypes, and the inhibitory D2, D3, and D4 receptors subtypes, although the highest affinity is shown especially for D2 receptors, which are highly expressed in ACTH-secreting pituitary tumors [ ].
Cabergoline is currently used as an off-label treatment for CD. It might be a therapeutic option in patients with CD who are pregnant, or desire pregnancy, as well as in patients with Nelson’s syndrome [ ].
It is orally administered at a dosage of 0.5–7 mg per week: it should be started with a low dose 0.5 mg per week and increased as tolerated to therapeutic dose up to 7 mg per week.
Prospective and retrospective studies showed that cabergoline induced UFC normalization in 25%–36.7% at 3–6 months [ ].
Cabergoline efficacy was also evaluated in long-term evidence studies, with UFC normalization reported in 30%–40% of patients at 12–60 months, with an escape rate of 18.2%–33.3% [ , ].
The cabergoline treatment was accompanied by an improvement in clinical signs of CD, such as facial mooning, plethora, as well as in body weight, waist circumference, muscle status, blood pressure and glucose metabolism [ ].
Notably, the treatment appeared also associated with a decrease in tumor volume >25% in 50% of patients [ ].
The most frequently reported AEs were dizziness and/or nausea (0%–35%), asthenia (0%–30%), hypotension (0%–10%), vertigo (3.2%), constipation (2.4%) together with diarrhea, edema, nasal congestion, personality change, and visual hallucination (0.8%) [ ].
Some specific safety issues related to cabergoline treatment should be considered in clinical practice, mainly impairment of behavior, and particularly impulse control disorders, hypotension-related AEs, and cardiac valve damage.
Impulse control disorders, such as hypersexuality, pathological gambling, excessive alcohol consumption, overeating, and uncontrolled shopping, may be observed in patients treated with cabergoline. Particularly, these events can occur within months after starting cabergoline therapy, or can occur later, and improve or resolve after treatment discontinuation [ , ]. Therefore, cabergoline should not be used in patients with history of impulse control disorders. Hypotension-related AEs may occur in patients treated with cabergoline; therefore, caution should be used in advising, monitoring, and instructing them on how to manage signs and symptoms of hypotension. Proper water intake, evening administration immediately after the last meal may ameliorate postural hypotension and specifically nausea [ , ].
Lastly, the increased risk of cardiac valve disease should not be underestimated, although more frequently reported at higher dosages used for neurological disorders. Therefore, an echocardiography is recommended prior to starting cabergoline treatment, annually when doses are higher than 2 mg per week, or every 5 years when doses are equal or lower than 2 mg per week [ , ].
Adrenal-blocking agents
The adrenal-blocking drugs, the historical drugs, ketoconazole and metyrapone, and the novel drugs levoketoconazole and osilodrostat, act at adrenal level, blocking cortisol production, through the inhibition of specific enzymes of the adrenal steroidogenesis.
Metyrapone
Metyrapone, a pyridine derivative, acts by inhibiting the 11β-hydroxylase, the cholesterol side-chain cleavage complex and, to a lesser extent, 17α-hydroxylase and aldosterone synthase [ , ].
Metyrapone is approved from EMA for the treatment of CS. It might be a therapeutic option in patients with CS who are pregnant [ ].
It is orally administered with a multiple schedule up to 4–6 times a day at a dosage of 500–6000 mg/day: it should be started at 750 mg/day. However, starting doses can vary from 250 to 1500 mg/day, depending on the severity of hypercortisolism and the cause of CS: for patients with severe CS, initial doses may be higher, whereas in cases of mild CD or CS due to adrenal adenoma or adrenal hyperplasia, initial doses may be lower [ ].
After 1–4 weeks of therapy, an intermediate efficacy evaluation should be performed. In case of UFC levels reduction without normalization, treatment should be adjusted until UFC normalization or until the maximum tolerated dose of metyrapone is reached. Thereafter, efficacy evaluations may be generally performed once a month or every 2 months or as indicated by individual clinical response, unless additional monitoring is recommended. In case of no response, metyrapone discontinuation should be considered. A down-titration, temporary interruption or discontinuation should be considered on the basis of safety profile [ ].
Retrospective studies showed that metyrapone induced UFC normalization in a mean of 63.4% of patients at a mean follow-up of 8.5 months [ ], with an escape rate of 0%–18.7% [ , , ].
Data of the first multicenter prospective study showed that metyrapone induced UFC normalization in 46.9% of patients at 12 weeks, in 52.5% of patients at 24 weeks and in 48.6% of patients at 36 weeks [ ].
These percentages appeared higher in a real-life study up to 64% after 3 months, with a sustained normalization up to 12 and 24 months [ ].
The metyrapone treatment was accompanied by improvement in body weight, blood pressure, glucose metabolism, lipid profile, muscle status, and psychiatric symptoms [ ].
Data on pituitary tumor in patients treated with metyrapone are limited. No pituitary tumor shrinkage was reported in the published literature.
The most frequently reported AEs were hirsutism and/or acne in women (4.5%–71.4%), hypertension (6%–48.4%), dizziness (9.7%–44.4%), nausea (5.3%–33.3%), fatigue (13%-26%), headache (22%), edema (6%–20%), arthralgia (8.7%-18.2%), and hypokalemia (6%–13.6%) [ ].
Some specific safety issues related to metyrapone treatment should be considered in clinical practice, mainly hypocortisolism-related AEs and adrenal hormone precursor accumulation-related AEs.
Hypocortisolism-related AEs may occur in patients treated with metyrapone; therefore, caution should be used in advising, monitoring, and instructing them on how to manage signs and symptoms of hypocortisolism as weakness, fatigue, anorexia, nausea, vomiting, weight-loss, and hypotension. If necessary, metyrapone dose reduction or interruption and/or a temporary exogenous steroid replacement therapy should be evaluated [ ].
Adrenal hormone precursor accumulation related-AEs, and particularly hypokalemia, hypertension and peripheral edema, hirsutism and acne may occur in patients treated with metyrapone. Therefore, a careful monitoring of hypertension is recommended during the 15 days after the beginning of treatment, and after any dose increase for 15 days [ ]. Concomitantly, hypokalemia must be corrected and monitored. Potassium supplementation and spironolactone might be useful prior to starting metyrapone treatment in case of severe hypertension and hypokalemia. Spironolactone might be useful also in case of hyperandrogenism, mainly including new onset or worsening of hirsutism or acne in women [ ].
Ketoconazole
Ketoconazole, a racemic mixture of two enantiomers (2S,4R-ketoconazole and 2R,4S-ketoconazole), acts by inhibiting the cholesterol side-chain cleavage complex, 17,20-lyase, 11β-hydroxylase, and 17β-hydroxylase [ ].
Ketoconazole is approved from EMA for the treatment of CS in adults and adolescents above the age of 12 years [ ].
It is orally administered twice-thrice daily at a dosage of 200–1200 mg/day: it should be started at 400–600 mg/day and increased rapidly to 800–1200 mg/day. After 1–4 weeks of therapy an intermediate efficacy evaluation should be performed. In case of UFC levels reduction without normalization, treatment should be increased by 200 mg/day every 1–4 weeks until UFC normalization or until the maximum tolerated dose of ketoconazole is reached. Thereafter, efficacy evaluations may be generally performed every 3–6 months or as indicated by individual clinical response, unless additional monitoring is recommended. In case of no response, ketoconazole discontinuation should be considered. A down-titration, temporary interruption or discontinuation should be considered on the basis of safety profile [ ].
Retrospective studies showed that ketoconazole induced UFC normalization in a mean of 64.3% of patients at a mean follow-up of 12.6 months [ , ], with an escape rate of 7.1%–22.7% [ ].
Ketoconazole efficacy was also evaluated in a long-term evidence study, with UFC normalization reported in 64.7% of patients treated for more than 24 months [ ].
The ketoconazole treatment was accompanied by improvement in body weight, muscle and bone status, blood pressure, glucose metabolism, potassium levels, psychiatric symptoms, as well as in hirsutism and menstrual cycles in women [ , , ].
Data on pituitary tumor in patients treated with ketoconazole are limited. No pituitary tumor shrinkage was reported in the published literature. Conversely, in specific experiences, new tumor appeared in 13.1%–13.8% of patients [ , ].
The most frequently reported AEs were hepatotoxicity (10.7%–18.7%), and particularly an increase in liver enzymes (2.6%–18.4%), gastrointestinal disturbances (3.7%–18.7%), adrenal insufficiency (5.3%–18.5%), gynecomastia (16.7%), and skin rash (3.6%–6.2%) [ , ].
Some specific safety issues related to ketoconazole treatment should be considered in clinical practice, mainly liver dysfunction, hypocortisolism-related AEs, QT prolongation, as well as drug-drug interactions.
Liver dysfunctions are commonly observed in patients treated with ketoconazole, generally occurring early after starting treatment or at dosage increase. Therefore, ketoconazole should not be used in patients with liver enzymes levels > 2xULN. A careful monitoring of liver enzymes (AST, ALT, gamma-glutamyltransferase, and alkaline phosphatase) and bilirubin is recommended, prior to starting ketoconazole treatment, weekly for the first month and at each dose increase, then monthly for 6 months on treatment, and thereafter periodically as clinically indicated. If liver enzyme levels increase < 3xULN, a more frequent monitoring is recommended and the ketoconazole dose should be decreased by at least 200 mg/day, or treatment should be withdrawn, if values persist elevated. If the patient develops clinical symptoms and signs of hepatitis or an increase in liver enzymes ≥ 3xULN, ketoconazole should be discontinued and should not be restarted due to the risk of serious hepatic toxicity [ ].
Hypocortisolism-related AEs may occur in patients treated with ketoconazole; therefore, caution should be used in advising, monitoring, and instructing them on how to manage signs and symptoms of hypocortisolism as weakness, fatigue, anorexia, nausea, vomiting, weight-loss, and hypotension. If necessary, ketoconazole dose reduction or interruption and/or a temporary exogenous steroid replacement therapy should be evaluated [ ].
QT prolongation is less frequently observed in patients treated with ketoconazole; however, an electrocardiography is recommended before, within 1 week after the beginning of the treatment, at each dose change and thereafter periodically as clinically indicated. Caution should be used in patients treated with concomitant medications known to prolong QT interval [ ].
Lastly, the increased risk of drug-drug interactions should not be underestimated. Indeed, considering that ketoconazole is a potent inhibitor of cytochrome CYP3A4, it can inhibit the metabolism of drugs metabolized by this enzyme, resulting in increase and/or prolongation of their effects, including adverse reactions. Concomitantly, considering that ketoconazole is mainly metabolized by cytochrome CYP3A4, its use with potent CYP3A4 inducers may significantly reduce its bioavailability and consequently its efficacy, as well as its use with potent CYP3A4 inhibitors may significantly increase its bioavailability and consequently adverse reactions. Therefore, in both conditions, ketoconazole dose should be adjusted accordingly [ ].
Levoketoconazole
Levoketoconazole, the 2S,4R enantiomer of ketoconazole, has a similar enzymatic inhibitory profile of ketoconazole, inhibiting the cholesterol side-chain cleavage complex, 11β-hydroxylase, 17α-hydroxylase, and aldosterone synthase [ ].
Levoketoconazole is approved from FDA for the treatment of CS adults, for whom surgery is not an option or has not been curative [ ].
It is orally administered twice daily at a dosage of 300–1200 mg/day: it should be started at 300 mg/day and increased up to 1200 mg/day. After at least 2–3 weeks of therapy, an intermediate efficacy evaluation should be performed. In case of UFC levels reduction without normalization, treatment should be increased by 150 mg/day until UFC normalization or until the maximum tolerated dose of levoketoconazole is reached. Notably, dose increases higher than 150 mg/day may be considered in patients with severe disease and many comorbidities requiring rapid control. Thereafter, efficacy evaluations may be generally performed at least every 1–2 months or as indicated by individual clinical response, unless additional monitoring is recommended. In case of no response, levoketoconazole discontinuation should be considered. A down-titration, temporary interruption or discontinuation should be considered on the basis of safety profile [ ].
The pivotal phase III study, SONICS, showed that levoketoconazole induced UFC normalization in 30.8% of patients at 6 months without dose up-titration. Regardless of dose increase, UFC normalization was obtained in 36.2% of patients at 6 months [ ]. These percentages appeared higher in the subsequent phase III study, LOGICS, up to 50% after approximately 6 months (22–27 weeks, corresponding to the end of the randomized withdrawal period) [ ], 55.1% at 9 months, and 40.9% at 12 months [ ].
The levoketoconazole treatment was accompanied by improvement in body weight, glucose metabolism, lipid profile, peripheral edema, QoL and depressive status, as well as in hirsutism and acne in women [ ].
Notably, data on pituitary tumor in patients treated with levoketoconazole showed stability in 87.1% of patients, increase in 9.6% of patients, and decrease in 3.2% of patients at 12 months or last follow-up [ ].
The most frequently reported AEs were increase in liver enzymes (11.7%–44.6%), nausea (29%–31.9%), headache (23%–27.6%), hypokalemia (10.6%–26%), and hypertension (17%–24%) [ ].
Some specific safety issues related to levoketoconazole treatment should be considered in clinical practice, mainly liver dysfunction, hypocortisolism-related AEs, QT prolongation, as well as drug-drug interactions.
Liver dysfunctions are commonly observed in patients treated with levoketoconazole; therefore, it should not be used in patients with cirrhosis, acute liver disease or poorly controlled chronic liver disease, AST or ALT > 3xULN, recurrent symptomatic cholelithiasis, a prior history of drug induced liver injury due to ketoconazole or any azole antifungal therapy that required discontinuation of treatment, or extensive metastatic liver disease. A careful monitoring of liver enzymes (AST, ALT, gamma-glutamyltransferase, and alkaline phosphatase) and bilirubin is recommended, prior to starting levoketoconazole treatment, weekly for at least 6 weeks, every 2 weeks for the subsequent 6 weeks, monthly for the subsequent 3 months, and thereafter periodically as clinically indicated. If liver enzyme levels increase < 3xULN, or AST or ALT elevations are ≥3 to <5 xULN and total bilirubin concentration is ≤ 2xULN, a more frequent monitoring is recommended and the levoketoconazole dose should be decreased by at least 150 mg/day, or treatment should be withdrawn, if values persist elevated. If the patient develops clinical symptoms and signs of hepatitis, or an increase in liver enzymes ≥ 5xULN, or an increase in liver enzymes ≥ 3xULN associated with increase in total bilirubin > 2xULN, levoketoconazole should be discontinued and should not be restarted due to the risk of serious hepatic toxicity [ ].
Hypocortisolism-related AEs may occur in patients treated with levoketoconazole; therefore, caution should be used in advising, monitoring, and instructing them on how to manage signs and symptoms of hypocortisolism as weakness, fatigue, anorexia, nausea, vomiting, weight-loss, and hypotension. If necessary, levoketoconazole dose reduction or interruption and/or a temporary exogenous steroid replacement therapy should be evaluated [ ].
The risk of QT prolongation, although less frequently observed in patients treated with levoketoconazole, should not be underestimated. Therefore, levoketoconazole is contraindicated in patients taking other drugs known to cause QT interval prolongation associated with ventricular arrhythmias, including torsades de pointes , in patients with a prolonged QTcF interval >470 msec, history of torsades de pointes , ventricular tachycardia, ventricular fibrillation, or long QT syndrome. Levoketoconazole should be used with caution in patients with other risk factors for QT prolongation, such as congestive heart failure, bradyarrythmias, and uncorrected electrolyte abnormalities. An electrocardiography is recommended before, within 1 week after the beginning of the treatment, at each dose change and thereafter periodically as clinically indicated. Concomitantly, hypokalemia and/or hypomagnesemia must be corrected and monitored. If QTcF >500 msec, levoketoconazole should be temporarily discontinued. After the QTcF interval returns to less than 500 msec and contributing factors are corrected, re-institution of levoketoconazole at a lower dose may be considered. If QT interval prolongation recurs after restarting levoketoconazole, levoketoconazole should be permanently discontinued [ ].
Similarly to ketoconazole, the increased risk of drug-drug interactions should not be underestimated.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree