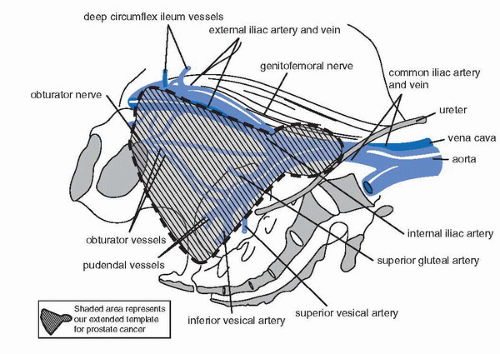
References
1. Holmberg L, Bill-Axelson A, Helgesen F, et al. A randomized trial comparing radical prostatectomy with watchful waiting in early prostate cancer. N Engl J Med 2002;347(11):781-789.
2. Bolla M, Gonzalez D, Warde P, et al. Improved survival in patients with locally advanced prostate cancer treated with radiotherapy and goserelin. N Engl J Med 1997;337(5):295-300.
3. D’Amico AV, Whittington R, Malkowicz SB, et al. Biochemical outcome after radical prostatectomy, external beam radiation therapy, or interstitial radiation therapy for clinically localized prostate cancer. JAMA 1998;280(11):969-974.
4. Quinlan DM, Epstein JI, Carter BS, et al. Sexual function following radical prostatectomy: influence of preservation of neurovascular bundles. J Urol 1991;145(5):998-1002.
5. Walsh PC, Partin AW, Epstein JI. Cancer control and quality of life following anatomical radical retropubic prostatectomy: results at 10 years. J Urol 1994;152(5 Pt 2):1831-1836.
6. Shipley WU, Thames HD, Sandler HM, et al. Radiation therapy for clinically localized prostate cancer: a multi-institutional pooled analysis. JAMA 1999;281(17):1598-1604.
7. Zietman AL, DeSilvio ML, Slater JD, et al. Comparison of conventional-dose vs highdose conformal radiation therapy in clinically localized adenocarcinoma of the prostate: a randomized controlled trial. JAMA 2005;294(10):1233-1239.
8. Rabbani F, Stapleton AM, Kattan MW, et al. Factors predicting recovery of erections after radical prostatectomy. J Urol 2000;164(6):1929-1934.
9. Catalona WJ, Carvalhal GF, Mager DE, et al. Potency, continence and complication rates in 1,870 consecutive radical retropubic prostatectomies. J Urol 1999;162(2):433-438.
10. Fowler FJ Jr, Barry MJ, Lu-Yao G, et al. Patientreported complications and follow-up treatment after radical prostatectomy. The National Medicare Experience: 1988-1990 (updated June 1993). Urology 1993;42(6):622-629.
11. Fowler FJ Jr, Barry MJ, Lu-Yao G, et al. Outcomes of external-beam radiation therapy for prostate cancer: a study of medicare beneficiaries in three surveillance, epidemiology, and end results areas. J Clin Oncol 1996;14(8):2258-2265.
12. Fowler FJ Jr, McNaughton Collins M, Walker Corkery E, et al. The impact of androgen deprivation on quality of life after radical prostatectomy for prostate carcinoma. Cancer 2002;95(2):287-295.
13. Joly F, Brune D, Couette JE, et al. Healthrelated quality of life and sequelae in patients treated with brachytherapy and external beam irradiation for localized prostate cancer. Ann Oncol 1998;9(7):751-757.
14. Litwin MS, Lubeck DP, Henning JM, et al. Differences in urologist and patient assessments of health related quality of life in men with prostate cancer: results of the CaPSURE database. J Urol 1998;159(6):1988-1992.
15. Sonn GA, Sadetsky N, Presti JC, et al. Differing perceptions of quality of life in patients with prostate cancer and their doctors. J Urol 2009;182(5):2296-2302.
16. Litwin MS, Hays RD, Fink A, et al. Quality-of-life outcomes in men treated for localized prostate cancer. JAMA 1995;273(2):129-135.
17. Wei JT, Dunn RL, Sandler HM, et al. Comprehensive comparison of health-related quality of life after contemporary therapies for localized prostate cancer. J Clin Oncol 2002;20(2):557-566.
18. Davis JW, Kuban DA, Lynch DF, et al. Quality of life after treatment for localized prostate cancer: differences based on treatment modality. J Urol 2001;166(3):947-952.
19. Chen RC, Clark JA, Talcott JA. Individualizing quality-of-life outcomes reporting: how localized prostate cancer treatments affect patients with different levels of baseline urinary, bowel, and sexual function. J Clin Oncol 2009;27(24):3916-3922.
20. Talcott JA, Manola J, Clark JA, et al. Time course and predictors of symptoms after primary prostate cancer therapy. J Clin Oncol 2003;21(21):3979-3986.
21. Ware JE Jr, Sherbourne CD. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med Care 1992;30(6):473-483.
22. Ware J Jr, Kosinski M, Keller SD. A 12-item short-form health survey: construction of scales and preliminary tests of reliability and validity. Med Care 1996;34(3):220-233.
23. Cella DF, Tulsky DS, Gray G, et al. The functional assessment of cancer therapy scale: development and validation of the general measure. J Clin Oncol 1993;11(3):570-579.
24. Aaronson NK, Ahmedzai S, Bergman B, et al. The European Organization for Research and Treatment of Cancer QLQ-C30: a quality-of-life instrument for use in international clinical trials in oncology. J Natl Cancer Inst 1993;85(5):365-376.
25. Barry MJ, Fowler FJ, Jr., O’Leary MP, et al. The American Urological Association symptom index for benign prostatic hyperplasia. The Measurement Committee of the American Urological Association. J Urol 1992;148(5):1549-1557; discussion 1564.
26. Rosen RC, Riley A, Wagner G, et al. The international index of erectile function (IIEF): a multidimensional scale for assessment of erectile dysfunction. Urology 1997;49(6):822-830.
27. Rosen RC, Cappelleri JC, Smith MD, et al. Development and evaluation of an abridged, 5-item version of the International Index of Erectile Function (IIEF-5) as a diagnostic tool for erectile dysfunction. Int J Impot Res 1999;11(6):319-326.
28. Streiner DL, Norman GR. Health measurement scales: a practical guide to their development and use, Oxford University Press, Oxford 2nd ed. 1995.
29. Litwin MS, Hays RD, Fink A, et al. The UCLA Prostate Cancer Index: Development, reliability, and validity of a health-related quality of life measure. Med Care 1998;36(7):1002-1012.
30. Borghede G., Sullivan M. Measurement of quality of life in localized prostatic cancer patients treated with radiotherapy. Development of a prostate cancer-specific module supplementing the EORTC QLQ-C30. Qual Life Res 1996;5(2):212-222.
31. Esper P, Mo F, Chodak G, et al. Measuring quality of life in men with prostate cancer using the functional assessment of cancer therapyprostate instrument. Urology 1997;50(6):920-928.
32. Wei JT, Dunn RL, Litwin MS, et al. Development and validation of the expanded prostate cancer index composite (EPIC) for comprehensive assessment of health-related quality of life in men with prostate cancer. Urology 2000;56(6):899-905.
33. Sanda MG, Dunn RL, Michalski J, et al. Quality of life and satisfaction with outcome among prostate-cancer survivors. N Engl J Med 2008;358(12):1250-1261.
34. Szymanski KMW, JT, Dunn, RL, Sanda MG. Development and validation of an abbreviated version of the expanded prostate cancer index composite instrument (EPIC-26) for measuring health-related quality of life among prostate cancer survivors. Urology 2010;76(5):1245-1250.
35. Clark JA, Talcott JA. Symptom indexes to assess outcomes of treatment for early prostate cancer. Med Care 2001;39(10):1118-1130.
36. Fransson P, Tavelin B, Widmark A. Reliability and responsiveness of a prostate cancer questionnaire for radiotherapy-induced side effects. Support Care Cancer 2001;9(3):187-198.
37. Mishel MH. Reconceptualization of the uncertainty in illness theory. Image J Nurs Sch 1990;22(4):256-262.
38. Patel MI, DeConcini DT, Lopez-Corona E, et al. An analysis of men with clinically localized prostate cancer who deferred definitive therapy. J Urol 2004;171(4):1520-1524.
39. Cooperberg MR, Lubeck DP, Meng MV, et al. The changing face of low-risk prostate cancer: trends in clinical presentation and primary management. J Clin Oncol 2004;22(11):2141-2149.
40. Befort CA, Zelefsky MJ, Scardino PT, et al. A measure of health-related quality of life among patients with localized prostate cancer: results from ongoing scale development. Clin Prostate Cancer. 2005;4(2):100-108. PMID: 16197610.
41. Roth AJ, Rosenfeld B, Kornblith AB, et al. The memorial anxiety scale for prostate cancer: validation of a new scale to measure anxiety in men with with prostate cancer. Cancer 2003;97(11):2910-2918.
42. Eastham JA, Scardino PT, Kattan MW. Predicting an optimal outcome after radical prostatectomy: the trifecta nomogram. J Urol 2008;179(6):2207-2210; discussion 2210-2211.
43. Donabedian A. The quality of medical care. Science 1978;200(4344):856-864.
44. Cleary PD, McNeil BJ. Patient satisfaction as an indicator of quality care. Inquiry 1988;25(1):25-36.
45. Lubeck DP, Litwin MS, Henning JM, et al. An instrument to measure patient satisfaction with healthcare in an observational database: results of a validation study using data from CaPSURE. Am J Manag Care 2000;6(1):70-76.
46. Hoffman RM, Hunt WC, Gilliland FD, et al. Patient satisfaction with treatment decisions for clinically localized prostate carcinoma. Results from the Prostate Cancer Outcomes Study. Cancer 2003;97(7):1653-1662.
47. Shah NL, Dunn RL, Greenfield TK, et al. Development and validation of a novel instrument to measure patient satisfaction in multiple dimensions of urological cancer care quality. J Urol 2003;169(Suppl 4):11.
48. Schroeck FR, Krupski TL, Sun L, et al. Satisfaction and regret after open retropubic or robot-assisted laparoscopic radical prostatectomy. Eur Urol 2008;54(4):785-793.
49. Potosky AL, Legler J, Albertsen PC, et al. Health outcomes after prostatectomy or radiotherapy for prostate cancer: results from the Prostate Cancer Outcomes Study. J Natl Cancer Inst 2000;92(19):1582-1592.
50. Stanford JL, Feng Z, Hamilton AS, et al. Urinary and sexual function after radical prostatectomy for clinically localized prostate cancer: the Prostate Cancer Outcomes Study. JAMA 2000;283(3):354-360.
51. Potosky AL, Davis WW, Hoffman RM, et al. Five-year outcomes after prostatectomy or radiotherapy for prostate cancer: the prostate cancer outcomes study. J Natl Cancer Inst 2004;96(18):1358-1367.
52. Madalinska JB, Essink-Bot ML, de Koning HJ, et al. Health-related quality-of-life effects of radical prostatectomy and primary radiotherapy for screen-detected or clinically diagnosed localized prostate cancer. J Clin Oncol 2001;19(6):1619-1628.
53. Korfage IJ, Essink-Bot ML, Borsboom GJ, et al. Five-year follow-up of health-related quality of life after primary treatment of localized prostate cancer. Int J Cancer 2005;116(2):291-296.
54. Potosky AL, Knopf K, Clegg LX, et al. Quality-of-life outcomes after primary androgen deprivation therapy: results from the Prostate Cancer Outcomes Study. J Clin Oncol 2001;19(17):3750-3757.
55. Downs TM, Sadetsky N, Pasta DJ, et al. Health related quality of life patterns in patients treated with interstitial prostate brachytherapy for localized prostate cancer—data from CaPSURE, prospectice, multicenter. J Urol 2003;170(5):1822-1827.
56. Arredondo SA, Downs TM, Lubeck DP, et al. Watchful waiting and health related quality of life for patients with localized prostate cancer: data from CaPSURE. J Urol 2008;179(5 Suppl):S14-S18.
57. Wu AK, Cooperberg MR, Sadetsky N, et al. Health related quality of life in patients treated with multimodal therapy for prostate cancer. J Urol 2008;180(6):2415-2422; discussion 2422.
58. Pinkawa M, Fischedick K, Asadpour B, et al. Toxicity profile with a large prostate volume after external beam radiotherapy for localized prostate cancer. Int J Radiat Oncol Biol Phys 2008;70(1):83-89.
59. Ferrer M, Suarez JF, Guedea F, et al. Healthrelated quality of life 2 years after treatment with radical prostatectomy, prostate brachytherapy, or external beam radiotherapy in patients with clinically localized prostate cancer. Int J Radiat Oncol Biol Phys 2008;72(2):421-432.
60. Smith DP, King MT, Egger S, et al. Quality of life three years after diagnosis of localised prostate cancer: population based cohort study. BMJ 2009;339:b4817.
61. Lara PN Jr, de Vere White R. Accrual issues in genitourinary cancer clinical trials. Urol Oncol 2006;24(5):379-383.
62. Steineck G, Helgesen F, Adolfsson J, et al. Quality of life after radical prostatectomy or watchful waiting. N Engl J Med 2002;347(11):790-796.
63. Johansson E, Bill-Axelson A, Holmberg L, et al. Time, symptom burden, androgen deprivation, and self-assessed quality of life after radical prostatectomy or watchful waiting: the Randomized Scandinavian Prostate Cancer Group Study Number 4 (SPCG-4) clinical trial. Eur Urol 2009;55(2):422-430.
64. Akakura K., Isaka S, Akimoto S, et al. Longterm results of a randomized trial for the treatment of stages B2 and C prostate cancer: radical prostatectomy versus external beam radiation therapy with a common endocrine therapy in both modalities. Urology 1999;54(2):313-318.
65. Akakura K, Suzuki H, Ichikawa T, et al. A randomized trial comparing radical prostatectomy plus endocrine therapy versus external beam radiotherapy plus endocrine therapy for locally advanced prostate cancer: results at median follow-up of 102 months. Jpn J Clin Oncol 2006;36(12):789-793.
66. Irani J, Celhay O, Hubert J, et al. Continuous versus six months a year maximal androgen blockade in the management of prostate cancer: a randomised study. Eur Urol 2008;54(2):382-391.
67. Bolla M, de Reijke TM, Van Tienhoven G, et al. Duration of androgen suppression in the treatment of prostate cancer. N Engl J Med 2009;360(24):2516-2527.
68. Fransson P, Damber JE, Tomic R, et al. Quality of life and symptoms in a randomized trial of radiotherapy versus deferred treatment of localized prostate carcinoma. Cancer 2001;92(12):3111-3119.
69. Widmark A, Klepp O, Solberg A, et al. Endocrine treatment, with or without radiotherapy, in locally advanced prostate cancer (SPCG-7/SFUO-3): an open randomised phase III trial. Lancet 2009;373(9660):301-308.
70. Fransson P, Lund JA, Damber JE, et al. Quality of life in patients with locally advanced prostate cancer given endocrine treatment with or without radiotherapy: 4-year follow-up of SPCG-7/SFUO-3, an open-label, randomised, phase III trial. Lancet Oncol 2009;10(4):370-380.
71. Giberti C, Chiono L, Gallo F, et al. Radical retropubic prostatectomy versus brachytherapy for low-risk prostatic cancer: a prospective study. World J Urol 2009;27(5):607-612.
72. Thompson IM Jr, Tangen CM, Paradelo J, et al. Adjuvant radiotherapy for pathologically advanced prostate cancer: a randomized clinical trial. JAMA 2006;296(19):2329-2335.
73. Moinpour CM, Hayden KA, Unger JM, et al. Health-related quality of life results in pathologic stage C prostate cancer from a Southwest Oncology Group trial comparing radical prostatectomy alone with radical prostatectomy plus radiation therapy. J Clin Oncol 2008;26(1):112-120.
74. Nguyen LN, Pollack A, Zagars GK. Late effects after radiotherapy for prostate cancer in a randomized dose-response study: results of a self-assessment questionnaire. Urology 1998;51(6):991-997.
75. Little DJ, Kuban DA, Levy LB, et al. Quality-of-life questionnaire results 2 and 3 years after radiotherapy for prostate cancer in a randomized dose-escalation study. Urology 2003;62(4):707-713.
76. Dearnaley DP, Sydes MR, Graham JD, et al. Escalated-dose versus standard-dose conformal radiotherapy in prostate cancer: first results from the MRC RT01 randomised controlled trial. Lancet Oncol 2007;8(6):475-487.
77. Yeoh EE, Holloway RH, Fraser RJ, et al. Hypofractionated versus conventionally fractionated radiation therapy for prostate carcinoma: updated results of a phase III randomized trial. Int J Radiat Oncol Biol Phys 2006;66(4):1072-1083.
78. Hoskin PJ, Motohashi K, Bownes P, et al. High dose rate brachytherapy in combination with external beam radiotherapy in the radical treatment of prostate cancer: initial results of a randomised phase three trial. Radiother Oncol 2007;84(2):114-120.
79. Donnelly BJ, Saliken JC, Brasher PM, et al. A randomized trial of external beam radiotherapy versus cryoablation in patients with localized prostate cancer. Cancer 2009;116(2):323-330.
80. Robinson JW, Donnelly BJ, Siever JE, et al. A randomized trial of external beam radiotherapy versus cryoablation in patients with localized prostate cancer: quality of life outcomes. Cancer 2009;115(20):4695-4704.
81. Jones JS, Rewcastle JC, Donnelly BJ, et al. Whole gland primary prostate cryoablation: initial results from the cryo on-line data registry. J Urol 2008;180(2):554-558.
82. Prestidge BR. A phase III study comparing combined external beam radiation and transperineal interstitial permanent brachytherapy with brachytherapy alone for selected patients with intermediate risk prostatic carcinoma. RTOG-0232. http://www.clinicaltrials.gov/ct2/show/N CT00063882?term=rtog+0232&rank=2.
83. Umbehr M, Kessler TM, Sulser T, et al. ProCOC: the prostate cancer outcomes cohort study. BMC Urol 2008;8:9.
84. Donovan J, Hamdy F, Neal D, et al. Prostate testing for cancer and treatment (ProtecT) feasibility study. Health Technol Assess 2003;7(14):1-88.
85. Wilt TJ. SPCG-4: a needed START to PIVOTal data to promote and protect evidence-based prostate cancer care. J Natl Cancer Inst 2008;100(16):1123-1125.
86. Namiki S, Ishidoya S, Tochigi T, et al. Healthrelated quality of life after intensity modulated radiation therapy for localized prostate cancer: comparison with conventional and conformal radiotherapy. Jpn J Clin Oncol 2006;36(4):224-230.
87. Namiki S, Ishidoya S, Ito A, et al. Five-year follow-up of health-related quality of life after intensity-modulated radiation therapy for prostate cancer. Jpn J Clin Oncol 2009;39(11):732-738.
88. Yoshimura K, Kamoto T, Nakamura E, et al. Health-related quality-of-life after external beam radiation therapy for localized prostate cancer: intensity-modulated radiation therapy versus conformal radiation therapy. Prostate Cancer Prostatic Dis 2007;10(3):288-292.
89. Lips I, Dehnad H, Kruger AB, et al. Healthrelated quality of life in patients with locally advanced prostate cancer after 76 Gy intensity-modulated radiotherapy vs. 70 Gy conformal radiotherapy in a prospective and longitudinal study. Int J Radiat Oncol Biol Phys 2007;69(3):656-661.
90. Talcott JA, Slater JD, Zietman A, et al. Longterm quality of life after conventional-dose versus high-dose radiation for prostate cancer: results from a randomized trial (PROG 95-09). J Clin Oncol 2008;26(Suppl):abstract 5058.
91. Coen J, Talcott JA, Houlihan RL, et al. Correlating patient reported symptoms after high dose proton irradiation for localized prostate cancer with dose volume histogram parameters. Int J Radiat Oncol Biol Phys 2009;75(3):S304-S305.
92. King CR, Brooks JD, Gill H, et al. Stereotactic body radiotherapy for localized prostate cancer: interim results of a prospective phase II clinical trial. Int J Radiat Oncol Biol Phys 2009;73(4):1043-1048.
93. Morgia G, De Renzis C. CyberKnife in the treatment of prostate cancer: a revolutionary system. Eur Urol 2009;56(1):40-42.
94. Ficarra V, Novara G, Artibani W, et al. Retropubic, laparoscopic, and robot-assisted radical prostatectomy: a systematic review and cumulative analysis of comparative studies. Eur Urol 2009;55(5):1037-1063.
95. Guazzoni G, Cestari A, Naspro R, et al. Intra- and peri-operative outcomes comparing radical retropubic and laparoscopic radical prostatectomy: results from a prospective, randomised, single-surgeon study. Eur Urol 2006;50(1):98-104.
96. Anastasiadis AG, Salomon L, Katz R, et al. Radical retropubic versus laparoscopic prostatectomy: a prospective comparison of functional outcome. Urology 2003;62(2):292-297.
97. Hara I, Kawabata G, Miyake H, et al. Comparison of quality of life following laparoscopic and open prostatectomy for prostate cancer. J Urol 2003;169(6):2045-2048.
98. Roumeguere T, Bollens R, Vanden Bossche M, et al. Radical prostatectomy: a prospective comparison of oncological and functional results between open and laparoscopic approaches. World J Urol 2003;20(6):360-366.
99. Touijer K, Eastham JA, Secin FP, et al. Comprehensive prospective comparative analysis of outcomes between open and laparoscopic radical prostatectomy conducted in 2003 to 2005. J Urol 2008;179(5):1811-1817; discussion 1817.
100. Namiki S, Egawa S, Terachi T, et al. Changes in quality of life in first year after radical prostatectomy by retropubic, laparoscopic, and perineal approach: multi-institutional longitudinal study in Japan. Urology 2006;67(2):321-327.
101. Hu JC, Gu X, Lipsitz SR, et al. Comparative effectiveness of minimally invasive vs open radical prostatectomy. JAMA 2009;302(14):1557-1564.
102. Menon M, Shrivastava A, Tewari A, et al. Laparoscopic and robot assisted radical prostatectomy: establishment of a structured program and preliminary analysis of outcomes. J Urol 2002;168(3):945-949.
103. Tewari A, Srivasatava A, Menon M. A prospective comparison of radical retropubic and robot-assisted prostatectomy: experience in one institution. BJU Int 2003;92(3):205-210.
104. Tseng TY, Kuebler HR, Cancel QV, et al. Prospective health-related quality-of-life assessment in an initial cohort of patients undergoing robotic radical prostatectomy. Urology 2006;68(5):1061-1066.
105. Shikanov S, Woo J, Al-Ahmadie H, et al. Extrafascial versus interfascial nerve-sparing technique for robotic-assisted laparoscopic prostatectomy: comparison of functional outcomes and positive surgical margins characteristics. Urology 2009;74(3):611-616.
106. Blana A, Walter B, Rogenhofer S, et al. Highintensity focused ultrasound for the treatment of localized prostate cancer: 5-year experience. Urology 2004;63(2):297-300.
107. Poissonnier L, Chapelon JY, Curiel, et al. Control of prostate cancer by transrectal HIFU in 227 patients. Eur Urol 2007;51(2):381-387.
108. Sanda MG and Kaplan. Clinical Crossroads: Review of Prostate Cancer Treatment. JAMA 2009; 301(20):2141-2151.
109. D’Amico AV, Denham JW, Crook J, et al. Influence of androgen suppression therapy for prostate cancer on the frequency and timing of fatal myocardial infarctions. J Clin Oncol 2007;25(17):2420-2425.
110. Shappley WV III, Kenfield SA, Kasperzyk JL, et al. Prospective study of determinants and outcomes of deferred treatment or watchful waiting among men with prostate cancer in a nationwide cohort. J Clin Oncol 2009;27(30):4980-4985.