Introduction
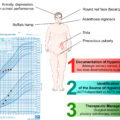
Cushing’s disease (CD) is the most common form of endogenous hypercortisolism and is characterized by an adrenocorticotropic hormone (ACTH)-secreting pituitary adenoma, with corticotroph tumors representing up to 15% of pituitary neoplasms [ ]. In terms of incidence, a nationwide Swedish study reported 1.6 cases per million persons between 1987 and 2003 and up to 2.0 cases per million between 2005 and 2013 [ ], while an even higher incidence of 6.2 and 7.6 cases per million persons per year in 2009 and 2010, respectively, was obtained from health insurance databases in the USA [ ]. The condition also has a significant female predominance with a 4:1 ratio.
Despite significant progress in terms of accuracy of radiological and biochemical testing, diagnosis of CD is still a challenging process, encompassing baseline and dynamic testing, invasive procedures, and different imaging techniques. Treatment strategies for CD are multifaceted and may include surgical, medical, and, in some cases, radiotherapeutic approaches, however, optimal management requires individualized, multidisciplinary care.
Differential diagnosis of Cushing’s syndrome
In the clinical suspect of hypercortisolism, several steps need to be taken. The diagnosis requires a stepwise approach, aimed at excluding exogenous Cushing’s syndrome (CS), confirming autonomous cortisol secretion, followed by investigating its origin. While the diagnosis of endogenous CS after exclusion of exogenous (Glucocorticoid) GC use has been previously described, to summarize, three tests are used:
-
1.
24-hour urinary free cortisol (UFC, average of 2–3 days collection): This test measures the amount of cortisol excreted in the urine over a 24-hour period. Elevated levels typically indicate hypercortisolism. It’s crucial to ensure the collection is complete and accurate, as improper collection can lead to false results. Contraindications to the test are mainly kidney-related, as cortisol excretion and metabolism are altered in renal failure. Overweight or obesity can also yield falsely positive results.
-
2.
Late-night salivary cortisol (LNSC, two or more tests): Thanks to the physiological midnight nadir of cortisol secretion, measuring cortisol in a salivary sample collected late at night (typically around 11–12:00 p.m.) can reveal autonomous secretion in case of nonsuppressed levels. This test is noninvasive and can be conveniently done at home. Night-shift work or other circadian-disrupting conditions can alter the results.
-
3.
Overnight dexamethasone suppression test (DST) (Nugent’s test): This test takes advantage of the HPA (hypothalamic-pituitary-adrenal) axis suppression by exogenous GCs. Patients take 1 mg of dexamethasone at 11:00 p.m., and a serum cortisol level is measured at 8:00 a.m. the next morning. Normally, dexamethasone suppresses cortisol production, but in CS, cortisol levels remain elevated above a 1.8 mcg/dL–50 nmol/L threshold. Contraindications are conditions that induce altered levels of cortisol-binding globulin (CBG), such as pregnancy or liver disease, or use of medications altering dexamethasone metabolism, such as CYP3A4 inductors, or malabsorption. False negatives can result from renal protein loss or inhibition of dexamethasone metabolism.
Other tests are serum midnight cortisol measurement, the low-dose 2-day dexamethasone test (LDDT) (Liddle’s test), and administering 0.5 mg of dexamethasone every 6 hours for 48 hours, followed by cortisol measurements.
To investigate the origin of CS, the first step is to measure ACTH levels. ACTH-dependent CS is characterized by normal or elevated ACTH levels, while in adrenal CS, pituitary suppression due to hypercortisolism leads to low levels of ACTH. Once the ACTH dependency is confirmed, further steps will be taken and will be discussed in the following section.
Investigating ACTH-dependent Cushing’s syndrome
Distinguishing CD from ectopic ACTH secretion (EAS) from a nonpituitary neoplasm and nonneoplastic hypercortisolism (pseudo-CS) poses a significant diagnostic challenge. In fact, all three conditions are characterized by signs and symptoms of cortisol excess and share biochemical and clinical features.
Nonneoplastic hypercortisolism : Several conditions, such as psychiatric disorders, polycystic ovary syndrome (PCOS), obesity, and alcohol use disorder, can chronically activate the HPA axis [ ]. Underlying mechanisms include alterations of the physiological cortisol rhythm, altered stress response, and hyperactive morning cortisol surge in neuropsychiatric disorders [ ]; reduced CBG and sex hormone binding globulin (SHBG) levels in PCOS; and increased corticotropin-releasing hormone (CRH) levels paired with decreased hepatic clearance of cortisol in alcohol use disorder.
Accordingly, DST, LDDT, and UFC results can be altered in all these conditions, and further testing is necessary. Two different dynamic tests are used to differentiate nonneoplastic and neoplastic hypercortisolism: the paired LDDT–CRH test (Dex-CRH) and the desmopressin test.
The first is based on the measurement of CRH-stimulated cortisol after 2 days of dexamethasone suppression, based on the observed increased response to CRH in corticotroph adenomas. It was first introduced in 1993 [ ], but further studies did not entirely confirm the reliability of its performance in identifying neoplastic hypercortisolism, even though the new cutoff of 15-minute cortisol levels has shown good results when compared to other tests [ ]. Variability in CRH formulation, cortisol and ACTH immunoassays, and characteristics of patients could contribute to the heterogeneity of results from different studies [ ].
The second consists of measuring ACTH and cortisol levels after desmopressin infusion, since due to the presence of hyperexpression of V2 and V1b (V3) vasopressin receptors on corticotroph adenomas, a marked increase in ACTH release can be stimulated by desmopressin [ , ].
Even though both tests have been evaluated independently with good specificity for CD, concordance between the two gives the best results in terms of avoiding false positives and detecting neoplastic hypercortisolism [ ].
Ectopic ACTH secretion : Acute and severe presentation of biochemical and clinical signs of CS is usually characteristic of EAS, but CD can be characterized by severe hypercortisolism as well, and the two conditions often have overlapping features. In the presence of life-threatening hypercortisolism, rapid diagnosis and treatment of complications are advised by guidelines [ ], with 2–3 ACTH determinations and UFC as the mainstays of a first-line evaluation. Severe hypokalemia, accelerated progression, and catabolic signs are usually suggestive for EAS rather than CD. UFC of more than 5-fold the upper limit of normality is also suggestive for an ectopic secretion. However, dynamic tests are often necessary [ ]. Most, but not all, nonpituitary neuroendocrine ACTH-secreting tumors lose CRH and vasopressin receptors, along with sensitivity to exogenous glucocorticoids. This is why CRH and desmopressin tests, along with high-dose DST, are used as differential diagnostic tools between CD and EAS. It is worth mentioning that well-differentiated neuroendocrine tumors (NETs) can show similar response to corticotroph adenomas.
Sensitivity and specificity of the CRH test for EAS across different studies were analyzed in a recent consensus [ ] with values ranging from 59% to 93% and from 70% to 100%, respectively. The desmopressin test has also been used in EAS, even though an aberrant expression of V3 receptors has been described, and false positives have been reported [ ]. Concordance between the tests seems to ultimately achieve the best performance.
On the other hand, the HDDST (High Dose Dexamethasone Suppression Test) seems to be losing ground, since its reliability has been challenged in several case series and cohorts, especially in well-differentiated NETs. A recent large retrospective study has confirmed the outperformance of the CRH test compared to HDDT [ ]. Interestingly, proposed cutoff values for CD diagnosis—a ≥35% ACTH and ≥20% cortisol increase for the CRH test and a ≥50% cortisol suppression for the HDDT test—showed sensitivity and specificity values ≤90%. Increasing thresholds to 40% for ACTH and 22% for cortisol in the CRH test and to ≥69% for the HDDT achieved the best results based on combined sensitivity and specificity. Lastly, the authors compared the diagnostic power of combined CRH and HDDT tests; however, the performance depended on whether a negative test—in case of discordant results—was interpreted as positive or negative. For example, if a positive CRH test and a negative HDDT test were considered compatible with the diagnosis of CD, the performance of the combined tests was similar to the CRH alone. On the other hand, interpreting any negative result in combined testing as suggestive for EAS increased specificity and positive predictive value, and was therefore the preferred approach. A “cautionary” approach, combined with the increased thresholds, achieved a 99% specificity in detecting EAS.
Invasive testing in Cushing’s disease: advantages and limitations
The differential diagnosis of ACTH-dependent CS remains a challenge, requiring a combination of clinical evaluation, biochemical testing, imaging, and, in selected cases, invasive procedures. Despite the progress in noninvasive diagnostic modalities, imaging and standard biochemical tests sometimes fail to provide definitive localization of the source of excess ACTH production. In such cases, bilateral inferior petrosal sinus sampling (BIPSS) represents the gold standard for distinguishing between CD and EAS [ ]. This procedure, which involves the catheterization of the inferior petrosal sinuses and measurement of ACTH levels with or without stimulation with CRH or desmopressin, offers high diagnostic accuracy. However, its invasive nature, potential risks, and limited availability require a careful assessment of its indications and limitations.
BIPSS remains the most reliable diagnostic tool when conventional tests fail to provide a clear diagnosis. In patients with ACTH-dependent hypercortisolism, distinguishing between pituitary and ectopic sources is essential to determine the appropriate therapeutic approach. Imaging techniques such as magnetic resonance imaging (MRI) frequently lack sensitivity, with studies showing that up to 50% of ACTH-secreting pituitary adenomas are not detectable on standard scans [ ]. Given that pituitary microadenomas may be very small or indistinguishable from incidental findings, BIPSS provides a direct assessment of ACTH secretion from the pituitary, helping to confirm or rule out CD. Several studies have demonstrated the high sensitivity and specificity of BIPSS in differentiating CD from EAS. The test relies on the measurement of an ACTH gradient between the inferior petrosal sinus and peripheral blood, and conventional ratios were ≥2 at baseline and ≥3 after CRH stimulation [ ]. A relatively recent systematic review and meta-analysis of over 1600 patients confirmed the high sensitivity and specificity of the test [ ], while a recent retrospective study has proposed new cutoffs of ≥1.9 at baseline and ≥2.1 5 minutes after CRH stimulation [ ], even though benefits on overall specificity were minor. In cases where imaging fails to identify an adenoma, BIPSS can be invaluable in confirming the presence of a corticotroph tumor.
Another important advantage of BIPSS is its role in preventing unnecessary pituitary surgery. Misdiagnosis may lead to incorrect transsphenoidal surgery (TSS); establishing a definitive diagnosis through BIPSS can prevent patients from undergoing an ineffective and invasive procedure. Furthermore, in patients with suspected EAS but without an identifiable tumor on imaging, BIPSS can provide clarity and direct further investigative efforts. Despite its advantages, BIPSS is an invasive procedure that carries risks and limitations. First and foremost, it requires highly specialized expertise, as successful catheterization of the inferior petrosal sinuses is technically demanding. This factor limits the availability of the test, as only centers with experienced interventional radiology teams can perform the procedure with high accuracy and minimal complications [ ].
In terms of procedural risks, BIPSS is associated with vascular complications, including thrombosis, hemorrhage, and, in rare cases, stroke. Catheterization of the venous system carries an inherent risk of thrombotic events, particularly in patients with hypercoagulability due to chronic hypercortisolism. Though uncommon, cases of dural sinus thrombosis following BIPSS have been reported, emphasizing the need for meticulous patient selection and postprocedural monitoring [ ].
Another important limitation of BIPSS is the potential for false-negative and false-positive results. False negatives may occur if catheterization is unsuccessful or if ACTH secretion is episodic, as seen in cyclic CD, where fluctuations in hormone levels may affect diagnostic results [ ]. Similarly, false positives can arise in cases where ectopic tumors retain partial CRH receptor expression, leading to an exaggerated response to CRH stimulation and an erroneous diagnosis of CD [ ]. These limitations highlight the necessity of correlating BIPSS findings with clinical presentation and other biochemical markers. Moreover, the cost and resource-intensive nature of BIPSS make it less accessible in many regions. The test requires the use of expensive CRH formulations, multiple ACTH assays, and specialized radiological expertise, factors that contribute to substantial financial and logistical burdens. Given the decline in availability of CRH in some countries, alternative stimulants such as desmopressin are increasingly employed [ ].
Given the complexity of diagnosing ACTH-dependent CS, the decision to perform BIPSS should be based on a comprehensive assessment of the patient’s clinical and biochemical profile. In cases where noninvasive tests provide conflicting results or where imaging fails to localize a pituitary adenoma, BIPSS remains an invaluable tool. However, its use should be limited to centers with appropriate expertise, and patients should be thoroughly informed of the potential risks and benefits. Recent studies have explored potential refinements to BIPSS, including the simultaneous measurement of prolactin levels, which has shown promise in improving diagnostic accuracy by confirming successful catheter placement [ ]. Additionally, novel noninvasive biomarkers and advances in molecular imaging may eventually reduce the reliance on invasive testing, though current methodologies remain insufficient to replace BIPSS in challenging diagnostic cases.
In conclusion, while BIPSS offers unparalleled accuracy in distinguishing between pituitary and ectopic ACTH sources, its invasive nature, potential complications, and limited availability necessitate careful patient selection. As research advances, efforts should focus on optimizing noninvasive alternatives while maintaining access to BIPSS for patients in whom it remains clinically essential.
Imaging modalities in the diagnosis of Cushing’s disease
The localization of ACTH-secreting pituitary adenomas in CD is crucial for determining appropriate treatment strategies. MRI remains the gold standard for identifying pituitary adenomas, but its sensitivity is variable, and alternative imaging techniques, including positron emission tomography (PET) and hybrid imaging methods, are increasingly being investigated.
MRI is the most widely used imaging technique for detecting pituitary adenomas in CD. The standard 1.5T MRI has a sensitivity of approximately 50% for detecting ACTH-secreting pituitary microadenomas, while 3T MRI improves this detection rate significantly [ ]. A systematic review reported an overall MRI sensitivity of 73.4%, with slightly lower sensitivity (70.6%) for detecting microadenomas, demonstrating that while MRI is useful, it fails to identify adenomas in a significant proportion of patients [ ].
Technical refinements, including thin-section acquisitions and the use of gadolinium contrast, particularly in dynamic sequences, enhance the ability to visualize adenomas. Fluid-attenuated inversion recovery (FLAIR) and newer 3D spoiled gradient-echo (SPGR) sequences may further improve sensitivity, though these techniques remain complementary to conventional spin-echo sequences [ ]. Additionally, ultra-high-field 7T MRI has been explored for its ability to improve lesion detection, but its availability is currently limited to research settings [ ]. Despite advances in MRI technology, a substantial proportion of ACTH-secreting adenomas remain undetected, leading to so-called MRI-negative CD. In such cases, additional imaging modalities are required to confirm the presence of a pituitary lesion or to explore EAS.
The role of PET imaging
Functional imaging with PET has been proposed as a complementary tool for identifying pituitary adenomas, particularly in cases where MRI is inconclusive. Studies have demonstrated that 18 F-fluorodeoxyglucose ( 18 F-FDG) PET/CT can localize corticotroph adenomas with a sensitivity comparable to that of MRI [ ]. However, prior stimulation with CRH has been shown to enhance 18 F-FDG uptake, thereby improving the detection of microadenomas [ ]. Another promising PET tracer, 11 C-methionine, offers a higher specificity for pituitary adenomas compared to 18 F-FDG, as it targets protein synthesis pathways that are upregulated in adenomatous cells [ ]. Hybrid imaging with PET/MRI has been explored as a way to combine the functional insights of PET with the anatomical precision of MRI. In a study involving patients with recurrent or persistent hypercortisolism, PET/MRI successfully localized adenomas in individuals with negative or equivocal MRI findings [ ].
Alternative and emerging imaging techniques
Several alternative imaging techniques have been evaluated for their role in localizing pituitary adenomas. Gallium-68 DOTATATE PET/CT, which detects somatostatin receptor (SSTR) expression, has been investigated for the identification of ectopic ACTH-secreting tumors but has limited utility in pituitary adenomas due to the low SSTR density in these lesions [ ]. Similarly, imaging targeting CRH receptors on corticotroph tumors has been proposed, though further studies are needed before this approach becomes widely available [ ]. Another innovation involves intraoperative MRI (iMRI), which allows for real-time imaging during TSS to confirm the extent of tumor resection. iMRI has been shown to improve gross total resection rates, thereby enhancing surgical outcomes [ ]. However, this technology is not yet widely available and remains limited to specialized centers.
Despite the improvements in imaging technology, certain challenges remain in the detection and localization of ACTH-secreting adenomas. First, the small size of these tumors and their potential for heterogeneous enhancement patterns make their identification difficult. Second, the presence of pituitary incidentalomas may confound interpretation, especially in cases of equivocal MRI findings [ ].
Looking forward, the development of molecular imaging techniques that specifically target corticotroph tumor receptors, along with machine learning-assisted image analysis, holds promise for improving diagnostic accuracy. Additionally, radiomics-based imaging approaches, which analyze quantitative features from imaging data, may provide novel insights into tumor characterization and localization [ ].
In conclusion, MRI remains the cornerstone of imaging in CD, with sensitivity enhanced by high-field scanners, contrast-enhanced sequences, and refined imaging protocols. However, MRI-negative cases necessitate the use of functional imaging modalities such as PET/CT and PET/MRI to improve localization. iMRI may further optimize surgical outcomes, and emerging molecular imaging strategies offer potential for future advancements. As technology continues to evolve, a multimodal approach that integrates anatomical and functional imaging will likely yield the highest diagnostic accuracy for CD.
Treatment
Surgery
The removal of the ACTH-secreting pituitary tumor via TSS is the first-line treatment for CD [ , ], except in cases where it’s not feasible or unlikely to substantially reduce hypercortisolism [ ].
Disease remission following TSS is typically defined as a serum cortisol level <55 nmol/L (<2 μg/dL). However, patients with mild hypercortisolism, cyclic CD, or those receiving preoperative medical therapy may achieve remission without marked postoperative hypocortisolism. The success rate of primary TSS varies widely (42%–97%) depending on tumor size and surgical expertise [ ]. Higher remission rates are generally observed in patients with microadenomas compared to macroadenomas (∼80% vs. ∼60%) [ ], with additional tumor characteristics, such as clear MRI detectability and absence of parasellar invasion, also being associated with better surgical outcomes [ , , ].
Although a recent meta-analysis has not detected a clear difference in surgical outcomes between the microscopic and endoscopic approach [ ], the implementation of an exoscopy-based imaging system to the microscopic approach has shown promise in reducing postoperative complications and improving remission rates in a recent retrospective series conducted on 388 surgically treated CD patients [ ]. Despite very low perioperative mortality (occurring in less than 1% of cases) [ , ], the most common surgical complications include new-onset hypopituitarism in approximately 10% of cases, as well as permanent diabetes insipidus, cerebrospinal fluid leaks, and venous thromboembolism, each occurring in <5% of cases, with complication rates being even lower with more experienced surgeons [ , ].
Patients in remission require glucocorticoid replacement until HPA axis recovery [ , ], which generally occurs within 2 years of successful surgery [ ]. Long-term monitoring for recurrence—defined as the reappearance of clinical and biochemical signs of hypercortisolism after initial remission—should begin following HPA axis recovery. It should be performed lifelong [ ] and continue annually or sooner if clinically indicated [ ]. Notably, the diagnosis of recurrence is often challenging due to it usually being a slow process, often with delayed clinical and biochemical manifestations: recent studies have estimated a 5-year recurrence rate of 10%–15%, increasing to 20%–25% at 10 years, or approximately 2% per year [ ]. Disease recurrence can indeed be detected via the same biochemical tests commonly used for initial CD diagnosis, albeit with reduced sensitivity and increased specificity in this context [ ]. LNSC is currently regarded as the earliest biochemical marker for detecting recurrence, exhibiting the highest sensitivity for detecting recurrence despite a relatively high interpatient variability [ ], followed by the 1 mg DST and UFC [ , ]. Annual LNSC assessment is therefore recommended after HPA axis recovery, although the choice of recurrence monitoring should be tailored to each patient’s abnormal test results at initial diagnosis [ ].
In cases of discordant clinical and biochemical findings, close monitoring with repeat testing and management of comorbidities is advised [ ]. To date, predictors of recurrence remain controversial. Despite being a defining criterion of surgical remission, low postoperative serum cortisol levels have not been consistently associated with long-term recurrence-free survival [ ], although some studies suggest an association with later recurrence [ , ]. Similarly, loss of response in postoperative dynamic testing (e.g., CRH stimulation or desmopressin testing, with or without dexamethasone) may indicate increased recurrence risk [ ]. For confirmed recurrence with a detectable tumor on MRI, repeat surgery should be considered [ , ]. Remission rates following repeat surgery widely vary across published studies (37%–88%), mainly due to the heterogeneity in remission criteria across studies [ , ]. Of note, some studies have reported a lower success rate [ ] and a higher incidence of surgical complications in reoperations [ , ], though this risk appears mitigated by surgical expertise. When recurrence occurs without a visible tumor on MRI, medical therapy targeting steroidogenesis can be an effective alternative for disease control. In select cases, repeat TSS may be considered [ ], particularly in high-volume centers with experienced surgeons, following evidence of a central ACTH gradient on IPSS or prior ACTH immunostaining on pathology [ , ].
Lastly, bilateral adrenalectomy is considered in cases of ACTH-dependent CS when the source of excess ACTH cannot be removed or in ACTH-independent CS with bilateral adrenal disease. While this approach offers immediate control of hypercortisolism, it also introduces the risk of Nelson syndrome and necessitates lifelong glucocorticoid and mineralocorticoid therapy [ ]. Some experts also propose unilateral adrenal surgery initially, but this method is associated with a high rate of hypercortisolism relapse [ , ].
Medical therapy
The role of medical therapy in treating patients with CS is increasingly recognized as significant, particularly for patients with persistent or recurrent hypercortisolism postsurgery or those who are not suitable candidates for surgery, or as a bridging treatment during the waiting period for radiation techniques to take effect [ ]. In certain instances, preoperative medical therapy is used to manage hypercortisolism, thereby improving the patient’s clinical status before surgery. Medical intervention is especially crucial for acutely ill patients, such as those suffering from severe infections, recent cardiovascular events, or acute psychosis. These patients typically require stabilization through medical means aimed at controlling hypercortisolism before they can safely undergo tumor-directed surgery. The spectrum of medical treatments for CD includes steroidogenesis inhibitors, agents targeting corticotropin-secreting tumors, and medications that inhibit the glucocorticoid receptor. Recently, the field has seen significant advancements with the development of novel pharmacological compounds. However, the absence of head-to-head studies means there is limited information available on the superiority of one drug over another.
Adrenal steroidogenesis inhibitors
Steroidogenesis inhibitors target the adrenal gland, blocking one or more enzymes involved in steroidogenesis, which leads to the production of aldosterone, cortisol, and testosterone. To date, ketoconazole, levoketoconazole, metyrapone, osilodrostat, mitotane, and etomidate are the available drugs used in clinical practice for all causes of CS.
Ketoconazole
Ketoconazole is a 50/50 racemic mixture of two enantiomers (2S,4R-ketoconazole and 2R,4S-ketoconazole) and blocks several steps of adrenocortical steroid biosynthesis through inhibition of cytochrome P450 enzymes, including the cholesterol side-chain cleavage complex, 17,20-lyase, 11β-hydroxylase, and 17β-hydroxylase, therefore inducing a decrease in glucocorticoid, mineralocorticoid, and adrenal androgen production and secretion [ ]. It is orally administered at dosages of 200–1200 mg/day in a twice- or thrice-daily administration schedule due to its relatively short half-life (3.3 hours) [ ], demonstrating a rapid onset of action, leading to a decrease in UFC in the first weeks of treatment in CD patients [ ]. Efficacy data for ketoconazole in treating hypercortisolism primarily derive from retrospective studies. At a median dosage of approximately 600 mg/day over 7.5–31 months, ketoconazole led to remission in 63%–71% of patients. This remission typically concurs with improvements in the clinical syndrome and comorbidities associated with CS [ , , ]. However, these results are influenced by considerable heterogeneity, including varying criteria for biochemical control (partial or complete), the timing of treatment initiation (pre- or postsurgery), the severity of hypercortisolism, and the dose of ketoconazole used. Higher remission rates are noted in patients who demonstrate a more favorable clinical response. An “escape phenomenon,” where patients relapse after an initial response, has been observed in 7.1%–22.7% of cases [ ]. Limited data suggest that ketoconazole has no significant impact on pituitary tumor shrinkage. Conversely, some studies have reported new tumor appearances in 13.1%–13.8% of patients [ ]. In terms of safety, the most common adverse events (AEs) include hepatotoxicity and increased liver enzymes, typically manifesting soon after treatment initiation or dose escalation. Other reported AEs are gastrointestinal disturbances, adrenal insufficiency, and skin rash, with some cases necessitating medication discontinuation due to intolerance [ ]. Notably, ketoconazole can reduce adrenal and gonadal androgen production, leading to hypogonadism; in men, gynecomastia has been reported. Drug interactions are another important consideration, as ketoconazole is a potent inhibitor of CYP3A4, which can affect the metabolism of many drugs. Inhibitors of CYP3A4 may elevate ketoconazole levels, while proton pump inhibitors could reduce its effectiveness. Lastly, although rare in clinical trials, ketoconazole is known to potentially affect the cardiac QT interval.
Metyrapone
Metyrapone, a potent inhibitor of 11β-hydroxylase and, to a lesser extent, 18-hydroxylase, is known for its rapid absorption and onset of action, generally achieving control of hypercortisolism within weeks of treatment initiation in patients with CD and CS. Metyrapone’s inhibition of cortisol synthesis typically results in a compensatory increase in ACTH levels, which can lead to overproduction of glucocorticoid and mineralocorticoid precursors, as well as adrenal androgens, causing typical AEs. It is administered orally in dosages ranging from 500 to 6000 mg/day, divided into multiple doses daily due to its short half-life of about 2 hours [ , ]. Similar to ketoconazole, the evidence supporting metyrapone’s efficacy mainly comes from retrospective studies [ , ] and the prospective PROMPT study (a prospective study to assess efficacy and safety of metyrapone in endogenous Cushing’s syndrome) [ , ]. A meta-analysis indicated a CS remission rate of 60%–75.9%, dropping to 48.5%–59.8% in sensitivity analysis for studies with a low risk of bias [ ], while preliminary data from the PROMPT study indicated that 47% of patients achieved remission from hypercortisolism after being treated with metyrapone over a 12-week period (median daily dose: 1500 mg/day), with an average time to normalize UFC levels of 34 days [ ]. Metyrapone treatment has also been shown to have a variable rate of escape, ranging from 0% to 18.7%, after an initial response [ ]. Treatment with metyrapone generally leads to improvements in both the clinical syndrome and its comorbidities, as supported by various studies [ , ]. In patients with CD, no significant effects on pituitary tumors have been observed [ ]. Regarding the safety profile, the most frequently reported AEs include gastrointestinal disturbances, nausea, hirsutism and/or acne in women, dizziness, and edema [ , ]. Additionally, AEs related to the increase in mineralocorticoid precursors, primarily hypertension and hypokalemia, have also been described [ ]. An interesting anecdotal case has been reported regarding the use of metyrapone in pregnant women with CS. There were no apparent fetal complications described, but maternal complications such as hypertension and preeclampsia were a concern [ ]. It is important to note, however, that metyrapone is not specifically licensed for use during pregnancy. Furthermore, when monitoring patients treated with metyrapone, it is crucial to use a cortisol assay that does not cross-react with 11-deoxycortisol. This ensures that any increase in plasma cortisol levels is not mistakenly interpreted, potentially leading to an erroneous increase in the drug dose [ ].
Mitotane
Mitotane acts as an adrenolytic agent, primarily inhibiting steroidogenesis at low doses. Its most significant effect is on 20,22-desmolase, but it may also impact other enzymes, such as 11β-hydroxylase and 18-hydroxylase. Although not widely used for treating hypercortisolism, mitotane has been reported to be effective in 72%–80% of patients [ ]. However, its use is limited by several factors: a slow onset of action (ranging from weeks to months), teratogenic and abortifacient properties, and potential for drug–drug interactions. The main AEs associated with mitotane include gastrointestinal disturbances, dizziness, cognitive changes, adrenal insufficiency, and elevated liver enzymes [ ].
Etomidate
Etomidate, an intravenously administered sedative, is a potent inhibitor of 11β-hydroxylase and, to a lesser extent, 20,22-desmolase. Its effectiveness in controlling hypercortisolism is notable, often achieving control within hours. Consequently, its administration requires meticulous monitoring for sedation and is typically limited to intensive care settings [ ]. Like metyrapone, etomidate’s inhibition of 11β-hydroxylase can lead to cross-reactivity with 11-deoxycortisol in cortisol immunoassays [ ].
Levoketoconazole
Levoketoconazole, the 2S,4R enantiomer of ketoconazole, has gained FDA approval for treating adults with CS when surgery is not an option or has been unsuccessful. It is favored over racemic ketoconazole for its enhanced efficacy and safety profile. Preclinical studies have shown that levoketoconazole more potently inhibits CYP11B1, CYP17, and CYP21 than ketoconazole. This allows for a lower dosage while having a weaker inhibitory effect on hepatic CYP7A, potentially reducing the risk of hepatotoxicity. Compared to ketoconazole, levoketoconazole also presents a more favorable pharmacokinetic profile, with higher plasma concentrations and a longer half-life of 4–6 hours, permitting twice-daily dosing of 300–1200 mg/day [ , ]. The phase III open-label SONICS (efficacy and safety of levoketoconazole in the treatment of endogenous Cushing’s syndrome) trial demonstrated that levoketoconazole controlled hypercortisolism in 46% of diabetic patients [ ]. The subsequent LOGICS (efficacy of levoketoconazole for endogenous Cushing’s syndrome) trial confirmed that levoketoconazole, used at the same doses as in SONICS, achieved a complete response in only 50% of patients [ ]. Additionally, levoketoconazole treatment is associated with improvements in clinical syndrome and CS comorbidities [ ].
One study evaluating pituitary tumors in levoketoconazole-treated patients found stable tumor appearance in 87% and progression in 10% of cases [ ]. The most common AEs reported were nausea, headache, hypokalemia, and hypertension [ , ], along with QT prolongation. Notably, while an increase in liver enzymes was observed in almost 30% of cases, hepatotoxicity was not reported [ , ].
Osilodrostat
Osilodrostat, a novel oral inhibitor of steroidogenesis, has been officially approved by the European Medicines Agency (EMA) for the treatment of adult endogenous CS and by the FDA for use in CD patients who are either unable to undergo or have not been cured by pituitary surgery. It effectively inhibits adrenal enzymes, including aldosterone synthase and 11β-hydroxylase, thereby reducing the production and secretion of glucocorticoids and mineralocorticoids [ ]. Administered orally, osilodrostat dosages range from 2 to 60 mg/day. Compared to metyrapone and ketoconazole, in vitro data indicate a higher potency and a longer half-life (approximately 4 hours) for osilodrostat, allowing for a twice-daily dosing schedule. Several multicenter prospective studies have assessed osilodrostat’s efficacy and safety in CS treatment. Results demonstrate an efficacy rate of 68%–79% in controlling hypercortisolism within the first 22 weeks, with long-term studies showing success in 50%–88% of patients up to 70 weeks [ ]. Remarkably, control of hypercortisolism can be achieved as quickly as within 4 weeks of starting treatment. Additionally, osilodrostat treatment has been associated with improvements in body weight, blood pressure, glucose metabolism, lipid profile, quality of life, and depressive symptoms, with no reported instances of treatment escape. In patients with measurable pituitary tumors at baseline and follow-up, changes in tumor volume (increase or decrease) were observed in 30% of patients. Regarding the safety profile, the most common AEs are fatigue, nausea, headache, diarrhea, and adrenal insufficiency (described in up to 30% of patients). These symptoms may also be indicative of withdrawal from excess cortisol. Androgenic symptoms or mineralocorticoid manifestations could arise due to the accumulation of adrenal steroid precursors with respective activities. In males, low-normal testosterone levels might return to the mid-normal range, whereas in females, mean testosterone levels may initially rise to the upper normal limit before normalizing with prolonged therapy. QT prolongation and arrhythmia-related episodes have been described, and therefore, caution should also be exercised with medications known to prolong the QT interval. As with metyrapone-treated patients, it is advised to measure cortisol levels using tandem mass spectrometry in patients receiving osilodrostat due to known cross-reactivity issues in immunoassays with 11-deoxycortisol. Osilodrostat can interact with drugs metabolized via the CYP3A4, CYP1A2, CYP2C19, and CYP2D6 pathways, necessitating adapted dosing and titration.
Pituitary-directed drugs
Pituitary-directed drugs have become a cornerstone of medical treatment in CD based on their ability to target different receptors expressed by corticotroph tumors, thus reducing ACTH secretion and exerting mass shrinkage. To date, pasireotide remains the only approved pituitary-directed drug for the treatment of CD, whereas cabergoline is widely used in clinical practice as an off-label alternative. Temozolomide is currently the only formally recommended option for aggressive and/or metastatic corticotroph tumors following the failure of conventional, multimodal treatment.
Pasireotide
Corticotroph adenomas have been demonstrated to express the SSTR subtypes SSTR1, SSTR2, and SSTR5, with the latter two playing key roles in the somatostatin-mediated inhibition of pituitary ACTH secretion [ ]. Nevertheless, first-generation, SSTR2-selective somatostatin analogs (i.e., octreotide, lanreotide) have been mostly ineffective in treating CD, generally failing to induce either UFC normalization or significant tumor shrinkage [ ]. Recent studies on corticotroph tumor cell cultures have shown that chronic exposure to glucocorticoids can downregulate SSTR2, but not SSTR5, expression, making the latter a potential therapeutic target for somatostatin analog treatment [ ]. In this regard, the second-generation analog pasireotide, displaying high affinity for SSTR1, 3, and five subtypes, demonstrated high efficacy in inhibiting ACTH and cortisol secretion in vitro [ ], leading to its investigation in a clinical setting in several high-quality, randomized clinical trials, as summarized in a recent meta-analysis [ ]. The first available formulation required twice-daily subcutaneous injections (0.3–0.9 mg twice daily); since then, a long-acting formulation (pasireotide long acting resease [LAR]) has been developed to alleviate the treatment-related burden, requiring monthly intramuscular injections with doses ranging from 10 to 60 mg per month. Overall, pasireotide has demonstrated moderate efficacy in CD patients, inducing long-term remission in up to 44% of cases [ ] with no evidence of resistance development [ , ]; interestingly, in a 12-month, phase three clinical trial involving 150 patients with CD, long-term remission rates were higher in those with mild disease (mUFC less than 2 times upper limit of normal [ULN]) at 52% in both the 10 and 30 mg groups of pasireotide LAR, compared to 35% and 37% in patients with mUFC of 2–5 times ULN [ ]. Additionally, a substantial reduction in tumor size has been observed in 41.2% of patients with visible pituitary tumors [ ]. Currently, pasireotide is the only pituitary-directed medication formally approved by the EMA and the FDA for the treatment of CD patients in which neurosurgery was not feasible or ineffective. It is noteworthy, however, that pasireotide treatment is also encumbered by a considerable negative impact on glucose metabolism, inducing hyperglycemia in up to 78% of patients, often requiring additional medical intervention. Among the frequently reported side effects are gastrointestinal issues, nausea, elevation of liver enzymes, and, in rare instances, QT elongation [ , ].
Cabergoline
Cabergoline (CAB), a potent D2-selective agonist, has been proposed in the medical treatment of CD patients following in vitro observations confirming the expression of D2 dopaminergic receptors, to a variable extent, in over 75% of corticotroph tumors [ ], along with its ability to inhibit ACTH secretion from corticotroph cell lines [ ]. It is orally administered as immediate-release 0.5 mg tablets, whose long half-life allows longer intervals between administrations compared to fellow dopamine agonists, such as bromocriptine. Therapeutic dosages in CD generally range from 0.5 to 7 mg/week. So far, cabergoline has been investigated as an off-label treatment for CD in nine studies, six of which have been included in a recent meta-analysis. Overall efficacy has been described as moderate, with long-term UFC normalization rates ranging from 25% to 40% of cases, with more encouraging results in patients with mild hypercortisolism (UFC 1.0–2.0 x ULN) [ ]. CAB treatment has also been associated with an improvement of clinical symptoms and, occasionally, tumor shrinkage [ , ]. While data on long-term outcomes are still few, treatment escape has been reported to occur in up to 40% of patients [ ], and no markers have been identified that are able to predict successful response [ ]. Moreover, the onset of action seems to be generally delayed, as highlighted by poor results demonstrated in a recent, prospective 6-week clinical trial [ ], supporting that cabergoline may not be suited for a prompt resolution of hypercortisolism. Several reports have indicated, however, that it may represent a safe therapeutic option in CD patients actively seeking or undergoing pregnancy [ ]. CAB has also been evaluated in combination with steroidogenesis inhibitor ketoconazole, enhancing the remission rate to 68.8% of cases (range 55.6%–78.6%) [ , ], and in combination with pasireotide, showing an additional remission rate of 33.3% of patients with CD in patients with recurring CD following surgery [ , ]. Cabergoline is generally well tolerated, with common adverse effects including nausea, dizziness, and, at higher doses, cardiac valvopathies [ , ].
Temozolomide
To date, CD constitutes the most prevalent type among aggressive pituitary tumors, accounting for 45% of adenomas and 47% of carcinomas [ ]. Currently, the only formally recommended drug for the treatment of aggressive pituitary tumors after the failure of standard treatments is temozolomide, an orally administered alkylating agent that exerts its cytotoxic action by inducing the uncoupling of guanine from thymine in the DNA replication cycle. Therapeutic regimens include monotherapy (150–200 mg/m 2 /daily for five consecutive days, every 28 days) or its combination with radiotherapy (RT, i.e., the Stupp protocol, consisting of daily RT and temozolomide 75 mg/m 2 for 6 weeks, followed by standard temozolomide monotherapy) in patients who did not previously reach maximal RT doses [ ]. The use of temozolomide has significantly enhanced the overall survival rate in responders; notably, a clinically functioning tumor, a low O 6 -methylguanine-DNA methyltransferase tumor expression, and the concomitant administration of RT have all been associated with a more favorable treatment response [ ]. Indeed, among aggressive pituitary adenomas and carcinomas, the radiological response rate to temozolomide has been estimated to be highest in corticotroph tumors, suggesting that they might be good candidates for such treatment [ ]. Hormonal response, characterized by significant decreases in ACTH levels, has been reported to occur in up to 67% of patients with functioning, aggressive corticotropinomas [ , ]. Temozolomide is generally well tolerated, with the most common side effects including pancytopenia, nausea, vomiting, and fatigue [ ]. These results suggest that temozolomide should be considered in the therapeutic algorithm for aggressive corticotroph adenomas and carcinomas after the failure of conventional therapies, although new treatment modalities are necessary for temozolomide-resistant tumors, which account for one-third of cases [ , ].
Radiotherapy
RT is primarily used as adjuvant therapy for patients with persistent or recurrent disease after TSS [ , ] or in cases of aggressive tumor growth [ ]. Treatment modalities employed in clinical practice include conventional RT, consisting of administration of small daily fractions over 6 weeks with a cumulative dose of 45–50 Gy, or stereotactic radiotherapy (SRT), administered as either single or several sessions through different modalities, including photon source (Gamma Knife, CyberKnife, and linear accelerators) or proton particles. The choice of radiation modality should include convenience for patients and proximity to critical structures. Gamma Knife radiosurgery (GKRS), administered as a single dose or a few fractions of approximately 20 Gy, is generally regarded as the more convenient option [ ], especially in case of a clearly identifiable lesion on MRI [ ], as fewer treatment sessions are required. Following RT, the rate of tumor control, defined by decreased or stable tumor volume on MRI, is generally very high, reaching up to 95% of treated patients [ , ]. Hormonal remission, generally defined by the normalization of UFC levels, has been documented in up to two-thirds of cases treated with conventional external-beam RT [ , ]. Of note, stereotactic radiosurgery (SRS) has been associated with even higher biochemical remission rates, occurring in 80% of cases and being maintained in the long term in 57% [ , ]. Conversely, whole-sellar RT might be the preferable option whenever a clear biological target is not identified, showing comparable initial and long-term remission rates to MRI-positive cases [ ]. SRS may also serve as a primary treatment option in selected cases, namely for patients with high surgical risk or those who decline surgery. In this setting, the 5-year endocrine remission rate was found to be 81% out of 41 patients [ ]. However, long-term monitoring is essential, as both recurrence and tumor progression have been reported following RT.
Importantly, normalization of hypercortisolism usually occurs within 3 years of radiation treatment, with a median time to remission of approximately 15 months [ ]. Given the latency between RT administration and initial remission, adjuvant medical therapy is often required to control hypercortisolism, with periodic discontinuations to assess treatment efficacy [ ]. To date, clear predictors of RT efficacy have yet to be identified, with preliminary data suggesting a potential role of age, high Ki67 index [ ], and the expertise of the treatment team [ ]. Conversely, cortisol-lowering medical therapies, such as ketoconazole [ , ] and cabergoline [ ], have been suggested to negatively affect RT outcomes, but definitive evidence is still lacking. According to a recent meta-analysis, recurrence rates have been reported to be as high as 16%–18% in previously controlled patients in the two largest available series, whereas smaller series did not describe the late failure of GKRS [ ].
New-onset hypopituitarism is the most frequent side effect of RT regardless of treatment modality, occurring in up to 50% of cases [ , ], with hypothyroidism being the most frequent deficiency [ ]. Conversely, severe neurologic complications, including stroke, cranial nerve damage, and risk of secondary brain tumor, are uncommon in radiation-naïve patients and even more so in those treated with SRS [ ].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree