David O. Freedman
Infections in Returning Travelers
Of the approximately 80 million people who travel from industrialized to developing countries each year, 22% to 64% of travelers report some illness.1,2 The approach to the patient requires knowledge of world geography, the epidemiology of disease patterns in 230 or so countries, and the clinical presentation of a wide spectrum of disorders.3 Most illnesses are mild, most are self-limited, and many are noninfectious. Up to 10% of travelers may consult a physician during or after a trip, and approximately 1 in 100,000 travelers will die.
The ill travelers that do come to the attention of infectious diseases clinicians are generally either the most seriously ill or are suspected of harboring infectious agents not familiar in their home country. Based on 42,173 ill returned travelers seen by the GeoSentinel Surveillance Network at 53 different clinical sites on six continents, in patients presenting to infectious or tropical diseases specialists after travel to the developing world, specific travel destinations are associated with the probability of the diagnosis of certain diseases.4 Diagnostic approaches and empirical therapies can be guided by these destination-specific differences. Important region-specific disease occurrence data indicate that febrile illness is most important from Africa and Southeast Asia; malaria is one of the top three diagnoses from every region, yet over the past decade dengue has become the most common febrile illness from every region outside sub-Saharan Africa; in sub-Saharan Africa, rickettsial disease is second only to malaria as a cause of fever; respiratory disease is most important in Southeast Asia and sub-Saharan Africa; and acute diarrhea is disproportionately from South Central Asia. When individual diagnoses are collected into syndrome groups and examined for all regions together, 233 of every 1000 ill returned travelers have a systemic febrile illness, 334 have acute or chronic gastrointestinal infection, 195 have a dermatologic disorder, and 209 have a respiratory disorder.
Travelers who become ill during, or any time up to several months after, a foreign trip will frequently associate that illness with a possible travel-specific cause. This may be the case, but often it is not. Routine disorders are common, and common disorders are common whether actually acquired during travel or at some time after the trip. Thus fever, sore throat, and cervical adenopathy in a college student who returned 2 weeks earlier from a developing country is still more likely to be streptococcal pharyngitis or infectious mononucleosis than diphtheria. Presented with an ill patient with a history of travel, the physician must maintain discipline in making two separate lists of differential diagnoses, the first with the travel history factored in and the second considering the same presenting symptoms and signs as if in any other patient. The approach and workup must then proceed in parallel, with appropriate priority given to the most urgent or the most treatable diagnoses at the top of each list.
In this chapter, travelers are considered to be those returning from short visits to developing countries, and the term does not include immigrants, refugees, and very long-term residents arriving from those countries. Constellations of exposures and clinical presentations highly suggestive of particular diagnoses in returned travelers are shown in Table 324-1. Highly exotic endemic diseases rarely acquired by travelers are not discussed. Low-frequency illnesses (<20 cases of the 42,173 listed in the “GeoSentinel Surveillance of Illness in Returned Travelers, 2007-2011”4), some potentially serious, were reported, including visceral leishmaniasis, east African trypanosomiasis, scrub typhus, relapsing fever, angiostrongyliasis, botulism, melioidosis, tularemia, hantavirus infection, and infection with Plasmodium knowlesi. No cases of yellow fever, Ebola virus, Lassa fever, Marburg virus, tetanus, polio, anthrax, or plague were reported in this 5-year cohort, thus attesting to their rarity.
TABLE 324-1
Constellations of Exposures and Clinical Presentations Suggestive of Particular Diagnoses in Returned Travelers*
| EXPOSURE SCENARIO | DISTINCTIVE FINDINGS | DIAGNOSIS |
| Any exposure in any area with documented malaria transmission | Fever with or without any other finding | Malaria |
| Most tropical countries | Fever and altered mental status | Malaria, meningococcal meningitis, rabies, West Nile virus |
| Budget travel to India, Nepal, Pakistan, or Bangladesh | Insidious-onset, high unremitting fever, toxic patient, paucity of physical findings | Enteric fever due to Salmonella Typhi or Salmonella Paratyphi |
| Freshwater recreational exposure in Africa | Fever, eosinophilia, hepatomegaly, negative malaria smear | Acute schistosomiasis (Katayama fever) |
| Bitten by Aedes aegypti in Central America, Southeast Asia, or the South Pacific | Fever, headache, myalgia, diffuse macular rash, mild to moderate thrombocytopenia | Dengue |
| Bitten by A. aegypti or Aedes albopictus in India, Malaysia, Singapore, the Caribbean, or an island in the Indian Ocean | Fever, headache, myalgia, diffuse macular rash, arthralgia, tenosynovitis often followed by chronic polyarthritis after the fever resolves | Chikungunya fever |
| Hunting or visiting game reserves in southern Africa | Fever, eschar, diffuse petechial rash | African tick typhus due to Rickettsia africae |
| Travel to Southeast Asia | Fever, eschar, diffuse petechial rash | Scrub typhus due to Orientia tsutsugamushi |
| Hiking, biking, swimming, rafting with exposure to fresh surface water | Fever, myalgia, conjunctival suffusion, mild to severe jaundice, variable rash | Leptospirosis |
| Cruise, elderly traveler | Influenza-like illness | Influenza A or B |
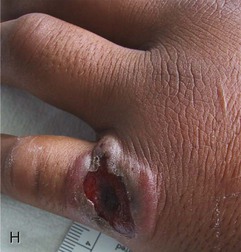
| Outdoor exposure anywhere in the Americas | Large, single furuncular lesion anywhere on body, with sense of movement inside | Myiasis due to Dermatobia hominis (botfly) |
| Clothing washed or dried out of doors in Africa | Multiple furuncular lesions around clothing contact points with skin | Myiasis due to Cordylobia anthropophaga (tumbu fly) |
| New sexual partner during travel | Fever, rash, mononucleosis-like illness | Acute human immunodeficiency virus infection |
| Travel to any developing country or to Western Europe | Coryza, conjunctivitis, Koplik spots, rash | Measles |
| Longer visit to humid areas of Africa, the Americas, or Southeast Asia | Asymptomatic eosinophilia or with periodic cough or wheezing | Strongyloidiasis |
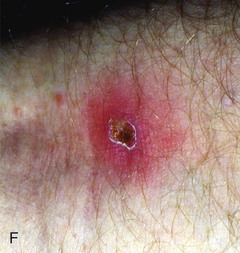
| Sand fly bite in either New or Old World tropical area | Painless skin ulcer with clean, moist base in exposed area | Cutaneous leishmaniasis |
| Resort hotel in southern Europe, ± exposure to whirlpool spas | Pneumonia | Legionnaires’ disease |
| Explored a cave in the Americas | Fever, cough, retrosternal chest pain, hilar adenopathy | Histoplasmosis |
| Ingestion of unpasteurized goat cheese | Chronic fever, fatigue | Brucella melitensis |
| Long trip to West/Central Africa | Afebrile, intensely pruritic, evanescent truncal maculopapular rash | Onchocerciasis |
| Long trip to West/Central Africa | Migratory localized angioedema or swellings over large joints, eosinophilia | Loiasis |
| Safari to game parks of East Africa | Fever, nongenital chancre, fine macular rash | East African trypanosomiasis |
| Travel to Australia | Fever, fatigue, polyarthritis | Ross River virus |
| Farming areas of India and Southeast Asia | Fever, altered mental status, paralysis | Japanese encephalitis |
| Forested areas of central and eastern Europe and across Russia | Fever, altered mental status, paralysis | Tick-borne encephalitis |
| Rodent exposure in West Africa | Fever, sore throat, jaundice, hemorrhagic manifestations | Lassa fever |
| Ingestion of sushi, ceviche, or raw freshwater fish | Migratory nodules in truncal areas with overlying erythema or mild hemorrhage | Gnathostomiasis |
| Returning Hajj pilgrim or family contact | Fever, meningitis | Meningococcal meningitis |
| Ingestion of snails, fish, or shellfish in Asia or Australia | Eosinophilic meningitis | Angiostrongyliasis, gnathostomiasis |
| Diabetic or compromised host with exposure to moist terrain in Asia or Australia | Fever, sepsis, pneumonia or multifocal abscesses | Melioidosis |
| Summertime exposure to rodent droppings in Scandinavia | Fever with decreased renal function | Puumala virus |
| Ingestion of undercooked meat of any animal in any country | Fever, facial edema, myositis, increased creatine phosphokinase, massive eosinophilia, normal erythrocyte sedimentation rate | Trichinosis |
| Unvaccinated, returning from sub-Saharan Africa or forested areas of Amazonia | Fever, jaundice, proteinuria, hemorrhage | Yellow fever |
| Exposure to farm animals | Pneumonia, mild hepatitis | Q fever |
| Possible tick exposure almost anywhere | Fever, headache, rash, conjunctival injection, hepatosplenomegaly | Tick-borne relapsing fever |
| Poor hygienic conditions with possible body louse exposure in Ethiopia or Sudan | Fever, headache, rash, conjunctival injection, hepatosplenomegaly | Louse-borne relapsing fever |
* The table includes illnesses of travelers (listed first) as well as less common diseases with presentations that should suggest the possibility of the appropriate diagnosis. Many diseases have a spectrum of presentation, and the table describes the most common presentations of these diseases. Many diseases have a spectrum of geographic origins, and the table describes the most common exposures seen in daily practice.
The focus is on the identification of infectious causes of the presenting illness, on travel-associated risk factors, and on manifestations of those diseases that are particular to travelers. Detailed discussions of pathophysiology, spectrum of clinical manifestations, and therapy for each infectious agent are found in the disease-specific chapters of this book. Fever, traveler’s diarrhea, and skin problems are the most common presenting illnesses in returned travelers. Eosinophilia is less common but is a frequent source of referral to the infectious diseases specialist.
Fever
Epidemiology
Fever occurs in 2% to 3%5–7 of European or American travelers to the developing world. The proportion of ill returned travelers who present to specialists with a febrile illness is 24%, with variation by region of travel: Americas, 14%; South-Central Asia (includes India), 13%; Southeast Asia, 18%; and sub-Saharan Africa, 43%.
Several large case series from busy tropical disease units indicate malaria to be the cause of the fever in 27% to 42%.5–7 The other most common tropical diseases specific to returning travelers are dengue, rickettsial disease, typhoid fever, and those caused by enteric pathogens. Less common but important considerations are leptospirosis, chikungunya, acute schistosomiasis, and amebic liver abscess. All of these diseases have widespread distribution in the tropics and need to be considered initially in all febrile travelers. Some may be ruled out quickly based on a detailed travel and exposure history and consultation with relevant information sources on disease distribution. Upper and lower respiratory tract infection, including streptococcal pharyngitis and influenza, as well as urinary tract infections, are cosmopolitan, nontropical febrile disorders that are remarkably common in travelers and should always be considered. In every series from sophisticated referral centers, up to 22% to 25% of those presenting with fever have self-limited illnesses that never have an etiologic diagnosis confirmed.7 These are mostly viral syndromes caused by one of hundreds of viral agents that exist outside developed countries for which diagnostic tests may not be available anywhere. In many cases, the time and expense of a large panel of viral isolation and serologic assays is not warranted outside the research setting. Fever due to deep venous thrombosis or pulmonary embolism may be related to travel, especially in those with preexisting conditions or underlying coagulopathy. Thromboembolic disease always needs to be considered from the outset but is not discussed further here.8,9
History
A good patient history is always important in clinical medicine, but nowhere is it as important as in the returning traveler. The cumulative list of infectious agents in 230 separate countries is daunting. A day-by-day travel itinerary, knowledge of risk factors and exposures for the common travel diseases, knowledge of usual incubation periods of those diseases, and knowledge of or access to the known geographic distribution of possible infectious diseases will lead to an appropriately focused workup.8–10 Much time, expense, and patient discomfort due to sometimes invasive diagnostic tests can be avoided when diagnoses that are not epidemiologically or chronologically possible are eliminated based on the patient history.
The fever pattern and clinical findings by themselves are often nonspecific and overlap greatly between many of the most common tropical infectious diseases. The history should include the key elements detailed in the following sections.
Detailed Travel Itinerary
A travel itinerary should include every locale visited in every country visited, including transit stops. Some individuals are frequent travelers, so all travel for at least the previous 6 months must be considered initially. If the diagnosis remains elusive, a more remote travel history, especially that involving malarious areas, may be sought. The exact date of arrival back in the home country is often crucial to ascertain the last possible exposure date to an exotic pathogen. These details are most efficiently ascertained using a waiting room questionnaire. For example, it is insufficient to know simply that the patient visited Peru. Some parts of Peru are malarious and others are not, only some have risk of yellow fever, high-altitude destinations have little risk of vector-borne disease, and there is no risk of strongyloidiasis along the desert coastal strip.
Chronology of Travel and Illness
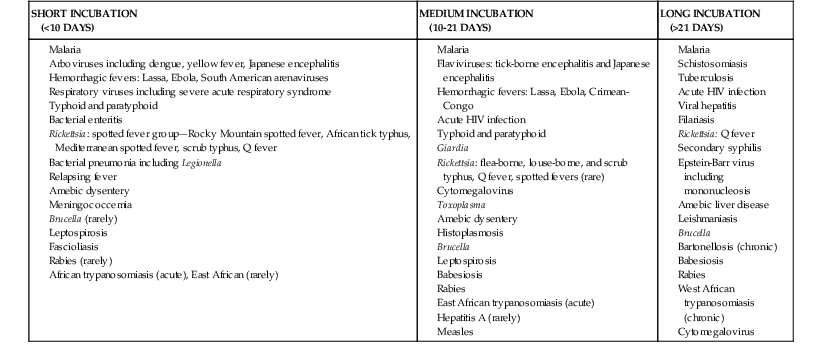
This should include the exact dates spent in each locale with respect to the onset of illness. Knowledge of typical incubation periods (Table 324-2) of possible infectious causes is a key tool in narrowing the differential diagnosis. Many agents are simply not biologically possible outside their usual incubation period. Arboviral diseases such as dengue uniformly have short incubation periods. Onset of illness more than 2 weeks after the last possible exposure effectively rules out this class of viral illness. None of the known hemorrhagic fever viruses has a possible incubation longer than 21 days. Long-incubation infections like schistosomiasis cannot present less than several weeks after first possible exposure. Some diseases such as malaria or enteric fever have more variable incubation periods but nevertheless have a typical incubation period during which time the majority of the patients present. A number of diseases, especially those that are arthropod borne, have a strict seasonality whereby transmission stops during either cold or dry weather. Examples would include malaria in nontropical countries such as Korea, Tajikistan, or northern China, as well as Lyme borreliosis or tick-borne encephalitis, all of which completely cease transmission during winter months. GeoSentinel surveillance data indicate that dengue cases in travelers show marked region-specific peaks for Southeast Asia (June, September), South-Central Asia (October), South America (March), and the Caribbean (August, October).11,12
TABLE 324-2
Incubation Periods of Common Travel-Related Infections*
| SHORT INCUBATION (<10 DAYS) | MEDIUM INCUBATION (10-21 DAYS) | LONG INCUBATION (>21 DAYS) |
* Diseases that commonly have variable incubation periods are shown more than once. However, most diseases may rarely have an atypical incubation period, and this is not shown here.
HIV, human immunodeficiency virus.
Exposures
A detailed dietary history is essential. Budget travel and associated high-risk eating habits predispose to a variety of common enteric pathogens. A history of specific foods associated with known pathogens also should be elicited. This includes unpasteurized dairy products (Brucella, Campylobacter, Salmonella, Mycobacterium tuberculosis), shellfish (vibrios, enteric viruses, viral hepatitis), uncooked beef such as carpaccio and steak tartare (Toxoplasma, Campylobacter, Escherichia coli O157-H7), undercooked fish such as sushi and ceviche (vibrios, Anisakis, Gnathostoma), and undercooked pork or game meat (trichinosis). Exposure to fresh water or surface water in recreational or other settings may be associated with schistosomiasis13–15 or leptospirosis.16,17 A history of exposure to mosquitoes and flies is generally unhelpful, but a history of tick bite (rickettsiae, relapsing fever, tick-borne encephalitis) or tsetse fly bite should be sought in the right setting. Exposures to new sexual partners,18,19,20 needles, or blood should be ascertained. Rodent exposure is associated with Lassa fever, hantavirus infection, murine typhus, and rat-bite fever. A history of contact with other sick people is especially important in the post-travel setting. Travelers usually move in groups or with families or companions, all of whom will likely have shared the same exposures.
Immunization History
The immunization history should include exact dates of the last dose of each vaccine received and in some instances whether an adequate primary series was completed in the first place. Most vaccines, with the notable exception of typhoid vaccines, are highly efficacious. Thus, hepatitis A or B, yellow fever, measles, and diphtheria are unlikely diagnoses in those travelers with a substantiated history of adequate and current immunization.
Antimalarial Prophylaxis or Treatment
If malaria is a possibility, a complete pill-by-pill history of ingestion of antimalarial drugs, including the name and dose of all drugs taken for prophylaxis or treatment, must be obtained. Patients often misunderstand the dosing or timing instructions given at the pretravel visit, or they may have been prescribed an inappropriate drug for their destination. Patients may have been treated with appropriate or inappropriate drugs en route for febrile illnesses. Some very efficacious drugs are not available in the United States, and an international pharmacopeia such as Martindale’s may need to be consulted by those unfamiliar with these drugs. A history of appropriate prophylaxis diminishes the possibility of malaria but does not eliminate the need for a malaria thick film, which may be preceded by a malaria rapid card test for any patient legitimately exposed to malaria.
Other Medications Ingested
Travelers who fall ill during travel often self-treat with antibiotics or see a local physician and are prescribed a broad-spectrum antibiotic. Again, an international pharmacopeia may need to be consulted. Recent ingestion of a 1-week course of a quinolone, tetracycline, or cephalosporin may alter the course of the illness or even affect the possibility of certain diagnoses. In particular, malaria may be suppressed by azithromycin, doxycycline, quinolones, or clindamycin.
Physical Examination
Common tropical infections often present as undifferentiated fever without focal findings. However, when a focal finding such as arthritis, meningitis, or pneumonia is present, the differential diagnosis can often be narrowed. Unfortunately, physical findings such as jaundice, hepatomegaly, splenomegaly, and lymphadenopathy occur at least a portion of the time in many of the most common travel-related infections and so are not specific enough to greatly narrow the differential diagnosis.5 Most imported febrile rash illnesses engender the same differential diagnosis as for nontravelers. However, arbovirus infections, typhoid, rickettsial illness, leptospirosis, measles, early stages of viral hemorrhagic fevers, relapsing fever, and acute African trypanosomiasis should always be kept in mind.
Considerations for the Common Travel-Related Febrile Illnesses
Malaria
Fever in a traveler returning from a malarious area is an emergency, and the instinctive performance of an immediate malaria smear will prevent unnecessary deaths. A malaria rapid diagnostic test is licensed in the United States for use in clinical laboratories and not at the point of care. The readout for this antigen detection test is Plasmodium falciparum, or non–P. falciparum, or mixed infection. Because it is highly specific, a positive test means immediate treatment is warranted.
The malaria rapid diagnostic test has excellent sensitivity for P. falciparum but may still miss low parasitemias so is not a definitive means to rule out malaria. The sensitivity is poor for non–P. falciparum malaria, which tends to have much lower parasitemias, but non–P. falciparum malaria is not usually life threatening.21 Thus, a negative test does not rule out malaria and must always be followed by a blood film. Malaria due to P. falciparum is easily treatable if diagnosed early but even with optimum treatment has a mortality rate of 20% or more if treatment is begun only after end-organ complications arise. Smears need to be repeated at least every 12 to 24 hours a minimum of three times to rule out malaria. Rapid deterioration can occur over a period of hours. Patients who are unreliable to care for themselves and have smear-negative results but a high index of suspicion for P. falciparum malaria may need to be admitted for inpatient observation.
Because malaria is overwhelmingly an African disease, with about 80% of all P. falciparum imported into developed countries originating there,22–25 suspicion of malaria is especially acute for Africa returnees. Beyond this, trends in the geographic origin of imported malaria cases do not always correlate well with regional transmission patterns because absolute numbers of cases from particular geographic areas may also mirror the intensity of travel to the affected region. Ethnic minority travelers returning home to visit friends and relatives in malarious areas have the highest risk for infection. Resources describing current country-specific malaria microepidemiology should be immediately accessible to those assessing tropical fevers. In general, malaria is a rural disease, but the cities of Africa and India are exceptions.
P. falciparum malaria in nonimmune travelers most commonly has an incubation period of 9 to 14 days, and 90% of cases occur within 1 month of last exposure. Non–P. falciparum malaria in travelers is only rarely life threatening but can present much later after arrival. Incubation periods are prolonged in those taking inadequate or incomplete chemoprophylaxis. Relapses of disease due to Plasmodium vivax or Plasmodium ovale may occur many months after travel in those whose initial attack was clinically silent because of suppressive chemoprophylaxis but in whom terminal prophylaxis with primaquine was not used (see Chapter 276).
The presenting signs and symptoms of imported malaria remain sufficiently protean so as to mimic a number of common tropical or nontropical conditions.22–25 No constellation of symptoms or signs differentiates P. falciparum from non–P. falciparum malaria. Classic periodic malarial fever is not a usual manifestation of imported malaria, although when fever does occur in discrete, repeated 48- or 72-hour cycles, the diagnosis is almost certain. The simian malaria parasite P. knowlesi,26 which is now known to commonly infect humans in Malaysia and throughout Southeast Asia, uniquely has a periodicity of 24 hours. Infected patients have high parasitemias (>1%) with a plasmodium that is morphologically almost identical to Plasmodium malariae. P. malariae typically has a parasitemia of less than 1%. Fever is absent at the exact time of the initial medical assessment in up to 40% of patients with malaria. Respiratory or gastrointestinal symptoms may be predominant. The presence of rash, lymphadenopathy, or leukocytosis indicates another diagnosis. Anemia is uncommon in travelers who present in the early days of their malarial illness. Thrombocytopenia occurs in over 50% and is a reliable if nonspecific indicator of malaria when present.
Many other serious infections are present in malarious areas. The search for malaria should not hamper the simultaneous workup for other pathogens in smear-negative patients. Similarly, semi-immune residents of endemic areas may be mildly parasitemic on a chronic basis with little ill effect, so a positive malaria smear in these patients should not hamper a search for any other clinically suspected infections.
Dengue
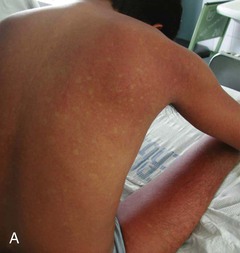
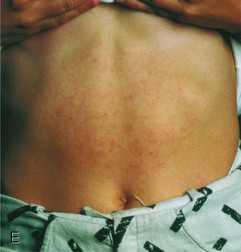
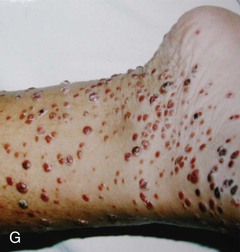
Dengue, transmitted by the day-biting Aedes aegypti mosquito, is an important travel-related problem most notably in heavily visited areas of Southeast Asia, the South Pacific, and Central America and the Caribbean.11,12 Travelers to Thailand seem particularly prone to infection, and dengue is relatively uncommon in Africa but has begun to emerge there. In contrast to many other tropical fevers, it is predominantly an urban infection so that it can even affect upscale business travelers in urban centers. The incubation period is usually 2 to 7 days after the mosquito bite, so many travelers initially become ill while still overseas. The clinical spectrum ranges widely from asymptomatic through a range of clinical manifestations up to the severe myalgia and arthralgia of “breakbone fever” (see Chapter 155). Malaria, other arbovirus diseases including chikungunya fever, leptospirosis, rickettsial disease, measles, or typhoid may present as similar initial findings. However, in cases in which one of several associated rashes manifests (Fig. 324-1A), dengue or chikungunya becomes more likely than the other possibilities.