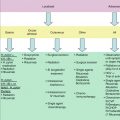
Fig. 7.1
(a) Extranodal NK/T-cell lymphoma of the nasal cavity. Small- to medium-sized tumor cells are intermingled with plasma cells. (b) EBER in situ hybridization revealed positive signal in the nuclei of tumor cells. (c) Tumor cells express cytoplasmic CD3. (d) Tumor cells express CD56
In the skin, the tumor presents as large ulcerating lesions or multiple erythematous skin rashes. Tumor cells infiltrate throughout the skin, including subcutaneous adipose tissue. Diffuse necrosis of the skin with ulceration of the epidermis is common (Kost et al. 2009; Chia et al. 2009). Pyogenic granuloma-like lesions have also been reported (Fernandez-Torres et al. 2009).
The immunophenotype of typical ENKL is positive for CD2, CD56, and cytoplasmic CD3ε and negative for surface CD3 (Fig. 7.1). Typical ENKL is also positive for cytotoxic granules including granzyme B, perforin, and T-cell–restricted intracellular antigen. Occasionally, ENKL is also positive for CD30 and CD7. Some cases are CD56 negative. Cases that are CD56 negative and cytoplasmic CD3ε positive are also classified as ENKL if they are positive for both cytotoxic granules and EBER. However, cases that are CD56 negative and cytoplasmic CD3ε positive but are negative for EBV and cytotoxic molecules should be classified as peripheral T-cell lymphoma, unspecified (Au et al. 2009).
7.4 Clinical Presentation
The most typical clinical presentation of ENKL is ulceration and destruction because of the characteristic angioinvasion and necrosis. The median onset age is the mid-40s to 50 years. This disease is quite uncommon in children and adolescents. There is a male predominance, especially nasal cases (Au et al. 2009; Lee et al. 2005a, b, 2006; Suzuki et al. 2010). Because around 80 % of cases occur in the upper airway tract, common symptoms are nasal obstruction, ulceration in the nasal cavity and palate, hemorrhage, and discharge. Severe destruction of the hard palate can cause midline perforation, which is why it was previously called “lethal midline granuloma” (Fig. 7.2a, b). Extranasal disease occurs in the skin, gastrointestinal tract, testis, and other organs (Au et al. 2009; Lee et al. 2005a, b; Suzuki et al. 2010). In cutaneous ENKL, the most typical lesion is nonhealing ulceration (Fig. 7.2c). Gut perforation, bleeding, and acute abdomen are common symptoms in cases of gastrointestinal tract involvement. The lesions are characterized endoscopically by erosion, ulceration, or ulceroinfiltrative lesions without mass formation (Fig. 7.2d) (Kim et al. 2007). Interestingly, there are clinical differences between nasal and extranasal ENKL even though they share the same histological features. Patients with extranasal ENKL have more adverse clinical features including advanced stage, elevated lactate dehydrogenase (LDH) level, presence of B symptoms, and poor performance status (Au et al. 2009; Lee et al. 2005a; Suzuki et al. 2010).
Hepatic dysfunction caused by cytokines and/or hepatosplenic involvement can occur even in clinically stage I or II disease. Hemophagocytosis presenting as fulminant multiorgan failure with elevated LDH level, fever, and pancytopenia can occur; this is sometimes misdiagnosed as sepsis due to secondary infection from the ulcerative lesions.
7.5 Staging and Prognostic Factors
No specific staging system for ENKL has been proposed. Ann Arbor staging is usually applied even though it is not satisfactory. For initial assessment, a complete history and physical examination are necessary (Kwong et al. 2009). Recent studies show the prognostic importance of local tumor invasiveness and regional lymph node involvement (Lee et al. 2006; Suzuki et al. 2010; Kim et al. 2005). The use of appropriate computed tomography or magnetic resonance imaging may provide good information about the extent of the disease. The role of positron emission tomography (PET) remains controversial. ENKL lesions are generally fluorodeoxyglucose avid. Therefore, it seems to be helpful to detect other systemic lesions before treatment. The role of posttreatment PET requires further studies (Kako et al. 2007; Fujiwara et al. 2011; Khong et al. 2008). To detect bone marrow involvement accurately, EBER in situ hybridization should be performed routinely in all patients with ENKL (Kwong et al. 2009; Lee et al. 2007). CNS involvement has been a concern for a long time because this lymphoma usually involves the nasal and paranasal sinuses. A recent report showed CNS involvement in <6 % of cases. Among patients at low risk (groups I and II by the KPI), CNS involvement was noted in <2 % of cases. Therefore, routine CNS evaluation is not recommended, although the role of CNS evaluation has not been determined for patients in KPI groups III and IV (Kim et al. 2010).
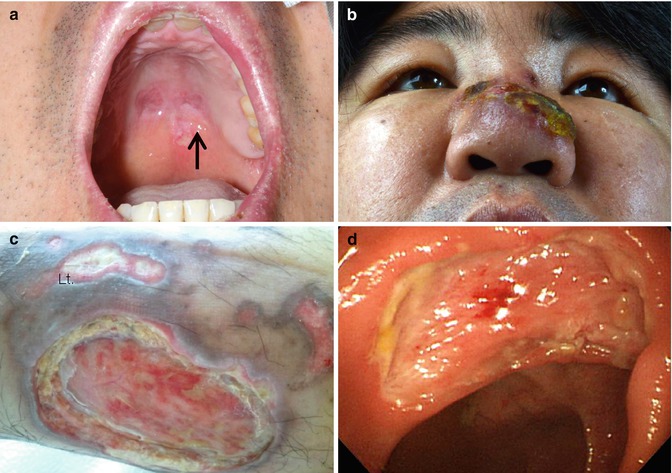

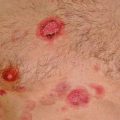
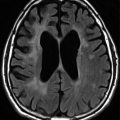
Fig. 7.2
(a, b) Nasal ENKL presented as palatal ulcer and perinasal skin infiltration. (c) Skin involvement as multiple nonhealing ulceration. (d) Intestine involvement as large ulceration. This lesion can easily bleed
EBV DNA can be released from the apoptotic tumor cells. Serial monitoring of circulating EBV DNA can be a valuable marker of tumor burden and disease control. The test is recommended for staging, if quantitative Polymerase chain reaction (PCR) is available (Au et al. 2004; Kim et al. 2009a; Suzuki et al. 2011).
The IPI has good power for predicting the prognosis. However, most patients are classified into low- and low-intermediate-risk groups by IPI even though they have a poor prognosis. Besides the IPI, the following parameters including local tumor invasiveness, extra-upper aerodigestive tract origin, high proliferation index of the tumor tissue, and quantification of circulating EBV DNA seem to be prognostically relevant for individual patients (Kim et al. 2002, 2005, 2009a; Au et al. 2004; Suzuki et al. 2011). Therefore, other prognostic models have been proposed (Lee et al. 2006; Suzuki et al. 2010). Currently, the KPI, which is based on B symptoms, stage, LDH level, and regional lymph node involvement, has the strongest predictive power (Fig. 7.3) (Au et al. 2009; Lee et al. 2006).
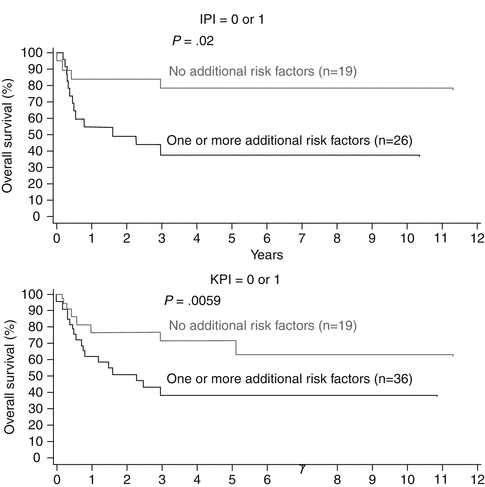

Fig. 7.3
Prognostic models (a) IPI form DLBCL: age/stage/extranodal/LDH/PS. (b) KPI: B symptom/stage/LDH/lymph node
7.6 Treatment
Anthracycline-based chemotherapies such as CHOP or CHOP-like regimens are not efficient because of high expression of P-glycoprotein. Instead, drugs that are not affected by P-glycoprotein are recommended as a part of a polychemotherapy regimen. l-asparaginase-containing chemotherapies have produced promising outcomes recently. In cases of localized disease, the addition of radiation is important.
7.6.1 Localized Disease
The upfront use of CHOP or CHOP-like regimens produces only a 35–44 % complete response (CR) rate (Kim et al. 2001a, 2003, 2006a; Wang et al. 2007). Frequent early disease progression during chemotherapy has been a problem. This lack of efficacy of anthracycline-based chemotherapy is associated with the high expression of P-glycoprotein (Yamaguchi et al. 1995).
The outcome is better for upfront use of radiotherapy than anthracycline-based chemotherapy (Kim et al. 2000; Chim et al. 2004; You et al. 2004). The largest series with radiotherapy alone produced a much higher CR rate (69 %) compared with previously reported CR rates (< 60%) of frontline chemotherapy (Kim et al. 2001b), and the frequency of early disease progression during radiotherapy is also lower than that of chemotherapy (22.2 % vs. 50.8 %) (Cheung et al. 2002). Therefore, radiotherapy is recommended as an initial treatment for localized ENKL (Suzuki et al. 2008; Kim and Kim 2010). However, there is no consensus on the radiation dose. Some studies reported that at least 52 or 54 Gy was required to obtain in-field control in patients with localized ENKL (Sakata et al. 2006; Huang et al. 2008), whereas a study of concurrent chemoradiotherapy showed that a median 40 Gy was enough to control the disease (Kim et al. 2009b). Considering the significant radiation-related toxicity, the adequate radiation dose should be defined more accurately.
Recent phase II trials demonstrated very impressive outcomes with concurrent chemoradiation (Table 7.1) (Kim et al. 2009b; Yamaguchi et al. 2009). In both trials, the response CR rates were >80 %, resulting in ~80 % long-term survival rates. Therefore, concurrent chemoradiation followed by nonanthracycline-based chemotherapy is now recommended as the treatment for localized ENKL.
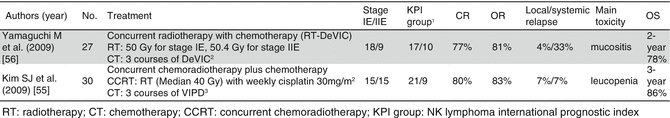
Table 7.1
Prospective trials with concurrent chemoradiotherapy for stage I/IIE ENKL

7.6.2 Advanced and Relapse/Refractory Disease
As mentioned above, anthracycline-based chemotherapy such as CHOP only yielded a response CR rate of <20 % (Lee et al. 2005b; Suzuki et al. 2010). Instead, l-asparaginase has been suggested for the treatment of ENKL because NK/T-lymphoma cells are unable to synthesize l-asparagine (Ando et al. 2005). Therefore, agents that are not affected by P-glycoprotein, such as methotrexate and ifosfamide, are recommended as part of polychemotherapy. A recent phase II study of patients with stage IV or relapsed/refractory ENKL demonstrated the efficacy and feasibility of SMILE (steroid, methotrexate, ifosfamide, l-asparaginase, and etoposide) chemotherapy (Yamaguchi et al. 2011). The overall response and CR rates after two cycles of SMILE were 79 and 45 %, respectively. Thus, SMILE chemotherapy can be recommended for both advanced disease and relapsed/refractory disease. l-Asparaginase-based regimens showed outstanding response rates of >80 % in patients with refractory and relapsed ENKL (Yong et al. 2008; Jaccard et al. 2008). Based on the results of these studies, current nonanthracycline-based intensive chemotherapy regimens including l-asparaginase can be recommended as frontline treatment.
7.6.3 Hematopoietic Stem Cell Transplantation (SCT)
High-dose chemotherapy followed by autologous SCT may be considered as a consolidation treatment for patients with a high risk of relapse. Initial experience suggested that patients with a poor prognosis should be considered for autologous SCT at the time of the first CR (Au et al. 2003; Kim et al. 2006b). A recent multinational, matched-control study of 47 patients who underwent autologous SCT suggested that autologous SCT could confer a survival benefit in patients who attained a CR with high-risk scores of KPI at diagnosis (Lee et al. 2008).
Allogeneic SCT may be another appealing treatment option for patients with advanced disease or relapsed/refractory disease. The largest series of 28 patients showed a 2-year overall survival rate of 40 % (Shustov et al. 2010). However, treatment-related mortality may present a hurdle to the application of allogeneic SCT for ENKL. Considering the limited data and heterogeneity of information, a firm recommendation cannot be made for allogeneic SCT in ENKL (Kwong 2010).
7.6.4 Prophylaxis Against Secondary CNS Involvement
Because ENKL frequently affects the nasal cavity and the paranasal area near the CNS, ENKL may have a risk of CNS involvement. Previous studies report a variable incidence of CNS involvement in 0–6 % of ENKL cases (Cheung et al. 1998; Cuadra-Garcia et al. 1999; Kim et al. 2004). Thus, it is unclear whether CNS prophylaxis should be included in the treatment of ENKL. A recent large retrospective analysis reported that 5.76 % (12/208) of cases had CNS involvement and that CNS involvement was associated with the KPI score (Kim et al. 2010). Therefore, routine CNS evaluation and prophylaxis is not necessary in patients at low risk of KPI. For high-risk patients with KPI, the role of CNS prophylaxis has not been determined yet.
References
Au WY, Weisenburger DD, Intragumtornchai T, Nakamura S, Kim WS, Sng I, Vose J, Armitage JO, Liang R (2009) Clinical differences between nasal and extranasal natural killer/T-cell lymphoma: a study of 136 cases from the International Peripheral T-Cell Lymphoma Project. Blood 113:3931–3937PubMedCrossRef
Aviles A, Diaz NR, Neri N, Cleto S, Talavera A (2000) Angiocentric nasal T/natural killer cell lymphoma: a single centre study of prognostic factors in 108 patients. Clin Lab Haematol 22:215–220PubMedCrossRef
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree