Squamoid, Spindle, and Giant Cell Lesions
UNDIFFERENTIATED (ANAPLASTIC) CARCINOMA
Anaplastic or undifferentiated carcinoma historically accounted for 5% to 10% of all primary malignant tumors of the thyroid [1], but in many centers, this number is decreasing, presumably due to earlier detection of antecedent disease. These are usually tumors of the elderly and show a slight female predominance. Their rapid growth and massive local invasion precipitate local symptoms that tend to clinically overshadow the early metastases to the lung, adrenals, and bone [2, 3]. They are highly lethal malignancies with a 5-year survival rate of only 7.1% [4] and a mean survival period of 6.2 to 7.2 months following diagnosis [4, 5].
The data suggest that anaplastic carcinoma most often originates in an abnormal thyroid gland. The tumor has a higher incidence in regions of endemic goiter, and a history of goiter is reported in over 80% of cases [4, 6]. However, it is difficult to document transformation of a benign lesion to a highly malignant tumor. The reported association between well-differentiated thyroid carcinoma and anaplastic carcinoma ranges from 7% to 89% of cases. The lower figures are likely underestimates, attributable to failure to detect a well-differentiated component due to inadequate sampling [4, 5, 6, 7, 8, 9, 10]. The association of papillary carcinoma, particularly the more aggressive tall cell variant, with anaplastic tumors has also been described [6, 10, 11].
Thyroid carcinomas can exhibit an entire spectrum of differentiation through insular to anaplastic foci. The significance of microscopic insular or anaplastic change is controversial. Some people have suggested that focal microscopic dedifferentiation does not alter prognosis, but others have shown that this finding alone is statistically significant as a marker of aggressive behavior. Poorly differentiated carcinoma is intermediate in the spectrum and may represent a transition form [10, 12].
The factors underlying dedifferentiation in thyroid tumors remain to be established, although age and exposure to radiation have been implicated [13, 14]. Clearly, the vast majority of well-differentiated thyroid lesions do not undergo transformation to anaplastic carcinoma. A pattern of genetic mutations resulting in oncogene activation or loss of tumor suppressor gene activity has been proposed to correlate with the stepwise
progression from adenoma to carcinoma and through the dedifferentiation process in thyroid [15, 16, 17]. The majority of anaplastic thyroid carcinomas have BRAF or RAS mutations with additional events accounting for progression [17, 18, 19]; ATCs have a greater mutation burden, including mutations in TP53, TERT promoter, PI3K/AKT/mTOR pathway effectors, SWI/SNF subunits, and histone methyltransferases. Tp53 mutations are common in anaplastic thyroid carcinomas [15, 16, 20, 21, 22, 23, 24] and are therefore often diffusely positive with immunohistochemistry [25, 26]. At the molecular level, the patterns of allelic loss show that the majority of cases have a core of conserved mutations in differentiated and undifferentiated areas with substantial increases in mutation rates in the anaplastic components [27], suggesting that additional genetic events promote tumor progression.
progression from adenoma to carcinoma and through the dedifferentiation process in thyroid [15, 16, 17]. The majority of anaplastic thyroid carcinomas have BRAF or RAS mutations with additional events accounting for progression [17, 18, 19]; ATCs have a greater mutation burden, including mutations in TP53, TERT promoter, PI3K/AKT/mTOR pathway effectors, SWI/SNF subunits, and histone methyltransferases. Tp53 mutations are common in anaplastic thyroid carcinomas [15, 16, 20, 21, 22, 23, 24] and are therefore often diffusely positive with immunohistochemistry [25, 26]. At the molecular level, the patterns of allelic loss show that the majority of cases have a core of conserved mutations in differentiated and undifferentiated areas with substantial increases in mutation rates in the anaplastic components [27], suggesting that additional genetic events promote tumor progression.
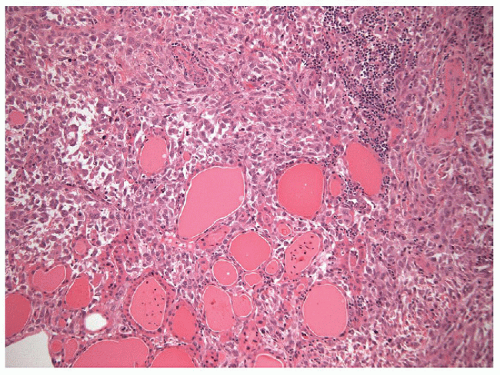
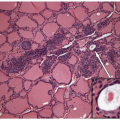
Anaplastic carcinomas are highly infiltrative tumors, and on histology, it is common to find the malignant cells entrapping normal thyroid elements (Fig. 12.1). The tumor also invades into skeletal muscle, adjacent adipose tissue, and other extrathyroidal structures. Blood vessel invasion and thrombosis is frequent, with or without tumor cell involvement.
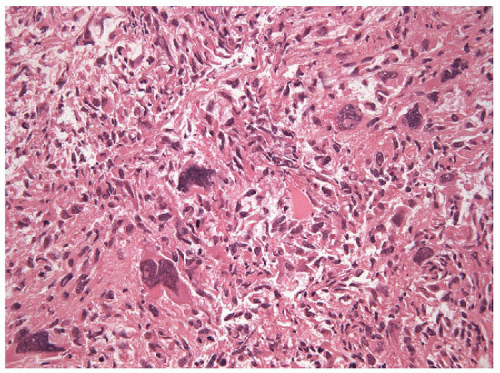
On both cytology and histology, anaplastic carcinomas exhibit wide variations in appearance with several morphologic patterns recognized and many tumors manifesting a mixed morphology. A common morphologic presentation, and one that is most easily recognized as an anaplastic carcinoma of the thyroid, is that of the biphasic spindle and
giant cell tumor (Fig. 12.2). Other tumors are dominated by bizarre, malignant giant cells and still others may show a more pure population of spindle cells (Fig. 12.3). Occasionally, the tumor is simply a highly malignant, undifferentiated, large cell carcinoma resembling a
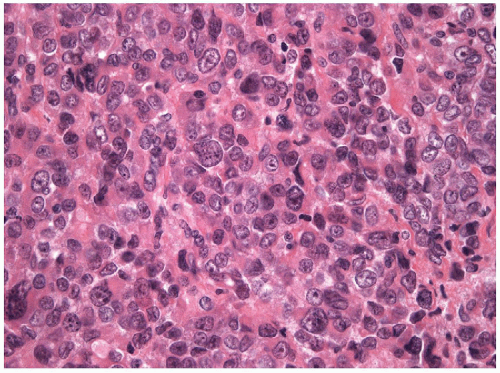
lymphoepithelioma-like carcinoma (Fig. 12.4). All variations of anaplastic carcinoma of the thyroid are highly proliferative with numerous mitotic figures and atypical mitoses (Fig. 12.5). There is usually extensive geographic necrosis (Figs. 12.6 and 12.7, e-Fig. 12.1), and in some cases,
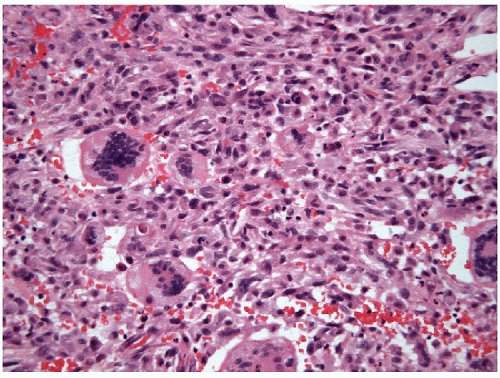
the necrosis may be so widespread that the only viable tumor is preserved around blood vessels. Inflammatory infiltrates are frequently seen with the necrosis. Osteoclast-like giant cells may be present (Fig. 12.8, e-Fig. 12.2) and have been shown by immunohistochemical studies to be
of the monocytic/histiocytic lineage [28, 29]. Neoplastic bone and cartilage may also be identified.
giant cell tumor (Fig. 12.2). Other tumors are dominated by bizarre, malignant giant cells and still others may show a more pure population of spindle cells (Fig. 12.3). Occasionally, the tumor is simply a highly malignant, undifferentiated, large cell carcinoma resembling a
lymphoepithelioma-like carcinoma (Fig. 12.4). All variations of anaplastic carcinoma of the thyroid are highly proliferative with numerous mitotic figures and atypical mitoses (Fig. 12.5). There is usually extensive geographic necrosis (Figs. 12.6 and 12.7, e-Fig. 12.1), and in some cases,
the necrosis may be so widespread that the only viable tumor is preserved around blood vessels. Inflammatory infiltrates are frequently seen with the necrosis. Osteoclast-like giant cells may be present (Fig. 12.8, e-Fig. 12.2) and have been shown by immunohistochemical studies to be
of the monocytic/histiocytic lineage [28, 29]. Neoplastic bone and cartilage may also be identified.
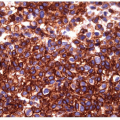
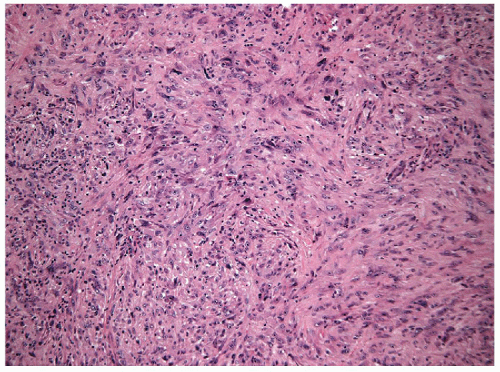 FIGURE 12.3 Spindle cell variant of anaplastic carcinoma. Spindle cells dominate the tumor (hematoxylin and eosin stain). |
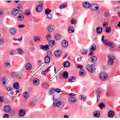
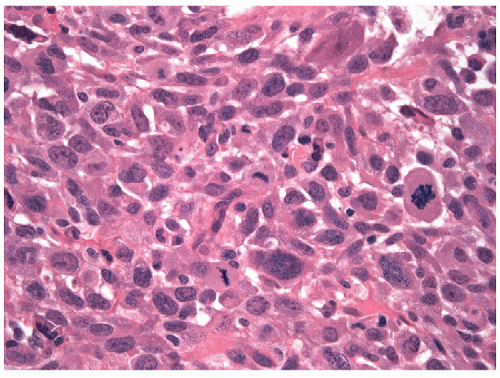 FIGURE 12.5 Anaplastic carcinoma. In keeping with their rapid growth, mitoses are frequent in anaplastic carcinomas, and atypical mitoses are commonly found (hematoxylin and eosin stain). |
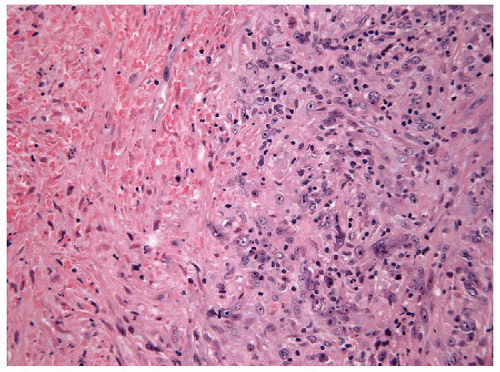 FIGURE 12.6 Anaplastic carcinoma of the thyroid. Necrosis is common and as in this example may induce an inflammatory reaction (hematoxylin and eosin stain). |
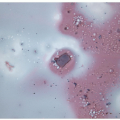
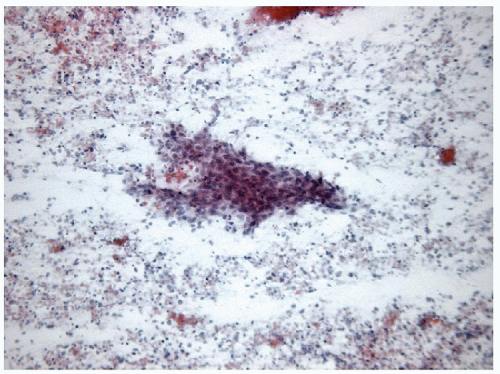 FIGURE 12.7 Anaplastic carcinoma of the thyroid. The necrotic background is often apparent in FNA samples admixed with a variable degree of inflammation (direct smear, Papanicolaou stain). |
On fine needle aspiration (FNA) or core biopsy, these tumors present a variety of differential diagnostic possibilities depending on the predominant morphologic pattern present in the sample. When tumor giant cells are numerous, the appearance is fairly characteristic and a high degree of certainty of an anaplastic carcinoma can be achieved. When the tumor presents in the form of a poorly differentiated, large cell carcinoma, the differential diagnosis includes medullary carcinoma, direct extension of a laryngeal squamous carcinoma, thymic carcinoma, metastatic carcinoma, melanoma, and large cell lymphoma (both diffuse large cell lymphoma and anaplastic large cell lymphoma). When the spindle cell morphology dominates, or the tumor appears biphasic with both epithelial and spindle cell components, the differential diagnosis once again includes medullary carcinoma, metastatic carcinoma (in particular a sarcomatoid renal primary), and melanoma but now must include sarcomas (both primary and metastatic) as well as involvement of the thyroid by a primary laryngeal sarcomatoid squamous carcinoma. Resolution of these diagnostic possibilities requires careful attention to the morphology, combined with immunohistochemical studies (which are often of limited help), and clinical information to arrive at the conclusion that the poorly differentiated malignancies is an anaplastic carcinoma of the thyroid. Given the large number of differential diagnoses, the most accurate diagnosis relies on the identification of a more differentiated thyroid carcinoma with evidence
of progression. It is not surprising that misdiagnosis is common [30] and reclassification is frequent on expert review.
of progression. It is not surprising that misdiagnosis is common [30] and reclassification is frequent on expert review.
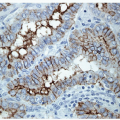
Immunohistochemistry is useful in only a limited fashion in the diagnosis of these lesions. Most anaplastic carcinomas do not contain convincing reactivity for TTF-1 or thyroglobulin, and the few that are positive have only a weak or focal reaction [5, 8, 29, 31, 32, 33, 34]. Pax-8 is a better marker [35, 36] that is more sensitive and does not stain lung tumors but still does not detect all anaplastic thyroid carcinomas. Focal positivity for Pax-8, TTF-1, or thyroglobulin must be interpreted carefully, since it may reflect trapped nontumorous follicles or follicular cells, or in the case of thyroglobulin alone, diffusion artifact. The epithelial nature of the tumor cells can be verified with stains for cytokeratins in some cases, but again most undifferentiated lesions are negative for this marker.
By electron microscopy [8, 28, 33, 37, 38], there may be formation of intercellular junctions, microvilli, and basal lamina, providing evidence of epithelial differentiation. However, many tumors do not exhibit evidence of any differentiation. Their large nuclei have prominent nucleoli and clumped chromatin; usually, the cytoplasm contains only poorly developed rough endoplasmic reticulum, scattered dense bodies, lipid droplets, numerous free ribosomes, mitochondria, and lysosomes. Intermediate filaments (keratin or vimentin) may form filamentous whorls that correspond to the acidophilic hyaline globules seen by light microscopy. Secretory granules are not seen in these tumors. Some tumors do not exhibit immunohistochemical or ultrastructural markers that allow classification as epithelial malignancies. Nevertheless, the diagnosis of anaplastic carcinoma should be favored for pleomorphic lesions in older patients if they arise in the thyroid. Most anaplastic thyroid carcinomas are aneuploid on flow cytometry; this abnormality correlates with poor outcome [39].
Giant Cell Variant of Anaplastic Carcinoma
The most common type of anaplastic carcinoma is the giant cell variant. As the name suggests, these tumors are predominantly composed of large cells with abundant amphophilic or eosinophilic, typically granular cytoplasm and bizarre, often multiple, hyperchromatic nuclei (Fig. 12.9). Some have round, densely acidophilic intracytoplasmic hyaline globules. These tumors grow in solid sheets, but artifactual tissue fragmentation may simulate an alveolar pattern (Fig. 12.10). FNA of the giant cell variant of anaplastic carcinoma is characterized by pleomorphic and often bizarre giant cells with multilobulated, single or multiple nuclei (Fig. 12.11, e-Figs. 12.3 and 12.4). The chromatin pattern is coarse with prominent parachromatin clearing and often multiple and bizarre nucleoli [40, 41, 42]. Necrotic material may be present in the background with a variable inflammatory reaction (Fig. 12.12). The malignant giant cells can be identified as epithelial since they express CK7, CK8, and CK18 [43].
An unusual variant of anaplastic thyroid carcinoma is the rhabdoid tumor [44, 45, 46]. This is a very rare and aggressive thyroid tumor with few
case reports appearing in the literature. These tumors are invasive, nonencapsulated solid lesions composed of large neoplastic cells supported by a delicate fibrovascular stroma. Areas of geographic necrosis and hemorrhage are seen (Figs. 12.13, 12.14, 12.15, e-Fig. 12.5). The neoplastic cells are large with abundant eosinophilic cytoplasm containing eosinophilic cytoplasmic inclusions and large eccentrically placed vesicular nuclei, frequently demonstrating a prominent nucleolus. Immunohistochemically, the rhabdoid cells may have focal positivity for Pax-8, TTF-1, vimentin, sarcomeric actin (e-Fig. 12.6), epithelial membrane antigen (EMA), and cytokeratin. They are negative for thyroglobulin, smooth muscle actin, desmin, myogenin, synaptophysin, chromogranin, and carcinoembryonic antigen [46, 47]. The cytoplasmic aggregates in the rhabdoid cells are often positive for epithelial markers [46] and by electron microscopy are seen to be composed of masses of intermediate filaments [40]. To our knowledge, the cytologic morphology of this lesion on aspiration biopsy has not been reported.
case reports appearing in the literature. These tumors are invasive, nonencapsulated solid lesions composed of large neoplastic cells supported by a delicate fibrovascular stroma. Areas of geographic necrosis and hemorrhage are seen (Figs. 12.13, 12.14, 12.15, e-Fig. 12.5). The neoplastic cells are large with abundant eosinophilic cytoplasm containing eosinophilic cytoplasmic inclusions and large eccentrically placed vesicular nuclei, frequently demonstrating a prominent nucleolus. Immunohistochemically, the rhabdoid cells may have focal positivity for Pax-8, TTF-1, vimentin, sarcomeric actin (e-Fig. 12.6), epithelial membrane antigen (EMA), and cytokeratin. They are negative for thyroglobulin, smooth muscle actin, desmin, myogenin, synaptophysin, chromogranin, and carcinoembryonic antigen [46, 47]. The cytoplasmic aggregates in the rhabdoid cells are often positive for epithelial markers [46] and by electron microscopy are seen to be composed of masses of intermediate filaments [40]. To our knowledge, the cytologic morphology of this lesion on aspiration biopsy has not been reported.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree