Introduction and Historical Perspectives
In late December 2019, an ophthalmologist named Li Wenliang sent a message to a group of fellow physicians in Wuhan, China, warning them of an emerging respiratory illness that he thought bore resemblance to severe acute respiratory syndrome (SARS). Weeks later, after local authorities admonished him for “spreading rumors,” Dr. Li contracted the virus from one of his glaucoma patients and passed away.
Ophthalmological manifestations of members of the Coronaviridae family were documented long before SARS-CoV-2 surfaced in humans. In the mid-20th century, a murine coronavirus was demonstrated to infect central nervous system cells and was later shown to cause a biphasic infection of the mouse retina characterized by early vasculitis and subsequent retinal degeneration. Later, with the first outbreak of the SARS pandemic in 2003, it was demonstrated that SARS-CoV-1 could cause conjunctivitis in humans , and was detectable in tear samples. In fact, the first diagnosis of a new SARS-CoV-1 strain was made in a 7-month-old child with conjunctivitis. It was also hypothesized that transmission of SARS-CoV-1 could occur through the ocular surface, because contact with SARS patient secretions without eye protection was an independent risk factor for transmission of the virus.
Since Dr. Li’s death, ophthalmologists have remained on the front lines of the COVID-19 pandemic and will continue to play an important role in prevention and detection of SARS-CoV-2 in the months and years to come. The physical examination is a critical component of an ophthalmology visit, and the close proximity necessitated by the slit-lamp examination is well within the range of droplets expelled by conversation and normal breathing that may harbor the virus. , Therefore it is essential that all health care workers who care for patients with eye conditions be prepared to recognize the role that the ocular surface plays in transmission of the virus, the ophthalmic manifestations of COVID-19, and the therapeutic interventions.
Role of the Eye in Transmission and Prevention
The surface of the eye is an important route of transmission for many pathogens, and coronaviruses are no exception. Surface proteins angiotensin-converting enzyme-2 (ACE2) and transmembrane protease serine 2 must be present for SARS-CoV-2 to invade human cells, and both receptors have been shown to be consistently expressed on the cornea, pointing to a possible route of ocular infection. ACE2 expression also has been demonstrated on the conjunctiva and in the aqueous humor.
In addition to being a potential site of inoculation, the eye also may play a role in viral shedding. Studies of a feline coronavirus have demonstrated that the offspring of infected cats have detectable live coronavirus on the conjunctiva, suggesting that ocular secretions are potentially infectious. Studies conducted in China early in the pandemic showed that a small number of patients had tear samples that were positive for SARS-CoV-2 antigens. , In a more recent study conducted in Italy, SARS-CoV-2 RNA was detected on the surface of the eye in 52 of 91 patients with COVID-19.
Lending further credence to the idea that SARS-CoV-2 transmission may occur through the ocular surface, a study conducted in China found that patients who wore eyeglasses daily may be less likely to contract the virus. This finding prompted many health care institutions to require eye protection for all employees who encounter patients during the pandemic. The decreased susceptibility to infection in eyeglass wearers suggests that the barrier provided by the glasses prevents contact of the ocular surface with droplets that may be expelled by patients as they cough, speak, or breathe. Alternatively, glasses could provide a deterrent to inadvertent touching of the eyes with hands that are contaminated with viral particles. Contact lens wearers are not afforded the same protection, but they also are not at any increased risk for infection, despite regularly touching their eyes to place and remove contact lenses.
| Structure | Pathology |
|---|---|
| Anterior segment | Conjunctivitis, episcleritis, keratitis, uveitis |
| Retina | Central retinal artery occlusion, central retinal vein occlusion, retinal nerve fiber layer edema, vitreous hemorrhage |
| Cranial nerves and optic pathway | Neuromyelitis optica spectrum disorder (NMOSD), Miller-Fisher syndrome, CN II–XII palsies, myasthenia gravis, encephalitis, opsoclonus-myoclonus, cavernous sinus thrombosis |
| Eyelids and orbit | Blepharitis, orbital cellulitis, orbital myositis |
Ocular Manifestations
Epidemiology
Reports of the prevalence of ophthalmic manifestations of COVID-19 vary across studies. Most systematic reviews estimate the range is from 4% to 11%, , but some case series place the prevalence as high as 47%. The most common ophthalmic manifestation is conjunctivitis. Ocular disease beyond conjunctivitis is rare and not widely reported in the literature. In one comprehensive review, only 8 of 43 studies reported additional ophthalmic manifestations, including keratitis, nodular scleritis, neuroophthalmological manifestations, and optic nerve findings. Other case series and reports have identified signs and symptoms of COVID-19 in the orbit, eyelid, retina, microvasculature, and uvea ( see Table 11.1 ). These reports are limited in their scope and must be approached with the understanding that more data are needed before it can be determined with certainty whether any given eye complaint is a result of a SARS-CoV-2 infection or if the patient was simply experiencing an unrelated eye condition when he or she contracted COVID-19.
The remainder of this section will discuss the current literature at the time of publication for reported manifestations of COVID-19 in the anterior segment of the eye, orbit, eyelid, retina, microvasculature, and uvea and neuroophthalmological manifestations and specific considerations for pediatric populations. The information here will undoubtedly change as the ophthalmology community learns more about the effects of SARS-CoV-2 on the eyes, and thus it will be updated accordingly in future editions.
Anterior Segment
The conjunctiva is a mucous membrane that covers the surface of the eye and the inner surface of the eyelids. Inflammation of the conjunctiva, called conjunctivitis, is the most common ocular manifestation of COVID-19, with an overall rate of 1.1% and representing an estimated 88% of ocular disease in COVID-19 patients. , Reported signs and symptoms of COVID-19 conjunctivitis range from mild tearing and foreign body sensation to severe symptoms such as photophobia, mucous secretions, and eyelid swelling. Other reported symptoms include eye pain, burning sensation, itching, conjunctival hyperemia, chemosis, and dry eye. , One systematic review reported findings of fibrin and inflammatory cell pseudomembranes on the tarsal conjunctiva, as well as tarsal petechiae, hemorrhages, and mucus filaments. Systemic findings associated with conjunctivitis, in addition to the known respiratory and constitutional symptoms of COVID-19, include lymphadenopathy in the preauricular, submandibular, and cervical chains.
One meta-analysis found an association between conjunctivitis and a higher severity of COVID-19, with conjunctivitis being present in 3% of severely ill patients and 0.7% of patients with milder symptoms. This finding indicates that providers should recognize conjunctivitis as a possible sign of more severe disease in COVID-19 patients and further underscores the importance of eye protection for health workers when caring for COVID-19 patients. More data are needed to evaluate the utility of such reports in the larger context of ocular manifestations of COVID-19.
Despite conjunctivitis being the most common ocular manifestation of COVID-19, the association between SARS-CoV-2 positivity on conjunctival swab and conjunctivitis remains ambiguous. In a study of 30 patients with COVID-19, only 1 patient had conjunctivitis and on polymerase chain reaction (PCR) evaluation was found to have SARS-CoV-2 RNA in the tear film. In a separate study that included 10 COVID-19 patients with ocular symptoms, SARS-CoV-2 was not detected in tear samples of any patients. Yet another study found a prevalence of ocular symptoms of 32%, with only 5% of the patients having SARS-CoV-2 nucleotides in conjunctival specimens. From these studies it is clear that ocular symptoms can occur even in the absence of viral shedding in tears.
Keratitis and episcleritis, or inflammation of the cornea and the episcleral coat of the eye, also have been noted in case reports. One case report notes pseudodendrites like those seen in herpetic keratitis that later evolved into dozens of subepithelial infiltrates with overlying epithelial defects across the cornea.
Uvea
The uvea is the middle layer of the eye involving the choroid, ciliary body, iris, and the associated vasculature of these structures. Uveitis is inflammation of the uvea and nearby structures such as the sclera, vitreous, and retina. The uveitides are a collection of about 30 different diseases. They are organized by location of primary inflammation (anterior, intermediate, posterior, and panuveitis) and whether they are infectious, associated with systemic autoinflammatory/autoimmune disease, or immune-mediated and limited to the eye without other signs of systemic inflammation. Although there have been reports of SARS-CoV-2 on the ocular surface, studies have yet to demonstrate active virus intraocularly. , Therefore it is suspected that any uveitis associated with COVID-19 is more likely a result of systemic or autoimmune inflammation.
Two case reports demonstrate autoinflammatory-mediated uveitis in the context of COVID-19. In the first report, a 54-year-old woman presented to an ophthalmology clinic with 2 weeks of bilateral blurry vision after being managed as an inpatient for COVID-19 multisystem inflammatory syndrome. Her ophthalmic examination had evidence of anterior chamber inflammation, keratic precipitates, corneal edema with Descemet folds, and decreased vision. She was managed successfully with topical steroids and cycloplegics. A second case report by Alonso et al. describes a patient with anterior uveitis associated with ocular hypertension, which was also thought to be secondary to multisystem inflammatory syndrome.
Although the described cases presented after a known infection of COVID-19, there have been cases in which the manifesting sign of COVID-19 was uveitis. A 60-year-old woman presented to the emergency department with ocular pain, redness, and decreased vision in her left eye. On examination she had anterior chamber inflammation, vitreous inflammation, and optic nerve edema consistent with acute panuveitis. Initial infectious serological studies for syphilis, toxoplasmosis, and human immunodeficiency virus were negative. She was presumed to have Vogt-Koyanagi-Harada disease and was started on oral steroids. Ten days later she developed a cough and systemic symptoms and tested positive for COVID-19. She was treated systemically and did well, including improved visual acuity in the left eye. Even in this case, anterior chamber PCR was negative for the SARS-CoV-2 virus.
Retina
In studies of coronavirus in animal models, the JMHV strain was demonstrated to affect cells that are found in the retina and posterior pole of mice, specifically glial cells, astrocytes, oligodendrocytes, and microglia. Moreover, COVID-19 infection has been associated with microvascular disease and coagulation disorders. With these neuronal and vascular manifestations of coronaviruses in mind, it is no surprise that retinal findings have been noted in some patients with COVID-19. In one cross-sectional study, patients with known COVID-19 (n = 54) demonstrated dilated veins (27.7%), tortuosity of vasculature (12.9%), retinal hemorrhages (9.25%), and cotton-wool spots (7.4%) on fundus photos. In addition, there was a significant increase in diameter of the retinal arteries and veins in the COVID-19 patients compared with those in controls, and the mean vein diameter was positively associated with COVID-19 in severe and nonsevere cases compared with unexposed controls. However, dilated veins are not always visible by clinical examination and require computer programming for accurate interpretation.
Overall, the retinal manifestations of COVID-19, like the uveitic manifestations, mostly likely are sequelae of the infection rather than acute infection itself. The most common retinal findings in COVID-19 patients are microangiographic changes and consequences of microvascular compromise. These findings are likely related to both hypoxia and the cytokines associated with the inflammatory response to the virus. Gonzalez-Lopez et al. reported a case of a 50-year-old patient in the late phase of acute COVID-19 infection receiving inpatient treatment for severe infection requiring antivirals, hydroxychloroquine, systemic steroids, and high-flow oxygen therapy. On day 7 of his care, he noted an inferior “crescent-shaped” blind spot that had been increasing without pain or other symptoms. Although his vision was good and the anterior segment examination was normal, fundus examination revealed peripapillary cotton wool spots in both eyes without hemorrhages. Optical coherence tomography (OCT) demonstrated retinal nerve fiber layer edema in the area associated with his scotoma.
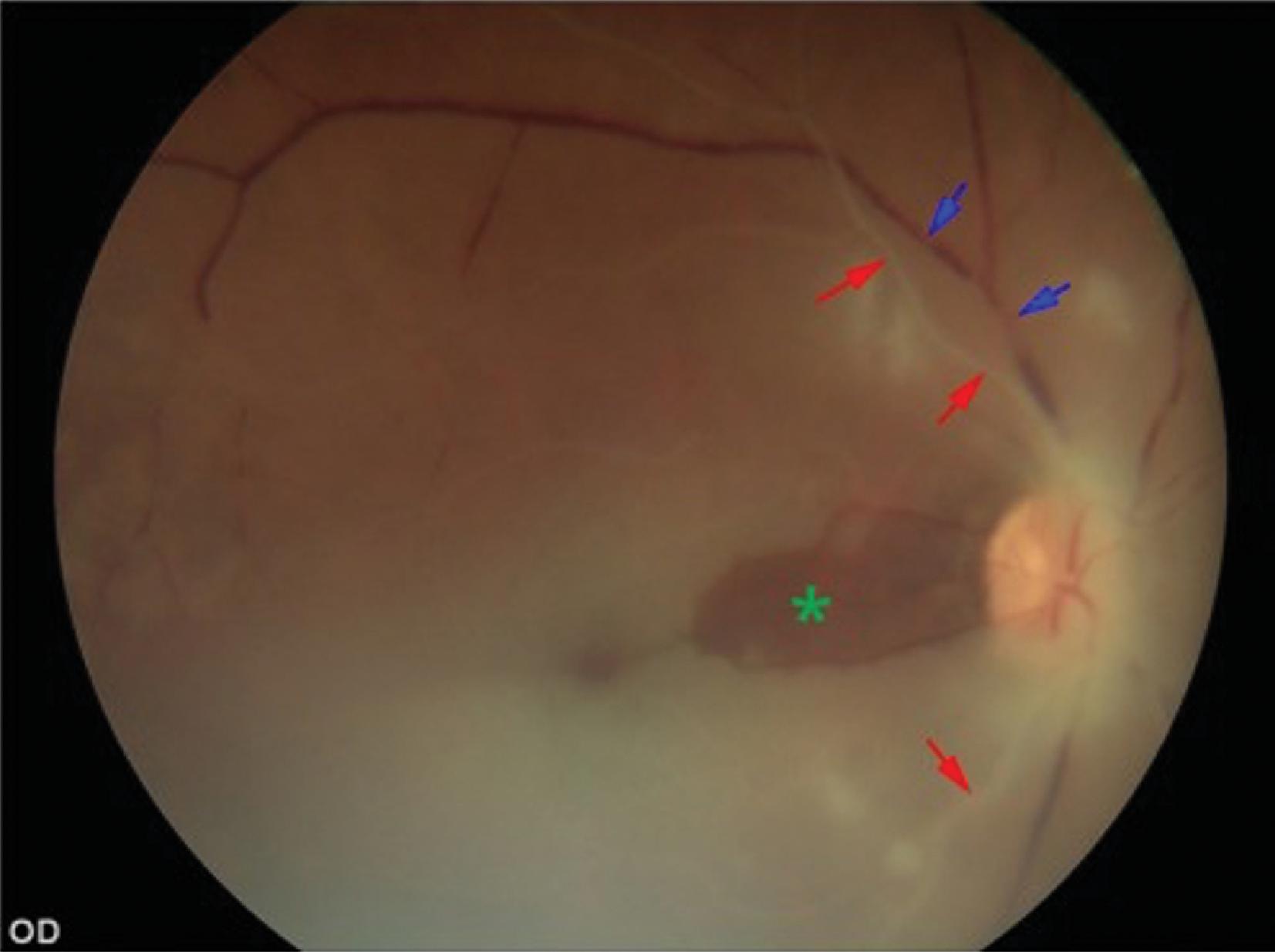
Other microvascular retinal findings in patients with COVID-19 include central retinal artery occlusion (CRAO) and central retinal vein occlusion (CRVO). CRAO is the ophthalmic equivalent of a stroke and is due to a lack of blood flow through the central retinal artery (see Figure 11.1 ). Usually, a CRAO manifests as monocular central vision loss and in one case report was the manifesting symptom of a patient with severe COVID-19 who required intensive care unit treatment with intubation. Other case reports describe CRAOs that developed in patients who were recovering from severe COVID-19 infections. , CRVOs, on the other hand, are due to venous outflow stasis from the retina. The suspected pathophysiology of CRVO in COVID-19 is related to endothelial cell damage and inflammation leading to stasis. CRVOs have been reported in a wide range of patients from a previously healthy 33-year-old to a case of bilateral CRVOs in an obese patient with COVID-19.
In addition, a case of paracentral acute middle maculopathy (PAMM), or irregularities seen in the middle layers on the retina on OCT, was diagnosed in a patient who developed a CRAO after a mild case of COVID-19. Two weeks after his COVID-19 diagnosis, this patient’s vision declined severely, and on fundoscopic examination a CRAO was seen. Of note, the hypercoagulable workup was negative. He had an OCT demonstrating diffuse reflectance and thickening of the inner nuclear layer of the retina consistent with PAMM.
Neuroophthalmology
Previous coronaviruses have demonstrated neurological complications in both animal and human models. A murine coronavirus has been shown to lead to demyelination, and a coronavirus that affects nonhuman primates has been found in high concentrations in perivascular neural tissue. Previous studies in humans discovered SARS in the cytoplasm of hypothalamic and cortical neurons, and there is one report of a child who was clinically diagnosed with acute disseminated encephalomyelitis and the only detected microbe in the cerebrospinal fluid was a coronavirus.
One of the most common symptoms of SARS-CoV-2 is loss of taste and smell, thought to be secondary to direct involvement of the virus with the olfactory nerve. As the virus commonly affects the first cranial nerve (CN I), it should come as no surprise that the optic nerve can also be affected. Each optic nerve is a collection of approximately 1.5 million nerve fibers that coalesce from the retinal nerve fiber layer and travel to the brain. Diseases of the optic nerve associated with COVID-19 include direct neuronal inflammation, such as optic neuritis, as well as microvascular compromise to the nerve, such as ischemic optic neuropathy (ION). There have been two reported cases of anterior ION, a disease associated with microvascular compromise of the anterior optic nerve head. ,
Neuromyelitis optica spectrum disorder (NMOSD) is an inflammatory condition of the nervous system that often involves the optic nerves and spinal cord with known causes (anti–aquaporin-4 and myelin oligodendrocyte glycoprotein–immunoglobulin G [MOG-IgG] antibodies, among others) and seronegative/idiopathic causes. As we are discovering, COVID-19 may be an inciting factor for NMOSD. In one report, a 15-year-old boy in the Netherlands presented with 7 days of worsening vision loss with associated flashing lights and headache a few weeks after being diagnosed with a mild nonspecific viral illness, which was presumed by the study authors to be COVID-19. He was found to have bilateral optic disc edema with decreased vision in both eyes. Magnetic resonance imaging (MRI) of the brain and orbits demonstrated longitudinal enhancement of bilateral optic nerves consistent with NMOSD, and follow-up serological testing confirmed MOG-IgG positivity. In another study, an 11-year-old boy presented with redness and pain in both eyes after a febrile illness. Two weeks later, he developed sudden severe vision loss in the right eye and was found to have right optic nerve edema with an associated relative afferent pupillary defect. He was later found to be positive for both COVID-19 and MOG-IgG, and MRI findings were consistent with NMOSD. The patients in both cases responded well to steroid therapy. In another case, a 39-year-old woman with previously diagnosed anti-MOG NMOSD was found to have a flare of her optic neuritis and confirmed COVID-19 positive on nasal swab.
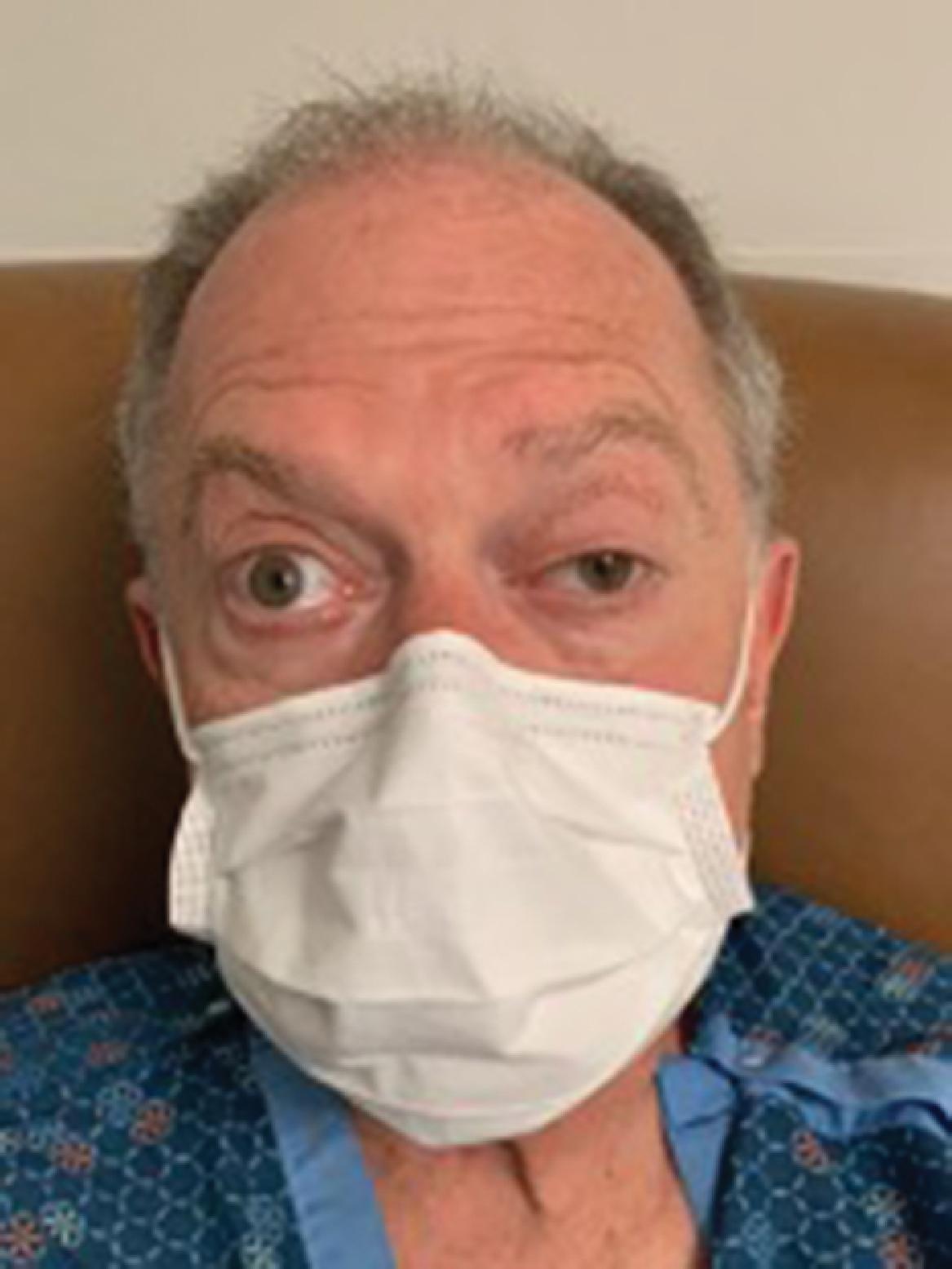
There have been multiple reports of CN palsies associated with COVID-19 infection, from both microvascular compromise and direct neuronal inflammation. MRI findings of enhancement and thickening of the nerves have been associated with direct inflammation or postinfectious nerve palsy. With respect to CN III palsies, they are most often reported as a postinfectious/inflammatory change associated with COVID-19 and/or associated with systemic inflammation in a Miller-Fisher syndrome subtype of Guillain-Barré syndrome. , There are also reports of CN III palsies resulting from microvascular compromise. In one case, a previously healthy 67-year-old man presented with double vision 1 month after being diagnosed with COVID-19 and was found to have a left CN III palsy (see Figure 11.2 ). An MRI of the brain demonstrated scattered nonspecific changes of the affected oculomotor nerve, which indicates microvascular ischemic changes. He was found to have an elevated d -dimer and otherwise negative workup, including systemic causes of microvascular disease. It is postulated that COVID-19 led to microvascular compromise of the left CN III. Similarly, there have been multiple case reports of CN IV and CN VI palsies in suspected microvascular and postinflammatory cranial neuropathy. , Finally, there have been cases of myasthenia gravis after infection with COVID-19, indicating an autoimmune mechanism. There are three reported cases of acetylcholine receptor antibody–positive myasthenia gravis with a wide array of manifesting symptoms, including ptosis and diplopia roughly 5 to 7 days after febrile illness from presumed COVID-19.
As we move further posteriorly in the anatomy of the ophthalmic system, there have been case reports of eye movement abnormalities associated with COVID-19 encephalitis, , postinfectious brainstem syndrome, and opsoclonus-myoclonus.
Larger vascular compromise and hypercoagulable disorders such as cavernous sinus thrombosis (CST) and cerebral venous sinus thrombosis (CVST) are a growing concern after COVID-19. CST has been reported in the acute infectious period and after recovery from the infection. Some providers recommend excluding the diagnosis of CVST in COVID-19–positive patients presenting with focal neurological deficits, progressive headaches, and/or other signs of increased intracranial pressure as a result of its prevalence.
Eyelids and Orbit
SARS-CoV-2 infection has been shown to have ophthalmic manifestations that extend beyond the globe to involve the eyelids and orbit. In one cohort study out of Italy, 11 of 29 patients hospitalized for COVID-19 presented with signs of blepharitis, including crusting of the eyelashes, lid margin hyperemia and telangiectasias, and alterations in the meibomian orifices. Other reports have noted that orbital infections can occur in the setting of COVID-19 secondary to nasal congestion and sinus obstruction. , Additionally, the immune response to SARS-CoV-2 has been linked in one study to the development of unilateral orbital myositis, or inflammation of the extraocular muscles.
Pediatric Ophthalmic Manifestations
The systematic course of COVID-19 infection in children has been milder than in adults, as have the ocular manifestations. However, there have been increasing reports of multisystem inflammatory syndrome, a syndrome similar to Kawasaki disease in which conjunctivitis is common. Furthermore, some newborns with suspected COVID-19 were found to have hemorrhagic conjunctivitis with serous chemosis and fundus findings of cotton wool spots and vitreous hemorrhage. As in adults, the relationship between ocular symptoms and SARS-CoV-2 positivity on conjunctival swab in pediatric patients is unclear; in one study, all pediatric patients with ocular manifestations of COVID-19 had negative PCR results of ocular samples, despite being positive for SARS-CoV-2 IgG by serology.
Conclusion
Eye care providers play an important role in the detection and prevention of COVID-19. In the context of the COVID-19 pandemic, and considering the data presented in this chapter, providers should proceed with an increased index of suspicion for patients presenting with common ocular complaints such as conjunctivitis. As we learn more about the natural history of COVID-19, our understanding of ocular manifestations of the disease will continue to improve, as will our strategies for treating its still emerging ophthalmic sequelae.
REFERENCES
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree