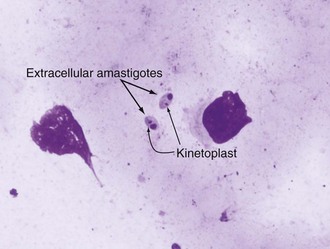
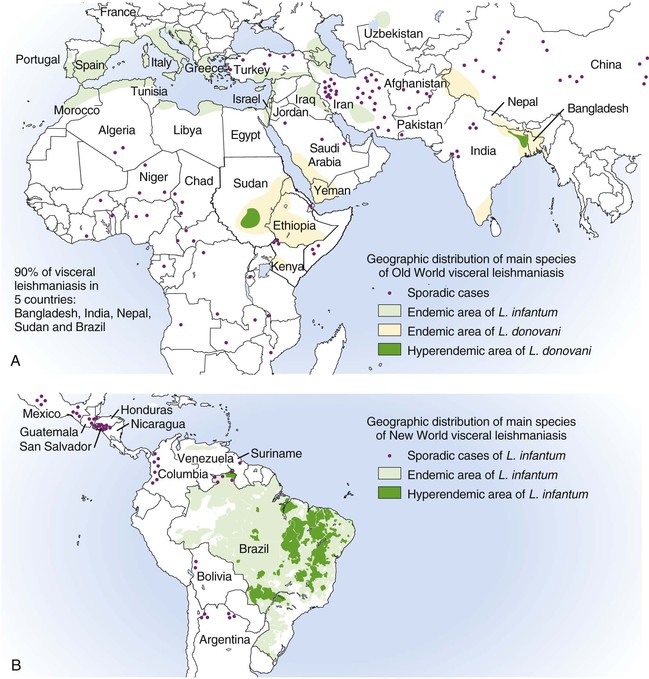
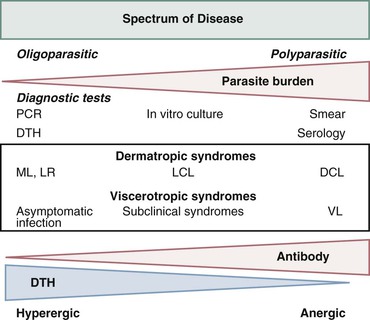
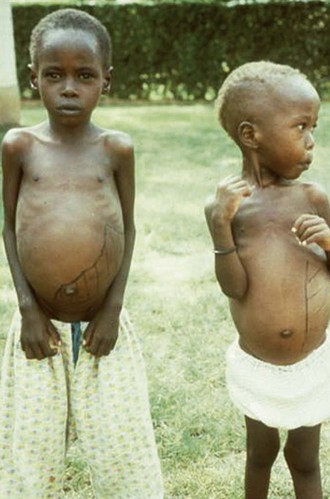
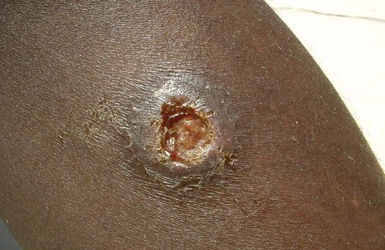
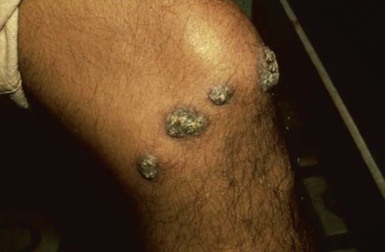
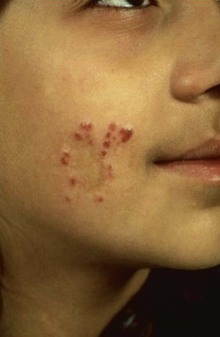
Alan J. Magill Leishmaniasis refers to a diverse spectrum of clinical syndromes caused by infection with protozoan parasites of the genus Leishmania. The clinical syndromes and manifestations of leishmaniasis vary widely but are often divided into the three clinically distinct syndromes of visceral leishmaniasis (VL), cutaneous leishmaniasis (CL), and mucosal leishmaniasis (ML). Cutaneous leishmaniasis is also divided into Old World cutaneous leishmaniasis and New World cutaneous leishmaniasis, referring to the Eastern and Western Hemispheres, respectively. Although there are features common to all Leishmania infections, there is also much that is unique and specific to each syndrome. A single Leishmania species can produce more than one clinical syndrome, and each of the syndromes is caused by more than one species. The outcome in any one patient is a result of parasite factors (invasiveness, tropism, and pathogenicity) and the host’s genetically determined cell-mediated immune responses. It is useful to view infection as leading to the leishmaniases, a heterogeneous collection of clinical diseases, each with its own relatively unique geographic distribution, biology, ecology, local mammalian reservoir, and sand fly insect vector. Aspects that are shared across the spectrum will be discussed initially, followed by syndrome-specific information. Leishmania species are diploid protozoa and have a dimorphic life cycle. A sexual stage has not yet been identified. The life cycle begins when the promastigote, an elongate motile form (1.5 to 3.5 by 15 to 200 µm) of the parasite found in the sand fly digestive tract and proboscis, is transmitted into the skin of a mammalian host by the bite of small, delicate, female sand flies when they take a blood meal. After inoculation by a sand fly, promastigotes are phagocytized by macrophages in the dermis and transform into intracellular oval or round amastigotes (3 to 5 µm in length) that lack an exteriorized flagellum. Amastigotes are found inside phagolysosomes, where they multiply by simple binary division, eventually rupturing the cell and invading other reticuloendothelial (RE) cells. Released amastigotes go on to infect other mononuclear phagocytes. In Wright- and Giemsa-stained preparations, the amastigote cytoplasm appears blue, and the nucleus is relatively large, eccentrically located, and red. Amastigotes have a distinct, rod-shaped, red-staining structure called a kinetoplast, a specialized mitochondrial structure that contains a substantial amount of extranuclear DNA arranged as catenated minicircles and maxicircles. Visualization of the kinetoplast, which has a characteristic appearance under oil-immersion microscopy, as seen in Figure 277-1, confirms the diagnosis of leishmaniasis. Additional mononuclear phagocytes are attracted to the site of the initial lesion and become infected. Amastigotes disseminate through regional lymphatics and the vascular system to infect mononuclear phagocytes throughout the reticuloendothelial system. The cycle is completed when female phlebotomine sand flies ingest parasitized cells. When in the digestive tract of the sand flies, Leishmania parasites develop through a series of flagellated intermediate stages to become infectious metacyclic promastigotes. The taxonomy of Leishmania parasites can be confusing and is constantly evolving. The genus Leishmania has been divided into two subgenera, Viannia and Leishmania. Species in the Viannia subgenus develop in the hindgut of the sand fly before migrating to the midgut and foregut (peripylaria), whereas those of the Leishmania subgenus develop in the midgut and foregut (suprapylaria).1 Species of the Viannia subgenus are endemic in Central and South America. Members of the Leishmania subgenus are found throughout the world. Different definitions, standards, and technical methods have been used over the past several decades to determine the species level characterization of Leishmania parasites, making clinically relevant correlation of a species designation to treatment study outcomes and prognosis challenging. The Leishmania spp. that infect humans, their geographic distributions, and the clinical syndromes they most commonly produce are summarized in Table 277-1.1,2,3,4 Historically, Leishmania parasites have been divided into different species based on clinical, biologic, geographic, and epidemiologic criteria. For example, a parasite isolated from a patient with a typical ‘‘dry ulcer’’ in an urban setting in the Middle East was called Leishmania tropica. TABLE 277-1 Leishmania Parasites, Major Clinical Syndromes, and Distribution Data from references 1, 2, 3, and 4. Beginning in the early 1970s, intrinsic characteristics, such as biochemical and molecular markers, were identified and used to develop classification systems. Isoenzyme analysis by electrophoresis, developed in the 1970s, is widely used as a typing system and still valuable as a reference technique for parasite characterization. The taxonomic profile determined by isoenzyme electrophoresis is called a zymodeme. Isoenzyme analysis is most often successful with culture-amplified parasites. It requires specialized laboratory expertise and usually cannot provide a result to the clinician in a meaningful time frame to affect the choice or duration of therapy. Further complicating isoenzyme interpretation, different research groups have adopted different techniques. The MON (for Montpellier, France) typing system, an authoritative standard from an acknowledged world reference center, uses 15 standardized isoenzyme loci analyzed by starch gel electrophoresis.5 In contrast, American researchers more commonly use a system of three standardized isoenzyme loci identified by cellulose gel electrophoresis.6 Intuitively, the use of more isoenzyme loci reveals greater differences between strains. The clinical implications of greater or lesser degrees of characterization are not clearly known but are likely to be important. For example, Leishmania panamensis and Leishmania guyanensis, considered different species in the past, may instead represent clusters within a spectrum of genetic diversity.7 From a clinician’s perspective, a useful classification is one that accurately predicts the natural history of infection and response to treatment. The use of polymerase chain reaction (PCR) assays with Leishmania species-specific oligonucleotide primers, other molecular and genetic information, and insights gained from the publication of the genomes of Leishmania braziliensis, Leishmania major, and Leishmania infantum will lead to an explosion of new knowledge.8–10 The leishmaniases are widely distributed across the tropical, subtropical, and temperate regions in 88 countries, 72 of which are in developing areas of the world (Figs. 277-2 and 277-3). Three-hundred fifty-million women, men, and children are at risk in widely scattered areas. An estimated 12 million people suffer from leishmaniasis, with 500,000 new cases of VL per year and 1 to 1.5 million new cases of CL per year, with 2.4 million disability-adjusted life-years.11 Approximately 90% of all cases of VL are found in three areas: the drainage basin of the Ganges river in eastern India and neighboring areas of southern Nepal (the “Terai”) and areas of Bangladesh that share the same ecology; the Sudan, where a large epidemic has occurred among displaced people12–14; and Brazil, where VL is endemic in rural areas and large periurban outbreaks have been reported from cities in the northeast.15,16 VL has also emerged as an important opportunistic disease in persons with acquired immunodeficiency syndrome (AIDS) in southern Europe and other areas of the world where the two diseases coexist, in persons who have had organ transplants, and in association with other conditions in which cell-mediated immunity is compromised. Approximately 90% of the world’s CL cases occur in Iran, Saudi Arabia, and Syria in the Middle East; in Afghanistan in Central Asia; and in Brazil and Peru in Latin America.11 CL is an important problem for residents, settlers, travelers, and military personnel visiting endemic areas. More than 2000 cases of CL have been reported among American troops serving in Iraq and Afghanistan since 2001. Other cases occur among North American civilians after exposure in endemic regions.17 Finally, 90% of the cases of ML occur in three Latin American countries: Bolivia, Brazil, and Peru. Female sand flies of the genus Lutzomyia in the Americas and Phlebotomus elsewhere transmit Leishmania spp. (Fig. 277-4).18 The sand flies are modified pool feeders, meaning that they feed on pooled blood from their bite. They breed in cracks in the walls of dwellings, in rubbish or rubble, or in rodent burrows. They are weak fliers and tend to remain close to the ground near their breeding sites. Promastigotes in the sand fly gut replicate and differentiate to metacyclic promastigotes over a period of approximately 1 week. Saliva from the sand fly enhances the infectivity of promastigotes through the effects of maxadilan, a potent vasodilator and immunomodulator, and possibly other factors. Depending on the Leishmania sp., the sand fly genus, and the geographic location, the major reservoirs are canines, rodents, or humans.19,20 Although most transmission is by sand fly bites, Leishmania can be transmitted by blood transfusions, sharing of needles by intravenous drug abusers, occupational exposures, congenital transmission, and rarely by sexual transmission.21–25 Leukodepletion effectively reduces or eliminates transfusion-associated risk of Leishmania infection.26 Leishmaniasis can be thought of as a polar disorder similar to other intracellular infections such as leprosy (Fig. 277-5).27 For example, the range of clinical features in CL parallels that of leprosy. At the polyparasitic end of the spectrum is diffuse cutaneous leishmaniasis (DCL), a relatively uncommon syndrome, in which there is little evidence of effective cell-mediated immune response. Heavily parasitized macrophages are abundant throughout the dermis, and few lymphocytes are present. Peripheral blood mononuclear cells neither proliferate nor produce interferon-γ (IFN–γ) nor interleukin-2 (IL-2) in response to leishmanial antigens in vitro and cutaneous delayed-type hypersensitivity (DTH) reactions are absent.28 DCL has been compared with lepromatous leprosy, in which there is a large number of mycobacteria in macrophages and no evidence of protective, Th1 cell–mediated immune responses. At the oligoparasitic end of the spectrum is leishmaniasis recidivans (LR), a hyperergic variant of CL caused by L. tropica infection in the Old World, in which chronic lesions slowly expand while healing at the center. Amastigotes are sparse, and a mononuclear cell infiltrate predominates. This is somewhat analogous to tuberculoid leprosy, in which there is an intense mononuclear infiltrate with few mycobacteria. But CL and leprosy differ in important ways. Although the character and organization of the granuloma in leprosy are invariably characteristic of the position in the clinical spectrum, this is not true in simple cutaneous leishmaniasis, in which lesions progress over time from a predominance of amastigote-containing macrophages and few lymphocytes to a granulomatous response with a predominance of lymphocytes and few parasites before healing.29 There are three ways to approach the diagnosis of all suspected Leishmania infections: clinical, parasitologic, and immunologic. A clinical diagnosis can have a very high pretest predictive probability in some settings. For example, a chronic ulcer present for many weeks with a typical appearance acquired in the jungles of Peru is very likely to be CL. Likewise, a person with fever, weight loss, pancytopenia, and hepatomegaly in known endemic areas of Bihar, India, is very likely to have VL. Because of the toxicity of some of the drugs used to treat leishmaniasis and the prognostic importance of knowing the species causing CL, a confirmed parasitologic diagnosis is often desirable. A parasitologic diagnosis is confirmed by visualizing amastigotes in tissue or smears, promastigotes in culture, or by amplifying Leishmania-specific DNA or RNA in a PCR assay. Knowing the infecting species may affect the choice to offer treatment, choice of treatments, and the regimen or duration of chemotherapy. Immunologic diagnosis is an adjunct in most cases, with various antibody tests, cell-mediated assays, and the use of skin tests, where available, to elicit DTH. The choice of the optimal diagnostic test or procedure depends on the parasite burden of the disease syndrome. Understanding the likely parasite burden of the various Leishmania syndromes allows the optimal choice of diagnostic testing. Syndromes with very high parasite burdens (polyparasitic), such as VL and later-stage DCL, are characterized by huge numbers of parasites, no cell-mediated immunity (anergy), and an easily detected antibody response. Syndromes with few or scanty numbers of parasites (oligoparasitic), such as ML and LR, are characterized by very few recognizable parasites, an exaggerated cell-mediated immune response, and a minimal antibody response. The utility of a relatively insensitive parasitologic test, such as a Giemsa-stained smear, in an oligoparasitic syndrome, such as ML, will be very low. A more appropriate parasitologic test in this syndrome would be PCR.30 Parasites in the Leishmania donovani complex are responsible for most cases of visceral leishmaniasis (see Table 277-1). Leishmania chagasi, once considered a separate species causing VL in the Americas, is now considered the same as L. infantum, which is endemic in the Mediterranean and was probably introduced into the New World by early explorers.31–33 Continued debate on the taxonomy of Leishmania spp. isolated from patients with VL continues, but the clinical relevance of such distinctions remains to be demonstrated. In the Indian subcontinent, late-stage VL is also known as kala-azar (Hindi for black or fatal sickness). VL is also known as Dum Dum fever and Assam fever. Mediterranean VL caused by L. infantum is also known as infantile splenomegaly. Leishmania spp. that are most commonly associated with cutaneous syndromes, such as Leishmania amazonensis in Latin America34,35 and L. tropica in Saudi Arabia, Kenya, Iran, and India, are rarely isolated from patients with visceral syndromes.36–39 The introduction of more sophisticated molecular analysis, such as multilocus microsatellite typing, allows greater discrimination and demonstrates the existence of hybrids and gene flow between Leishmania populations.40,41 VL is a spectrum of symptoms and findings. At one extreme are persons with asymptomatic, inapparent, or self-resolving infections. At the other end are those with classic VL (kala-azar), who present with a characteristic pentad of prolonged fever, weight loss, hepatosplenomegaly, pancytopenia, and hypergammaglobulinemia (Fig. 277-6). Ratios of self-resolving infection to late-stage VL vary geographically, with a person’s age (likely a partial surrogate for immunity) and the tests used to identify asymptomatic infection. Depending on the definition of asymptomatic infection and the sensitivity and specificity of the various serologic, parasitologic, and skin tests used in surveys, the number of estimated asymptomatic infections varies widely.42 Ratios of asymptomatic infection to VL cases are reported to range from 6.5 : 1 in children to 18 : 1 in adults with L. infantum/L. chagasi in northeastern Brazil.43–45 Using sensitive quantitative (qPCR) and serial testing of peripheral blood in longitudinal cohort studies, investigators have demonstrated an unexpectedly high positive rate of 20% to 25% in India46 and 14% in Brazil.47 Although many of those infected have no clinical manifestations, some experience mild symptoms or develop splenomegaly before either progressing to frank visceral leishmaniasis or undergoing spontaneous resolution. Persons who are immunocompromised by AIDS, neoplasm, or immunosuppressive therapy are at increased risk of asymptomatic parasitemia48 and developing progressive disease. VL occurs in widely dispersed areas of the world (see Table 277-1). Transmission depends on the sand fly vector, the presence of a suitable reservoir, and susceptible humans. In the Indian subcontinent, humans serve as the reservoir, and transmission is by Phlebotomus argentipes and other anthropophilic Phlebotomus spp. Persons with post–kala-azar dermal leishmaniasis (PKDL) may serve as the reservoir during interepidemic periods.49 VL caused by L. infantum also occurs in Central Asia and historically in southern China, where dogs and other canines are reservoirs. In East Africa, VL occurs in Eritrea, Ethiopia, Kenya, Somalia, Sudan, and Uganda. L. donovani has been responsible for a very large epidemic among displaced persons in southern Sudan.50–52 VL is endemic and sporadic in other areas of East Africa. Putative reservoirs include rats, gerbils, other rodents, and small carnivores. Humans may also be a reservoir during epidemics. Although all visceralizing Leishmania parasites from East Africa are now considered to be L. donovani,53 molecular methods demonstrate a very high degree of strain heterogeneity.54,55 Zoonotic VL occurs sporadically in the Mediterranean littoral and the Middle East, where rodents, such as the black rat, and dogs are reservoirs for L. donovani and L. infantum, respectively. The importance of asymptomatic infection in humans as a reservoir of L. infantum, for parasite transmission by blood donation and for development of clinical disease in immunocompromised individuals is increasingly recognized.56 Clinically apparent cases are typically encountered among infants, young children, and immunocompromised persons. VL emerged as an important opportunistic disease among persons with AIDS in southern Europe in Spain, France, and Italy.57,58 Sharing of contaminated needles and syringes by intravenous drug users was implicated in artificial anthroponotic transmission of Leishmania in Spain.59 In Latin America, L. infantum/L. chagasi is endemic and broadly distributed. Most areas have focal disease risk with a background of asymptomatic or subclinical infection with sporadic clinical cases in rural areas. The clustering of cases in households suggests that humans may also be reservoirs in these settings. Children are most frequently affected. Lutzomyia longipalpis is the major vector. Domestic dogs and wild foxes are reservoirs of infection.43 Major periurban outbreaks of VL have been reported from cities in northeastern Brazil, where suburbs have extended into endemic areas.15,16,60 On occasion, Leishmania spp. that usually cause CL, such as L. amazonensis, L. tropica, or other Leishmania spp., are isolated from persons with visceral disease. For example, a small group of American military personnel who served in the Persian Gulf War acquired a “viscerotropic” form of L. tropica infection.39 The pathogenesis of VL is dependent on a combination of vector, parasite, and host factors.61 Skin lesions at the site where promastigotes are inoculated are usually not apparent in persons with VL. Neutrophils, the earliest cells recruited to the bite site, ingest parasites, and both free parasites and infected neutrophils are then taken up by dendritic cells and macrophages, which then spread to visceral organs. Increasing numbers of amastigote-infected mononuclear phagocytes in the liver and spleen result in progressive hypertrophy of these organs, leading to clinically apparent hepatosplenomegaly. The spleen often becomes massively enlarged as splenic lymphoid follicles are replaced by parasitized mononuclear cells. In the liver, there is a marked increase in the number and size of Kupffer cells, many of which contain amastigotes. Autopsy studies demonstrate infected mononuclear phagocytes are also found in the bone marrow, lymph nodes, and skin and are widely disseminated in other organs in this disseminated disease.62,63 Host determinants associated with the outcome of infection and the manifestations of visceral disease include genetically determined human immune responses, host nutritional status, and immunocompetence. There is evidence of both protective and disease-enhancing immune responses. Cytokines and chemokines play key roles in mediating the outcome of infection, but despite extensive studies in murine models and in naturally infected humans, the precise sequence of events that determines the outcome of infection has not been fully characterized. Sixty-six studies during an outbreak of VL in the Sudan showed that one locus on chromosome 22q12 and probably two loci on chromosome 2q22-23 control susceptibility to VL in this population.64–71 Malnutrition has long been recognized as a risk factor for progression of infection to disease.72–74 Persons with self-resolving infection with L. donovani or L. infantum/L. chagasi and those who have undergone successful chemotherapy develop protective immune responses, but VL disease can develop years later if the infected person becomes immunocompromised. The resolution of infection and the development of immunity are associated with expansion of Leishmania-specific Th1-type CD4+ T cells that secrete IFN-γ and IL-2 in response to parasite antigens. IL-12 also plays an important early role in the development of protective immune responses. Evidence of Leishmania-specific Th1 responses is missing in progressive disease. At the cellular level, IFN-γ activates macrophages to kill amastigotes through l-arginine–dependent nitric oxide production, which follows induction of nitric oxide synthase, and oxidative killing mechanisms. IL-1 and tumor necrosis factor-α (TNF-α) prime macrophages for activation by IFN-γ. In persons with progressive infection who go on to develop VL, development of Leishmania-specific Th1 responses is inhibited. Peripheral blood mononuclear cells neither proliferate nor produce IFN-γ or IL-2 in response to leishmanial antigens in vitro. There is no evidence of cutaneous DTH responses to leishmanial antigens. Paradoxically, antileishmanial antibodies are produced in high titer during progressive visceral leishmaniasis, but they are not protective. Progressive disease in humans is associated with production of IL-10 and transforming growth factor (TGF)-β. IL-10 is known to suppress the development of Th1 responses, and the activation of macrophages by IFN-γ and TGF-β appears to play an important role early in infection.75–83 Why Th1 responses arise and dominate in some persons and not in others is not understood. The sequence of early cytokine responses, the manner in which leishmanial antigens are presented by macrophages and dendritic cells, parasite virulence factors, and the size of the infecting inoculum may all be important variables to identify the precise sequence of events that leads to the development of immunity and to identify the immunogenetic determinants of human disease. Vector determinants associated with progressive disease are the vasodilatory effects of sand fly saliva, the size of the inoculum, and presence of immune responses to sand fly salivary proteins. Vasodilation at the bite site, small inoculums, and absence of antibodies to sand fly salivary antigens may contribute to visceral disease.61 Parasite determinants associated with progressive disease include species-specific genes, gene polymorphism, pseudogenes, and expression levels for stress and virulence genes. Completion of the L. major, L. donovani, L. braziliensis, and Leishmania mexicana genomes highlights only that of more than 8000 genes, only 19 L. donovani–specific genes are absent or found as pseudogenes in cutaneous species.84 Insertion of some of these L. donovani–specific genes into L. major promotes the survival of L. major in the viscera of mice in murine models.61 The fully developed clinical manifestations of VL are similar in all endemic areas. The incubation period is typically several (2 to 8) months but has a wide range, from as short as 10 days or longer than a year. Knowing the exact time of exposure in endemic areas is not possible; incubation periods are often extrapolated from travelers with known exposure periods. Clinical disease may first become symptomatic years after exposure in persons who become immunocompromised. Immunocompromise secondary to a disease process, such as infection with human immunodeficiency virus (HIV) leading to AIDS; immunosuppressive drugs, such as chronic use of corticosteroids, methotrexate, or the new class of TNF inhibitors; or use of immunosuppressive agents after solid-organ transplantation has been well described.85–93,94,95 Anyone with a history of birth or residence in, or travel to, a Leishmania-endemic area is at risk of late reactivation if he or she becomes immunocompromised. In cases with a subacute or chronic course, there is an insidious onset of fever, weakness, loss of appetite, weight loss, failure to thrive, and abdominal enlargement caused by hepatosplenomegaly. In endemic areas, low-grade symptoms may persist for weeks to months before progressing to full-blown VL or slowly resolve. Because these symptoms may not be sufficiently severe to warrant medical attention in poverty-stricken endemic areas, patients may be called “subclinical” when they should be more appropriately be called oligosymptomatic. Fever may be intermittent, remittent with twice-daily temperature spikes (double quotidian), or, less commonly, continuous. Fever is relatively well tolerated, and older clinical references routinely noted the patients were not acutely ill or “toxic” in appearance. Acute presentation in nonimmune persons comprises abrupt onset with high fever and chills, sometimes with a periodicity that suggests malaria.96 Chills, but seldom rigors, accompany the temperature spikes. As time passes, the spleen can become massively enlarged. It is usually soft and nontender. The presence of a hard spleen suggests a hematologic disorder or another diagnosis, such as schistosomiasis. The liver also enlarges; it usually has a sharp edge, soft consistency, and a smooth surface. Lymphadenopathy is common in patients in Sudan52 but uncommon in other geographic areas. Elevated liver enzymes and bilirubin may be observed. Peripheral edema may be seen late in disease, particularly in malnourished children. Hemorrhage can occur from one or more sites; epistaxis and gingival bleeding may be noted as well as petechiae and ecchymoses on the extremities in late-stage disease. Many patients with visceral leishmaniasis become cachectic. This appears to be mediated in part by TNF-α and other cytokines that are known to have catabolic and anorectic effects.97 Secondary bacterial infections are common in persons with advanced VL. Patients can present with coinfection or acquire secondary bacterial infections, such as nosocomial pathogens during hospitalizations. It is important to recognize and treat clinically significant bacterial coinfections.98–100 Death may result from pneumonia, septicemia, tuberculosis, dysentery, or measles, or it may be the consequence of malnutrition, severe anemia, or hemorrhage. The laboratory findings include anemia, leukopenia, thrombocytopenia, and hypergammaglobulinemia. Anemia is almost always present and may be severe. It is usually normocytic and normochromic and appears to be due to a combination of factors, including hemolysis, marrow replacement with Leishmania-infected macrophages, hemorrhage, splenic sequestration of erythrocytes, hemodilution, and marrow suppressive effects of cytokines such as TNF-α. Leukopenia is also prominent, with white blood cell counts occasionally as low as 1000/mm3. It is not known whether the observed neutropenia is due to increased margination, splenic sequestration, an autoimmune process, or a combination of those factors. Eosinopenia (absence of eosinophils) is frequently observed. Of note, anemia and neutropenia have not been prominent in patients with VL who have undergone splenectomy. Hypergammaglobulinemia, circulating immune complexes, and rheumatoid factors are present in the sera of most patients with VL.101,102 The globulin level may be as high as 9 g/dL; the ratio of globulin to albumin is typically high. The erythrocyte sedimentation rate is usually elevated.103 Renal involvement in VL is common.104 Acute renal failure, nephrotic syndrome, and proteinuria have been reported.105–107 Acute glomerulonephritis,105 proliferative glomerulonephritis,107 collapsing focal segmental glomerulosclerosis,108 acute interstitial nephritis,107 and tubular cell necrosis and tubulitis109 have all been described. In a recent retrospective study of 57 patients with VL caused by L. infantum/L. chagasi in Brazil, an elevated serum creatinine was seen in 26% of patients, and the only three deaths occurred in this group.110 In a prospective study of 50 patients with VL caused by L. infantum/chagasi in another region of Brazil, abnormalities in glomerular filtration, urinary concentration, and acidification were consistently observed.111 In patients with HIV coinfection, Leishmania infection is persistent and asymptomatic infections are common. When immunosuppression occurs in late-stage HIV infection, clinical VL can present as an opportunistic infection (OI). VL may be the presenting syndrome of AIDS or another late-stage OI in the terminal stages of disease. VL usually presents with typical combinations of fever, hepatomegaly, splenomegaly, weight loss, and pancytopenia when patients have CD4+ counts greater than 50 cells/mm3, but atypical presentations and localization of parasites are more common when CD4 cells are less than 50 cells/mm3.112,113 AIDS patients have extensive gastrointestinal tract involvement that includes oral mucosa, esophagus, stomach, and small intestine and may present with chronic diarrhea.58,114,115 Lung and pleural involvement presenting as pleural effusions, and bone marrow involvement presenting as aplastic anemia, also have been described.116,117 The incidence of leishmanial OIs has decreased with the introduction of modern antiretroviral drug regimens,118–121 but the number of cases of VL-HIV coinfection worldwide may increase dramatically as HIV infection continues to spread into Leishmania-endemic regions.122–124 Also, post-treatment relapse of VL remains problematic.125 Predictors of relapse include CD4+ T-cell count greater than 100 cells/mm3, an increase in CD4+ T-cell count on follow-up, use of secondary prophylaxis, and prior VL relapse.126 HIV viral load and response to antiretrovirals do not seem to predict relapse. An unusual systemic syndrome associated with L. tropica infection, called viscerotropic leishmaniasis, was described in returning U.S. military personnel who served in the Persian Gulf War of 1990 to 1991.38,39 The symptoms included chronic low-grade fever, malaise, fatigue, and, in some cases, diarrhea. Mild splenomegaly was observed in some. None of the troops developed classic kala-azar or progressive visceral leishmaniasis. Subsequent studies failed to show an association between L. tropica infection and chronic fatigue and other symptoms associated with the Gulf War syndrome.127 Similar oligosymptomatic syndromes have been reported in Brazil and Italy and likely are more common than currently recognized because of the difficulty in making a parasitologic diagnosis in these syndromes.44,128–130 PKDL follows the treatment of VL caused by L. donovani in 5% to 10% of persons within 2 to 4 years after treatment in India and approximately 50% of persons within 0 to 6 months of treatment in the Sudan (Fig. 277-7).131–134 PKDL is rarely seen after treatment of VL in Latin America or in the Mediterranean when the infection is with L. infantum/L. chagasi and, when reported, has been seen in patients with concurrent AIDS.135 PKDL is more commonly seen after treatment with pentavalent antimony (SbV) compounds and appears to be less common after successful treatment with amphotericin B formulations,136 although a definitive demonstration of this effect awaits a prospective clinical trial. The skin lesions of PKDL are chronic and may persist for as long as 20 years in India, whereas in the Sudan, they persist for only a few months to a year. The skin lesions vary from hyperpigmented or hypopigmented macules progressing to papules, nodules, and verrucous forms.137 The lesions are found on the face, trunk, extremities, oral mucous membranes, and occasionally on the genitalia. Lesions may be confused clinically and pathologically with leprosy. Patients generally feel well. The diagnosis is mainly clinical, but amastigotes can be detected in the skin in more than 80% of cases in the Sudan. Antileishmanial treatment is generally indicated for Indian PKDL. In the Sudan, most cases cure spontaneously, but chronic or severe cases are treated. In a few instances in India, VL has recurred in patients with PKDL. Patients with PKDL are thought to be infectious and serve as reservoirs for continued anthroponotic infection.138 Treatment of PKDL patients, at least to render them noninfectious to sand flies, is likely an important part of a future successful control strategy.139 The positive predictive value of a clinical diagnosis of VL in patients with the pentad of prolonged fever, progressive weight loss, pronounced hepatosplenomegaly (especially splenomegaly), pancytopenia, and hypergammaglobulinemia from a known endemic area is very high. In patients with late-stage HIV infection or AIDS, returning travelers who may present with clinical symptoms months or years after exposure in low-transmission areas, such as the Mediterranean, those with oligosymptomatic or viscerotropic syndromes, and in those with atypical presentations, clinical diagnosis without supporting parasitologic confirmation is unsatisfactory. The parasitologic diagnosis of VL can be confirmed by demonstrating amastigotes in tissue, by isolating promastigotes in culture, or with a positive PCR assay. Splenic aspiration, liver biopsy, lymph node aspirates, and bone marrow aspirates have all been used with success. The spleen is the most sensitive location, and splenic aspiration to obtain a few drops of fluid for Wright-Giemsa–stained smears, culture, and for PCR assay is the most sensitive method for parasite identification.140 Splenic aspirations are reported positive in greater than 95% when compared with bone marrow aspirations. The sensitivity of a bone marrow aspirate approaches that of a splenic aspirate when microscopists spend more time reviewing the smear.141–143 Quantifying the parasite burden from splenic aspiration is widely used to stage patients and monitor response to treatment.144 Splenic aspirations are routinely performed in some areas, but it can be associated with substantial hemorrhage, particularly in patients with advanced stages of disease who have undergone aspirations by inexperienced operators. The risk is less when aspiration is performed quickly by an experienced person using a small-bore needle in patients with no laboratory evidence of coagulopathy.140 Bone marrow aspiration is safer and preferred in nonendemic settings but is less sensitive. Amastigotes are seen in 40% to 90% of patients when compared with a positive splenic aspirate. The wide range reflects the stage of disease and the rigor of microscopic review because of the lower parasite burden in the bone marrow. Liver biopsy is less likely to yield the diagnosis than is splenic puncture or bone marrow biopsy but occasionally is positive when bone marrow is negative.145 Lymph node aspiration or biopsy may be diagnostic when enlarged nodes are present, as is often the case in Sudan. Amastigotes may also be seen within mononuclear cells in Wright- and Giemsa-stained smears of the buffy coat146 or in biopsy specimens of various organs. The latter is particularly true in patients with AIDS in whom amastigotes in macrophages have been identified in bronchoalveolar lavage fluid, pleural effusions, or biopsy specimens of the oropharynx, stomach, or intestine. On occasion, parasites have been cultured from the buffy coat or blood. When the diagnosis of VL is suspected, parasitologic confirmation may require more than one technique and repeated procedures.96 Whenever possible, aspirates from the spleen, bone marrow, liver, or lymph node should be cultured. Specimens can be inoculated into one of several media (Schneider’s modified media, Novy-MacNeal-Nicolle [NNN] media, and others) and maintained at ambient temperatures: 22° to 26° C. Amastigotes must transform into motile promastigotes and then multiply in vitro to expand to a number that can be microscopically visualized. When the initial parasite inoculum is high, they may be seen in culture within a few days, but it may take several weeks for the concentration to reach the level of detection. Culturing parasites is an art and is unlikely to be performed in routine microbiology laboratories. In the United States, culture media and expert assistance are available from the Centers for Disease Control and Prevention (CDC). Isolates can be forwarded to the CDC or other World Health Organization reference laboratories for speciation. High-titer antileishmanial antibodies are typically present in immunocompetent patients with VL. A number of serologic tests using different antigens and assays are available. Currently, enzyme-linked immunosorbent assay and dipstick tests using L. infantum/L. chagasi recombinant k39 (rk39), a kinesin-like antigen,147 have demonstrated excellent sensitivity and specificity for the diagnosis of VL in immunocompetent persons in India and Brazil but less so in East Africa,148,149,150 likely because of the extensive diversity of kinesin-related proteins found in East African isolates.151 Proper interpretation of the rk39 test results requires an understanding of the antibody response. Titers to rk39 and other serologic tests remain elevated and detectable after successful treatment.152 False-positive results may occur because of cross-reacting antibodies in patients with leprosy, Chagas’ disease, CL,153 and other infections. Antileishmanial antibodies may be absent or present at low titer in patients with AIDS, resulting in false-negative serologic results. Other serologic test formats, such as direct agglutination and urine antigen detection, have been used with variable success.154,155 Each test format has its relative advantages and disadvantages, depending on the intended use, resources, and cost considerations. The sensitivity of a positive rk39 antibody dipstick test in individuals with suspected VL in India and with a therapeutic response to amphotericin B was shown to be 99% (119/120) in India.156 The use of sensitive point-of-care diagnostic tests in patients with clinically suspect VL, along with the expected therapeutic response to a specific treatment, such as amphotericin B, promises to restrict otherwise invasive procedures, such as splenic aspiration, only to those negative for rk39 or those who fail to improve with amphotericin B. The leishmanin (Montenegro) skin test is usually negative in patients with active VL, becoming positive in the majority of those in whom infection spontaneously resolves and in patients who have undergone successful chemotherapy.157 However, there are no commercially available skin test antigens for leishmaniasis, and skin test preparations used in endemic countries have not been rigorously qualified for cross-reactivity, potency, stability, cross-strain sensitivity, and the ability to sensitize nonexposed individuals.158 The leishmanin skin test may be negative or positive in persons with PKDL, depending on the stage of disease.131 Molecular approaches have become increasingly relevant because of improved sensitivity, specificity, and flexibility in choice of samples. The qPCR assay is a highly sensitive and specific tool used in referral laboratories for detection/assessment of parasite load in VL patients and subsequently in monitoring treatment response to antileishmanial agents.159,160 The method displays potential to provide a threshold for distinguishing asymptomatic persons in endemic areas.47 Currently, improvement in VL diagnostics is required for successful decentralized (point-of-care) testing in field conditions and to detect VL-HIV coinfection. Techniques such as loop-mediated isothermal amplification may offer a reliable molecular diagnostic method for field application.161 The differential diagnosis of late-stage VL with the full complement of symptoms and signs is limited to hematologic and lymphatic malignancies and, occasionally, disseminated histoplasmosis and tropical splenomegaly syndrome. Acute VL has a much broader differential, including malaria, enteric fevers, bacterial endocarditis, sarcoidosis, hemophagocytic syndromes, typhus, acute Chagas’ disease (in Latin America), acute schistosomiasis, miliary tuberculosis, and amebic liver abscess. Subacute or chronic VL may be confused with brucellosis, prolonged Salmonella bacteremia, histoplasmosis, infectious mononucleosis, lymphoma, leukemia, myeloproliferative disease, hepatosplenic schistosomiasis, and chronic malaria. PKDL must be differentiated from leprosy, yaws, and syphilis. Cutaneous leishmaniasis (CL) is endemic in widely scattered areas throughout the world. The classic form of Old World CL is the “oriental sore,” also known by a variety of colorful local expressions, such as bouton d’orient, bouton de Crete, bouton d’Alep, bouton de Biskra, Aleppo evil, Baghdad boil, and Delhi boil, in various regions of the Middle East, the Mediterranean littoral, Africa, India, and Asia. Old World cutaneous leishmaniasis is most frequently caused by L. major, L. tropica, or Leishmania aethiopica, but L. donovani and L. infantum/L. chagasi can also cause simple cutaneous leishmaniasis. The resulting skin lesions range from troublesome and unsightly to severe and complicated, but they generally are not life threatening. Severe cases and facial lesions are very stigmatizing. DCL caused by L. aethiopica infection is reported from Ethiopia and adjacent areas of Africa. New World CL is endemic in widespread areas of Latin America. The causative species include L. braziliensis, L. mexicana, L. panamensis/L. guyanensis, and many others (see Table 277-1). L. infantum/L. chagasi is associated with simple nodular CL in Central America. Depending on the clinical presentation and geographic location, New World CL is variously known as pian bois (bush yaws), uta, or chiclero’s ulcer. The spectrum of New World CL disease includes single or multiple, localized, cutaneous ulcers (Figs. 277-8 to 277-11), diffuse cutaneous leishmaniasis, and mucosal disease (espundia) caused by L. braziliensis or, less commonly, other related Leishmania spp. (Fig. 277-12). The spectrum of Old World CL disease includes single or multiple, localized, cutaneous ulcers (Figs. 277-13 and 277-14), diffuse cutaneous leishmaniasis, post–kala-azar dermal leishmaniasis (see Fig. 277-7), and leishmaniasis recidivans (Fig. 277-15). In the Old World, CL is usually a sporadic disease in endemic areas, but on occasion it occurs in an epidemic pattern, particularly when large groups of susceptible persons are exposed during road construction, refugee movements, or military activities.162 L. major is an infection of desert rodents, primarily gerbils, and affects humans in arid and rural regions of the Middle East, North Africa, and Central Asia. CL caused by L. major has been a major problem for Western military personnel operating in endemic regions in the Middle East.163,164 The lesions tend to be larger and “wet” with an overlying exudate. Phlebotomus papatasi and other Phlebotomus spp. are the vectors. L. tropica infects dogs and humans in urban areas of the Middle East, such as Baghdad, Teheran, Kabul, and Damascus, as well as cities in the Mediterranean littoral, India, and Pakistan.165 The lesions tend to be crusted and “dry.” The vectors include Phlebotomus sergenti and P. papatasi. L. aethiopica is endemic in Ethiopia, Kenya, and southwest Africa. The primary reservoirs are the rock hyrax; rodents are secondary reservoirs. Phlebotomus longipes and Phlebotomus pedifer are the vectors. On occasion, L. donovani and L. infantum/L. chagasi cause simple CL. New World CL is usually a rural zoonosis.3 The main reservoirs are forest rodents, except in the case of Leishmania peruviana, for which dogs are the primary reservoir. The vectors are ground-dwelling or arboreal Lutzomyia spp. Disease is common in persons working at the edge of the forest and among rural settlers. Outbreaks occur when areas of forest are cleared for roads, villages, or farms, or when military personnel or tourists enter endemic regions. L. mexicana is responsible for New World CL from northern Argentina to Texas, where a small number of autochthonous cases have been reported.166–168 New World CL is an occupational hazard of gum (chicle) collectors in Central America as well as persons living, working, or touring in endemic areas. Lesions typically appear on exposed areas of the extremities, face or ears (known as chiclero’s ulcer when lesions involve the ear.).169 A number of sylvatic rodents are reservoirs. Lutzomyia species are the vectors. L. amazonensis produces a spectrum of disease in South America that includes simple cutaneous, diffuse cutaneous, and visceral leishmaniasis.35 The vectors are Lutzomyia spp., and the reservoirs are forest animals. L. braziliensis is found in widely scattered areas of Central and South America. It is responsible for cutaneous as well as ML. CL caused by L. braziliensis has been diagnosed among American tourists returning from Belize and other Latin American areas. L. panamensis is found in Panama and adjacent countries. It was an important problem for U.S. military personnel training in jungle areas of Panama. L. guyanensis is responsible for pian bois or bush yaws in the northern Amazon basin. L. peruviana is the cause of uta in Peru. It typically causes dry lesions.
Leishmania Species
Visceral (Kala-Azar), Cutaneous, and Mucosal Leishmaniasis
Overview and General Principles
Life Cycle
Taxonomy and Classification
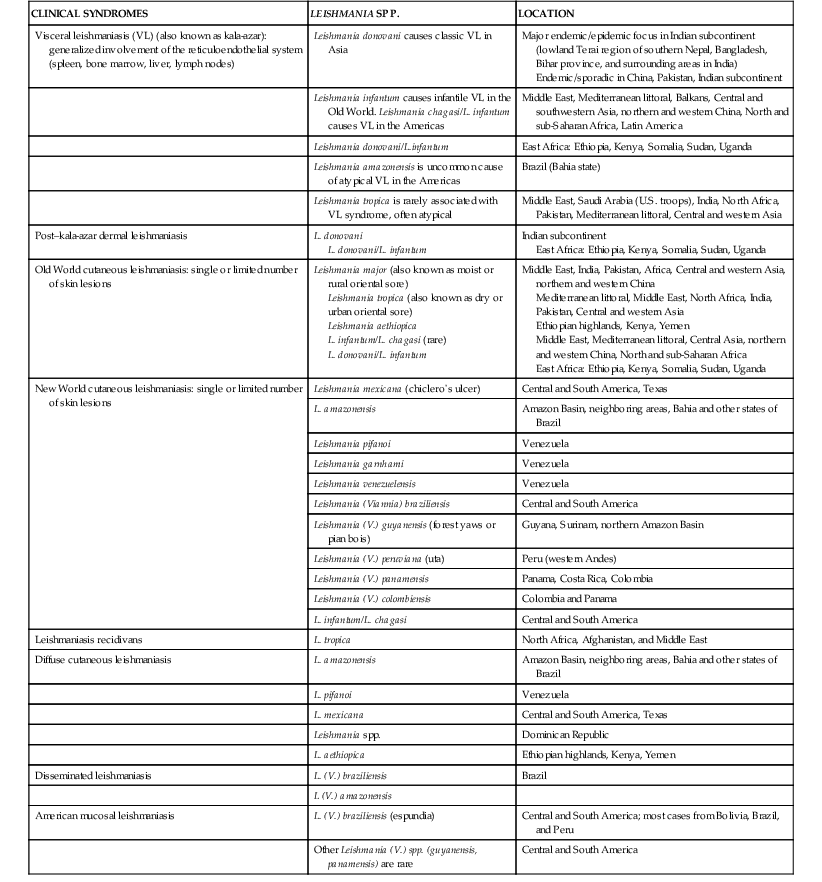
CLINICAL SYNDROMES
LEISHMANIA SPP.
LOCATION
Visceral leishmaniasis (VL) (also known as kala-azar): generalized involvement of the reticuloendothelial system (spleen, bone marrow, liver, lymph nodes)
Leishmania donovani causes classic VL in Asia
Major endemic/epidemic focus in Indian subcontinent (lowland Terai region of southern Nepal, Bangladesh, Bihar province, and surrounding areas in India)
Endemic/sporadic in China, Pakistan, Indian subcontinent
Leishmania infantum causes infantile VL in the Old World. Leishmania chagasi/L. infantum causes VL in the Americas
Middle East, Mediterranean littoral, Balkans, Central and southwestern Asia, northern and western China, North and sub-Saharan Africa, Latin America
Leishmania donovani/L.infantum
East Africa: Ethiopia, Kenya, Somalia, Sudan, Uganda
Leishmania amazonensis is uncommon cause of atypical VL in the Americas
Brazil (Bahia state)
Leishmania tropica is rarely associated with VL syndrome, often atypical
Middle East, Saudi Arabia (U.S. troops), India, North Africa, Pakistan, Mediterranean littoral, Central and western Asia
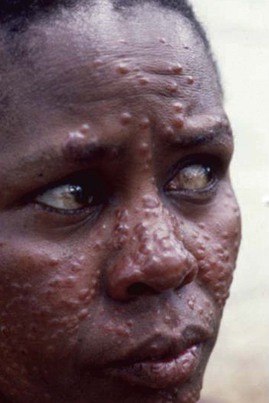
Post–kala-azar dermal leishmaniasis
L. donovani
L. donovani/L. infantum
Indian subcontinent
East Africa: Ethiopia, Kenya, Somalia, Sudan, Uganda
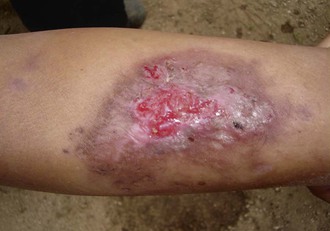
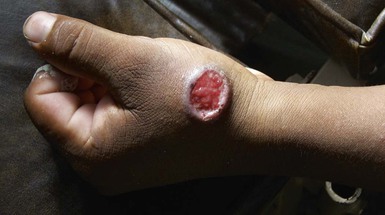
Old World cutaneous leishmaniasis: single or limited number of skin lesions
Leishmania major (also known as moist or rural oriental sore)
Leishmania tropica (also known as dry or urban oriental sore)
Leishmania aethiopica
L. infantum/L. chagasi (rare)
L. donovani/L. infantum
Middle East, India, Pakistan, Africa, Central and western Asia, northern and western China
Mediterranean littoral, Middle East, North Africa, India, Pakistan, Central and western Asia
Ethiopian highlands, Kenya, Yemen
Middle East, Mediterranean littoral, Central Asia, northern and western China, North and sub-Saharan Africa
East Africa: Ethiopia, Kenya, Somalia, Sudan, Uganda
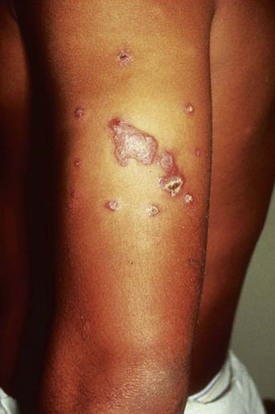
New World cutaneous leishmaniasis: single or limited number of skin lesions
Leishmania mexicana (chiclero’s ulcer)
Central and South America, Texas
L. amazonensis
Amazon Basin, neighboring areas, Bahia and other states of Brazil
Leishmania pifanoi
Venezuela
Leishmania garnhami
Venezuela
Leishmania venezuelensis
Venezuela
Leishmania (Viannia) braziliensis
Central and South America
Leishmania (V.) guyanensis (forest yaws or pian bois)
Guyana, Surinam, northern Amazon Basin
Leishmania (V.) peruviana (uta)
Peru (western Andes)
Leishmania (V.) panamensis
Panama, Costa Rica, Colombia
Leishmania (V.) colombiensis
Colombia and Panama
L. infantum/L. chagasi
Central and South America
Leishmaniasis recidivans
L. tropica
North Africa, Afghanistan, and Middle East
Diffuse cutaneous leishmaniasis
L. amazonensis
Amazon Basin, neighboring areas, Bahia and other states of Brazil
L. pifanoi
Venezuela
L. mexicana
Central and South America, Texas
Leishmania spp.
Dominican Republic
L. aethiopica
Ethiopian highlands, Kenya, Yemen
Disseminated leishmaniasis
L. (V.) braziliensis
Brazil
L (V.) amazonensis
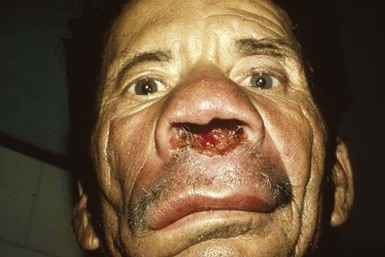
American mucosal leishmaniasis
L. (V.) braziliensis (espundia)
Central and South America; most cases from Bolivia, Brazil, and Peru
Other Leishmania (V.) spp. (guyanensis, panamensis) are rare
Central and South America
Epidemiology
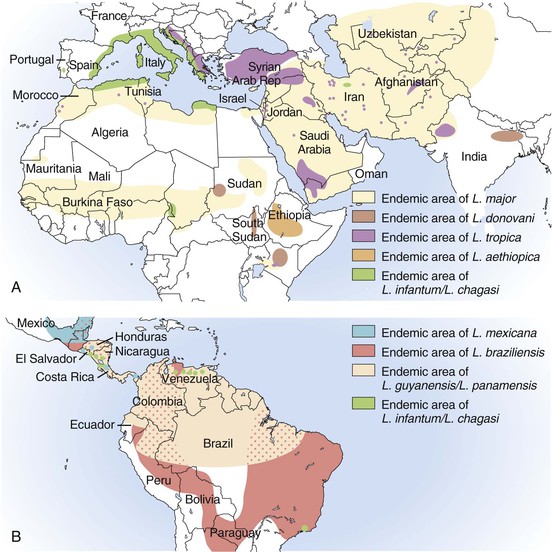
Pathogenesis
Diagnosis
Visceral Leishmaniasis
Epidemiology
Pathogenesis and Natural History of Visceral Leishmaniasis
Clinical Manifestations of Kala-Azar
Visceral Leishmaniasis in Patients with AIDS
Viscerotropic Leishmaniasis
Post–Kala-Azar Dermal Leishmaniasis
Diagnosis
Differential Diagnosis
Cutaneous Leishmaniasis
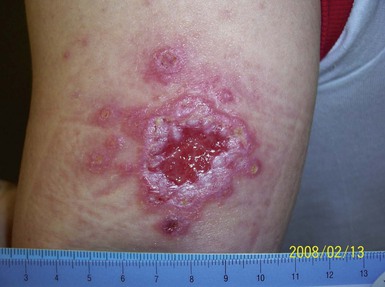
Epidemiology
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Leishmania Species
277