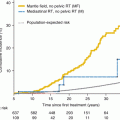
Minimum ovarian dose (GY)
Effect
0.6
None
1.5
No deleterious effect in most young women. Some risk of sterilization especially in women aged >40
2.5–5.0
Variable. Aged 15–40 years: about 60 % sterilized permanently, some with temporary amenorrhea. Aged >40: usually 100 % permanently sterile
5–8
Variable. Aged 15–40 years: about 70 % sterilized permanently; of the remainder, some temporary amenorrhea
>8
100 % permanently sterilized
13.1.4 Cytotoxic Effects of Chemotherapy
The effects of chemotherapy on ovarian function are both agent- and dose-dependent, and this effect may be additive to that resulting from abdominopelvic radiotherapy. Alkylating agents affect the resting oocyte in a dose-dependent, cell cycle-independent manner. Thecal cells and ova are depleted, as are the primordial follicles, resulting in arrest of follicular maturation and decreased estrogen secretion. Again, as was the case with radiotherapy, the effects are more pronounced in postpubertal as compared with prepubertal females, due to the fact that postpubertal females have fewer remaining viable oocytes. The effects worsen with age, as the normal aging process is accompanied by an ongoing depletion of oocytes. Risks of menstrual irregularity, ovarian failure, and infertility increase with age at treatments. Conversely, younger females can tolerate higher doses of alkylating agents without impairment of fertility, compared with adult females [12–17].
13.2 Clinical Manifestations
13.2.1 Effects of Radiotherapy on Ovarian Function
The clinical relationship between ovarian failure and the dose of radiation to the ovary is well illustrated by Stillman’s study of 182 girls treated at less than 17 years of age with 12–15 Gy of abdominal radiotherapy. Overall, primary ovarian failure occurred in 22 girls (12 %). However, ovarian failure was noted in 68 % of the girls whose ovaries received the full irradiation dose, but in only 14 % of those who had at least one ovary at the edge of the abdominal treatment volume (estimated dose 0.9–10 Gy, with a mean of 2.9 Gy). Conversely, none of 34 girls who received an estimated ovarian dose of 0.5–1.5 Gy (mean: 0.54 Gy) to at least one ovary outside the direct treatment volume had ovarian failure. Covariate and multivariate revealed that the location of the ovaries relative to radiation treatment fields was the only risk factor for ovarian failure [18].
In considering the risk of ovarian failure related to radiotherapy, other fields than the abdomen and pelvis must be considered. Direct or scattered irradiation from the spinal component of craniospinal radiotherapy may also produce ovarian damage [8, 19]. With the expanded use of hematopoietic stem cell transplantation in pediatric oncology, it is important to recall that total body irradiation (TBI) utilized in the conditioning regimen is commonly associated with primary ovarian failure or premature menopause, with prevalence rates as high as 90–100 %. Fraction size is of importance as well as the age of the patient at the time of radiotherapy [20–24].
13.2.2 Effects of Chemotherapy on Ovarian Function
The dose–response relationship of alkylating agents, and the effect of age, is a recurring theme in studies of fertility following chemotherapy. Amenorrhea and ovarian failure occur more commonly in adult women treated with cyclophosphamide and other alkylating agents than with adolescents, with prepubertal females tolerating cumulative cyclophosphamide doses as high as 25 g/m2 [13, 25]. In examining protocols with common chemotherapy, 86 % of women >24–30 years have been shown to have ovarian failure, compared with 28–31 % of younger women [3, 25].
It is clear that the sterilizing effects of all alkylating agents are not equal. Mechlorethamine and procarbazine together are perhaps the most damaging of the alkylating agents. These chemotherapy agents were used in the past together for the treatment of Hodgkin lymphoma, often in combination with radiotherapy, resulting in impaired fertility, among other adverse long-term effects [26]. Newer risk-adapted protocols for Hodgkin lymphoma have been developed to avoid mechlorethamine or procarbazine and to limit cumulative doses of other gonadotoxic alkylating agents, without negatively impacting the efficacy of the chemotherapy regimens [27–30].
Newer studies have also been designed to collect long-term follow-up data, and investigators are starting to collect data on the impact of these changes.
In recent years, ifosfamide, a congener of cyclophosphamide, has been used for a variety of solid tumors and lymphoma. The effects of ifosfamide on reproductive function are only beginning to be evaluated. A case report of successful pregnancies in two young women treated with high-dose ifosfamide and cyclophosphamide at Memorial Sloan Kettering was reported in 2001 [31]. In 2008, a small case series, which included 13 females treated for sarcoma with ifosfamide as the only alkylating agent, there was no primary ovarian failure reported. However, AMH levels were lower than an age-matched reference group, suggesting risk for early menopause [32].
Due to improved survivorship from childhood cancer noted as early as the 1970s–1980s, large cohorts of female survivors have reached the third and fourth decades of life, where the risk for infertility and premature menopause has been examined. In this era of treatment for these cohorts, the use of both radiotherapy and chemotherapy together was common, and thus it is not possible to fully separate the effects of the two modalities of therapy.
Two large studies of these survivors demonstrated elevated risks for infertility and premature menopause [11, 33]. A study of 2,498 female survivors, treated between 1945 and 1975, showed a 7 % deficit in fertility, compared with siblings. Between ages 21 and 25 years, survivors had a risk of premature menopause four times greater than that of siblings. Treatment-related risk factors included radiotherapy alone (RR = 3.7), alkylating agents alone (RR = 9.2), or a combination of both (RR = 27). By age 31, 42 % of these women had reached menopause, compared with 5 % of siblings [11]. In a study of 719 survivors treated between 1964 and 1988, 15.5 % of women were unable to conceive. Women treated with abdominopelvic radiotherapy alone had a fertility deficit of 23 %, compared with those treated with surgery. As with the previous study, the risk of infertility and premature menopause increased with increasing dose of abdominopelvic radiotherapy and amount of alkylating agent [33].
Several studies have been conducted within the Childhood Cancer Survivor Study (CCSS), a cohort of survivors treated between 1970 and 1987. In an analysis of 3,390 female survivors, acute ovarian failure was self-reported in 215 (6.3 %). Risk factors in multivariate analysis included increased dose of ovarian radiation, exposure to cyclophosphamide in those 13–20 years of age, and any exposure to procarbazine [34]. Premature menopause was also noted in 8 % of participants studied compared to 0.8 % in a sibling cohort. Risk factors for premature menopause were higher-attained age, increased dose of radiotherapy to the ovaries, increased alkylating agent exposure as determined by the alkylating agent score, and a diagnosis of Hodgkin lymphoma [35]. In another CCSS analysis of 5,149 female participants and 1,441 female siblings, the relative risk for survivors ever being pregnant was 0.81 compared to the siblings. Risk factors in multivariate analysis included ovarian/uterine radiation dose of >5 Gy, hypothalamic/pituitary dose of ≥30 Gy, and alkylating agent dose score of 3 or 4 [36].
Similar to what has been done with conventional chemoradiotherapy protocols, transplant conditioning protocols without TBI are being utilized to avoid some of the associated adverse long-term sequelae. The use of high-dose cyclophosphamide without TBI or other alkylating agents is associated with a lower risk of ovarian failure than conditioning regimens with TBI or multiple alkylating agents. In a study by Sanders, 100 % of women (n = 15) younger than age 26 and three of nine older than age 26 who were treated with 200 mg/kg cyclophosphamide recovered normal gonadotropin levels and menstruation posttransplantation [37]. However, many transplant protocols use high doses of alkylating agents together, most commonly busulfan and cyclophosphamide, which are associated with similar degrees of ovarian failure in females as protocols containing TBI [38].
13.2.3 Effects of Radiotherapy and Chemotherapy on Reproductive Outcomes
Many survivors of childhood cancer previously treated with cytotoxic therapy will remain fertile, and, therefore, pregnancy outcomes and the risk of cancer or genetic disease in offspring must be addressed. Young women who have been exposed to radiotherapy below the diaphragm are also at risk of impaired uterine development, which can adversely affect pregnancy outcomes, often resulting in premature labor and low-birth-weight infants. The magnitude of the risk is related to the radiotherapy field, total dose, and fractionation schedule. Female long-term survivors treated with total body irradiation and marrow transplantation are at risk for impaired uterine growth and blood flow, and, if pregnancy is achieved, for early pregnancy loss and premature labor. Despite standard hormone replacement, the uterus of the childhood cancer survivor may be impaired in its development and measure only 40 % of normal adult size, the ultimate uterine volume correlating with the age at which radiotherapy was received [7, 20].
With more childhood cancer survivors retaining fertility, pregnancy outcome data is now available. Of 4,029 pregnancies occurring among 1,915 women followed in the Childhood Cancer Survivor Study (CCSS), there were 63 % live births, 1 % stillbirths, 15 % miscarriages, 17 % abortions, and 3 % unknown or in gestation. Risk of miscarriage was 3.6-fold higher in women treated with craniospinal radiotherapy and 1.7-fold higher in those treated with pelvic radiotherapy. Chemotherapy exposure alone did not increase the risk of miscarriage. Compared with siblings, however, survivors were less likely to have live births and more likely to have medical abortions and low-birth-weight babies [39]. In an updated analysis of this cohort, it was noted that offspring of women who receive uterine radiation doses of >5 Gy were more likely to be small for gestational age [35]. In another analysis from the CCSS, Signorello also found that uterine and ovarian radiation doses of >10 Gy increased risk of stillbirth or neonatal death, and furthermore, for girls treated prior to menarche, uterine or ovarian doses as low as 1.0–2.49 Gy increased the risk of stillbirth or neonatal death [40].
In a Danish population-based cohort, in analysis of 34,000 pregnancies, which included 1,479 pregnancies of childhood cancer survivors, there were no significant differences noted in the proportions of live births, stillbirths, or all kinds of abortions combined between survivors and women without cancer. However, survivors had a 23 % increased risk for spontaneous abortion, with ovarian and uterine radiotherapy as the major significant risk factor [41].
In a Finish population-based cohort, in an analysis of 3,501 and 16,908 children of female cancer patients and siblings, respectively, the risk of stillbirth or early neonatal death was not significantly increased among offspring of cancer survivors as compared to offspring of siblings [42]. In a case-cohort study conducted involving 472 Danish survivors of childhood and adolescent cancer and their 1,037 pregnancies, no statistically significant associations were found between genetic disease in children and parental treatment with alkylating drugs or preconception radiation doses to the testes in male and ovaries in female cancer survivors. A statistically significant association between abdominopelvic irradiation and malformations, stillbirths, and neonatal deaths was not seen in the children of female survivors overall or in the children of mothers receiving high uterine doses [43].
In the National Wilms Tumor Study, records were obtained for 427 pregnancies of >20 weeks duration. In this group, there were 409 single and 12 twin live births. Early or threatened labor, malposition of the fetus, lower-birth-weight (<2,500 g), and premature delivery (<36 weeks) were more frequent among women who had received flank radiotherapy, in a dose-dependent manner [44].
Preservation of fertility and successful pregnancies may occur following HSCT. Sanders and colleagues evaluated pregnancy outcomes in a group of females treated with bone marrow transplant. Among 116 treated before puberty and 23 treated after the onset of puberty who retained ovarian function, 32 (28 %) and 9 (30 %), respectively, became pregnant. Of the 32 pregnancies in those treated with TBI, 16 resulted in early termination, compared with a 21 % prevalence of early termination in those treated with cyclophosphamide alone. There were no pregnancies among the women treated with busulfan and cyclophosphamide [37].
For childhood cancer survivors who have offspring, there is the concern about congenital anomalies, genetic disease, or risk of cancer in the offspring. In the report from the National Wilms Tumor Group, congenital anomalies were marginally increased in the offspring of females who had received flank radiotherapy [44]. However, this risk was not observed in a study of 247 offspring of 148 cancer survivors treated at a single institution [45] or in several larger cohort studies. In a study that compared a group of 2,198 offspring from adult survivors treated for childhood cancer between 1945 and 1975 with a group of 4,544 offspring from sibling controls, there were no differences in the proportion of offspring with cytogenetic syndromes, single-gene defects, or simple malformations. There was no association of type of childhood cancer treatment used and the occurrence of genetic disease in the offspring [46]. In the CCSS, among the 1,915 female survivors who reported 4,029 pregnancies, there was no increased risk of offspring with simple malformations, cytogenetic syndromes, single-gene defects, or congenital malformations [35]. In a subsequent analysis from the CCSS, among children of 1,627 female cancer survivors, there was no increased risk for congenital anomalies and no increase conferred from ovarian radiation or alkylating exposure [47].
Similar results were reported in a study of 5,847 offspring of survivors of childhood cancers treated in five Scandinavian countries. In the absence of a hereditary cancer syndrome (such as hereditary retinoblastoma), there was no increased risk of cancer [48]. In an updated analysis from Finland among 26,331 children of pediatric and young adult cancer survivors and 58,155 children of siblings, there was no increased risk of cancer in the offspring of the cancer survivors in the absence of a known cancer predisposition syndrome [49].
Further follow-ups are needed to determine whether patterns of cancer or genetic disease in offspring change with changes in cancer treatments, further elapsed time, and studies of greater numbers of offspring.
13.3 Detection and Screening
All prepubertal females who are treated with potentially gonadal toxic radiotherapy or chemotherapy should be rigorously assessed for appropriate progression through puberty. The average age for menarche is 12.7 years ± 1.0 year [50]. An evaluation should include a complete history, a physical examination that includes an assessment of sexual development and pubertal milestones (Tables 13.2 and 13.3) and selected laboratory studies (Table 13.4), as summarized in Table 13.5. Reduced ovarian volume and low inhibin B and anti-Mullerian hormone concentrations in survivors with regular menses may be markers of incipient ovarian failure [2, 7]. In conjunction with the evaluation of gonadal effects, attention must be paid to growth. Cranial radiotherapy confers significant risk for growth hormone deficiency. Once patients have reached full sexual maturity, linear growth will stop. Linear and sexual development must, therefore, be monitored simultaneously (see the chapter on “Neuroendocrine Late Effects” for further details). Patients who received radiotherapy to the central nervous system or the neck are also at risk for thyroid dysfunction that can negatively impact gonadal function and linear growth. Even after successful progression through puberty, it is important to monitor the frequency and characteristics of menstrual periods, due to risk for premature menopause. Females with ovarian failure, either primary or secondary, should undergo assessments for impaired bone mineral density. Calcium intake, weight-bearing exercise, a history of fractures, and a family history of osteopenia/osteoporosis should be evaluated. The determination of bone mineral density, using dual-energy X-ray absorptiometry (DXA) scan, and comparison of results with the well-established adult normative values, is indicated for all adult females. Screening in children is less defined. Several different measurement techniques and standards have been applied, but none has been well validated in large pediatric populations (much less in pediatric oncology patients). However, some monitoring is indicated, and trends over time may be of greater value than a single DXA scan. The Children’s Oncology Group has published guidelines (www.survivorshipguidelines.org) [51] that are helping in determining surveillance for adverse long-term ovarian or ovarian-associated outcomes, as reviewed by Metzger and colleagues [52].
Stage | Age (mean ± SD, years) |
|---|---|
I. Preadolescent. Only papilla is elevated | |
II. Breast and papilla are elevated as small mound. Areolar diameter is enlarged | 10.0 ± 1.0 |
III. Areola and papilla project to form a secondary mound above the level of the breast | 11.9 ± 1.0 |
IV. There is projection only of papilla because of recession of the areola to the general contour of the breast | 12.9 ± 1.2 |
Stage | Age (mean ± SD, years) |
|---|---|
I. Preadolescent vellus over pubis is no further developed than that over anterior abdominal wall (i.e., no pubic hair) | |
II. There is sparse growth of long, slightly pigmented, downy hair, straight or only slightly curled, appearing chiefly along the labia | 11.2 ± 1.1 |
III. Hair is considerably darker, coarser, and more curled. Hair spreads sparsely over pubic junction | 11.9 ± 1.1 |
IV. Hair is now adult in type but area covered by it is still considerably smaller than in most adults. There is no spread to medial surface of the thighs | 12.6 ± 1.1 |
Table 13.4
Laboratory assessment for ovarian function
Testing | Treatment exposure | Time and frequency of evaluations |
|---|---|---|
LH, FSH, estradiol | Alkylating agents | Baseline at 11 years of age or older, and then yearly |
Abdominopelvic, cranial, or total body radiotherapy | Assessment also of whether the following are present: delayed puberty, irregular menses or amenorrhea, clinical signs or symptoms of estrogen deficiency | |
Free T4, TSH | Neck, cranial, or total body radiotherapy | Yearly |
Assessment also of presence of signs or symptoms of thyroid dysfunction |
Table 13.5
Pertinent history and physical examination
History | Physical examination |
|---|---|
Doses and types of chemotherapy agents received | Height, weight, and height velocity |
Doses and fields of radiotherapy | Complete examination of all organ systems, with particular attention to pubertal status and thyroid gland |
Surgical history, especially for patients with CNS and GU tumors | Gynecologic examination in postpubertal females as indicated by treatment history, sexual activity, and overall developmental status |
Patient and maternal history of menarche and thelarche | |
Menstrual periods – timing and tempo | |
Symptoms of estrogen deficiency (hot flashes, dry skin, leg cramps, reduced libido) | |
Parental heights | |
Family history of infertility, pregnancy, labor complications, assisted fertilization |
Pediatric endocrinologists and reproductive endocrinologists/gynecologists are essential consultants in the monitoring, prevention, and management of ovarian late effects in childhood cancer survivors.
13.4 Management of Established Problems
13.4.1 Prevention Strategies
Reduction in the dose or use of alkylating agents and abdominopelvic radiotherapy is the most effective means of preserving ovarian function and promoting positive reproductive outcomes. There are, however, many instances where cytotoxic and gonadal toxic chemotherapy and radiotherapy are still required for long-term cure. As a result, additional strategies need to be employed to minimize adverse long-term outcomes. To shield the ovaries from direct irradiation during abdominal or pelvic radiotherapy, an oophoropexy may be performed if it is possible to move the ovaries to a location that can be safely shielded without jeopardizing the patient for tumor recurrence. Typically, with abdominal radiotherapy for Hodgkin lymphoma that targets lymph nodes, the ovaries are moved to a midline position in front of or behind the uterus. For pelvic radiotherapy, they may be moved laterally to the iliac wings. This may also be helpful for young girls or adolescents undergoing cranial spinal radiotherapy for brain tumors using historic radiation techniques, though current approaches with intensity modulation allow ovarian sparing. However, if such techniques are not possible, then the ovaries should be marked by the surgeon with clips that can later by identified by a simulator film. Central pelvic blocking at the time of “inverted Y” field will prevent direct irradiation, although scatter dose and transmitted dose will be inevitable. Medial or lateral transposition of the ovaries results in ovarian doses of 8–10 % and 4–5 %, respectively, of the pelvic dose [53]. For most patients, this will be compatible with the preservation of fertility, although there may be temporary amenorrhea.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree