Abstract
Among breast cancer survivors, a contralateral breast cancer (CBC) is the most frequent second-cancer event, with a reported incidence rate of 0.5% to 1.0% per year. CBC risk is even higher among carriers of BRCA1 or BRCA2 mutations and noncarriers with a significant family history of breast cancer. However, recent studies have shown a favorable decline in CBC in the United States since 1985, which is largely due to the widespread adoption of tamoxifen in the mid-1980s for the adjuvant treatment of breast cancer. Despite the documented declining incidence of CBC during this time period, a dichotomous shift toward more aggressive surgical therapy with contralateral prophylactic mastectomy (CPM) has occurred. Reasons for this aggressive approach are multifactorial and include an overestimation of CBC risk among patients, increasing use of breast magnetic resonance imaging, availability of postmastectomy reconstruction, and overestimation of benefit of CPM. In the era of modern systemic therapy, the subject of bilateral breast cancer and, in particular, CBC, is of particular importance when considering the appropriate management and counseling of patients with unilateral breast cancer. In this chapter, we review risk factors for CBC, including age at diagnosis, genetic risk assessment, family history, prior radiation exposure, invasive lobular histology, and a personal history of lobular carcinoma in situ (LCIS). We also discuss the impact of advanced imaging on detection of synchronous CBC and discuss the effects of adjuvant systemic therapy on declining CBC risk. We review prognosis among breast cancer patients who have developed a CBC. Finally, we discuss the CPM epidemic that has developed in the United States since the early 2000s, and we review the benefits and risks associated with more aggressive surgery.
Keywords
Bilateral breast cancer, contralateral breast cancer, contralateral prophylactic mastectomy
In 1945 Foote and Stewart memorably stated that “the most frequent antecedent of cancer of one breast is the history of having had cancer in the opposite breast.” In fact, among breast cancer survivors, a contralateral breast cancer (CBC) is the most frequent second-cancer event. The incidence of CBC among breast cancer survivors has traditionally been reported as a constant incidence rate of 0.5% to 1.0% per year. CBC risk is even higher among carriers of BRCA1 or BRCA2 mutations and noncarriers with a significant family history of breast cancer. However, recent studies have shown a favorable decline in CBC in the United States since 1985, which is largely due to the widespread adoption of tamoxifen in the mid-1980s for the adjuvant treatment of breast cancer. The Early Breast Cancer Trialists’ Collaborative Group’s (EBCTCG) overview of 12 randomized trials demonstrated a 39% reduction in CBC development among women with estrogen receptor (ER)-positive or ER-unknown breast cancers who took tamoxifen for 5 years. Despite the documented declining incidence of CBC, a dichotomous shift toward more aggressive surgical therapy with contralateral prophylactic mastectomy (CPM) has occurred. A review of the Surveillance, Epidemiology and End Results (SEER) database demonstrated that CPM rates more than doubled from 1998 to 2003. Reasons for this aggressive approach are multifactorial and include an overestimation of CBC risk among patients, increasing use of breast magnetic resonance imaging (MRI), availability of postmastectomy reconstruction, and overestimation of benefit of CPM. In the era of modern systemic therapy, the subject of bilateral breast cancer, and, in particular, CBC, is of particular importance when considering the appropriate management and counseling of patients with unilateral breast cancer.
In this chapter, we review risk factors for CBC, including age at diagnosis, genetic risk assessment, family history, prior radiation exposure, invasive lobular histology, and a personal history of lobular carcinoma in situ (LCIS). We also discuss the impact of advanced imaging on detection of synchronous CBC and discuss the effects of adjuvant systemic therapy on declining CBC risk. We review prognosis among breast cancer patients who have developed a CBC. Finally, we discuss the CPM epidemic that has developed in the United States since the early 2000s, and we review the benefits and risks associated with more aggressive surgery.
Risk Factors for Bilateral Breast Cancer
Age
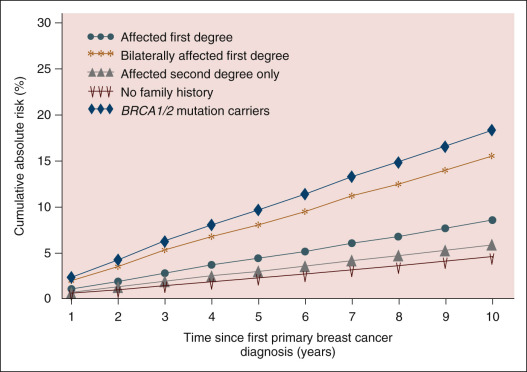
Studies from the 1970s, before the availability of genetic testing, initially demonstrated an association between young age at first breast cancer diagnosis and CBC risk. The results of large population-based studies from England and Connecticut (and the large personal experience of Haagensen) demonstrated the greatest risk of CBC in the youngest patients. However, lack of knowledge of genetic mutations in these patients limits the findings in these early studies. A recent report from the Women’s Environmental Cancer and Radiation Epidemiology (WECARE) Study demonstrated that among noncarriers of BRCA1 and BRCA2 mutations with no family history, the 10-year cumulative risk of CBC was approximately 4.6% (95% confidence interval [CI] 4.0%–5.1%) in women 25 to 54 years of age at first diagnosis. This was substantially lower than the 18.4% 10-year cumulative risk of CBC among BRCA1 / BRCA2 mutation carriers or the 15.6% 10-year cumulative risk of CBC in noncarriers with a family history of bilateral breast cancer in the same age group ( Table 75.1 ). Furthermore, among young breast cancer patients, molecular phenotype also contributes to risk of CBC. In a population-based study using SEER data, Nichols and coworkers demonstrated that current age-specific CBC rates (per 100/year) for an ER-positive cancer were 0.45 for first cancers diagnosed before age 30 compared with 1.26 for ER-negative cancers. Although young age at first diagnosis may be an indication for genetic testing, young women who are noncarriers with no family history can be counseled that their risk of CBC is low, particularly among women with ER-positive first breast cancers.
| Age at First Diagnosis (Years) | NONCARRIERS WITH NO FAMILY HISTORY | NONCARRIERS WITH BILATERALLY AFFECTED FIRST-DEGREE RELATIVE | BRCA1 OR BRCA2 MUTATION CARRIERS | |||
|---|---|---|---|---|---|---|
| 10-Year CBC Risk (%) | 95% CI (%) | 10-Year CBC Risk (%) | 95% CI (%) | 10-Year CBC Risk (%) | 95% CI (%) | |
| 25–29 | 6.3 | 4.4–8.7 | 21.7 | 11.1–42.3 | 28.2 | 16.0–50.0 |
| 30–34 | 7.0 | 5.4–8.7 | 23.7 | 12.8–44.2 | 30.7 | 18.4–51.5 |
| 35–39 | 5.2 | 4.1–6.3 | 18.1 | 9.8–33.4 | 23.7 | 14.3–39.3 |
| 40–44 | 4.2 | 3.3–5.1 | 14.8 | 8.0–27.2 | 19.4 | 11.8–32.1 |
| 45–49 | 4.5 | 4.1–5.6 | 15.1 | 8.2–27.7 | 12.2 | 6.5–22.9 |
| 50–54 | 4.0 | 3.6–4.9 | 13.4 | 7.3–24.7 | 10.2 | 5.8–20.3 |
| All ages (25–54) | 4.6 | 4.0–5.1 | 15.6 | 8.5–28.5 | 18.4 | 16.0–21.3 |
Hereditary Breast Cancer
Approximately 5% to 10% of breast cancers are hereditary and associated with an inherited germline mutation. Mutations in the tumor-suppressor genes BRCA1 and BRCA2 account for most hereditary breast cancers and confer a 36% to 84% lifetime risk of first primary breast cancer. The risk of CBC after a first primary breast cancer among BRCA1 or BRCA2 mutation carriers is substantially higher than the risk in sporadic breast cancer survivors. A recent population-based, nested case-control study of CBC risk among 705 patients with CBC and 1398 controls with unilateral breast cancer (from the WECARE study) demonstrated that BRCA1 and BRCA2 mutation carriers had a 4.5-fold (95% CI 2.8-fold–7.1-fold) and 3.4-fold (95% CI 2.0-fold–5.8-fold) increased risk of CBC, respectively, compared with noncarriers. Furthermore, the relative risk of CBC among BRCA1 carriers increased as age at first diagnosis decreased, with an 11-fold increased CBC risk among women first diagnosed before 35 years of age, compared with 2.6-fold increased risk in women 45 to 54 years of age at first diagnosis. Similar risk estimates were reported in a retrospective, multicenter, cohort study evaluating CBC risk in 978 BRCA1 and BRCA2 positive families. The cumulative risk of CBC at 25 years after first diagnosis was 48.1% (95% CI 38.3%–57.9%) in BRCA1 patients and 47.1% (95% CI 28.9%–65.3%) in BRCA2 patients. BRCA1 patients younger than 40 years of age at first diagnosis had a 25-year CBC incidence of 62.9% (95% CI 50.4%–75.4%) compared with 43.7% (95% CI 24.9%–62.5%) in women 41 to 50 years of age and 19.6% (95% CI 5.3%–33.9%) in women older than 50 years. This substantial risk of CBC among BRCA1 and BRCA2 patients, particularly in women younger than 40 years at first diagnosis, should be considered at the time of first diagnosis when counseling women regarding surgical treatment options.
Multigene, or panel testing, has led to the identification of additional breast cancer genes, although the magnitude of breast cancer risk for many of these mutations remains poorly understood. Among patients with non- BRCA deleterious mutations, the most common pathogenic mutations include PALB2, CHEK2, and ATM, with a prevalence of 1.3%, 0.9%, and 0.8%, respectively. There is strong evidence that protein-truncating mutations in these three genes are associated with a moderate breast cancer risk among unaffected carriers, with mutations in PALB2 conferring the highest risk. However, data regarding reliable risk estimates for CBC in these patients are lacking, and it is not clear that surveillance strategies or surgical treatment should be altered in these patients on the basis of the presence or absence of the mutation alone. Patients with moderate penetrance genes should be counseled on CBC risk according to their individual personal risk factors and family history of breast cancer; more importantly, decisions for risk-reduction strategies in these patients should be individualized because the magnitude of risk for future breast cancer is far less clear than in patients with high-penetrance genes.
Multigene panel testing has also identified variants of uncertain significance (VUS) in which the predicted amino acid sequence of a protein has been altered, without information as to whether this genetic variant impairs the function of the protein. As a result, these variants are not clinically actionable, and VUS results should not be used in isolation to counsel patients about breast cancer risk or CBC risk and should not alter patient management.
Family History
A family history of breast cancer is a well-established and significant risk for breast cancer, even in the absence of a known genetic mutation. Seventy-four studies before 1997 link family history to the risk of breast cancer and are the subject of a comprehensive meta-analysis. Taken together, these studies confirm increased breast cancer risk based on a family history in any relative (relative risk [RR] 1.9), a mother (RR 2.0), a sister (RR 2.3), a daughter (RR 1.8), a mother and sister (RR 3.6), and a second-degree relative (RR 1.5).
CBC risk among non- BRCA women with a family history of breast cancer was assessed in the WECARE study. Those with a first-degree relative with breast cancer had an almost two-fold greater risk of developing CBC (RR 1.9, 95% CI 1.4–2.6) compared with women without a family history. Risk of CBC was also influenced by the age at diagnosis of the affected individual, age at diagnosis of the family member, and the nature of the family history (see Table 75.1 ; Fig. 75.1 ). Notably, a history of bilateral breast cancer in an affected relative was one of the most important predictors of CBC risk, with a 10-year cumulative risk of CBC of 15.6%. This CBC risk in women with a first-degree family history of bilateral breast cancer was similar to the 10-year risk seen in BRCA mutation carriers (15.6% vs. 18.4%, respectively), suggesting that these women should be considered for preventive counseling at first diagnosis of breast cancer.
Radiation Exposure
Increasing use of conservative surgery plus radiotherapy (RT) for early-stage breast cancer has raised concern about the possibility of CBC induced by low-dose radiation scatter. This has not been observed. A 20-year follow-up of the National Surgical Adjuvant Bowel and Breast Project (NSABP) B-06 trial comparing total mastectomy, lumpectomy, and lumpectomy and irradiation demonstrated no difference in CBC rates among the treatment groups (8.5% [total mastectomy] vs. 8.8% [lumpectomy] vs. 9.4% [lumpectomy plus irradiation]). Another large breast conservation trial from Milan, Italy, showed no statistically significant difference in the 20-year cumulative incidence of CBC among women treated with radical mastectomy compared with breast conservation therapy ( p = .5).
These data do not absolutely rule out an effect of RT on the risk of CBC. In the 2005 EBCTCG overview (a meta-analysis of 46 randomized trials of RT vs. no RT and 17 trials of RT vs. more surgery, in 32,800 patients), the incidence of CBC at 15 years was marginally but significantly higher in the RT arm than in the control arm (9.3% vs. 7.5%, p = .002). Notably, the absolute difference in CBC between the two groups was small. Furthermore, these data do not take into account the increased precision of contemporary RT techniques or the effects of current systemic treatments (chemotherapy and/or hormonal) in reducing the incidence of CBC, which is addressed elsewhere in the chapter.
A striking exception to the preceding findings is the increased risk of breast cancer noted in young women after mantle RT for Hodgkin’s lymphoma. Early reports from Aisenberg and colleagues suggested the following: (1) that a relationship existed between age at RT exposure and risk of breast cancer, with an RR of 56 (95% CI 23.3–107) for those 19 years of age or younger at the time of treatment compared with an RR of 0.9 (95% CI 0–5.3) for those 30 years of age and older, and (2) that breast cancer onset occurred approximately 11 to 25 years after initial chest irradiation. Recently, a longitudinal cohort study of childhood cancer survivors demonstrated that the cumulative incidence of breast cancer was 30% by 50 years of age among 1230 female childhood cancer survivors who received chest irradiation, which is similar to the incidence of breast cancer in BRCA1 carriers (estimated cumulative risk: 31% by age 50). Furthermore, women treated with mantle RT have a higher risk of bilateral breast cancer, with Yahalom and colleagues and Basu and colleagues reporting an incidence of bilaterality of 22% and 34%, in their respective studies. Basu and colleagues additionally reported that the time to bilateral breast cancer after first primary breast cancer was short (12–34 months) in their patient cohort. The incidence of bilaterality among female patients treated with mantle RT is substantially more common than in sporadic breast cancer cases, and treatment of first primary breast cancer in these patients should include discussion regarding risk for CBC.
Invasive Lobular Carcinoma
On the basis of data before the routine use of adjuvant systemic therapy for the treatment of breast cancer, lobular histology was believed to be an independent risk factor for metachronous CBC (compared with ductal histology). More recent data from the SEER registry comparing CBC incidence in 134,501 women with unilateral breast cancer showed no difference in CBC rates based on histology of the index cancer; specifically, actuarial rates of CBC at 5, 10, 15, and 20 years were 2.6%, 6.0%, 9.1%, and 12.1% for invasive ductal cancers (IDCs) compared with 3.2%, 6.4%, 9.0%, and 11.7% for invasive lobular cancers (ILCs), respectively. Similarly, a single-institution review of a consecutive cohort of 1182 patients with breast cancer demonstrated no difference in the incidence of metachronous CBC between patients with ductal (12 of 1011; 1%) and lobular (2 of 171; 1%) histology ( p = not significant) at a median follow-up of 4 years. In contrast to metachronous CBC, the incidence of synchronous bilateral breast cancer is reported to be slightly higher in patients with ILC ; however, a modern study using the Netherlands Cancer Registry demonstrated the absolute difference in CBC within 6 months of diagnosis of the index lesion in patients with lobular versus ductal histology was only 0.6%, and did not justify the routine use of preoperative breast MRI in patients with ILC to evaluate the contralateral breast. On the basis of the aforementioned studies, imaging evaluation and surgical recommendations for the contralateral breast in women with ILC should be no different from that for women with IDC.
Lobular Carcinoma in Situ
Women with LCIS have a 7- to 10-fold increase in breast cancer risk compared with the general population. The classic studies of Haagensen and Rosen and colleagues both document an invasive breast cancer risk in patients with LCIS of approximately 30% at 20 to 25 years’ follow-up, with half of the cancers in the ipsilateral breast and half in the contralateral breast. In a study of 4853 patients with LCIS from the SEER database, Chuba and colleagues report a slightly lower but similar risk of invasive breast cancer of 11.1% at 15 years. In one of the largest single-institution experiences with almost 30 years’ follow-up, King and colleagues reported on 1060 patients with LCIS; the annual cancer development rate was 2% per year for the first 6 years after LCIS diagnosis, with an overall 15-year cumulative cancer incidence of 26%. Twelve percent of the cancers were bilateral; importantly, breast cancer incidence was significantly reduced with chemoprevention (10-year cumulative risk: 21% no chemoprevention; 7% with chemoprevention) highlighting the effect of systemic therapy on breast cancer risk reduction in high-risk patients.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree