FREE-LIVING AND PARASITIC AMEBIC INFECTIONS
SHANNON MOONAH AND WILLIAM A. PETRI, JR.
Central nervous system (CNS) infection with free-living and parasitic amebae is rare but life threatening. Three separate clinical syndromes occur from infection with free-living amebae (1). Primary amebic meningoencephalitis is due to infection with Naegleria fowleri and occurs almost exclusively in previously healthy children and young adults following exposure to freshwater. Pathologic examination of brain tissues is consistent with infection via direct invasion of the brain from the olfactory nerves (2–11) (Table 45.1). Granulomatous amebic encephalitis is due to hematogenous or direct infection of the brain with Acanthamoeba species (including Acanthamoeba castellani, Acanthamoeba polyphaga, Acanthamoeba culbertsoni, Acanthamoeba palestinensis, Acanthamoeba astronyxis, Acanthamoeba hatchetti, and Acanthamoeba rhysodes) (12,13) or Balamuthia mandrillaris (formerly misidentified as leptomyxid amebae) (14–27). Although granulomatous amebic encephalitis due to Acanthamoeba has been observed predominantly in patients with known immunodeficiencies, B. mandrillaris infection has also been observed in nonimmunocompromised individuals (Table 45.2). Acanthamoeba keratitis is seen predominantly in individuals who wear contact lenses and is not discussed further in this chapter (28,29). Brain abscess is a rare complication of Entamoeba histolytica infection, an otherwise common infectious disease in the developing world. E. histolytica brain abscess is most commonly seen in association with E. histolytica hepatic and/or lung abscess (30–40) (Table 45.3). As opposed to infection with the parasitic ameba E. histolytica, infection with free-living amebae is not linked to fecally contaminated food or water and is therefore no more common in developing nations than in the industrialized West. Encephalitis may also be caused by ameba belonging to the genus Sappinia. Only one case of amebic encephalitis due to Sappinia pedata infection has been reported (41,42).



ORGANISMS AND EPIDEMIOLOGY
N. fowleri is the only recognized pathogenic species of Naegleria. Trophozoites of N. fowleri are 10 to 30 µm in diameter and the nucleus has a prominent central dense nucleolus (Fig. 45.1A and B). The trophozoites can transform into a flagellated form (seen in vitro upon transfer to distilled water). The cyst form of the parasite is an approximately 9-µm diameter sphere with two pores in the cyst wall that serve as an egress point for the trophozoite upon excystation. N. fowleri have been isolated from freshwater lake and river water and soil in all parts of the world (3). Warm temperatures appear to favor their growth; for example, in Florida, it is not uncommon to isolate N. fowleri from freshwater lakes and streams. Most cases of N. fowleri infection have occurred in the summertime, and in the winter, the organism is identified in lake and river sediments. The cyst form of the parasite is stable for months in the environment but is not found in human tissues. It is estimated that in Florida, there are more than 100 million exposures to the parasite for every case of primary amebic meningoencephalitis (3). In fact most adults in the United States appear to have been exposed to the parasite as evidenced by the presence of serum antibodies against N. fowleri. The factors leading to N. fowleri infection in only a tiny minority of exposed individuals is unknown. Primary amebic meningoencephalitis has been reported in the central and southern United States, Central America, Europe, Japan, Africa, Australia, and New Zealand. Outbreaks have been linked to common environmental exposures (2,6,10).
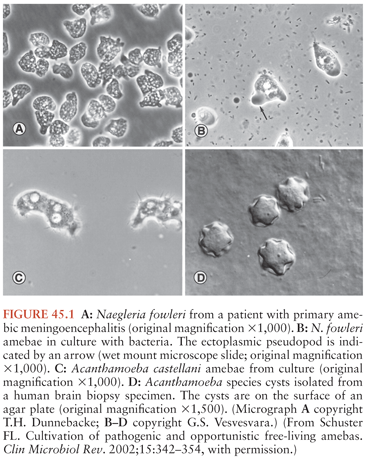
Acanthamoeba species consist of cyst and trophozoite stages, with the trophozoites approximately 14 to 40 µm in diameter and cysts with diameters of 12 to 16 µm (Fig. 45.1C and D). Both cysts and trophozoites can be observed in infected brain. The nucleus has a central nucleolus similar in appearance to that of Naegleria. Acanthamoeba species have also been isolated from soil, water, and air from diverse geographic locations (1,12,13). In contrast to N. fowleri, Acanthamoeba may be spread through the air. For example, 2% of nasopharyngeal swabs from healthy individuals were culture positive for Acanthamoeba species in one study. Again, it is common to find antibodies against Acanthamoeba in healthy individuals, although almost all cases of granulomatous amebic encephalitis due to Acanthamoeba species have been observed in immunocompromised individuals (1,12,13). The cause of immunosuppression has included HIV/AIDS, renal and bone marrow transplantation, cancer chemotherapy, steroid therapy, diabetes mellitus, and liver disease.
B. mandrillaris trophozoites have an average size of 30 µm with cysts of 15 µm and therefore can be distinguished from Acanthamoeba only by hematoxylin-eosin stains using specific antisera (1,20,26). It is an inhabitant of soil and freshwater with the highest concentration of organisms observed in freshwater in the spring and fall. Unique to B. mandrillaris is that it can cause CNS infection in both healthy and immunocompromised individuals (14–17,19–21,25,26).
E. histolytica trophozoites have an average diameter of 15 to 30 µm, and the 10-µm quadrinucleate cysts of the parasite have never been found in brain. E. histolytica infection is estimated to occur in hundreds of millions of people annually (30,31). With the use of modern diagnostic tests, the burden of amebic disease is becoming better appreciated. In Dhaka, Bangladesh, preschool-aged children had a 2.2% frequency of amebic dysentery during 3 years of prospective community observation (32). In Hue City, Vietnam, an incidence of amebic liver abscess of 21 cases per 100,000 inhabitants was observed (33). In the United States where fecal-oral transmission is unusual, amebiasis is most commonly seen in immigrants from and travelers to developing countries. Brain abscess is seen usually in association with an amebic liver or lung abscess (32–40). Amebic liver abscess in turn is predominantly (but not exclusively) a disease of young men (30,31), leaving one to have the highest suspicion for an E. histolytica brain abscess in a male patient with hepatic abscess, risk factors of E. histolytica infection, and CNS signs or symptoms.
PATHOLOGY
N. fowleri appears to enter the CNS after disrupting the olfactory mucosa. It produces a diffuse meningoencephalitis with purulent leptomeningitis. Cortical edema and hemorrhage with cerebellar or uncal herniation are observed. The olfactory bulbs are commonly hemorrhagic or necrotic and trophozoites can be observed in the olfactory nerves and in the perivascular spaces and adventitia of arterioles and midsize arteries. A neutrophilic myocarditis has been observed in some cases (2,5,6,8). Trophozoites of N. fowleri can be observed in wet preparations of cerebrospinal fluid (CSF) but are commonly misidentified as monocytes.
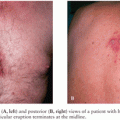
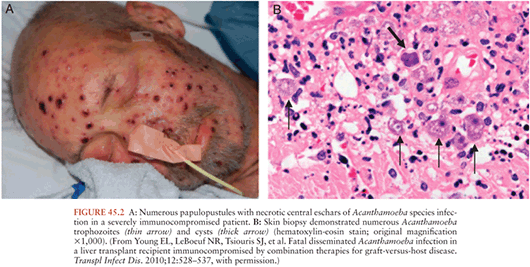
Granulomatous amebic encephalitis due to Acanthamoeba species usually presents with focal neurologic deficits in immunocompromised individuals. Cerebral edema resulting in bilateral cerebellar or uncal herniation occasionally occurs. Areas of cortical involvement include the cerebellum, midbrain, and brainstem (12,13). Histologically, the lesions spare the leptomeninges and contain necrotizing granulomas with amebic cysts and trophozoites. Multinucleated giant cells may be seen within the granulomas. Occasionally in immunodeficient individuals, the granulomatous reaction may not be present. The parasites are observed most often in a perivascular location. Identification of Acanthamoeba in the skin (Fig. 45.2), lung, adrenals, and lymph nodes of patients with CNS infection suggests hematogenous dissemination of infection to the brain.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree