Introduction
Cushing’s syndrome (CS) is a well-established endocrine disorder resulting from persistent exposure to glucocorticoids, originating either endogenously or, more commonly, from exogenous sources [ , ]. The subclinical form of CS may be overlooked or diagnosed late. Because it is associated with significant morbidity and mortality, it is crucial to diagnose early and accurately subtype CS to prevent disease-related complications [ , ].
Exogenous or iatrogenic CS is associated with the prolonged or excessive use of glucocorticoids. This could be related to different forms of glucocorticoid intake, including oral, topical, injectable, or inhaled. Given the widespread application of glucocorticoids in the management of inflammatory, autoimmune, and neoplastic disorders, obtaining a comprehensive medication history is essential in the first diagnostic step of CS [ , ]. Endogenous CS is categorized into ACTH (adrenocorticotropic hormone)-dependent and ACTH-independent subtypes. The predominant etiology of the ACTH-dependent subtype is represented in Cushing’s disease (CD), which is often due to an ACTH-secreting pituitary neuroendocrine tumor (PitNET) of T-box pituitary transcription factor (TPIT) lineage (known as corticotroph tumor), accounting for 70%–80% of cases of endogenous CS. Additional instances of the ACTH-dependent subtype may result from pituitary corticotroph cell hyperplasia (rare) and ectopic ACTH secretion (10%–20%) originating from various neuroendocrine neoplasms (NENs) located in different anatomical sites (lung, thymus, thyroid, adrenal medulla, appendix, and pancreas). Furthermore, a subset of NENs can also manifest with CS by secreting corticotropin-releasing hormone (CRH) [ ], occurring in less than 1% of CS. ACTH-independent manifestations resulting from primary cortisol-producing adrenocortical lesions account for around 15%–25% of overall manifestations.
This chapter provides an update on the clinicopathological correlates of ectopic ACTH- and CRH-producing tumors manifesting with CS.
Epidemiology and etiology
The incidence of CS is approximately 2–3 per million people annually [ , ]. Ectopic CS (ECS) was first identified and studied in detail as early as the 1960s [ , ]. ECS is a rare disease, accounting for 10%–20% of all CS and around 15%–25% of ACTH-dependent CS patients [ , , ].
ECS is most frequently caused by ACTH-producing epithelial NENs in the lungs, mediastinum, pancreas, and thyroid, as well as nonepithelial NENs originating from extra-adrenal paraganglia or the adrenal medulla. Particularly, small cell lung carcinoma (SCLC), pancreatic NENs, thymic NENs, pheochromocytoma and paraganglioma (PPGL), and medullary thyroid carcinoma (MTC) account for the vast majority of ECS. Less than 1% of cases can occur in the context of ectopic CRH production, mainly by epithelial or nonepithelial NENs [ , , ].
Besides PitNETs, hypothalamic gangliocytomas can lead to CS [ , ]. Gangliocytomas, whether pure or in association with adenohypophyseal tumors (e.g., PitNETs), constitute a small fraction of sellar tumors (approximately 0.25%–1.25%). In addition to their capability to produce ACTH and CRH, gangliocytomas have also been observed in association with other hormone production, such as growth hormone-releasing hormone (GHRH), prolactin (PRL), and vasopressin [ ]. Among these, vasopressin secretion by gangliocytomas can also result in corticotroph hyperplasia and CD [ ].
There is no specific etiology responsible for the development of ECS; however, a subset of patients may be seen in the context of familial endocrine tumor syndromes (see below).
Genetics and molecular findings
Patients with ECS may harbor pathogenic constitutional variants related to familial endocrine tumor syndromes that manifest with NENs. For instance, MTC (C-cell NEN) can be a manifestation of multiple endocrine neoplasia type 2 (MEN2), and PPGLs can be components of genetic syndromes, including but not limited to MEN1 ( MEN1 ), MEN2 ( RET ), MEN5 ( MAX ), succinate dehydrogenase (SDH) deficiency syndrome ( SDHA, SDHB, SDHC, SDHD, SDHAF2 ), von Hippel-Lindau ( VHL ) disease, and neurofibromatosis type 1 ( NF1 ) syndrome [ , ]. However, no somatic variants have been directly linked to the development of ECS.
There are no distinct molecular features of NENs that are directly linked to ACTH/CRH secretion. However, a recent study identified different molecular mechanisms in ACTH-secreting pancreatic NENs compared to their nonpancreatic counterparts [ ]. In that study of four pancreatic and seven nonpancreatic tumors, targeted RNA sequencing showed that only the four pancreatic NENs had fusions consisting of EWSR1::BEND2 in two tumors and KMT2A::BCOR and TFG::ADGRG7 in the other two tumors [ ].
Clinical, radiological, and biochemical findings
CS can result from a variety of pathogenetic mechanisms, but the primary cause is chronic exposure to high levels of cortisol. While there is no single pathognomonic symptom for adult CS, an elevated cortisol level alone is insufficient to distinguish the source of hypercortisolemia. Therefore, a comprehensive patient evaluation, including clinical, radiological, and biochemical findings, is crucial for accurate diagnosis. This excess cortisol can arise from different sources, including but not limited to PitNETs, adrenal tumors, ectopic ACTH or CRH production, and exogenous corticosteroid use. Although ACTH-producing PitNETs are the most common cause of CS, distinguishing between ACTH-dependent and independent CS and ACTH-producing ectopic tumors poses challenges due to overlapping clinical phenotypes. These groups differ in clinicopathological, radiological, and biochemical features despite some similarities.
The common clinical findings of CS include weight gain, central obesity, round face, facial plethora, dorsocervical fat pad, purple striae, hypertension, hirsutism, osteoporosis, muscle weakness, glucose intolerance, menstrual irregularities, easy bruising, and emotional changes [ , ]. The typical signs and symptoms of CS do not occur in all patients. In patients with a suspicion of CS, the use of exogenous glucocorticoids should be excluded, and an appropriate diagnosis should be made subsequently. On the other hand, pseudo-CS is characterized by physiological and nonneoplastic endogenous hypercortisolism causes such as psychiatric conditions, alcohol dependence, obesity, diabetes mellitus, chronic kidney disease, and pregnancy, which should be excluded as well [ , ].
ECS also presents diagnostic challenges due to chronic exposure to excessive ACTH/CRH from nonpituitary neoplasms. The gender distribution in ECS patients is equal, and they typically tend to be older than most ACTH-dependent manifestations [ ]. The time of diagnosis of CS is 14 months (95% CI 11–17) for ECS, which is earlier than adrenal CS and pituitary CS, 30 months (95% CI 24–36), and 38 months (95% CI 33–43), respectively [ ].
The clinical manifestations of ECS relate to the cortisol level and how long the patient has been exposed to hypercortisolemia. Symptoms of ECS-related hypercortisolemia encompass muscle weakness, hypertension, menstrual irregularities, hirsutism, osteoporosis, central obesity, increased body weight, psychiatric disorders like anxiety and depression, hypokalemia, hyperglycemia with insulin resistance, and central obesity [ , ]. Hypertension, diabetes mellitus, metabolic syndrome, myopathy, and osteoporotic fractures have a higher prevalence in ECS patients [ ]. Severe hypercortisolemia, often characterized by rapid onset, is primarily attributed to ACTH secretion from SCLC. This condition typically manifests with profound weakness and hypokalemia, while weight gain is minimal and classical Cushingoid features are absent [ , ].
The two clinical phenotypes that have been described for ECS include aggressive and indolent forms of ECS [ ]. Aggressive ECS is related to SCLC (a poorly differentiated pulmonary neuroendocrine carcinoma (NEC) with small cell cytomorphology) and often presents with atypical symptoms and markedly elevated cortisol and ACTH levels [ , , ]. Rapid clinical onset occurs over a few months, and the underlying tumor is often diagnosed in advanced stage, and treatments are largely ineffective due to high recurrence rates. Protein catabolism-related symptoms such as severe myopathy, hypokalemia, therapy-resistant hypertension, hyperglycemia, and hyperpigmentation characterize the clinical profile of aggressive ECS [ , ]. In contrast, indolent ECS is usually related to ACTH-producing bronchial well-differentiated epithelial NET, and its clinical finding is similar to ACTH-dependent CS, which is characterized by slightly elevated cortisol and ACTH levels [ ].
In patients with ECS, SCLC and other pulmonary epithelial NENs (e.g., carcinoids) are the most common sources of ectopic ACTH secretion. Therefore, a significant part of the primary tumor is above the diaphragm (approximately 30%–40% in the lung). Therefore, for patients with a biochemical investigation suggestive of ECS, it is recommended that patients undergo contrast-enhanced thin-section computed tomography (CT) from neck to abdomen to investigate the underlying neoplastic source of ECS [ ]. Lung NETs are typically small, round lesions located peripherally and close to the pulmonary vessels and may be confused with pulmonary vascular shadows [ ]. The diagnostic performance of CT is considered to be superior to magnetic resonance imaging (MRI) in the ECS, especially for tumors in the lung [ ]. However, bronchial NETs may not be detected on imaging because they may be occult tumors. Therefore, functional imaging studies are very useful for staging, localizing the tumor, and confirming the diagnosis of NEN. To detect the tumor localization, 111 In-pentetreotide scintigraphy (Octreoscan), and more frequently, 68 Gallium-somatostatin receptors ( 68 Ga-SSTR) PET/CT are considered [ ]. In a systematic review, lung lesions were detected with the highest sensitivity by CT (79.4%), 68 Gallium-SSTR-PET/CT (77.8%), F-DOPA-PET (71.4%), and MRI (66.7%). Tumors with high metabolic activity, such as pancreatic high-grade NETs or NECs, or SCLCs, are frequently positive by fluorodeoxyglucose positron emission tomography (FDG-PET) [ ]. Ultrasound and CT can also be used as the first option to detect MTC (C-cell NENs) and PPGLs. However, functional imaging with PET/CT, such as 68 Gallium-DOTATATE and 18 F-DOPA-PET/CT, has higher sensitivity in detecting tumors than conventional imaging [ , ].
The initial test for biochemical evaluation includes a 24-hour urine-free cortisol test, a late-night salivary cortisol measurement, and a dexamethasone suppression test (DST) [ ]. The abnormal results of at least two tests are usually warranted for the accurate diagnosis of CS. When high plasma ACTH levels are detected, CS is confirmed to be ACTH-dependent, and adrenal CS is excluded. The distinction between pituitary and ectopic sources of ACTH can be determined through three tests: pituitary MRI, high-dose DST, and desmopressin or CRH stimulation test [ , , ]. Detecting a tumor larger than 6–10 mm on MRI favors the diagnosis of CD; in such cases, further testing with bilateral inferior petrosal sinus sampling is unnecessary [ , ]. In CD, strong responses for ACTH and cortisol to CRH and desmopressin, and suppression with the high-dose dexamethasone test are expected. Conversely, in patients with ECS, ACTH levels remain unchanged upon CRH or desmopressin stimulation or in response to dexamethasone [ ]. CRH stimulation remains a valuable test since, unlike ectopic ACTH-related ECS, corticotroph tumors have CRH receptors that result in response to CRH via excess secretion of ACTH. As a result, the majority of CD patients exhibit elevated plasma ACTH and cortisol concentrations following CRH stimulation. Typically, patients with ECS do not respond to tests. However, some tumors, such as rare bronchial well-differentiated NETs (also known as carcinoids), may express CRH receptors [ , ]. If all three tests do not support a pituitary source, inferior petrosal sinus sampling may also be considered for accurate diagnosis. Cyclic CS, characterized by changing cortisol levels, can be more challenging and may require repeat tests [ ].
Neoplasms causing ectopic ACTH or CRH production
In addition to clinical, biochemical, and radiological findings, pathological examination is critical in diagnosing CS. Various tumors with ectopic ACTH/CRH production have been described ( Table 9.1 ). The pathological classification of these tumors is essential for the clinical management of patients with ECS. Since most patients with ECS have an underlying NEN, it is important to recognize the modern World Health Organization (WHO)/IARC nomenclature framework applied to NENs. The term “NEN” represents an umbrella term to cover the three groups of neoplasms that were defined based on the epithelial-paraneuronal origin (e.g., epithelial, nonepithelial), differentiation, and proliferative features. These include (i) epithelial NENs consisting mainly of well-differentiated NETs and poorly differentiated NECs (large cell NEC, small cell NEC, and Merkel cell carcinoma), (ii) nonepithelial NENs consisting of paragangliomas and adrenal paragangliomas (pheochromocytomas), and (iii) paraganglioma-like NENs consisting of COGNETs (composite gangliocytoma/neuroma and NET) and cauda equina NETs [ ].
Table 9.1
Neoplasms that can cause ectopic Cushing’s syndrome.
| Tumor type | |
|---|---|
| Epithelial neoplasms |
Epithelial neuroendocrine neoplasm
Bronchial NET/NEC Small cell lung cancer (small cell NEC of the lung) Pancreatic NET/NEC Medullary thyroid carcinoma Thymic NET/NEC Epithelial NET/NEC of various sites Non-neuroendocrine neoplasm (paraneoplastic syndrome) Colorectal adenocarcinoma Prostatic adenocarcinoma Others |
| Nonepithelial neoplasms |
Paraganglioma
Pheochromocytoma (adrenal paraganglioma) Neuronal neoplasms (e.g., gangliocytoma) |
NET , Neuroendocrine tumor; NEC , Neuroendocrine carcinoma.
PPGLs are paraneuronal NENs originating from paraganglia that are aligned with components of the autonomous nervous system. While all NENs are malignant neoplasms irrespective of their types, epithelial well-differentiated NENs are defined as NETs (graded from grade 1 to 3 based on proliferation rate in the gut, pancreas, and upper aerodigestive tract; criteria of grading vary based on the anatomic sites and are not discussed in this chapter), and epithelial poorly differentiated NENs are defined as NECs [ , ]. MTC with the carcinoma suffix is a rule breaker in the nomenclature framework of epithelial NENs, since MTCs are mostly well-differentiated NETs of C-cell origin, but poorly differentiated forms have also been recognized [ , ].
From a morphological perspective, well-differentiated NETs demonstrate nested, trabecular, solid, pseudo-glandular, or varying growth combinations of these patterns, which are indistinguishable from their nonepithelial counterparts. Typically, the cells exhibit relatively small, uniform features with regularly shaped, round-to-oval nuclei and have a low nuclear-to-cytoplasmic ratio. Tumor nuclei have granular chromatin, often described as a “salt and pepper” appearance. The cytoplasm ranges from pale to moderately eosinophilic depending on the characteristics of the hormone content of neurosecretory granules. In contrast, NECs are aggressive malignancies characterized by frequent mitotic activity (in the lung, thymus, and aerodigestive tract exceeding 10 mitoses per 2 mm 2 , and in the gut and pancreas exceeding 20 mitoses per 2 mm 2 ) along with severe cell polymorphism, nuclear molding, high nuclear/cytoplasmic ratio, abundant necrosis, and elevated Ki67 (MIB1) labeling index [ ]. While the cytomorphological features are distinctive for most NENs, immunohistochemistry is essential to distinguish NENs and their subtypes from other neoplasms, assist the confirmation of the anatomic origin, and provide prognosticators and insights into their pathogenesis. Therefore, the detailed assessment of the morphology and application of appropriate immunohistochemical biomarkers are essential for the accurate diagnosis and appropriate subtyping of neoplasms leading to ECS. Ectopic ACTH/CRH-secreting NENs include the well-differentiated pulmonary NETs as well as poorly differentiated NECs (e.g., SCLC), pancreatic epithelial NENs ( Fig. 9.1 ), thymic epithelial NEN, MTC (C-cell NENs), and PPGLs ( Figs. 9.2 and 9.3 ) [ ].
Ectopic Cushing’s syndrome due to ACTH- and CRH-producing well-differentiated pancreatic neuroendocrine tumor with high-grade features (Grade 3 pancreatic NETs).
This composite photomicrograph illustrates the characteristics of a well-differentiated pancreatic neuroendocrine tumor (A). Consistent with an epithelial neuroendocrine neoplasm, the tumor is diffusely positive for the low molecular weight keratin CAM5.2 (B), INSM1 (C), synaptophysin (not shown herein), and chromogranin-A (not shown herein), and is negative for PHOX2B (D). The tumor is diffusely positive for ACTH (E), whereas focal reactivity for CRH (F) is also noted. Among pancreatic hormones, the tumor shows focal reactivity for pancreatic polypeptide/PP (G). The Ki67 (MIB1) labeling index is 21% in hot spots (H). The tumor shows neither abnormal p53 reactivity (not shown here) nor retinoblastoma (RB) loss (not shown here).
Ectopic Cushing’s syndrome due to ACTH-producing adrenal paraganglioma (pheochromocytoma).
This composite photomicrograph illustrates the morphological features of a pheochromocytoma (A) that shows near diffuse reactivity for ACTH (B). Consistent with the diagnosis of pheochromocytoma, the tumor shows diffuse reactivity for GATA3 (C) and tyrosine hydroxylase (D).
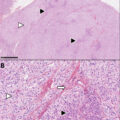
Ectopic Cushing’s syndrome due to CRH-producing adrenal paraganglioma (pheochromocytoma).
This photomicrograph illustrates focal reactivity for CRH in the tumor. This finding underscores that hormone expression can be variable in neuroendocrine neoplasms. Therefore, any reactivity should be carefully recorded given its clinical significance.
The assessment of the peptide profile of a NEN is of clinical significance, as it can serve as a biochemical screening tool and also assess the status of aberrant hormone expression, as in the case of ECS. The pathological confirmation of ACTH/CRH production in a NEN often relies on immunohistochemical testing of ACTH/CRH to confirm aberrant hormone expression ( Fig. 9.1–9.3 ) rather than relying solely on morphological features. There is no cytologic or morphological harbinger of ectopic hormone expression, and the morphology of tumors with ECS is no different than their nonsecreting counterparts. The extent of ACTH/CRH reactivity can also be variable in tumors ( Fig. 9.1–9.3 ). Furthermore, it is important to recognize that the immunohistochemical expression of CRH and/or ACTH does not necessarily indicate the presence of ECS. Therefore, the clinical and laboratory findings are essential when assessing the correlates of aberrant ACTH/CRH reactivity in NENs. In some tumors, both ACTH and CRH are expressed together in varying proportions ( Fig. 9.1 ). Isolated CRH-producing tumors are rare ( Fig. 9.3 ); however, around half of pure CRH-secreting tumors are MTC and PPGLs [ , , ].
In the setting of appropriate cytomorphological characteristics, a diagnosis of NEN is confirmed when diffuse neuroendocrine differentiation is confirmed using at least two of the following biomarkers, including INSM1, synaptophysin, and chromogranin [ , ]. The next step relies on the assessment of the status of epithelial and nonepithelial NENs. Most epithelial NENs are virtually all positive for low molecular weight keratins (CAM5.2, CK8, CK8/18) ( Fig. 9.1 ) and express endoderm-related transcription factors depending on their developmental origins. Unlike epithelial NENs, nonepithelial NENs are negative for keratins but express special transcription factors including PHOX2B, HAND2, and GATA3, as well as enzymes in the catecholamine biosynthesis (e.g., commonly used rate-limiting enzyme “tyrosine hydroxylase”) ( Fig. 9.2 ) [ , ].
Unlike NECs, most NETs tend to preserve their developmental differentiation pathways related to transcription factor expression. For instance, MTCs and pulmonary NETs are TTF1-positive epithelial NENs. Both can express calcitonin, but diffuse carcinoembryonic antigen (CEA) reactivity distinguishes MTCs from pulmonary NETs in a patient with ECS and synchronous lung and thyroid lesions. Similarly, the pituitary corticotroph origin is confirmed using TPIT transcription factor reactivity. In a case of poorly differentiated NEC, tumor cells may show any combination of common transcription factor expression irrespective of their anatomic site of origin. For instance, a small cell NEC in the pancreas can also express TTF1, similar to an SCLC. Nevertheless, TPIT reactivity is not regarded as a feature of poorly differentiated NECs of visceral organs.
From a morphological perspective, ACTH-secreting pheochromocytomas can be suspected when assessing the nontumorous adrenal cortex ( Fig. 9.4 ). Unlike pituitary ACTH-dependent diffuse hyperplasia, ectopic ACTH secretion can result in diffuse compact cell adrenal cortical hyperplasia leading to an expanded cortex with a lipid inversion pattern ( Fig. 9.4 ). This distinctive feature can serve as a morphological harbinger of ECS related to an ACTH-secreting pheochromocytoma.
Ectopic ACTH-producing adrenal paraganglioma (pheochromocytoma) and diffuse adrenal cortical hyperplasia.
This composite photomicrograph illustrates morphological features of a pheochromocytoma ( A) that shows diffuse reactivity for ACTH (not shown here). The adjacent nontumorous adrenal cortex shows diffuse adrenal cortical hyperplasia with a lipid depletion pattern characterized by compact cell diffuse hyperplasia (B). This is a morphologic harbinger of an ectopic ACTH-related pathogenesis.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree