Polycythemia Vera
Robert T. Means, Jr.
DEFINITION AND HISTORY
Polycythemia vera (PV), also called polycythemia rubra vera, is a chronic clonal myeloproliferative disorder characterized by a striking absolute increase in the number of red blood cells and in the total blood volume, and usually by leukocytosis, thrombocytosis, and splenomegaly. The bone marrow is typically hypercellular and exhibits hyperplasia of myeloid, erythroid, and megakaryocyte lineages.
In 1892, Vaquez described persistent polycythemia, as distinguished from relative and transient forms, in a man whom he believed to have a congenital cardiac lesion even though no auscultatory signs were noted. At autopsy, a year after the patient was first examined, his heart was found to be normal.1 In 1903 and 1908, Osler recognized that various reports of polycythemia in the literature represented a single disorder and further clarified the clinical picture of the disease,2, 3 and 4 and Türk, in 1904, called attention to the occurrence of leukocytosis as well as to immature forms of cells of the red and white series, suggesting a hyperplastic disorder of blood formation involving the marrow as a tissue and not merely erythrocytes.5 Thus, the general clinical picture of the disease was established in the earliest publications.6
Synonyms for PV used in the older literature include erythremia, splenomegalic polycythemia, Vaquez disease, Osler disease, polycythemia with chronic cyanosis, myelopathic polycythemia, erythrocytosis megalosplenica, and cryptogenic polycythemia.
EPIDEMIOLOGY
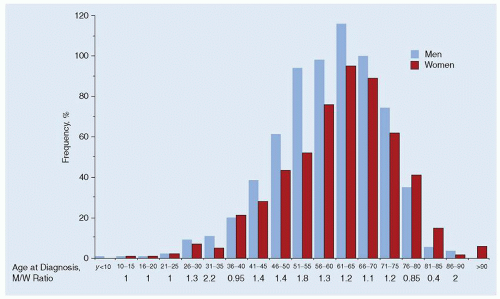
The age- and sex-adjusted incidence rate of PV was 1.9 per 100,000 person-years in Olmstead County, Minnesota, during the years 1935 to 1989,7 and 2.6 per 100,000 person-years in Malmo, Sweden, during 1980 to 1984.8 PV appears to be somewhat more common in men than in women, with reported male-to-female ratios ranging from 1.2 to 2.2 in various studies.7,9, 10 and 11 PV in younger patients reportedly shows less marked male predominance.10 Some8 but not all7 studies have suggested that the incidence of PV is increasing over time. The frequency of PV according to age and sex is shown in Figure 82.1.
PV tends to be a disease of older individuals, with peak incidence observed at 60 to 80 years of age,10,11 with maximum incidence exceeding 20 per 100,000 person-years.7 The mean age at diagnosis has increased steadily since the 1920s.11, 12, 13, 14 and 15 Cases of PV have also been reported in patients younger than the age of 40 years.16,17
Racial and ethnic factors influence the incidence of PV. PV is significantly less common in African Americans than Americans of European ancestry.9,13,18 A number of investigators (beginning with Türk) have reported that the incidence of PV is higher in individuals of Jewish origin.9,13,19,20 Ashkenazi Jews in Northern Israel exhibit a higher incidence of PV than do their neighbors of Arab or Sephardic Jewish origins.19
Familial occurrence of PV has been reported by 6% of patients enrolled in the protocols of the Polycythemia Vera Study Group (PVSG) and in intermittent sporadic cases.21, 22, 23, 24 and 25,26,27
It was previously claimed that PV affected primarily those of slender body habitus, but there is little evidence to support this statement.13
It was previously claimed that PV affected primarily those of slender body habitus, but there is little evidence to support this statement.13
CLINICAL FEATURES
The symptoms and signs of PV can be attributed in large part to the expanded total blood volume and to the slowing of the blood flow as a result of increased blood viscosity, as discussed in Chapter 44. At the time of diagnosis, it becomes apparent that the disorder has been present for a long time in most cases. Patients may complain of headache, dizziness, tinnitus, visual disturbances, dyspnea, lassitude, or weakness. Although the color of the skin often is acknowledged to have been abnormal for a long time, this complaint alone rarely brings the patient to the physician. Skin and mucous membrane hemorrhages are not uncommon; these, or a sense of weight or swelling in the abdomen owing to enlargement of the spleen, may be the initial symptoms. The lack of specificity of symptoms may in fact contribute to the delay in diagnosis. On the other hand, some patients have no complaints whatsoever, and polycythemia is discovered incidentally. Table 82.1 lists the frequency of common symptoms and physical findings in patients with PV.
Skin and Mucous Membranes
The color of the face is not cyanotic but is rather “ruddy,” as might be produced by severe sunburn or a profound blush. The face also often appears swollen. This “rubor” may be so intense that it produces a startling appearance. The face, particularly the lips, cheeks, tip of the nose, ears, and neck, show this color (Fig. 82.2), but the skin of the trunk usually is not particularly affected. The distal portions of the extremities exhibit these changes more than the proximal portions and may be more truly cyanotic. The skin capillaries are distended, and the capillary loops are enlarged. The degree of red or blue coloration depends on the state of dilation of the peripheral vascular network and on the speed of circulation through these areas, because these factors determine the quantity of reduced hemoglobin present.28 This is responsible for the frequent description in earlier reports that PV produced “cyanosis.” These findings are not unique to PV, but are also observed in patients with an elevated hematocrit from secondary erythrocytosis.
TABLE 82.1 PHYSICAL FINDINGS AND SYMPTOMS IN POLYCYTHEMIA VERA | ||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||
Ecchymoses and purpura of various sizes are common as the disease progresses. Red or dark-violet spots or brownish pigmentation of the skin may be found, and a great variety of skin lesions29 have been observed, including dry skin, eczema, acneiform or urticarial changes, acne rosacea, acne urticata,30 urticaria pigmentosa,31, 32, 33 and 34 and neutrophilic dermatosis (Sweet syndrome).35, 36, 37 and 38 The eyes may appear bloodshot. The mucous membranes may be a deep raspberry-red, and epistaxis and/or gingival bleeding may occur.
A common complaint is aquagenic pruritus, intense itching after exposure to water (most typically in a bath or shower). This may be the initial presentation of PV and is reported in up to 60% of PV patients younger than the age of 40 years.17,39,40 The itching may be so troublesome that bathing with hot or even warm water is avoided. The reaction is less frequent after the use of cold water. This complaint tends to disappear as the polycythemia is treated, but may require specific intervention. Reddening, swelling, and pain in the digits (erythromelalgia) may occur and are typically associated with extreme platelet elevations.41,42
Cardiovascular System
Cardiac symptoms are not particularly prominent, and cardiac hypertrophy is typically absent. The circulatory minute volume is reduced, and the velocity of blood flow is greatly lowered,43 but the cardiac output and work are normal.44,45 Electrocardiograms may show prolonged QT duration.46 When echocardiographic abnormalities are observed, they are typically associated with previous thromboembolic events and consequent pulmonary hypertension.47,48 PV occurs in the population generally considered to be at highest risk for myocardial infarction, so whether PV increases this risk, and if it does, to what degree, is unclear.49 Increased blood viscosity related to polycythemia, however, may contribute to symptoms in patients with atherosclerotic
cardiovascular disease.50,51 Thrombotic events, in both the arterial and venous circulations, are common in patients with PV. Nineteen percent of the 1,213 patients followed by the Gruppo Italiano Studio Policitemia (GISP) experienced a thrombotic event. Of these, 50.5% of nonfatal thrombotic events were documented as having occurred in the arterial circulation and 38.5% in the venous circulation. More than 80% of fatal thromboses were arterial.10 Other abnormalities of the venous system include varicosities and phlebitis.52 Venous thrombosis is more common in women.53 Moderate or significant thickening of the peripheral arteries is found in patients with PV, and coronary thrombosis, claudication without occlusion, arterial occlusion with gangrene, acroparesthesia, Raynaud phenomenon, and thromboangiitis obliterans have been described.54,55
cardiovascular disease.50,51 Thrombotic events, in both the arterial and venous circulations, are common in patients with PV. Nineteen percent of the 1,213 patients followed by the Gruppo Italiano Studio Policitemia (GISP) experienced a thrombotic event. Of these, 50.5% of nonfatal thrombotic events were documented as having occurred in the arterial circulation and 38.5% in the venous circulation. More than 80% of fatal thromboses were arterial.10 Other abnormalities of the venous system include varicosities and phlebitis.52 Venous thrombosis is more common in women.53 Moderate or significant thickening of the peripheral arteries is found in patients with PV, and coronary thrombosis, claudication without occlusion, arterial occlusion with gangrene, acroparesthesia, Raynaud phenomenon, and thromboangiitis obliterans have been described.54,55
As noted in Table 82.1 and in other studies,56 hypertension is relatively common in patients with PV. It is unclear whether this reflects the increased incidence of hypertension in the middle-aged and elderly or is a consequence of increased blood viscosity. Certainly, improvement of blood viscosity by reduction of the red cell volume aids in the control of blood pressure.
Gastrointestinal System
In addition to nonspecific gastrointestinal symptoms experienced by everyone at some time, such as feelings of fullness, thirst, gas pains, and constipation, patients with PV have an increased frequency of peptic ulcer, gastrointestinal bleeding, or thrombosis of mesenteric vessels. When compared with dyspeptic controls, PV patients had a three- to fourfold greater frequency of upper gastrointestinal erosions or ulcers.57,58 The relationship of blood basophil concentration, histamine content, gastric acid secretion, and dyspepsia on peptic ulceration has also been examined in a small group of patients with PV.59 No convincing correlations were demonstrated. The frequency of Helicobacter pylori infection is increased in PV patients.58
Massive hemorrhage from varices in the esophagus, stomach, or bowel may be observed.60, 61 and 62 Thrombosis in the mesenteric veins and arteries may be mistaken for peritonitis or the perforation of an ulcer. Hepatomegaly is common (Table 82.1), and cirrhosis has also been reported.63 Mosse syndrome is a term applied by some to the co-existence of cirrhosis and PV.64 Budd-Chiari syndrome also occurs,65,66,67 particularly in patients with a concurrent hypercoagulable state.68, 69, 70 and 71
Splenomegaly
Palpable splenomegaly occurs in more than two thirds of PV patients (Table 82.1).12,72 The size of the spleen varies greatly between individual patients and occasionally may extend to the pelvic brim.73 It is usually hard and smooth. Patients may experience pain in the splenic region, and after infarction a friction rub can be heard in this area. The general assumption is that polycythemia antedates the enlargement of the spleen and that engorgement of this organ with blood and extramedullary hematopoiesis are the major contributors to splenomegaly. Splenomegaly appreciated only by radiographic techniques was considered a minor diagnostic criterion in most pre-2007 diagnostic schemes for PV.74,75 Experience suggests that most practitioners regard it as the equivalent of splenomegaly observed on physical examination.
Respiratory System
Dyspnea on severe exertion is common, and hoarseness is not unusual. Chest radiographs often reveal prominent vascular markings. Before the establishment of the PVSG diagnostic criteria, which require a reasonably normal arterial oxygen saturation for the diagnosis of PV, it was noted that the vast majority of patients with PV had normal arterial oxygen saturation, even when the hemoglobin levels were high, indicating that the high viscosity of the blood does not prevent normal blood oxygenation; oxygen dissociation studies were also normal.76, 77, 78 and 79 High diffusing capacities, which were reduced after phlebotomy, were also noted in a number of patients with PV.80 In another study, hypoxia, as evidenced by a low arterial oxygen tension and saturation, was observed in the absence of demonstrable co-existent cardiorespiratory disease.81 In these individuals, ventilation-perfusion ratios were altered, and the diffusing capacity was low. The authors postulated that these findings might be the result of an alteration of the pulmonary vasculature caused by unrecognized thromboembolism. Pulmonary hypertension is reported in patients with PV and other myeloproliferative disorders.48,82
Genitourinary System
Neuromuscular System
Headache is the most common neurologic symptom87 but lassitude, vertigo and giddiness, transitory syncope, insomnia, weakness, and a sensation of fullness in the head and numbness and tingling in the fingers (less often in the feet) are also common.
Visual disturbances are common and include transitory dimness of vision, or even temporary blindness, scotomas, specks and bright points in front of the field of vision, diplopia, and temporary paralysis of one of the eye muscles. On examination of the eye grounds, the vessels may be engorged, tortuous, and irregular in diameter; the veins may be dark purple, and the retina deeply colored. Papilledema and embolism of the central retinal artery have been reported.88
The cerebrospinal fluid pressure may be increased.89 Ringing and roaring in the ears are exceedingly common when the hematocrit is significantly elevated. Ménière syndrome and chorea90 have also been reported.
Vascular lesions of the brain constitute the most serious complication of PV.91, 92, 93 and 94 A variety of neurologic syndromes, ranging from hemiparesis to seizures to alteration of cerebral function, have been reported in patients with PV. All such symptoms and signs presumably are secondary to increased blood volume and/or decreased blood flow. Investigators have shown clearly that cerebral blood flow is greatly diminished at hematocrit levels between 0.53 and 0.62,95 and that elevated hematocrit is a risk factor for cerebrovascular accidents.96,97 Central nervous system vascular events represented 30% of the nonfatal thrombotic events and 10.3% of deaths observed in the GISP study.10
Pain in the limbs may be troublesome and severe. It has been attributed to pressure on the bone by swollen, hyperplastic bone marrow. Unusual paresthesias may be encountered, but anatomic evidence of spinal cord changes has not typically been found at autopsy.
BLOOD AND LABORATORY FINDINGS
Hematologic Findings
Erythrocytes
Hemoglobin concentration typically is in the range of 18 to 24 g/dl. Red cell counts of 7 to 10 × 1012/L are common when patients with this disease are first evaluated, and values as high as 12 and even 15 × 1012/L have been recorded.98 The individual erythrocytes usually appear normal. Slight anisocytosis may be evident, but poikilocytosis is unusual. Polychromatophilia and,
occasionally, basophilic stippling may be found. An occasional normoblast may be observed in the blood smear, and, in the presence of a relatively normal or definitely increased red cell count, should arouse suspicion of PV. The reticulocyte percentage is not significantly increased. After hemorrhage, however, the reticulocytes may be increased, and a number of other immature forms of the red cell series may be encountered. If bleeding occurs repeatedly, iron-deficient erythropoiesis may develop. This raises an interesting semantic point: these patients are iron deficient in that iron stores are absent, but the total body iron content, including the iron present as hemoglobin in red cells, may be normal. Increased resistance to osmotic lysis has been reported for PV erythrocytes.99
occasionally, basophilic stippling may be found. An occasional normoblast may be observed in the blood smear, and, in the presence of a relatively normal or definitely increased red cell count, should arouse suspicion of PV. The reticulocyte percentage is not significantly increased. After hemorrhage, however, the reticulocytes may be increased, and a number of other immature forms of the red cell series may be encountered. If bleeding occurs repeatedly, iron-deficient erythropoiesis may develop. This raises an interesting semantic point: these patients are iron deficient in that iron stores are absent, but the total body iron content, including the iron present as hemoglobin in red cells, may be normal. Increased resistance to osmotic lysis has been reported for PV erythrocytes.99
Leukocytes
Leukocyte counts of 25.0 × 109/L are not uncommon and values in the 50.0 × 109/L range are occasionally seen.100 The myeloid leukocytes are both relatively and absolutely increased, metamyelocytes are increased in number, and 1% or 2% of myelocytes, sometimes more, are found. Myeloblasts usually are not observed. Basophil, eosinophil, or monocyte concentrations may be increased and provide a marker of an underlying myeloproliferative disorder. Leukocytes from patients with PV exhibit increased metabolic activity.101 A total leukocyte count greater than 12.5 to 15.0 × 109/L is a risk factor for thrombosis.102
Neither Vaquez nor Osler appreciated the significance of the leukocytosis with a “shift to the left” in the myeloid series of leukocytes that often is present in this disease initially described by them. Türk, in 1904, called attention to this significant finding, which suggested that the whole bone marrow, rather than the erythropoietic tissue alone, was hyperactive. The leukocyte counts were >10.0 × 109/L in 50% of Osler’s patients. In larger series, leukocytosis is seen in more than 80% of patients, and counts greater than 12.0 × 109/L in approximately 40%.12,100
Platelets
The platelet count frequently is increased, usually in the 500 to 1,000 × 109/L range, but counts as high as 3,000 and even 6,000 × 109/L have been reported.103 Bleeding time and conventional coagulation parameters usually are normal, but the clot may retract poorly. An artifactual elevation of protime and activated partial thromboplastin time may be observed in patients with erythrocytosis. The standard citrated tube used for coagulation studies contains a fixed quantity of anticoagulant for a fixed volume of blood. In polycythemia, there is a relative reduction of plasma, meaning that there will be excess anticoagulant for the volume of plasma. Functional assays of coagulation factors will thus be prolonged. This prolongation has no clinical significance other than provoking panic on the part of the individual reviewing the laboratory results. The leukocyte and platelet counts are not always increased above normal in patients with otherwise typical disease. Normal values were found in 20% of the patients in one series.15
Morphologic and qualitative functional platelet abnormalities often are detectable. Platelets may appear to be abnormally large, and even bizarre-shaped, and megakaryocyte fragments sometimes are seen in the blood smear. The hemorrhagic complications of this disease suggest that a hemostatic defect may be present, although physical distention of the vascular bed undoubtedly contributes to bleeding when it occurs. Platelet function defects reported in PV include shortened platelet survival in patients with erythromelalgia,104 altered von Willebrand factor multimers105 and acquired von Willebrand syndrome,106 deficient platelet-aggregating factor-induced aggregation,107 increased platelet thromboxane production,108 and platelet factor 3 deficiency.109 Plasminogen activator inhibitor-1 levels have been reported to be both elevated109 and decreased110 in patients with PV. Platelet glycoprotein IIIa expression has also been reported to be decreased in PV patients.111
Total Blood Volume
The total blood volume characteristically is increased. The enormous increase in blood volume, which distends even the smaller vessels of the whole body, no doubt accounts for many of the symptoms of this disease. In a group of 30 patients in whom the hematocrit was 0.55 or greater, the total red cell volume, measured by the radioactive phosphorus (32P)-labeled red cell method, was 38.8 to 91.9 ml/kg body weight as compared with the normal average of 29.9 ml/kg.112 In two thirds of this patient group, the plasma volumes were below the lower limits of normal, and in none was the plasma volume above normal. Similar observations have been made using the chromium isotope method.11 Because of variations in plasma volume, the packed cell volume (or hematocrit) gives only a rough indication of the size of the red cell mass.
In the early phases of PV, the red cell volume may not be elevated into this range. It has been proposed that a red cell volume > 125% of predicted is an appropriate criterion for polycythemia.113 In other circumstances, blood loss may result in “autophlebotomy”; that is, the patient becomes iron deficient, and the hemoglobin, hematocrit, and red cell volume fall into the normal range.
Erythrocyte Kinetics
Erythrokinetic studies have shown active hemoglobin production, but otherwise, findings have been diverse. Erythrocyte survival may be normal114 or shortened,115 and splenic sequestration may or may not be present. As measured by the 15N-glycine method, the rate of hemoglobin production was approximately two times the normal rate. The plasma iron turnover rate was also increased.115 This value was not reduced to normal by oxygen administration, as occurs when polycythemia is caused by anoxia.
Increased serum bilirubin99 and increased urine and stool urobilinogen116 levels have been demonstrated in some patients with PV, but when adjusted for the increase in the total amount of hemoglobin that must be degraded, fecal urobilinogen values rarely are increased above expected values and may be somewhat reduced.
Neutrophil Kinetics
Studies of neutrophil kinetics in patients with PV in whom neutrophil counts ranged from normal to 23.9 × 109/L showed a blood neutrophil pool that ranged from normal to 12 times normal, with increased margination and a normal or slightly prolonged half disappearance time. The blood neutrophil turnover rate (effective neutrophil production) usually was increased and varied from normal to five times normal mean values.117 Increased polymorphonuclear leukocyte activation is also observed in PV patients, and is associated with evidence of endothelial damage.118
Platelet Kinetics
Studies of platelet kinetics in five patients with PV in whom the platelet concentration was increased revealed effective production rates that ranged from 2 to 13 times normal. The marrow megakaryocyte mass was increased in all five patients studied.119
Fibrinogen Turnover
Results of studies involving three patients with erythremia indicated that fibrinogen was consumed in the course of chronic disseminated intravascular coagulation120; similar findings were
observed in several patients with erythrocytosis secondary to pulmonary insufficiency. No correlations between fibrinolytic parameters and thrombotic complications of PV have been reported.121
observed in several patients with erythrocytosis secondary to pulmonary insufficiency. No correlations between fibrinolytic parameters and thrombotic complications of PV have been reported.121
Other Laboratory Findings
The viscosity of the blood may be five to eight times greater than normal.98,122 The specific gravity is 1.075 to 1.080, compared with the normal range of 1.055 to 1.065. The degree of abnormality varies with the relative quantity of red corpuscles. The viscosity and specific gravity of the serum were actually less than normal. The erythrocyte sedimentation rate of polycythemic blood is low.123 The urine may be normal, but albuminuria is found occasionally, and, less often, casts are present.100 The increased urobilinogenuria noted in some of the patients has been mentioned. Studies of renal hemodynamics suggested that glomerular filtration, despite the decreased fraction of plasma in the blood, is kept at almost normal values by an increase in renal blood flow and in the proportion of plasma filtered.124 The amount of uric acid in the serum may be normal or increased. Hyperuricemia was present in 70% of 127 patients in one series125 and in 55% of 325 patients11 in another series. Values ranged from 2.8 to 11.7 mg/dl (average, 6.6 mg).11 Secondary gout occurs in 5% or more of these patients,13,17,125, 126 and 127 and symptoms often are atypical.98 The occurrence of hyperuricemia in association with PV is attributable to overproduction of uric acid.126,128 A vitamin B12-binding protein, which may be an altered form of transcobalamin I (Chapter 36), has been found in the plasma of patients with PV and in a variety of conditions involving leukocytosis.129,130 The presence of this protein may explain the observation that, whereas serum B12 content may be within the normal range or only moderately elevated, the capacity of the serum to bind additional vitamin B12 added in vitro (unsaturated B12-binding capacity, UB12BC) is increased. Plasma homocysteine levels in polycythemic patients are higher than nonpolycythemic subjects but still in the normal range.131 The leukocyte alkaline phosphatase score is normal or elevated in 80% to 100% of PV patients.113,125
Serum erythropoietin concentration is typically low in PV and may be elevated in secondary polycythemia. An elevated erythropoietin level in a polycythemic patient with reduced arterial oxygen saturation would be evidence against PV. Birgegard and Wide have proposed that PV can be distinguished from secondary polycythemia by the erythropoietin response to phlebotomy: the erythropoietin concentration rises after phlebotomy in secondary erythrocytosis but remains unchanged in PV.132 A decreased serum erythropoietin concentration is a minor criterion for the diagnosis of PV in the WHO criteria, and a clinical criterion in the European Clinical and Pathological (ECP) criteria.74,113,133
Spurious hyperkalemia has been noted when platelets are greatly increased in number.134 Hyperhistaminemia and hyperhistaminuria were reported in patients with PV compared with normal controls and secondary polycythemia subjects, but did not correlate with the pruritus often present.135 The basal metabolic rate may be increased moderately.136 Gastric acidity ranges from absent to increased.
Patients with PV and thrombosis exhibit a greater frequency of procoagulant abnormalities in antithrombin III, protein C, protein S, and resistance to activated protein C than do PV patients without thrombosis.137 Studies of prothrombin and factor V gene polymorphisms with a thrombotic diathesis showed no increased incidence of these abnormalities in PV patients.138 However, polymorphisms of the PlA2 allele of platelet glycoprotein IIIa were associated with increased arterial thrombosis in PV and essential thrombocythemia patients.138
Bone Marrow
For most of the last 40 or 50 years, bone marrow examination was not one of the criteria included in the PVSG standard for the diagnosis of PV.11 The value of bone marrow examination in the differentiation of PV from other myeloproliferative disorders, from secondary polycythemia, from spurious polycythemia, or from the normal state was debated. Some investigators believed it to be an invaluable aid for making this differentiation;139,140 others regard it as an important diagnostic tool but not one that is capable of definitively establishing the diagnosis.12 However, both currently accepted diagnostic requirements for PV, the World Health Organization (WHO) criteria and the ECP criteria, formally include bone marrow examination.133,141
The marrow typically is hypercellular142,143 (Fig. 82.3), but normal cellularity is noted at the time of diagnosis in approximately 13% of cases.144 Hyperplasia involves all of the marrow elements and displaces marrow fat. Cellularity can be estimated with considerable accuracy by low-power examination of clot sections and needle or trephine biopsies. In several series, the mean cellularity of the marrow was 80% to 90% compared to approximately 30% in normal subjects and 40% in patients with secondary erythrocytosis.139,140,145 As mentioned, a few patients with untreated PV may have normal marrow cellularity.144,145 Thus, the lack of hypercellularity does not exclude the diagnosis but should make it suspect.
An increase in megakaryocyte number and size is well documented in association with PV and was reported in 95% of 175 cases. When the number of megakaryocytes was expressed in terms of fat-free marrow, however, the megakaryocyte hyperplasia was proportional to overall marrow cellularity.144 The ratio of the different cell types in the marrow is not strikingly different from normal. Clumps of pronormoblasts and basophilic erythroblasts are seen,143 and the percentage of nucleated red cells may be moderately elevated.146 Myelocyte and myeloblast numbers may be greater than normal, and an increase in eosinophils147 and basophils may be found.146
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree