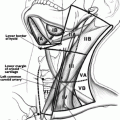
Fig. 19.1
Anatomy of the larynx (Permission granted from Operative techniques in Laryngology by Clark A. Rosen and C. Blake Simpson. Published by Springer. See enclosed permission)
Knowledge of the anatomy of the RLN and EBSLN is of upmost importance to avoid injury during thyroid surgery. The vagus nerve exits the cranial cavity via the jugular foramen along with the glossopharyngeal and hypoglossal nerves. It descends in the carotid sheath just deep to and between the internal jugular vein and carotid artery. The right RLN branches from the vagus in the upper mediastinum and recurs around the right subclavian artery before traveling cranially in the tracheoesophageal groove toward its laryngeal insertion. The left RLN branches from the left vagus at the level of the aortic arch and recurs around the ligamentum arteriosum and travels cranially in the tracheoesophageal groove to insert into the larynx (Fig. 19.2 – Anatomy of recurrent laryngeal and superior laryngeal nerves). The cervical portion of the right RLN tends to have a slightly more oblique course from the mediastinum toward the laryngeal insertion point compared to the left RLN which travels more directly in the tracheoesophageal groove posterior to the left thyroid lobe. The RLN inserts into the larynx by passing under the inferior constrictor of the pharynx to innervate the intrinsic laryngeal muscles.
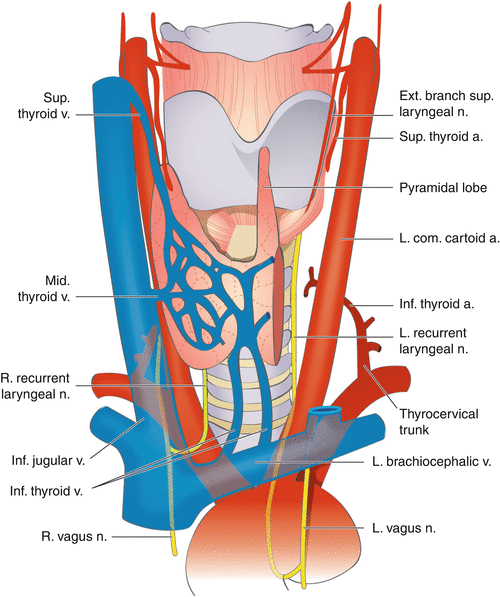

Fig. 19.2
Anatomy of recurrent laryngeal and superior laryngeal nerves (Permission granted from Color atlas of thyroid surgery by Yeo-kyu Youn, Kyu Eun Lee and June Young Choi. Published by Springer. See enclosed permission)
A non-recurrent RLN is an embryological anomaly present in approximately 0.5 % right RLNs and 0.04 % left RLNs. A non-recurrent right RLN is associated with the arteria lusoria vascular abnormality whereby the innominate artery is absent and the right common carotid and right subclavian arteries originate directly from the aortic arch. In this anomaly the right subclavian artery is the most distal branch of the aortic arch and travels in a retroesophageal course. This may be associated with dysphagia due to compression of the posterior esophagus (dysphagia lusoria). A left non-recurrent RLN is a rarer anomaly and is associated with a right-sided aortic arch as seen in situs inversus with displacement of the ligamentum arteriosum to the right side.
Identification of the RLN during thyroidectomy should be considered a necessity in order to prevent injury. The typical diameter of the RLN ranges from 1 to 3 mm. Identification of the RLN inferior to the lower pole of the thyroid gland allows for visualization as dissection progresses from the lower pole toward the tubercle of Zuckerkandl and the ligament of Berry. Large thyroid goiters or those with retrosternal extension can make identification of the RLN difficult, and identification of the nerve from a cranial to caudal direction after ligating the superior pole vessels can be useful. The association of the RLN to the inferior thyroid artery can vary significantly and care should be taken during ligation of the inferior thyroid artery and its braches near the ligament of Berry (Fig. 19.3). The tubercle of Zuckerkandl is a posterior extension of thyroid tissue near the ligament of Berry which often has close association with the RLN, and in cases of enlargement or nodules, the nerve can often course anterior to the tubercle and is prone to injury. The RLN frequently will bifurcate near its insertion point into the larynx into posterior and anterior branches which typically have a sensory and motor function, respectively [1]. Both of these branches need to be fastidiously followed and preserved.
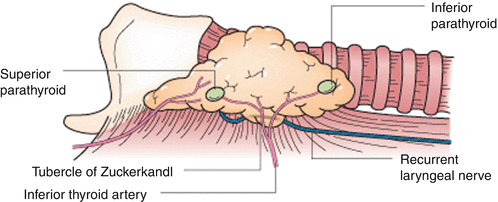

Fig. 19.3
Recurrent laryngeal nerve anatomic association with ligament of Berry (Permission granted from Greenfield’s surgery: scientific principles and practice 5th ed. See enclosed permission)
Intraoperative Considerations
Operations commonly associated with a risk of RLN injury include thyroidectomy, parathyroidectomy, anterior approach cervical spine procedures, cervical esophageal surgery, and upper mediastinal surgery. Most series of thyroidectomy in high-volume centers report an injury rate ranging from 1 to 5 %; however, RLN injury may be unrecognized in patients with minimal voice changes following surgery, and therefore actual injury rates are likely higher due to under reporting. This is one argument for routine perioperative laryngoscopy.
Neuropraxia or stretch injuries to the RLN typically occur during dissection in the area of the tubercle of Zuckerkandl and the ligament of Berry near the insertion of the RLN into the larynx. Traction of the RLN due to medial and anterior retraction of the thyroid can occur due to the close association of the inferior thyroid artery and its distal branches to the RLN near the ligament of Berry. Should a neuropraxia occur and an anatomically intact RLN in confirmed, neural function typically will return in a matter of weeks to months after surgery. Intraoperative nerve monitoring can allow for recognition of such an injury manifesting as a loss of RLN function with nerve stimulation proximal to the site of injury whereas without nerve monitoring observation of an anatomically intact nerve would erroneously presume normal nerve function. The decision to proceed with contralateral thyroidectomy in the setting of a neuropraxia injury should be carefully considered, and in some cases a staged procedure which would allow the RLN function to recover can prevent bilateral nerve injury. Continuous vagal nerve stimulation may be useful in detecting real-time impending neuropraxia which is evident as decreasing nerve waveform amplitude and latency and loss of nerve signal [2]. Gentle dissection of the RLN, constant attention to tissue or iatrogenic tension on the nerve, avoidance of cautery or energy devices in close proximity of the nerve, and avoidance of constricting ligatures will avoid traumatic injury in most cases.
Transection of the RLN during thyroidectomy can occur unintentionally or intentionally in the setting of invasive thyroid cancer. Recognized unintentional transection of the RLN without loss of nerve length should be addressed intraoperatively with an attempt at neural repair. Neural repair is performed by adequate mobilization of the distal and proximal segments of the RLN with reanastomosis and epineural repair in an attempt to preserve neural tone. Canine models of recurrent laryngeal transection and reanastamosis report restoration of laryngeal adduction function in around 50 % [3]. A long segment transection which would not allow for direct neural anastomosis has been treated with anastomosis of the distal RLN to the ipsilateral ansa cervicalis nerve with some success [4, 5]. A preoperative paralyzed vocal cord associated with malignant infiltration should be sacrificed at the time of surgery in an attempt to achieve an oncologic complete resection since postoperative recovery of function would not be expected. However, in the setting of a normally functioning RLN preoperatively with the discovery of tumor involvement from differentiated thyroid cancer at the time of thyroidectomy, attempts should be made to anatomically preserve the nerve with resection of all gross disease. A grossly negative margin with persistent microscopic disease can often be adequately treated with postoperative therapy with radioiodine, TSH suppression and in some cases external beam radiation therapy. Magnification for sharp neurolysis and separation from the tumor can be very helpful in this situation.
Intraoperative Nerve Monitoring
Intraoperative neural monitoring of the RLN and EBSLN during thyroidectomy can be implemented with severally commercially available nerve monitoring systems. Nerve monitoring involves measurement of vocal cord movement or laryngeal muscle action potentials with either electrodes on the external surface of the endotracheal tube or with needle electrodes implanted intraoperatively into the cricothyroid muscle. Nerve stimulation of the RLN with 0.3–3 mA stimulation current will produce adduction of the true vocal cord and an evoked potential measured by the electrodes. Stimulation of the EBSLN will produce a muscular twitch of the cricothyroid muscle and can variably produce an evoked potential measured by endotracheal tube electrodes. Nerve monitoring can aid in identification of the RLN and EBSLN during thyroidectomy; however, it cannot replace surgical skill and knowledge of normal anatomy and anatomic variants which is required to consistently identify the nerves and preserve vocal cord function. Neuropraxia and subsequent loss of function of the RLN in the setting of an anatomically intact nerve can be accurately determined with intraoperative nerve monitoring. Intraoperative stimulation of the RLN that produces an evoked potential above 200 μV correlates with normal postoperative vocal cord function in nearly 100 % of patients (true negative), while loss of signal intraoperative can be related either to nerve dysfunction to technical failures of the monitoring system commonly due to malposition of the endotracheal monitoring tube, and therefore loss of intraoperative signal correlates with vocal fold paresis in about 72 % of patients [6]. Knowledge of a neuropraxic injury provided by nerve monitoring (after troubleshooting the system of confirm true loss of signal) can be important in intraoperative decision making as to whether to proceed with contralateral thyroidectomy and risk bilateral nerve dysfunction or completing the hemithyroidectomy with plans for completion thyroidectomy after nerve function has recovered [7]. The suspected pathology (i.e., benign vs. malignant) is important in this decision making.
Multiple studies and meta-analyses have shown no difference in nerve injury rates with intraoperative neural monitoring; however, it may improve recognition of nerve injury with subsequent change in intraoperative management and decision making as discussed previously [8–12]. Most advocates also suggest the technology that allows the operation to move forward more confidently and expeditiously as well as being a valuable teaching aid. The use of nerve monitoring can be especially helpful in reoperative thyroid procedures given the increased risk of nerve injury and the altered anatomic planes present in reoperative cases.
Diagnosis of Nerve Dysfunction
Unilateral RLN dysfunction generally presents as hoarseness (dysphonia) with a weak and “breathy” voice. This is often accompanied with patient complaints of running out of air during speech due to lack of complete adduction of the vocal folds during phonation. The cough has a “bovine” quality without a sharp percussive initiation since the vocal cords cannot tightly approximate. Aspiration most commonly with drinking of liquids is also common especially in the acute phase of unilateral vocal cord dysfunction or in older patients. The contralateral vocal cord compensation in its degree of adduction can often result in a nearly normal voice despite unilateral RLN palsy, and therefore the reliance of symptoms and subjective voice quality alone can often be inaccurate in elucidating nerve function. Other etiologies of dysphonia with normal nerve function including viral illnesses, reflux pharyngitis, laryngeal polyps or cancer, and voice overuse should be considered in cases of confirmed of normal vocal fold movement on laryngoscopy.
There are three primary modalities for visual inspection of the vocal cords: direct laryngoscopy, indirect mirror laryngoscopy, and flexible laryngoscopy. Direct laryngoscopy typically requires general anesthesia but can be combined with rigid scopes for interventional purposes to treat cord pathology. Indirect mirror laryngoscopy can visualize the larynx in about 50 % of patients using a dental mirror, flashlight, and anterior retraction of the tongue. Flexible laryngoscopy, either with or without video, provides consistent and complete visualization of the vocal cords without need for sedation, but with the added cost of equipment. Flexible nasolaryngoscopy is easily taught and can be accomplished in the office setting with nasal application of aerosolized topical anesthetic often combined with oxymetazoline or phenylephrine for their vasoconstrictive properties.
The typical findings on laryngoscopy in unilateral RLN paralysis include a true vocal fold in the paramedian position with anterior displacement of the arytenoid cartilage and lack of movement with vocalization (Fig. 19.4 – Flexible laryngoscopy). EBSLN dysfunction results in some bowing of the involved true vocal fold with loss of tension and is somewhat more difficult to elucidate. Bilateral RLN paralysis will result in medialization of both the right and the left true vocal folds with a narrow orifice between the immobile cords, leading to stridor and airway compromise in some patients.
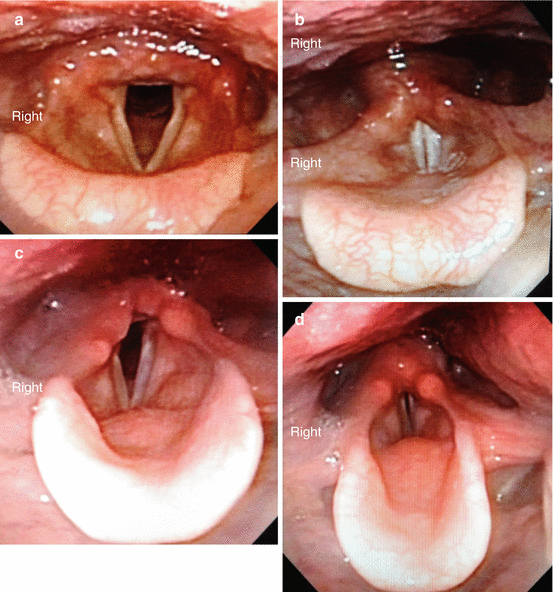

Fig. 19.4
Flexible laryngoscopy demonstrating normal vocal cord positioning (a, b) and paralyzed right vocal cord (c, d) with quite respiration (a, c) and phonation (b, d). Note the paramedian position of the paralyzed right vocal cord, the anterior positioned arytenoid, the shortened vocal cord, and the glottic gap with right recurrent laryngeal nerve paralysis. (a). Normal with respiration. (b) Normal with phonation. (c) Right RLN paralysis with respiration. (d) Right RLN paralysis with phonation
Stroboscopy (phonoscopy) is a visualization of the vocal cords during vibration and may increase the diagnostic sensitivity of laryngoscopy especially in the evaluation of EBSLN injury. During stroboscopy light flashes just below or above the frequency at which the object is vibrating, therefore making the object appear at a standstill or as if it is vibrating in slow motion. High-speed digital recordings have increased the capability of stroboscopy to allow for diagnostic analysis. In cases of EBSLN injury, the affected true vocal cord appears flaccid, bowed, and shortened compared to the functional contralateral cord.
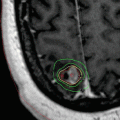
Transcutaneous laryngeal ultrasound has recently been described as an additional modality useful in assessing vocal cord function. Using a linear ultrasound probe similar to that used for thyroid ultrasound, but at lower frequency (8–10 mHz) and increased gain settings, the true and false vocal cords can be visualized in static and active motion with ultrasound (Fig. 19.5 – Transcutaneous laryngeal ultrasound). Visualization of the TVC’s is necessary to judge vocal fold movement appropriately. The sensitivity of ultrasound is notably lower than laryngoscopy and is unlikely to replace laryngoscopy; however, ultrasound may be a useful, non-invasive method of screening for RLN function in the perioperative period for thyroidectomy patients [13, 14].
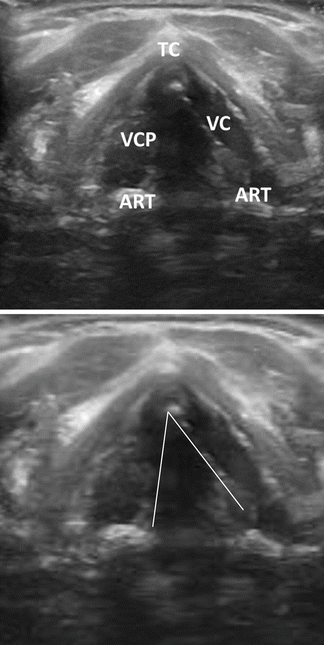

Fig. 19.5
Transcutaneous laryngeal ultrasound. Right vocal cord paralysis during respiration with paramedian location of the right true vocal fold. TC thyroid cartilage, VC vocal cord, ART arytenoid, VCP vocal cord paresis (Permission granted from Carneiro-Pla et al. [14]. See enclosed permission from Elsevier)
Preoperative confirmation of vocal cord function can be applied on a routine or on a case-by-case basis. Patients with evident preoperative voice dysfunction, a risk of invasive thyroid malignancy or a past history of neck surgery, where the RLN was at risk for injury are situations where preoperative vocal cord assessment can be especially useful. Generally, patient reported voice complaints have a poor predictive value for nerve dysfunction, whereas experienced surgeons documenting voice abnormalities may be more specific as a screening method for selecting patients for laryngoscopic examination [15].
Treatment of Vocal Cord Dysfunction
Treatment modalities for vocal cord dysfunction related to RLN or EBSLN injury include voice therapy and operative treatments. Voice therapy is typically provided by speech language pathologists and includes interventions that help with pitch alteration, increasing breath support and loudness, and techniques finding the correct head and neck position for optimal voice production such as turning head to one side or manipulating the thyroid cartilage. Voice therapy can be especially useful in the immediate postoperative period in cases of neuropraxia and allow time for spontaneous recovery of the injured nerve. In patients who have not achieved acceptable voice quality with speech therapy and have persistent vocal cord paralysis after an appropriate period of time to allow for recovery of neuropraxia (typically 6 months), an operative approach can be considered. Swallowing therapy may also be necessary if the tendency for aspiration is apparent.
Operative treatment options for vocal cord paralysis include injection laryngoplasty and laryngeal framework surgery. The primary goal of these treatments is to correct the phonatory gap between the paramedian paralyzed vocal cord and the contralateral functioning vocal cord. Medialization of the paralyzed cord to a near median position can improve the phonatory gap and help produce more normal voice and can improve aspiration due to laryngeal incompetence. Thyroplasty (injection laryngoplasty) is an injection of material laterally into the vocal fold which acts to displace the cord medially thus improving the glottic gap during phonation (Fig. 19.6). Injection materials can be temporary (hyaluronic acid, acellular human cadaveric dermis, gelfoam, and collagen) or permanent (calcium hydroxyapatite, fat, Teflon). Injection laryngoplasty can often be accomplished with local anesthesia and a flexible laryngoscope in a clinical office setting. Medialization thyroplasty (laryngeal framework surgery) involves operative placement of a Silastic or Gore-tex implant lateral to the vocal fold via a window cut in the thyroid cartilage (Fig. 19.7). This acts to displace the paralyzed vocal fold medially, ensuring adequate glottic closure. Some will add medialization and anterior rotation of the ipsilateral arytenoid to address the vocal cord gap at the posterior aspect of the vocal cords. Thyroplasty procedures result in a vocal cord in the medial position with improved vocal cord length which then generates a more normal voice. Thyroplasty should not be considered until surgical treatment of the thyroid pathology is complete (i.e., not before completion thyroidectomy if that is necessary).
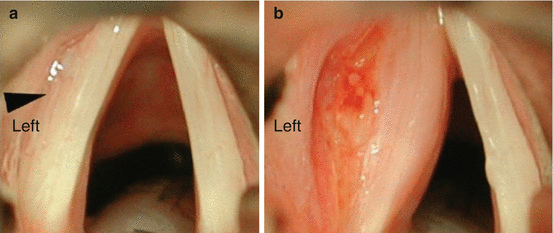
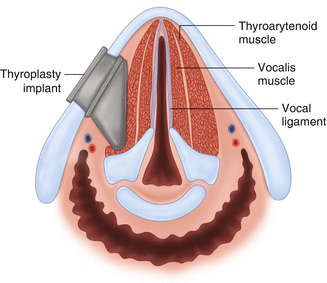

Fig. 19.6
Injection thyroplasty (a) Arrowhead indicates paralyzed left vocal cord with bowed appearance. (b). After injection thyroplasty the paralyzed cord has a more medial location to allow for improved glottic closure with phonation

Fig. 19.7
Type 1 thyroplasty – A right-sided thyroplasty implant with medialization of the right vocal fold
In cases of bilateral vocal cord paralysis, the primary problem to address is the small airway opening between the bilateral paramedian paralyzed vocal cords. Tracheostomy should be considered the standard of care, especially in the early postoperative period, as many nerve injuries which are neuropraxic in nature will recover over time, and patients will recover normal cord function and eventually be able to be decannulated. For patients with persistent bilateral vocal cord paralysis with persistent stridor, operative intervention directed at the airway obstruction should be considered. Cordotomy involves incising the paralyzed vocal cord with a transverse, curvilinear incision resulting in traction away from the arytenoid process resulting in enlargement of the airway through tissue retraction. Cordotomy should aim to provide both respiratory and phonatory function with a goal of tracheostomy tube decannulation with some level of preserved phonatory function. Cordotomy can be used in the acute nerve injury setting as no significant vocal cord tissue is removed.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree