Introduction
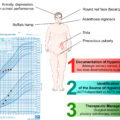
Endogenous Cushing’s syndrome (CS), or hypercortisolism, is a rare medical condition (up to 10 per million) caused by glucocorticoid excess [ ]. This condition may be difficult to recognize owing to overlap with common diagnoses such as obesity, diabetes, and depression, which may lead to delayed diagnoses, even in cases of overt disease. Endogenous CS is most commonly (∼80%) due to pituitary microadenomas which secrete ACTH. These tumors are benign, but there is a substantial risk of recurrence after surgical removal. Cortisol-producing adrenal tumors cause endogenous CS in about 15% of cases; although typically benign adrenocortical adenomas, adrenal tumors may also be malignant (adrenocortical carcinoma, ACC), in which case they may secrete other adrenal hormones or hormone precursors. There is also growing recognition of an entity known as mild autonomous cortisol secretion (MACS), in which an adrenal nodule secretes cortisol in an unregulated fashion, but the symptoms are not sever enough to cause frank CS. This condition may occur in up to 40% of patients with an adrenal nodule [ ]. Lastly, excess ACTH may be secreted from nonadrenal tumors such as neuroendocrine tumors (NETs), small-cell lung cancers (SCLCs), or carcinoid tumors [ ]. In these cases of ectopic ACTH secretion (EAS), removal of the primary tumor is curative, although small NETS or carcinoid tumors may be difficult to localize, whereas SCLCs frequently present at advanced stages.
Lastly, although endogenous CS is rare, the use of steroids is a common medical practice, including indications for dermatologic or inflammatory/rheumatologic conditions, and glucocorticoids may be a part of many anticancer treatments or as antirejection medications after transplant. Although the association of CS with oral or intravenous steroids may be obvious, the same effects can be seen on occasion with intraarticular or inhaled steroids, with a similar set of symptoms if absorbed doses are high enough.
The actions of cortisol at the organismal level are complex and tissue specific. To understand these actions, there has been significant interest in developing animal models that will allow a detailed dissection of the actions of glucocorticoids at the tissue and cellular level, in addition to assessing overall effects on animal health. In this review, I will summarize prior work on the development of animal models for Cushing syndrome. The bulk of the review will discuss rat and mouse models, but I will include some information about larger animal species that develop CS and may be suitable for studies in certain settings ( Table 3.1 ) .
Table 3.1
Translatability of manifestations of CS to animal models.
| Rodent | Dog | Horse | |
|---|---|---|---|
Metabolic
|
+ | + | + |
| Osteoporosis | + | ? | + |
| Neuropsychiatric (e.g., anxiety) | + | + | ? |
| Increased infections | ? |
+
(skin) |
+
(skin) |
| Skin/hair | + | + | + |
Rodent models for Cushing’s syndrome
Rats and mice have been used for many years to attempt to dissect the effects of glucocorticoids on physiology. It is important to recall that rodents do not produce cortisol, the end-product glucocorticoid that is produced in humans. Instead, corticosterone is the steroid that binds to the glucocorticoid receptor (GR) in the common laboratory rat and mouse species [ ]. However, the biologic effects of corticosterone are generally similar to cortisol in humans. Multiple approaches have been used to study CS in rodents, which model the different etiologies of CS in human patients ( Fig. 3.1 ).
Etiologies of Cushing’s syndrome and mouse modeling approaches. See text for details.
Assessing the phenotype of CS in rodents
Humans with CS exhibit a number of symptoms, including many that are common in the general population and thus present a challenge in diagnostic specificity [ ]. Mice present the advantage of a genetic homogeneity (in many models) and a controlled environment, such that more subtle changes can be identified. For example, objective findings like weight changes can readily be assessed in rodent populations, so that even changes of a few percent can be identified. Glucose intolerance/diabetes in rodents frequently presents with polyuria, polydipsia, and hyperphagia, and these can all be quantified in the experimental setup. The diagnosis can also be confirmed using intraperitoneal (IP) glucose tolerance testing; measurement of area under the curve (AUC) for glucose and insulin can provide sensitive assessments for these biochemical derangements. Bone mineral density can be measured using high-resolution X-rays or via microCT, whereas bone formation and bone turnover can be measured by direct measurement of cortical bone deposition using fluorescent labeling. To assess neuropsychologic effects of steroids in rodents, there is a well-established array of behavioral tests which can assess parameters such as anxiety, depression, muscle strength, and other similar measures which have human correlates. Lastly, rodents can be sacrificed and analyzed pathologically for measures of fat deposition, muscle mass, and so on. Thyroid atrophy has also been identified as a marker of CS in rodents, and they do not normally undergo thymic involution.
Production of CS by administration of exogenous steroids
Rats can be induced to develop CS by the administration of exogenous glucocorticoids in several ways. The earliest studies relied on the oral administration of glucocorticoids, typically dexamethasone (dex) in the drinking water [ ]. Dex can also be administered by other means, including IP or subcutaneous injections, or via osmotic mini-pump [ ]. There has also been the use of cortisol-carrying subcutaneous pellets, which can be implanted under the skin of a rat. The severity of CS produced by these means generally reflects the treatment dose for the steroids, and higher doses induce more severe fat accumulation, glucose regulation, and others [ ].
Similar to rats, mice can be forced to develop CS by a variety of means of providing exogenous steroids [ ]. Administration of 10–30 mg/L of corticosterone can provide replacement steroid dosing in a mouse [ , ], whereas higher doses in the range of 75–100 mg/L will induce a CS phenotype [ ]. Other methods of administration include the use of slow-release pellets [ ] or injection of steroids, either by the intraperitoneal (ip) or subcutaneous route [ , ], or osmotic minipumps [ ]. For these approaches, both in mice and rats, experimental decisions are based on degree and duration of hypercortisolism being modeled. For example, to study osteoporosis or long-term effects of steroids might require treatment over an extended period of time.
Cushing’s syndrome by overexpression of CRH: the power of genetically engineered mouse models
However, the real value of mice models is the capability to use genetically engineered mouse models (GEMM) to model specific aspects of tumorogenesis. Experimental genetically modified mice can be generated by multiple means, including through the use of random mutagenesis. via introduction of transgenes, or through targeted knock-ins (including through utilization of clustered regularly interspaced short palindromic repeats [CRISPR] technology).
Mutagenesis screens
As an example of this approach, one interesting model was derived from an ethyl-nitrosourea (ENU) screen in mice in which progeny were selected for metabolic phenotypes [ ]. This approach identified a mouse with obesity and glucose intolerance; genetic analysis led to the identification of a mutation in the promoter region of the gene encoding CRH at position −120. The mutation led to enhanced activity of the corticotrophin-releasing hormone (CRH) promoter, with clear biochemical evidence for excess glucocorticoid production. Interestingly, ACTH levels were not elevated, and there were no gross abnormalities of the pituitaries or adrenal glands, although adrenal glands were mildly hypertrophic. Analysis of steroid rhythm showed that it was disrupted in the animals. Metabolic analysis confirmed marked obesity (as a heritable trait) as well as diabetes and osteoporosis. The mice also exhibited thin skin and hair loss, as is common in mouse models for CS. This line has been subsequently used for further metabolic analysis of diabetes caused by CS [ ].
Models of CRH-driven Cushing’s syndrome by transgenes
The use of transgenesis in murine models allows a fairly straightforward mechanism for the expression of any gene under the control of an alternate promoter. This approach has been applied to generate multiple models of CS driven by ectopic expression of CRH under a variety of promoters. Human tumors producing ectopic CRH have been described as causing CS, although these are very rare. These tumors are all neuroendocrine in origin and include pheochromocytomas [ ], medullary thyroid carcinomas [ ], and other types of neuroendocrine tumors, some of which cosecrete ACTH or other hormones [ ].
In the first mouse model generated using this approach, ectopic expression of CRH was driven by the metallothionein 1 (MT-1) promoter [ ]. Normally, expression of MT1 is induced by zinc administration, although in this model, no changes were induced by administering zinc to the animals. The mice displayed features of Cushing’s syndrome, including metabolic abnormalities and obesity. More detailed analysis showed that the animals exhibited other features of CS, including muscle atrophy [ ], osteoporosis [ ], and anxiety behaviors [ ].
A generalized approach to tissue-specific expression of CRH has also been produced by placing the CRH structural gene under the control of the widely expressed Rosa26 promoter, but subject to silencing by a lox-STOP-lox cassette placed upstream of the gene. By making these mice with animals expressing cre recombinase in a tissue of interest, CRH may be produced at moderate-high levels in any tissue for which a cre line exists [ ]. When the nestin-cre line was used to drive expression in the CNS, mice were observed to have altered anxiety behavior [ ]. Interestingly, basal ACTH and corticosterone levels were unchanged, although stressing the mice led to enhanced biochemical responses.
The same animals were used to make generalized CRH overexpressing mice, or those solely producing excess CRH in the anterior pituitary [ ]. When CRH was overexpressed globally, mice exhibited signs of CS by 3 weeks, including hair loss and thinning of the skin. Obesity was common as the mice age, and the animals consistently showed increased adrenal weight, as would be expected because of chronic trophic stimulation from CRH-stimulated ACTH. Measurement of corticosterone showed consistent elevation as well, although stressing the animals enabled the littermate controls to produce equivalent amounts of steroid. When the transgene was expressed only in the anterior pituitary (under the control of the Pomc-cre allele), mice developed similar but milder phenotypes, with findings not evident until 5–6 months of age. Body weight were actually very slightly decreased in the transgenics, although AM cortisol was elevated. PM cortisol was similar in the Pomc-cre line. Interestingly, these mice also exhibited sex differences in effects, with males exhibiting more prominent changes. Behaviorally, the generalized overexpressed mice exhibited anxiety-related behaviors, whereas the centrally expressed (Pomc-cre) mice did not. There was also better stress coping activity, similar to what was observed in the MT1-CRH transgenic mice [ ].
A different model for CRH overexpression was generation by placing the rat CRH gene under the control of the Thy1.2 promoter, which drives expression primarily in postmitotic neurons [ ] although expression in other tissues has been described for this promoter [ ]. In this model of CNS-expressed exogenous CRH, animals did not exhibit signs of anxiety or depression [ ] but exhibited a paradoxical decrease in startle reflexes which the authors suggested might be related to observations observed in human schizophrenic patients [ ]. Biochemical analysis did reveal elevated systemic ACTH and steroid levels, as well as nonsuppressibility to dexamethasone [ ]. Interestingly, these animals were found to have reduced body weight, likely as a result of hyperactivity associated with the anxiety behaviors [ ].
To enable both spatial and temporal control of CRH expression, mice were generated in which the coding region for Rat CRH was placed behind a binding site for the tet operon [ ]. When these mice are bred to animals carrying the reverse tet TransActivator protein (rtTA), the gene of interest is expressed when tetracycline is administered to mice. For this study, the CamKII promoter, which directs expression primarily to areas of the forebrain, was used to spatially and temporally direct expression of CRH. When CRH expression was driven in postpubertal mice by 3 weeks of doxycycline, CRH and corticosterone levels were observed to rise. Similar to some of the other models mentioned before, changes in plasma ACTH were not detected in these animals. Modest behavioral changes were observed, which were more subtle compared with other models, possibly related to either the shorter duration of exposure to elevated CRH or the lower maximal CRH production. A similar model [ ] was used to probe the long-term effects of CRH exposure when it was limited to a developmental time window (from conception until postnatal day 15). These mice showed elevation of corticosterone when the transgene was on, but these biochemical abnormalities reverted when the transgene was turn off. However, even after normalization of the biochemical parameters, the adult animals exhibited anxiogenic and “despair-like” behavior, confirming the important developmental timing effects of altering the HPA axis.
Models for pituitary Cushing’s syndrome (Cushing’s disease)
In humans, ACTH-secreting pituitary adenomas causing are the most common causes of CS [ , ]. However, reports of these tumors arising spontaneously are absent from the mouse literature. Thus, most of the mouse models rely on GEMM models to produce pituitary tumors. Because these are gene KO or gene overexpressers, the exact relevance to tumorigenesis is uncertain, although the mice develop clinical features of CS.
In humans, ACTH-secreting pituitary tumors have been associated with MEN4, caused by CDKN1B mutations [ ], whereas they can also be found associated with germline mutations in the genes MEN1 and AIP [ ]. Analysis of sporadic tumors has revealed mutations in the ubiquitin specific protease 8 (USP) in up to 50% or 60% of tumors, and mutations in the related gene USP48 in a smaller subset. Mutations in BRAF and in the PI3K catalytic subunit p110A have also been described albeit very rarely [ ].
Although MEN4 is a very rare condition, it is associated with Cushing’s syndrome in about 10%–15% of cases [ ]. Mice that lack CDKN1B have been generated, and these animals develop pituitary tumors primarily affecting the intermediate lobe (pars intermedia) [ , ], which is a vestigial structure in human pituitaries. The tumors in these mice were similar to those in pRb mutant mice, with large and degenerating tumors [ ]. This observation is not surprising given the fact that pRb and CDKN1B function in the same growth inhibitory pathway. In CDKN1B mice, intermediate lobe tumors stained for α melanocyte-stimulating hormone, β-endorphin, and adrenocorticotropic hormone, presumably indicated POMC overproduction [ ], although no blood measurements of ACTH or steroid hormones were performed. In addition, mice were not described as having notable obesity or glucose intolerance, although a detailed investigation of the glucocorticoid axis was not performed in any of these models.
Another common genetic cause of pituitary CS in humans is MEN1, which can have its origin from the pituitary, the adrenals themselves, or via ectopic hormone secretion from hormonally active neuroendocrine tumors of the GEP system [ ]. Although MEN1+/− mice develop pituitary, adrenal, and neuroendocrine tumors [ ], stigmata of CS were not observed in animals; further, pituitary tumors did not stain for ACTH in animals tested [ , ]. Aip heterozygous mice have been described [ ] and shown to develop GH-secreting tumors exclusively; although human pedigrees with AIP mutations tend to be GH-predominant, they frequently include individuals with pituitary tumors that secrete other hormones, including ACTH.
Usp8 KO mice have been made and characterized [ ]. Hetero- and homozygote mice were viable and had no increased mortality up to 40 weeks. Characterization of the pituitary glands of the animals showed mild increases in ACTH staining by immunofluorescence, and adrenal glands also exhibited elevations in ACTH. However, serum ACTH and urinary steroids were unchanged among the three groups. Careful analysis showed modest elevations of blood glucose but no differences in weight or blood pressure. KO mice had modestly elevated heart rates, although no cardiovascular discase was observed. Usp48 KO mice have not been reported to date.
An interesting and unexpected model for mouse Cushing’s disease (pituitary Cushing) comes from the dopamine receptor 2 (D2R) knockout mouse [ ]. Animals null for the D2R exhibit hyperplasia of the pituitary intermediate lobe, including increases in ACTH and corticosterone elevations associated with adrenocortical hyperplasia. This study indicated that dopamine can play a significant role in the inhibition of pituitary ACTH secretion in the intermediate lobe, which can be prominent in many rodent models. There is also clearly applicability to humans as well, as the use of an SSTR5 targeted dopamine agonist (pasireotide) can be used to treating residual pituitary CD, whereas those agents that only target SSTR2 are largely ineffective.
On a related note, a knockout model of the prohormone convertase 2 chaperone protein known as 7B2 also produced a complex phenotype that included elements of Cushing’s syndrome [ ]. Ablation of 7B2 prevents maturation of PC2, which is responsible for intracellular processing of many neuroendocrine hormones, including POMC, as well as many other neuropeptide hormones. The phenotype of the 7B2 mice was complex due to the multiple functional defects in hormones and included both hypercortisolism and hyperinsulinism and animals died before 3 months of age [ ]. These animals also exhibited intermediate lobe hyperplasia. Interestingly, the phenotype could be ameliorated by adrenalectomy although the physiology of this change is unclear [ ]. Unlike other models, the physiology of the complex hormonal disturbances likely makes them unsuitable for use as a good model for CS.
Lastly, a model for ACTH-producing pituitary adenomas was generated by the introduction of a polyoma large T antigen driven by the polyoma early region promoter [ ]. These animals expressed the transgene in the pituitary and testes. Mice were observed to exhibit features of CS, most notably weight gain, around 9–12 months. Pathologic examination showed the presence of pituitary tumors which stained for ACTH, and serum hormone levels were also elevated. If mice were allowed to age further, they began losing weight, although it was not made clear if this was due to progression of CS or loss of other pituitary function. Interestingly, these authors demonstrated that the tumors could be resected and retransplanted into a syngenic strain. After this procedure, recipient mice developed further elevated ACH and a more severe form of CS compared with the original tumor donors [ ]. Mechanistically, polyoma large T antigen primarily causes transformation by binding to the tumor suppressor Rb [ ]. Based on this consideration, it is not surprising that these animals developed tumors in a fashion similar to the Cdkn1b KO or Rb overexpressing mice described before, as inactivation of Rb is the common downstream feature.
Ectopic ACTH syndrome
Although neuroendocrine tumors including small-cell lung cancers can secrete ACTH, the genetic factors that cause specific tumors to secrete ACTH remain occult. One successful model for ectopic ACTH secretion is the use of xenografts of the AtT-20 cell line using immunocompromised host animals. This cell line was initially isolated from a mouse pituitary tumor and avidly secretes ACTH [ ]. Nude mice that receive AtT-20 implants develop features of CS within a few weeks, which can be readily detected by the 4% weight gain [ ]. This model has been used as a preclinical model to test new potential therapies for ACTH-secreting pituitary adenomas, as well as to understand transcriptome changes that may be associated with treatment [ ].
In a related model, mice were infused for 2 weeks with ACTH via an osmotic pump [ ]. The animals were observed to have hypertension related to altered sodium handling, which was partly revered by both glucocorticoid and mineralocorticoid antagonists. Parameters related to other effects of chronic ACTH stimulation were not evaluated.
Models of adrenal Cushing’s syndrome
In humans, adrenal CS is associated with a number of specific genetic defects. Among these are diseases that lead to activation of the protein kinase A pathway in the adrenal cortex. It is also associated with cortical tumors MEN1, Beckwith–Wiedeman syndrome, and bilateral macronodular adrenal disease due to mutations in the ARMRC5 gene.
Adrenal CS is a common feature of the Carney Complex, an inherited tumor predisposition which includes spotty skin pigmentation, myxomas, endocrine tumors, and schwannomas [ , ]. The CS in these patients arises from primary pigmented nodular adrenocortical disease (PPNAD), in which the adrenal glands are studded with pigmented and highly active micronodules. The syndrome is caused by mutations in PRKAR1A [ , ], which encodes the type 1A regulatory subunit for protein kinase A, or cAMP-dependent protein kinase. This subunit is the mostly highly and widely expressed for the 4 PKA regulatory subunits, and loss of this protein causes increased PKA activity. Patients with the disease are heterozygous for inactivating mutations, and CS is observed in approximately 60% of the patients [ ]. In mice, heterozygotes for Prkar1a mutations develop tumors, but these do not involve the adrenal glands, and mice do not show evidence for hypercortisolism [ ]. When tissue-specific knockouts were made, the animals did exhibit evidence for adrenal hyperplasia and excess glucocorticoid production which was resistant to dex suppression [ ]. The same gene was targeted by an inducible antisense strategy. Mice had a subtle phenotype with late (>1 year) increases in weight, as well as elevated corticosterone [ , ], both pre- and postdexamethasone. Analysis of the adrenal glands showed cortical hypertrophy, but they lacked pigmentation seen in PPNAD adrenals.
In human tumors, the PKA pathway can also be activated by mutation [ , ] or overexpression [ , ] of PKA subunits, and these phenomena have been observed in adrenal tumors causing CS. These genetic alterations have not yet been modeled in mice. GNAS1, which is activated in the noninherited McCune Albright syndrome, is an upstream activator of PKA. Although a conditional activating allele of GNAs1 have been generated [ ], mouse models using this gene to model adrenocortical tumorigenesis have not yet been described.
An alternative approach to PKA activation involves removal of the phosphodiesterase (PDE) enzymes, which are responsible for degradation of cAMP. Mutations of the PDE11A [ ] or PDE8B [ ] enzymes have been identified in human tumors. Pde11a knockout mice were generated to determine if this phenotype could be modeled [ ]. Analysis of the animals revealed that the “knockout” allele actually retained partial Pde11a activity. There were minimal changes in corticosterone levels or reactivity, and also a lack of clinical changes, including normal body weight and normal lifespan. In contrast, mice that were lacking Pde8b exhibited hypercortisolism despite the fact that discrete adrenal tumors were not observed [ , ]. Mice with heterozygous mutation in Pde8b did not exhibit a significant phenotype, even when combined with heterozygous mutations in Prkar1a or activation of the Wnt pathway [ ].
Armc5 mice were also generated to model the genetic defect in MMAD. Armc5 KO mice exhibited an early embryonic lethality effect. Heterozygote mice developed mild hypercorticosteronemia, but this was not until 18 months and was only observed in about one-third of animals. In these mice, there was aberrant activation of the Wnt/ beta-catenin pathway in the zona fasciculata, reflecting the complex signaling interplay between PKA and Wnt signaling during adrenal development [ ].
Cushing’s syndrome in other animal specials
Although rodents provide an attractive model species to understand CS because of their ease of maintenance and the well-developed tools for genetic manipulation, they have some limitations. Specifically, rodents use corticosterone as their primary glucocorticoid, whereas humans use cortisol. In addition, the mouse adrenal gland lacks a discrete zona reticularis, instead including a fetal X zone, which regresses during maturation [ ]. Thus, there is some rationale to consider other animals as better models for the biology of CS, particularly adrenal tumors.
Canine Cushing’s syndrome
CS in dogs is a well-known phenomenon, with veterinary clinics providing the opportunity to study large populations of animals. Although higher incidence estimates for hypercortisolism in dogs have been reported in the past, a study of UK primary care veterinary clinics including over 900,000 dogs showed an incidence of CS was 0.06% with a 1-year prevalence of 0.17% [ ]. A similar but much smaller study (>21,000 dogs) from Italy derived an incidence of 0.2% [ ]. Thus, the reported incidence estimate for endogenous CS in dogs (600–2000 per million) is significantly higher than rates reported in humans (1–2.4 per million) [ ]. Certain breeds do seem to have higher risks, although different studies have identified different breeds as being at higher risk, so these effects may be population/country dependent.
As in humans, CS is most commonly caused by ACTH-secreting pituitary tumors, with adrenal tumors contributing the bulk of the remaining cases. Dogs affected by this condition present with a variety of features related to the multisystem effects of steroids. Canines commonly present with signs of diabetes, including the “classic” triad of polydipsia, polyuria, and polyphagia. They may also exhibit panting. They frequently exhibit abdominal distension and hepatomegaly, both due to abnormal fat deposition. Like humans, dogs may also exhibit hypertension, muscle weakness, and skin/hair changes, including alopecia [ ]. Based on the typical clinical presentation of CS in dogs, a scoring system has been developed based on clinical features [ ] and been largely validated in an external cohort [ ].
Like in rodents, CS can also be induced exogenously by administration of steroids. Administration of prednisone at a dose of 2 mg/kg for up to 84 days was used to model the effects of chronic steroid exposure on cardiac function valvular morphology in beagle dogs [ , ].
Although the genetics/genomics of canine tumors have not been well studied, this is beginning to change with advanced high-throughput sequencing and canine-based genetic databases. For example, a screening analysis of protein kinase A signaling pathways in canine adrenal tumors identified activating mutations in the canine GNAS1 gene in 14/44 (32%) of tumors tested [ ]. Molecular analyses including a recent transcriptome analysis of canine adrenal tumors identified two subsets of cortisol-producing tumors, although causative mutations were not identified [ ]. Interestingly, these data were shown to translate to an adrenal cancer dataset from The Cancer Genome Atlas.
Equine Cushing’s syndrome
The development of CS in horses is a phenomenon that has been well described in the equine veterinary literature. It appears to be a phenomenon that is restricted to horses of at least 15–18 years of age (about half of a horse’s normal age of 25–30 years), and affects both sexes equally. The incidence of equine CS has been reported to be in the range of 0.1%–2% of all horses but rises to the 10%–20% range in studies where the horses are at least 15 years of age. One study, in which the median age of the animals was 26.5 years, reported an incidence of 39% [ ].
Clinically, horses with CS present in a similar to fashion as dogs, with weight changes and fat deposition in the abdomen and other specific locations such as the neck crest, shoulders, and around the mammary glands or prepuce. These may be associated with polyuria and polydipsia, likely as a result of hyperglycemia. Horses also tend to exhibit changes in their coats, with development of a curly or wavy coat, as well as a risk for sinus and skin infections. Horses may also experience a weakening of their connective tissues, leading to the debilitating condition known as “laminitis,” caused by weakening of the tissues (laminae) anchoring the hoof wall to the underlying bone. This progressive condition may lead to lameness and require euthanasia of the animals if not treated [ ].
In horses, CS is almost exclusively caused by excess ACTH production arising from cells in the pars intermedia of the pituitary gland and is typically designated as pituitary pars intermedia dysfunction (PPID). Overproduction of ACTH is thought to be caused by loss of dopamine inhibition of ACTH secretion and thus can be characterized as a neurodegenerative disease caused by loss of dopaminergic neurons, analogous to Parkinson’s disease. From a therapeutic viewpoint, horses can be effectively treated with the dopamine agonist pergolide [ , ].
Efforts to characterize the molecular aspects and genetics of equine Cushing’s disease are in early stages, but tools are being developed [ ].
Summary
CS represents a unique human disease, with multiple exogenous and endogenous causes. Because endogenous CS is rare, advances in understanding the biology and physiology of the condition have been greatly aided by the use of model systems. Administration of exogenous steroid to small animals (including rodents and beagle dogs) has facilitated tools for the analysis of the extensive and tissue-specific molecular changes that accompany excess glucocorticoid signaling, both in the acute and chronic phases.
Genetic manipulation of the mouse genome has enabled similar studies using endogenous models, which have readily been generated by use of GEM models to drive hormonal overexpression. Most of the extant mouse models rely on overexpression of CRH, so the correlation between these animals and human CD must be carefully considered. Unfortunately, of the genes that have been shown to cause human hypercortisolism, only adrenal-specific ablation of PRKAR1A or generalized ablation of PDE8B , which is an exact genetic model for hypercortisolism associated with the Carney Complex, recapitulates this aspect of the phenotype.
To date, genes associated with other forms of adrenal Cushing have not been developed, and genes associated with pituitary Cushing (CD) have generally not successfully modeled hypercortisolism. The only exception may be CDKN1B, which is clearly associated with CS in a significant portion of the admittedly rare patients with MEN4, and appears to cause proliferation of ACTH-producing cells in the pituitary of mice, although it is less clear if there is “clinical” CS in the animals.
Larger animals like dogs and horses develop CS at a far higher frequency than that observed in humans, but this seems primarily driven by pars intermedia tumors under the control (or loss of control) of dopaminergic neurons. Humans retain only a rudimentary pars intermedia, and dopamine agonists are rarely effective in human CD, again suggesting that there are limits to either of these species as models for the human disease.
Nonetheless, a substantial amount of valuable data about the biochemistry and physiology of Cushing disease has been generated through animal modeling, and improved models should continue to be developed to allow new approaches to understanding and treating this complex and poorly understand hormonal disease.
References
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree