Fig. 8.1
Anatomy of the ear (Illustration by Aimee Ermel)
Sound is described in terms of its intensity or loudness (measured in decibels) and frequency or pitch (measured in Hertz). Normal hearing thresholds are between −10 and 25 dB, and hearing loss is graded on a scale of “mild” to “profound” (Table 8.1). The speech frequencies are between 250 and 2,000 Hz, but higher frequencies (>2,000–8,000 Hz) are critical for speech discrimination, since many of the consonant sounds are in the high-frequency range; therefore, even a “mild” degree of high-frequency hearing loss can have a profound effect on a young child who is just acquiring language. Conductive hearing loss occurs when transmission of sound from the environment is impaired due to a pathological process in the outer or middle ear. Sensorineural hearing loss occurs as a result of pathology involving the cochlea or auditory nerve. Mixed hearing loss encompasses elements of both conductive and sensorineural hearing loss. In general, hearing loss occurring as a consequence of ototoxic pharmacologic agents is sensorineural in nature, whereas hearing loss resulting from radiation, tumor, or surgical procedures is often multifactorial and may include both conductive and sensorineural components.
Table 8.1
Degrees of hearing loss
Hearing threshold | Degree of loss |
|---|---|
−10 to 25 dB | Normal |
26–40 dB | Mild |
41–55 dB | Moderate |
56–70 dB | Moderately severe |
71–90 dB | Severe |
>90 dB | Profound |
8.2.2 Ototoxic Effects of Tumor and Therapy: Risk Factors and Prevalence
8.2.2.1 Surgery and Tumor
Hearing loss is rarely a presenting symptom in tumors that affect children. Exceptions include nasopharyngeal cancer, parameningeal rhabdomyosarcoma, tumors of the base of skull (chordoma), vestibular schwannoma, and metastases that affect the temporal bone, most notably neuroblastoma. Although CNS tumors may come in contact with vital aspects of the auditory system (including the VIIIth nerve and associated vasculature and the brainstem and vestibular nuclei, as well as cortical tracts essential to hearing), hearing loss is not commonly recorded at diagnosis. Surgical management of CNS tumors or musculoskeletal tumors of the head and neck can have a profound and irreversible effect on hearing, especially when essential components of the auditory system are involved with the tumor and surgical resection is required. Transcranial surgery in the region of the middle cranial fossa and suboccipital region can result in bony complications that may lead to mastoiditis and infections. Fortunately, most pediatric head and neck tumors are sensitive to radiation and chemotherapy. Tumors of the CNS account for nearly 20 % of all neoplasms in children, with the posterior fossa one of the most common locations. Children with medulloblastoma, ependymoma, cerebellar astrocytoma, and other tumors that gain access to the lateral recesses and cranial nerves are at risk for transient or permanent hearing loss at the time of resection. Great skill is required to remove tumor from the lateral aspect of the brainstem, through which the VIIIth nerve and vessels may run (Fig. 8.2). Recent efforts in surgery include intraoperative neurophysiologic monitoring of cranial nerves during tumor resection to limit neurologic impairment [84].
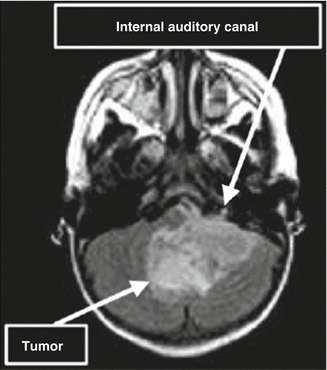

Fig. 8.2
Left cerebellopontine angle ependymoma encasing CN VIII with extension into the internal auditory meatus
Children with CNS tumors often present with hydrocephalus that requires emergent evaluation and treatment with temporary ventriculostomy and resection. When the natural flow of CSF cannot be reestablished, permanent ventriculoperitoneal shunting may be required. Rapid loss of intracranial pressure, which may occur after profound blood loss, lumbar puncture, ventriculostomy, and tumor resection, has been associated with hearing loss. The mechanism is likely related to the anatomic connection between the CSF spaces and the perilymph of the cochlea (cochlear aqueduct) (Fig. 8.3). Hydrocephalus and its management can influence radiation and chemotherapy-related hearing loss. In one study that included children with localized brain tumors treated with conformal radiation therapy, patients with CSF shunts and pre-irradiation ototoxic chemotherapy had the greatest change in hearing thresholds. Patients with shunts and supratentorial tumors who received radiation doses exceeding 32 Gy (without chemotherapy) over a 6-week period had hearing impairment at low and intermediate frequencies. Additionally, patients with shunts and suprasellar, hypothalamic, or thalamic tumors developed intermediate-frequency hearing loss after radiation regardless of dose [54]. In a recent study of children with medulloblastoma with and without ventriculoperitoneal shunting, the odds of hearing loss for patients with a CSF shunt were 23.5-fold higher (95 % confidence interval, 4.21–131.15) than for those without a shunt, suggesting an independent association between shunting and hearing loss [26].
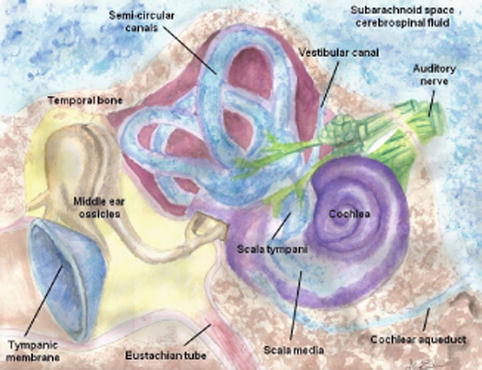

Fig. 8.3
Drawing of cochlear duct and anatomic relationships with CSF spaces and auditory system (Illustration by Aimee Ermel)
8.2.2.2 Radiation Therapy
Hearing loss is a potential complication of radiation therapy in children. Serous otitis media, with associated conductive hearing loss, may be noted as a complication in patients receiving radiation in doses ≥30 Gy to the posterior nasopharynx and mastoid [95]. The problem can be long-standing, as in the case of patients with nasopharyngeal carcinoma or rhabdomyosarcoma, although the risk appears to be increased in those with middle ear effusions prior to irradiation [48] (Fig. 8.4). Mucociliary dysfunction is thought to be the primary cause of middle ear effusions during and after radiation therapy; this can be self-limited in nature or, in severe cases, may require treatment with myringotomy [100]. Direct structural damage to the conductive system, including osteoradionecrosis, is rare with modern radiation therapy, although patients who receive high-dose irradiation to the ear and temporal bone may be at high risk for complications from localized bone and soft tissue infections arising in the region of the external auditory canal. For this reason, soft tissue infections of the ear should be treated aggressively and instrumentation of the external auditory canal should be undertaken cautiously. Clinical experience suggests that patients treated with irradiation encompassing the ear canals may have chronic difficulty with production of excessive and/or dry cerumen, leading to accumulation of inspissated debris that can interfere with hearing and require periodic removal.
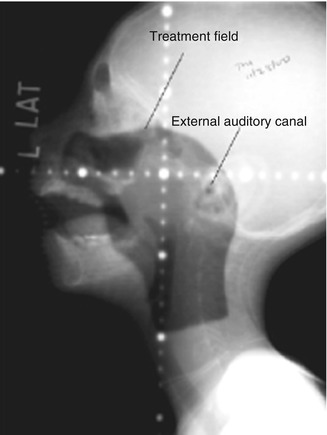

Fig. 8.4
Lateral radiation portal film of a child with nasopharyngeal rhabdomyosarcoma
Sensorineural hearing loss can occur as a result of radiation effects from the auditory cortex to the cochlea. Because the doses administered to neural tissue directly or indirectly are generally accepted as safe and below the threshold for neurologic impairment, sensorineural effects of radiation are most likely to occur in the cochlea and are usually noted months or years after treatment. Despite recognition of the cochlea as the primary component in sensorineural hearing loss [23], the prevalence and time course for injury due to radiation is unknown. Because most children at risk for radiation-related hearing loss also receive ototoxic chemotherapy, the pathophysiology and clinical course of hearing loss is most often described after combined modality therapy. Similar to the effects of ototoxic chemotherapy, the effects of radiation appear to be dose related, with a threshold dose for sensorineural hearing loss in the range of 35–45 Gy [31, 54]. The combined effects of radiation and chemotherapy – specifically cisplatin and carboplatin – for the treatment of medulloblastoma and other CNS tumors is better understood, with the high prevalence of hearing loss reported among children receiving combined modality therapy [82].
8.2.2.3 Pharmacologic Therapy
The primary pharmacologic agents implicated in ototoxicity include platinum chemotherapy, aminoglycoside antibiotics, and loop diuretics. These agents are all capable of causing sensorineural hearing loss; however, ototoxicity related to carboplatin alone is uncommon [15, 34, 43, 89], except when administered at myeloablative doses [44, 67], in combination with cisplatin [37, 42], or possibly in very young children when dosed according to body surface area rather than weight [72]. The mechanism of aminoglycoside and platinum-related ototoxicity is oxidative stress resulting in destruction of cochlear sensory hair cells [16, 81]. These specialized hair cells are arranged tonotopically (in order of pitch) in four rows (one inner and three outer rows) along the organ of Corti, and each hair cell is sensitive to a limited frequency range [51].
Cisplatin and aminoglycoside antibiotics damage the outer hair cells, whereas carboplatin selectively damages only inner hair cells [92]. The initial hearing loss associated with ototoxic pharmacologic agents usually affects the high-frequency ranges. This is because destruction of the sensory hair cells typically begins at the base of the cochlea, where high-frequency sounds are processed, and proceeds toward the apex, where the processing of low-frequency sound occurs [77]. Individuals are generally born with a full complement of auditory sensory hair cells. Once destroyed, these cells cannot regenerate; therefore, hearing loss occurring as a result of sensory hair cell loss is almost always irreversible.
The mechanism of ototoxicity associated with loop diuretics is thought to be related to changes in the fluid and electrolyte balance within the inner ear, resulting in tissue edema within the cochlea and decreased endocochlear potential [16, 79]. Hearing loss resulting from diuretics typically occurs following rapid intravenous administration. Fortunately, this type of hearing loss is usually transient. However, if loop diuretics are administered simultaneously with or shortly after the administration of platinum chemotherapy or aminoglycosides, the likelihood of permanent auditory damage increases as a result of synergism between these agents [3, 90].
Factors placing survivors at highest risk for hearing loss related to pharmacologic therapy include very young age (less than 5 years) at the time of cancer therapy, diagnosis of central nervous system tumor, treatment with multiple ototoxic agents, and/or treatment with platinum chemotherapy in combination with radiation to the ear or brain [33, 47, 65, 67, 82, 97]. Many childhood malignancies, including germ cell tumors, central nervous system tumors, osteosarcoma, and neuroblastoma, frequently require treatment with platinum-based chemotherapy protocols, and supportive care regimens often employ aminoglycoside antibiotics and loop diuretics [27, 42, 46, 65]. Therefore, the index of suspicion for treatment-related hearing loss should be high for any survivor who received potentially ototoxic therapy, including patients treated for malignancies such as acute myeloid leukemia, where administration of platinum compounds is rare, but use of aminoglycosides for infectious complications is common.
Factors contributing to the risk for ototoxicity include diminished renal function at the time of treatment [25], rapid intravenous administration of ototoxic agent(s) [73], prolonged elevated serum trough drug levels [90], coadministration of other ototoxic drugs (e.g., chelating agents, quinine, salicylates [16, 77]), and excessive noise exposure [7]. Further, the potential impact of new agents with ototoxic properties (e.g., oxaliplatin [16]) should be kept in mind when assessing patient risk.
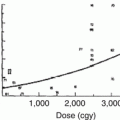
The prevalence of ototoxicity is well documented in children receiving cisplatin chemotherapy [83] and is dose related, with an inverse relationship to age at therapy [96]. Brock et al. [5] studied 29 children off therapy for at least 2 years who had received “standard-dose” (60–100 mg/m2/course) cisplatin-containing chemotherapy regimens without brain or ear irradiation. Median age at diagnosis was 2 years, 2 months (range: 1 month–13.5 years) and median cumulative dose was 540 mg/m2 (range: 400–1,860 mg/m2). Moderate to severe sensorineural hearing loss was detected in 48 % of these children, with 33 % requiring hearing aids; no child demonstrated any recovery of hearing at median 4-year follow-up. Schell et al. [82] studied 177 children and young adults receiving cisplatin (median cumulative dose 360 mg/m2, range 90–1,260 mg/m2) with and without prior cranial radiation (median dose: 5,050 cGy; range 2,880–8,260 cGy). In irradiated patients, doses as low as 270 mg/m2 were associated with a high probability of substantial hearing loss, whereas nonirradiated patients demonstrated negligible loss at doses up to 360 mg/m2; however, as cumulative doses increased to 720 mg/m2, the risk of substantial shifts in hearing threshold increased to 25 %. In a study of 49 patients with osteosarcoma receiving 400 mg/m2 cisplatin with or without ifosfamide, Meyer et al. [55] reported a significant increase in the incidence of hearing loss (≥30 dB at 2,000 or 3,000 Hz) in the cisplatin/ifosfamide group, indicative of a synergistic effect between the two agents. Although a small early study of patients receiving carboplatin reported that 11 of 22 children who received carboplatin at a median dose of 2,409 mg/m2 demonstrated sensorineural hearing loss at 4,000–12,000 Hz [50], most subsequent studies of carboplatin in pediatric oncology populations have demonstrated only rare, isolated cases of moderate to severe hearing loss at conventional doses [43, 58, 68, 89]. However, in one recent study, sustained ototoxicity was documented in 10 of 60 (17 %) infants and toddlers treated with carboplatin for retinoblastoma, a finding potentially explained by the relatively large exposure of 3,850 mg/m2 (range, 2,580–4,580 mg/m2) that resulted from dose calculation by body surface area rather than weight [72]. In children receiving myeloablative doses of carboplatin and in those receiving carboplatin in combination with cisplatin, moderate to severe hearing loss is commonly seen [42, 67, 85].
Ototoxicity is also a potential complication of therapy with aminoglycoside antibiotics [53] and loop diuretics [79]. Prospective studies by Fee [19] and Smith et al. [87] reported ototoxic rates of 10–16 % for gentamicin and tobramycin. Lerner et al. [45] reported an 11 % incidence of ototoxicity associated with gentamicin and a 13 % incidence in amikacin-treated patients. An augmented ototoxic effect has been reported with the concurrent administration of cisplatin and gentamicin [76]. Brookhouser [7] described a 6.4 % incidence of furosemide-associated ototoxicity and a 0.7 % incidence for ethacrynic acid; however, the risk increased significantly when loop diuretics were administered concurrently with aminoglycoside antibiotics [3].
8.2.3 Genetic Susceptibility
Variations in individual susceptibility suggest that genetic factors may play a role in predisposing certain patients to drug-induced ototoxicity [2, 64, 69, 75, 78]. Currently, only a few studies have explored genetic variants that may render certain patients at high risk for ototoxicity. Early work is based on candidate genes linked plausibly to the physiologic mechanisms of cisplatin ototoxicity (i.e., generation of reactive oxygen species (ROS) leading to depletion of the normally protective cochlear antioxidant enzymes involved in detoxification of superoxides). Polymorphisms of genes coding for glutathione S-transferase (GST) proteins (crucial enzymes involved in detoxification) have been implicated in cisplatin-related ototoxicity. For example, Peters et al. reported a protective effect of the GSTM3*B allele on ototoxicity [69], and Oldenburg et al. reported the presence of both alleles of 105Val-GSTP1 to be protective against ototoxicity, while the GSTM1 variant was associated with increased susceptibility to cisplatin-related hearing loss in testicular cancer patients [64]. Recently, Rednam et al. reported an increased risk for radiation-associated ototoxicity among patients with the GSTP1-105G allele in pediatric medulloblastoma, as well as a strong interaction with radiation dose [74]. In a discovery cohort of 54 children, strong associations between cisplatin-induced ototoxicity and genetic variants in thiopurine S-methyltransferase (TPMT) and catechol-O-methyltransferase (COMT) have also been reported; for the combined mutation, the odds ratio was 42.2 (p = 1.1 × 10e9), with positive and negative predictive values of 92.9 % and 48.6 %, respectively [78]. Additional plausible candidate genes identified to date include megalin, a low-density lipoprotein-related protein 2 strongly expressed within the stria vascularis in the cochlea; ERCC1, a nucleotide excision repair gene; and mutations in mitrochonrial DNA associated with aminoglycoside-induced deafness [6, 57]. A genome-wide approach to identification of genetic variants associated with susceptibility to ototoxicity is also underway [57]. In principle, identification of genetic variants that place patients at high risk for ototoxicity could inform future strategies to tailor chemotherapy and/or utilize preventive measures in order to maximize efficacy while minimizing risk. However, there are currently contradictory findings, and further validation of data is needed in larger groups in order to make informed recommendations.
8.2.4 Preventive Measures
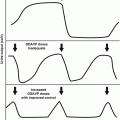
The administration of platinum chemotherapy prior to, rather than following, cranial irradiation in children with CNS tumors has been shown to reduce ototoxicity [40, 82]. Newer radiation delivery techniques (e.g., intensity-modulated and proton beam radiation) hold the promise of reducing radiation-related ototoxicity by conforming the prescribed dose to the region at risk and sparing the cochlea [31, 54, 56]. Using these techniques, the dose to the cochlea can be estimated more accurately to optimize treatment and to collect dose information, which can be correlated with other factors that influence hearing after treatment [31, 32, 54, 56] (Fig. 8.5).
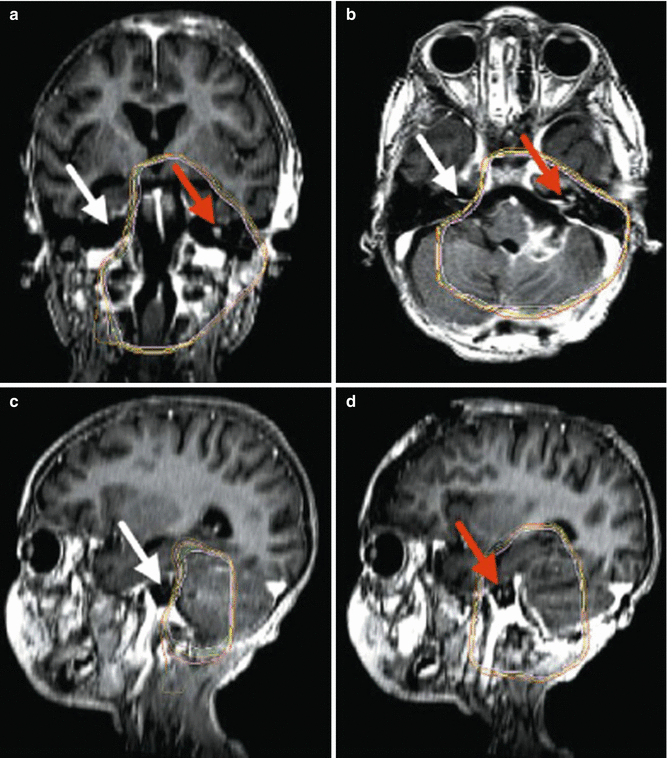

Fig. 8.5
Coronal (a), transverse (b), and sagittal (c, d) MR images with three-dimensional radiation dosimetry, showing the ability of conformal treatment techniques to spare auditory structures not adjacent to tumor bed in a child with cerebellopontine angle ependymoma. (White arrows represent spared cochlea; red arrows represent irradiated cochlea)
Careful monitoring and appropriate dose modification earlier in the course of therapy, before severe hearing loss has been sustained, are effective in decreasing auditory morbidity; however, decisions regarding dose modification or substitution of alternate therapeutic agents in the face of identified hearing loss must be weighed carefully against concerns regarding disease control and survival [47, 91]. Thus, identification of effective otoprotective strategies holds promise for reducing ototoxicity without compromising potentially curative therapy.
To date, only two agents have been the subject of study in clinical trials. Amifostine, a reactive oxygen scavenger, was originally developed as a radiation protectant [80] and has been evaluated in several recent trials to assess its potential for otoprotection. Although otoprotection was observed in a study of amifostine in adults with advanced ovarian cancer [36], studies of amifostine in children receiving platinum chemotherapy for hepatoblastoma [35], germ cell tumors [52], and osteosarcoma [22, 70] failed to demonstrate significant otoprotection. However, a recent comparative cohort study using a more dose-intensive amifostine schedule demonstrated a significant protective effect against severe ototoxicity in children receiving cisplatin for average-risk medulloblastoma [20]. Studies of sodium thiosulfate, a free-radical scavenger thiol compound, have been reported to provide otoprotection in limited institution studies [17, 63], and pediatric cooperative group otoprotection studies of this agent are currently underway [21, 91]. Other potential otoprotectants at various stages of investigation include N-acetyl-cysteine [62, 93, 99], D-methionine [10], and ebselen [49]. Another approach that is conceptually appealing is the intratympanic administration of otoprotectants as compartmental therapy to avoid risks attributed to systemic exposure, but to date experience with this has been limited [30, 98].
Additional otoprotective strategies include counseling patients regarding the importance of avoiding other potentially ototoxic agents, including medications (e.g., chelating agents, salicylates) and loud noises (especially ≥85 dB). Patients should also be advised to use protective measures (e.g., ear plugs) when in noisy environments and avoid excessively loud volumes with headphones in order to prevent comorbid noise-induced hearing loss [24, 71].
8.3 Clinical Manifestations
8.3.1 Clinical Manifestations of Ototoxicity Related to Surgery or Tumor
Tumor-related hearing loss is not a common presenting symptom. Notable exceptions include patients with nasopharyngeal tumors or rhabdomyosarcoma involving the middle and inner ear, patients with eustachian tube dysfunction secondary to mass effect or lymphadenopathy, and the rare patient with neurofibromatosis type II who develops a neurofibroma involving the otoacoustic nerve (CN VIII). In the scope of possible side effects and complications first noted after surgery for brain tumors, hearing loss is usually not a priority. Children at risk for hearing loss from surgery often have acquired other deficits that are more apparent or life-threatening, which can delay the diagnosis of surgery-related hearing loss. These patients often have deficits involving the abducens (CN VI) or facial nerve (CN VII) and, in extreme cases, lower cranial nerves affecting speech and swallowing. Children who require temporary or permanent CSF shunting may experience altered hearing, either temporary or permanent loss, or heightened sensitivity (hyperacusis). Recovery from surgery-related hearing deficits can occur in some cases despite adjuvant irradiation, provided that the nerve has not been transected or the vascular supply permanently disrupted.
8.3.2 Clinical Manifestations of Radiation-Related Ototoxicity
Radiation-related effects on the auditory system may occur during or after treatment. Acute effects are more likely to involve the external auditory canal (radiation dermatitis of the epithelium lining the canal leading to otitis externa) and middle ear (otorrhea with otalgia or mucociliary dysfunction of the middle ear with resultant eustachian tube dysfunction). Cerumen production appears to be increased in some patients during and after radiation, although the contribution from other causes cannot be excluded. Atrophy of the sebaceous glands may occur and is dose dependent. Soft tissue fibrosis, otosclerosis, and even cholesteatoma have been reported. Because radiation therapy is often given in conjunction with ototoxic chemotherapy, separating the effects of the two treatments can be difficult. Since most instances of treatment-related hearing loss occur in close temporal proximity to chemotherapy or combined modality therapy, more ototoxicity is known or observed in this setting. However, radiation therapy alone may result in hearing loss with an onset many years following treatment; therefore, long-term survivors remain at risk for hearing loss. Radiation-related hearing loss may occur during the first year after treatment in patients who also received chemotherapy and is usually seen 2 or more years after treatment in patients treated with radiation alone. With newer means of delivering radiation therapy, namely, noncoplanar, individually shaped beams to avoid the middle ear and cochlea, some of the acute and late effects of treatment appear to be reduced. With additional considerations regarding the timing of radiation and chemotherapy, further reductions in ototoxicity seem to be feasible.
8.3.3 Clinical Manifestations of Ototoxicity Related to Pharmacologic Agents
Hearing loss resulting from ototoxic medications is generally bilateral and symmetrical, progressive with continued therapy, and irreversible. Early symptoms may include tinnitus, vertigo, and difficulty hearing in the presence of background noise, which is indicative of vestibular injury and high-frequency (>2,000 Hz) hearing loss. Because consonant sounds are primarily high frequency and vowel sounds are primarily low frequency, a person with high-frequency hearing loss will be able to hear vowel sounds better than consonants. The English language relies heavily on high-frequency consonant sounds (such as “th,” “f,” “s,” and “k,”) to convey the meaning of words (Fig. 8.6); therefore, the inability to hear high-frequency sounds often results in poor speech discrimination, or a perception of “hearing but not understanding.” For people with high-frequency hearing loss, understanding high-pitched voices (e.g., females and children) may be particularly problematic. Increasing the volume of the speaker’s voice (e.g., by shouting) is generally not helpful, since this raises the intensity level of vowels but not consonants. As hearing loss progresses, patients may experience difficulty in hearing sounds within the speech ranges (250–2,000 Hz). It is important to understand that even mild high-frequency hearing loss is clinically significant, particularly in young children. It may result in difficulties with speech discrimination and language acquisition that adversely affect both cognitive and social development.
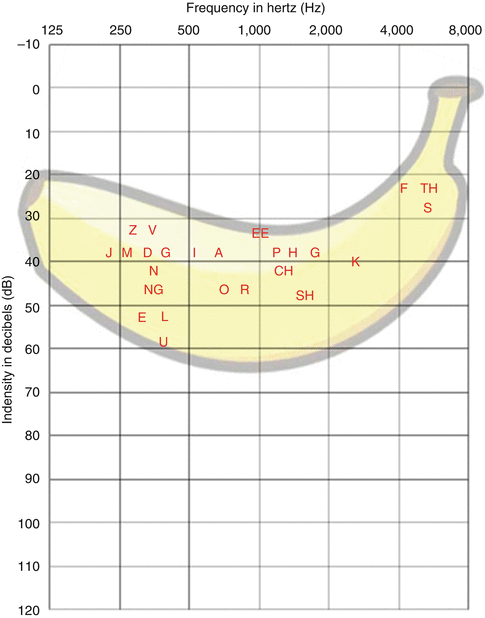

Fig. 8.6
Audiogram showing the speech frequencies, which cluster in a banana-shaped region, often referred to as the “speech banana”
8.4 Detection and Screening
8.4.1 Auditory Screening
It is important to distinguish between auditory “screening” and “testing.” Conventional auditory screening typically evaluates the patient’s ability to hear pure tones at 1,000, 2,000 and 4,000 Hertz (Hz) in each ear; a score of “pass” or “fail” is given (hearing threshold ≤20 or >20 decibels [dB], respectively). Measurement of otoacoustic emissions (OAEs) is another auditory screening technique often utilized for newborns or very young children. OAEs are sounds generated by the cochlea’s outer hair cells in response to stimulation by auditory signals [88]. A score of “pass” on OAE screening verifies that a patient has near-normal hearing (threshold ≤30 dB). However, inasmuch as carboplatin selectively damages inner rather than outer hair cells, OAE screening is not appropriate for monitoring carboplatin-related ototoxicity [28]. In general, auditory screening is used to select individuals who need referral for formalized testing, and OAEs may be more sensitive in detecting initial ototoxic damage; however, the gold standard for ototoxicity monitoring is formal diagnostic audiometric assessment using measurement of pure-tone thresholds [38].
8.4.2 Diagnostic Audiometry
8.4.2.1 Pure-Tone Audiometry
The standard method used for diagnostic evaluation of hearing in cooperative patients is pure-tone audiometry. The aim is to establish hearing thresholds across a wide range of sound frequencies (usually 250–8,000 Hz, up to 12,000 or even higher with ultra-high-frequency testing). This testing is generally done in a soundproof booth; air and bone conduction thresholds are measured and results graphed on an audiogram (Fig. 8.7a), which indicates the sound intensity needed to produce a response (the hearing threshold, dB) at a given frequency (Hz). Often, a “stairstep” pattern of hearing loss, progressing from the high frequencies down to the speech ranges, is evident on the audiogram of patients with sensorineural hearing loss resulting from ototoxic therapy (Fig. 8.7b). For patients with a developmental age less than 4–5 years, pure-tone audiometric testing can be performed using behavioral modification techniques, such as conditioned play audiometry or visual-reinforcement audiometry [9].
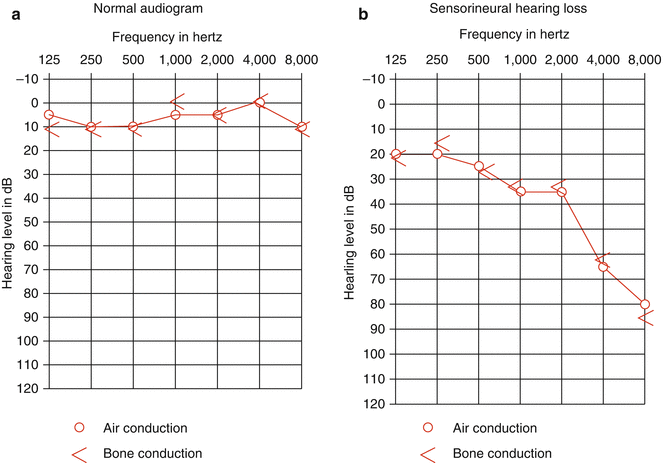

Fig. 8.7
(a) Normal audiogram. (b) Audiogram demonstrating severe high-frequency sensorineural hearing loss (85 dB at 8,000 Hz and 60 dB at 4,000 Hz). Hz Hertz, dB decibels
8.4.2.2 Brainstem Auditory-Evoked Response
For patients unable to cooperate with behavioral testing due to young age, cognitive impairment, and/or medical illness, hearing can be assessed via brainstem auditory-evoked response (BAER, ABR). Electrodes are placed on the skull, and sound is presented at intensity levels that elicit an electrophysiological response, recorded as a waveform. The intensity of the stimulus is then lowered until the response is no longer measurable; this level corresponds roughly to the threshold measurements used in pure-tone audiometry. In order to determine hearing sensitivity for purposes of ototoxicity monitoring, frequency-specific tone-burst stimuli (rather than broadband clicks) should be used during the assessment [11]. The patient must remain motionless throughout this test; therefore, sedation is almost always required for infants and young children (as reviewed in [88]).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree