Evaluation
Growth
Height, bone age, somatomedin C, IGFBP-3
TBI and/or cranial irradiation: refer to endocrinologist for GH provocative testing; growth hormone may be indicated
Thyroid
Physical assessment, free T4, T3, TSH
Elevated TSH: may repeat in 3–6 months if mild elevation; thyroxine replacement
Reproductive
Tanner assessment
Postpubertal females: estrogen replacement; prepubertal children: refer to endocrinologist if delayed puberty or low testosterone/estradiol
Males: FSH, LH, testosterone (>13 years)
Females: FSH, LH, estradiol (>12 years)
Glucose metabolism
Height, weight, fasting cholesterol, triglycerides
There have been some reports following chemotherapy-only conditioning of growth disturbances [1, 4]. However, most children who receive chemotherapy-only conditioning regimens have relatively normal growth, although both the long-term use of steroids for chronic GVHD and prior cranial irradiation negatively affect growth [38, 72, 73].
18.2.2 Thyroid
Thyroid dysfunction is a well-described complication after HSCT. Although it occurs most commonly after TBI, there are reports of thyroid dysfunction in patients who received chemotherapy alone. The developing thyroid gland may be more susceptible to damage as the incidence is increased in children below the age of 10 years and the highest risk group is patients undergoing mantle radiation for treatment of Hodgkin’s lymphoma with an overall incidence of 73 % [13]. The incidence varies widely in studies, depending upon the technique of TBI. Patients who receive fractionated TBI have a 15–20 % incidence of hypothyroidism [6, 32, 68]. Compensated primary hypothyroidism has a median time of onset of 12 months post HSCT and may be transient [32]. Replacement therapy is indicated for persistent elevation of TSH. Patients who received TBI should have annual physical assessments of the thyroid gland as radiation increases the risk of thyroid cancer.
Compensated primary hypothyroidism has been noted in 10–15 % of patients who received chemotherapy without radiation therapy [2, 70]. The etiology of hypothyroidism in this group is unclear; but it is believed that the high doses of drugs associated with chemotherapy may be involved. Chemotherapy-only regimens have been associated with euthyroid sick syndrome, in which the T3 and T4 are low, while the TSH is either normal or low. This syndrome has been reported in 29 % of patients at 14 months post therapy [70]. The clinical significance of this is not clear.
18.2.3 Diabetes and Metabolic Syndrome
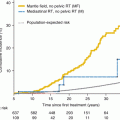
Survivors of HSCT may be at risk for insulin resistance, impaired glucose tolerance, and type 2 diabetes [66]. Risk factors relevant to the development of these problems include obesity, family history of diabetes, inactivity, inadequate diet, use of growth hormone, and race. In one study of 748 patients evaluated for type 2 diabetes, 34 had developed this condition at a median follow-up of 11 years. The prevalence of type 2 diabetes was 9 % among the survivors of leukemia, with CML patients at highest risk [23]. The prevalence was age-related, with 12 % occurring among leukemia survivors 20–39 years old and 43 % occurring among survivors 40–49 years. The prevalence of diabetes type 1, although less common, was three times higher than in the general population. Most patients evaluated were not obese and experienced a relatively early onset of type 2 diabetes. Racial minorities were more likely to develop diabetes; TBI was not a risk factor in this analysis (Table 18.2).
Table 18.2
Organ systems: suggested follow-up
Organ | Suggested evaluation |
|---|---|
Heart | ECG, echocardiogram, exercise stress test: yearly to every 3–5 years |
Pulmonary | PFTs: yearly to every 3–5 years |
Renal | Blood pressure, urinalysis, BUN, creatinine, erythropoietin level if evidence of thrombotic microangiopathy |
Dental | Semiannual dental evaluation |
Hearing | Audiogram for patients at risk (prior cisplatin, carboplatin, cranial irradiation) |
Eyes | Annual ophthalmologic evaluation, artificial tears if sicca syndrome |
Bone | Physical assessment, bone density evaluation (DXA, pQCT) |
Brain | Neuropsychologic assessment 1–3 years post BMT in patients at risk (<6 years with TBI, hearing loss) |
Hyperinsulinemia and hypertriglyceridemia [23] have been described post HSCT. Therefore, post-HSCT patients, particularly those who were treated for leukemia, merit close observation for the development of diabetes, as well as lipid abnormalities.
18.2.4 Reproductive
Gonadal dysfunction is common following HSCT, a finding attributable to the use of alkylating agents, such as cyclophosphamide, and radiation therapy. Busulfan, as a stem cell toxin, also causes a high incidence of gonadal failure [2, 67]. Gonadal dysfunction results in infertility in most affected patients, with some patients also having difficulties with pubertal development.
In males, the Sertoli and germ cells are more vulnerable to radiation and chemotherapy than the Leydig cells. FSH levels are usually elevated, with normal LH levels. Testosterone levels may be normal with reduced or absent spermatogenesis. Most boys will undergo spontaneous pubertal development without the addition of testosterone. Testicular irradiation used for treatment of leukemic testicular relapse is associated with damage to Leydig cells and low testosterone levels. Boys who have undergone testicular irradiation should be followed closely as most will need long-term testosterone replacement.
Estrogen is necessary for breast development and for the normal pubertal growth spurt. In prepubertal females who undergo HSCT, the recovery of ovarian function may be more likely than in postpubertal [55], but approximately 70 % will have hypergonadotropic hypogonadism and require estrogen replacement therapy [56]. Failure of progression through puberty is often an indication for the need for estrogen replacement. Patients without breast development who have increased FSH and LH levels should be treated with estrogen/progestin. The dose of hormonal replacement will require adjustment with age in order to ensure progression through puberty and cyclic menstruation. Estrogen replacement for postpubertal females should be initiated 3–6 months after HSCT. It has been suggested that replacement be stopped for 2 months at around 1 year post HSCT to evaluate for ovarian recovery [67]. The incidence of pregnancy is less than 3 % for females who received TBI or busulfan, although there are a few reports of successful pregnancies following TBI [54, 55]. Most males will also be sterile after TBI. However, recent studies show that male recipients younger than 25–30 years at time of transplant and without chronic GVHD have a reasonable likelihood of spermatogenesis recovery even those receiving myeloablative TBI based conditioning. Recovery can be incomplete, resulting in oligospermia [74]. Options for preservation of fertility should be discussed prior to HSCT. For pubertal males, sperm banking should be encouraged. Prior therapy may limit this option for some males. Cryopreservation of testicular tissue in prepubertal males is being done on research basis only as well as cryopreservation of ovarian tissue for prepubertal females. For postpubertal females, cryopreservation of ovarian tissue or oocytes is feasible. Gamete preservation is not yet available on a non-research basis. There have been reports of pregnancy from embryos with donated oocytes, with hormonal support [53]. Pregnancies that do occur following TBI are more likely to result in miscarriage, preterm labor, and low-birth-weight infants [55].
With non-myeloablative conditioning regimens, the endocrine effects, particularly gonadal function, are unknown. Effects may be dependent upon the dose and timing of the agents used.
18.3 Pulmonary
The assessment of the effects of HSCT conditioning on pulmonary function is usually limited by the age of the patient, as very young children cannot perform pulmonary function tests. The spectrum of long-term pulmonary complications differs among patients who received allogeneic vs. autologous HSCT. This is due to the effects of chronic GVHD, which may result in changes called bronchiolitis obliterans. Other factors that may impact pulmonary function post HSCT include prior chemotherapies such as bleomycin, TBI during conditioning, and thoracic irradiation.
There are three categories of PFT abnormalities: obstructive, restrictive, and those that result from a decrease in diffusion capacity. Obstructive abnormalities result in decreased FEV1 and in a decreased FEV1/FVC ratio. These occur as a result of small airway closure or obstruction of expiration. Bronchiolitis obliterans is the most common cause of obstructive abnormalities post HSCT. Radiation, pulmonary infection, and pneumonitis may all result in restrictive lung disease, with decreased total lung capacity (TLC) and preserved FEV1/FVC ratio. Decreased DLCO, or diffusion capacity for carbon monoxide, may be a result of an abnormal alveolar–capillary interface. Anemia will result in a low DLCO as well.
Up to 85 % of patients will have abnormal PFTs 3–6 months post HSCT, with a restrictive pattern being the most common abnormality [9]. Late abnormalities are often associated with chronic GVHD, although prior aggressive therapy for advanced-stage disease may also have a negative impact on pulmonary function [9]. The most common late abnormality is a decrease in DLCO, followed by restrictive defects [37]. GVHD is associated with the risk of chronic aspiration pneumonia, particularly in patients with esophageal involvement. GVHD is also associated with the risk of bronchiolitis obliterans (BO) in up to 37 % [59] of post-HSCT patients and with the risk of bronchiolitis obliterans organizing pneumonia (BOOP).
Patients with BO do not present with fever, but have complaints of exercise intolerance, cough, and wheezing. Chest radiograph may be normal, and PFTs will show obstructive defects. Immunosuppressant agents are usually not therapeutically helpful. Patients with bronchiolitis obliterans organizing pneumonia (BOOP) present with fever, cough, dyspnea, and rales. BOOP may be present as early as 1 month post HSCT, but is more common after 3 months. In patients suffering from this condition, there is a patchy distribution of granulation tissue plugs filling the lumens of airways and extending into the alveoli [17]. BOOP is strongly associated with prior acute or chronic GVHD, as well as prior leukemia [17]. Radiographic findings may include patchy alveolar opacities and asymmetric infiltrates.
In a small study of children who received autologous HSCT, restrictive impairment was found in 20 % of patients at 5–10 years [18], but there was stabilization after the first year. Obstructive impairment was rare, but diffusion impairment was found in over 50 % of patients at 10 years. TBI was associated with decreased lung volumes.
18.4 Cardiac
Cardiotoxic drugs, particularly the anthracyclines, used for disease treatment prior to HSCT conditioning may increase the risk of cardiotoxicity post HSCT, although most patients are asymptomatic despite changes on ECG and echocardiograms. Factors that may increase the risk of cardiotoxicity include young age at the time of treatment, high-dose cyclophosphamide, and chest irradiation. In addition, HSCT survivors are at an increased risk for developing cardiovascular risk factors such as hypertension and metabolic syndrome [4, 42]. Posttransplant echocardiograms have been reported as normal in most children [38], but some studies have reported decreased systolic function [32, 48]. However, pre-transplant anthracycline administration and TBI increase risk for reduced cardiac function [71]. Exercise testing may be a more sensitive tool for detecting changes in cardiovascular function over time; however, it is limited to patients who are old enough to perform such testing. A retrospective study of serial cardiopulmonary exercise tests noted that despite decreases in all parameters of exercise performance, both aerobic and physical working capacity increase over time [24]. This study suggests that oxygen extraction becomes more efficient with recovery and that it may compensate for impaired cardiac ability.
18.5 Renal
Renal toxicity post HSCT may occur as a result of TBI or as a result of nephrotoxic drugs commonly used such as cyclosporine or tacrolimus, a history of cisplatin administration, or conditioning with carboplatin. Thrombotic microangiopathy, a syndrome which includes endothelial injury resulting in microangiopathic hemolytic anemia and platelet consumption, may increase the risk of chronic renal injury [34]. This syndrome may be associated with GVHD and TBI, and patients requiring dialysis have a poor prognosis [21]. Nephrotic syndrome has also been associated with chronic GVHD [52].
18.6 Ocular
Cataract formation and keratoconjunctivitis sicca (dry eye) syndrome are the two most common ocular complications for patients post HSCT. Risk factors for cataracts include TBI schedule, type of transplant, development of GVHD, and prolonged use of steroids. Cataracts are usually posterior subcapsular, in contrast to those seen in older adults, which appear in the central part of the lens. Cataracts are seen in up to 80 % of patients who received unfractionated TBI, but are less common in patients receiving fractionated TBI, with incidences of approximately 20 % [38]. They may often occur after 4 years (median, 98 months after fractionated TBI); annual follow-up is, therefore, extremely important. Surgical repair may be necessary for some patients, but this is not commonly required. Clinically significant cataracts are noted only occasionally in patients who received non-TBI regimens, and more often than not, this may be related to corticosteroid exposure.
The incidence of keratoconjunctivitis sicca syndrome reaches 20 % 15 years after stem cell transplantation. The ocular manifestations include reduced tear flow, conjunctivitis, corneal defects, and corneal ulcerations. Chronic GVHD is the greatest risk factor, with late-onset keratoconjunctivitis occurring in 40 % of patients with chronic GVHD, versus 10 % of patients without GVHD. Other risk factors for late-onset keratoconjunctivitis include female gender, age greater than 20 years, single-dose TBI, and the use of methotrexate for GVHD prophylaxis [3].
18.7 Dental
Damage to dentition associated with HSCT is generally the result of irradiation. Side effects that have been reported include disruption in normal enamel development, hypoplasia, microdontia of the crowns of erupted permanent teeth, and thinning and tapering of the roots of erupted permanent molars. Cranial irradiation prior to TBI may further increase the risk of tooth agenesis [68] as can chemotherapy in young children.
Chronic GVHD may result in damage to the oral cavity and salivary glands. A significant reduction in the show of saliva, as measured by sialometry and salivary gland scintigraphy, has been noted in patients with acute and chronic GVHD. This reduction may persist in patients who received TBI [41]. Decreased salivation and poor oral hygiene post HSCT may increase the risk of dental caries.
18.8 Ototoxicity
Children who receive platinum-based agents prior to, or as part of, the conditioning are at the highest risk for developing hearing loss after transplantation, and more than 80 % may experience significant loss [46, 49]. Depending on the chemotherapy history, local or total body irradiation can accentuate hearing loss in children with solid tumors such as neuroblastoma and brain tumors. For patients with neuroblastoma, hearing loss prior to transplant portends significant hearing loss after a regimen containing high-dose carboplatin [46 ]. However, even those children who did not have a hearing loss prior to transplant developed hearing loss after receiving carboplatin as part of the conditioning.
18.9 Bone Mineral Density
There are increasing data on the effects of HSCT upon bone mineral density (BMD). Factors that may impact BMD include prior therapy for malignancies, conditioning regimens, lack of physical activity, poor nutrition, and post-HSCT therapy with calcineurin inhibitors and corticosteroids [44]. Post-HSCT hypogonadism may also negatively impact bone mineral density. One recent study showed nadir BMD at month 24 for total body and femoral neck [58] in patients who received allogeneic HSCT. BMD continuously declined at the femoral neck sites. Steroids and cyclosporine use, as well as loss of muscle mass, were associated with low BMD. Only very young patients were protected from bone loss. The relationship between BMD changes and fracture risk is not yet established post HSCT.
Osteochondromas are benign bone tumors that consist of projecting mature bone capped by cartilage. Radiation, including TBI, is generally believed to be the cause of osteochondromas. The pathogenesis of these bone tumors is not well understood. The mean latent time from HSCT to the development of osteochondromas was 4.6 years in one study [65] (NBL paper by Ginsberg et.al most recent), and younger patients (less than 5 years old) were at increased risk. Of patients less than 5 years old at the time of TBI, an osteochondroma occurred in 24 % [65]. There is a low malignant potential for these tumors, but they do cause a great deal of anxiety when found and can be painful depending on the location.
Osteonecrosis (ON) is a condition that presents as vague, diffuse bone pain, most likely because of increased intraosseous pressure, or joint-related pain because of an effusion. It was first reported as a posttransplant complication in 1980s. Once subchondral collapse occurs, arthritic-type joint pain predominates and is accompanied by decreased range of motion. The most common sites of destruction in children are knees, hips, and shoulders [8].
There are multiple risk factors for the development of ON which are TBI-based conditioning regimens and the use of corticosteroids both before and after HSCT [14]. Treatment involves minimizing exposure to corticosteroids, use of analgesics for pain, and physical therapy designed to focus on non-weight-bearing exercise [31]. If symptomatic, treatment is targeted to preserve the joint and control the pain. Some of these include core decompression, use of bisphosphonates, and joint replacement [50]. Guidelines and consensus on treatment are lacking.
18.10 Neuropsychologic
Conditioning regimens containing high drug doses and radiation that may be neurotoxic can result in neuropsychologic sequelae. Agents that are associated with neurotoxicity include busulfan, thiotepa, and melphalan. Although much is known about the neuropsychologic effects of cranial irradiation for leukemias and brain tumors, there is relatively little known about the outcomes of children post HSCT. Doses of radiation used for central nervous system leukemia are between 1,800 and 2,400 cGy and for brain tumors up to 6,000 cGy. Although the total doses used for TBI are may be lower (1,000–1,400 cGy) than used in other situations, the biologic effect enhanced by the shorter duration of radiation over 3–4 days compared to radiation therapy for leukaemia or solid tumors. Children of different ages may be impacted by TBI in different ways; children under 3 years old may be at higher risk than older children, and they may experience declines in cognition and school performance [33, 60]. Other small studies have suggested that no cognitive impairment occurs even with TBI [47, 61]. However, executive functioning has not been well studied, and it is suggested that this may be affected by conditioning. Chemotherapy-only regimens rarely show a detrimental effect of cognitive function, although hearing loss in children with neuroblastoma may have a significant effect on verbal IQ [43]. Prior cranial radiation increases the risks.
18.11 Other Issues Post HSCT
18.11.1 Chronic GVHD
The incidence of chronic GVHD is lower in children compared with adults, but is responsible for significant post-HSCT morbidity. Chronic GVHD occurs in approximately 20 % of children who receive matched sibling donor transplants and 40–60 % who receive unrelated donor hematopoietic stem cells. The use of mobilized peripheral stem cells increases the risk of chronic GVHD [36, 57]. The most significant risk factor for chronic GVHD is prior acute GVHD; other risk factors include older age, a female multiparous donor, or an unrelated or partially matched related donor [35]. Chronic GVHD may be classified as progressive, evolving from acute GVHD; quiescent, following a period of resolution from acute GVHD; or de novo, in patients who had no prior acute GVHD. Chronic GVHD is associated with a decrease in leukemic relapse risk, although it may come at a price with a significant impact upon quality of life and increase in transplant-related mortality. Classification of chronic GVHD is difficult and currently undergoing revision.
Current classification of chronic GVHD is “limited” or “extensive.” Limited GVHD refers to localized skin involvement with or without hepatic test abnormalities, and extensive refers to generalized skin involvement or involvement of other organs. Poor prognostic factors include progressive onset, platelets <100 × 109/ml, and poor performance status.
The etiology of chronic GVHD is not well understood. Many manifestations resemble autoimmune diseases, with loss of normal T-cell regulation considered to be a possible cause.
18.11.2 Clinical Features
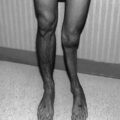
The manifestations of chronic GVHD usually occur after day 100 post HSCT. Almost all patients are diagnosed within the first year post HSCT. The effects of chronic GVHD vary, depending upon location and severity of involvement (Table 18.3). The skin is the most commonly affected organ, with over 50 % of patients with chronic GVHD having some degree of skin involvement. Diagnosis may be confirmed with a biopsy if other diagnoses (particularly infection) are being considered.
Table 18.3




Chronic GVHD: clinical features
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree