Coronavirus Infections in Children and Adolescents
Coronaviruses are enveloped single-stranded RNA viruses comprising four genera: alpha, beta, gamma, and delta. They infect humans and animals and were first described in the 1960s as causative agents of mild upper respiratory tract infections. Four human coronaviruses caused up to 35% of upper respiratory tract infections in humans: 2229E, NL63 (both alpha coronaviruses), and HKU1 and OC43 (beta coronaviruses). OC43 was thought to cause the most disease and was prevalent in children younger than 5 years. In rare instances, severe infections have been described in immunosuppressed children with NL63. These endemic human coronaviruses are often identified in coinfections with other respiratory viruses, making it difficult to determine their true role in disease. ,
In evaluations of children with respiratory symptoms and respiratory virus testing, approximately 2% to 10% had human coronavirus. , Human coronavirus was also detected in 10% of asymptomatic control children. Additionally, 33% to 68% had coinfections of human coronavirus and another virus, most commonly rhinovirus or respiratory syncytial virus (RSV). , , Human coronavirus infections were present throughout the year but peaked in the winter months in North America and Europe. , Clinical manifestations of human coronavirus were indistinguishable from those of other respiratory viruses. , In rare cases, human coronavirus could cause lower respiratory tract infection in children with underlying medical conditions.
Over the past 20 years, three new beta coronaviruses emerged and caused severe, sometimes fatal illness in humans: severe acute respiratory syndrome coronavirus-1 (SARS-CoV-1), Middle Eastern respiratory syndrome coronavirus (MERS-CoV), and, more recently, SARS-CoV-2. SARS-CoV-1 was described in 2003 in Southeast Asia and is thought to have originated from bats. It caused over 8000 cases and more than 700 deaths and had a mortality rate of 10% in adults but caused no deaths in the 135 pediatric cases reported. No further cases of SARS-CoV-1 have been reported since 2004. MERS-CoV is thought to have been transmitted from bats to camels to humans and was identified in 2012 in Saudi Arabia. It infected more than 2400 people and caused severe respiratory illness and a mortality rate of 36%. However, in children most cases of MERS-CoV infection were asymptomatic. MERS-CoV continues to occur sporadically, primarily in the Middle East.
In both SARS-CoV-1 and MERS-CoV outbreaks, children had less severe manifestations than adults and infected children were commonly identified during contact investigations of adult patients. , The clinical manifestations of children infected with SARS-CoV-1 or MERS-CoV were nonspecific and ranged from asymptomatic to mild, nonspecific respiratory symptoms such as fever, cough, and rhinorrhea. Some infected children developed opacities on their chest radiographs and laboratory abnormalities, including lymphopenia, thrombocytopenia, and elevated transaminases. ,
Epidemiology of SARS-CoV-2 Infection in Children and Adolescents
SARS-CoV-2 was first recognized as the cause of the COVID-19 pandemic by the World Health Organization (WHO) in March 2020, at which time 118,000 cases had occurred in 114 countries and nearly 4300 people had died. By September 27, 2021, approximately 18 months later, there had been more than 231 million confirmed cases of COVID-19 and more than 4 million deaths globally. Our understanding of SARS-CoV-2 transmission dynamics, infectiousness, clinical manifestations, and outcomes continues to evolve during the pandemic.
Throughout the pandemic there has been great geographic variation in the burden of COVID-19. At the time of this writing, the highest number of COVID-19 cases has been reported by the United States, at more than 42 million cases, followed by India with more than 33 million cases and Brazil with 21 million cases. These three countries also report the greatest number of COVID-19–associated deaths. In contrast, by September 2021 all of Europe reported 69 million cases, Southeast Asia reported 42 million cases, and Africa reported 6 million cases. All of these numbers are underestimates because of imperfect reporting systems and limited capacity for SARS-CoV-2 testing in many parts of the world. Additionally, the true burden of infection in children is likely to be much higher, because many places preferentially tested for SARS-CoV-2 among symptomatic, hospitalized patients early in the pandemic, and these patients tended to be adult patients.
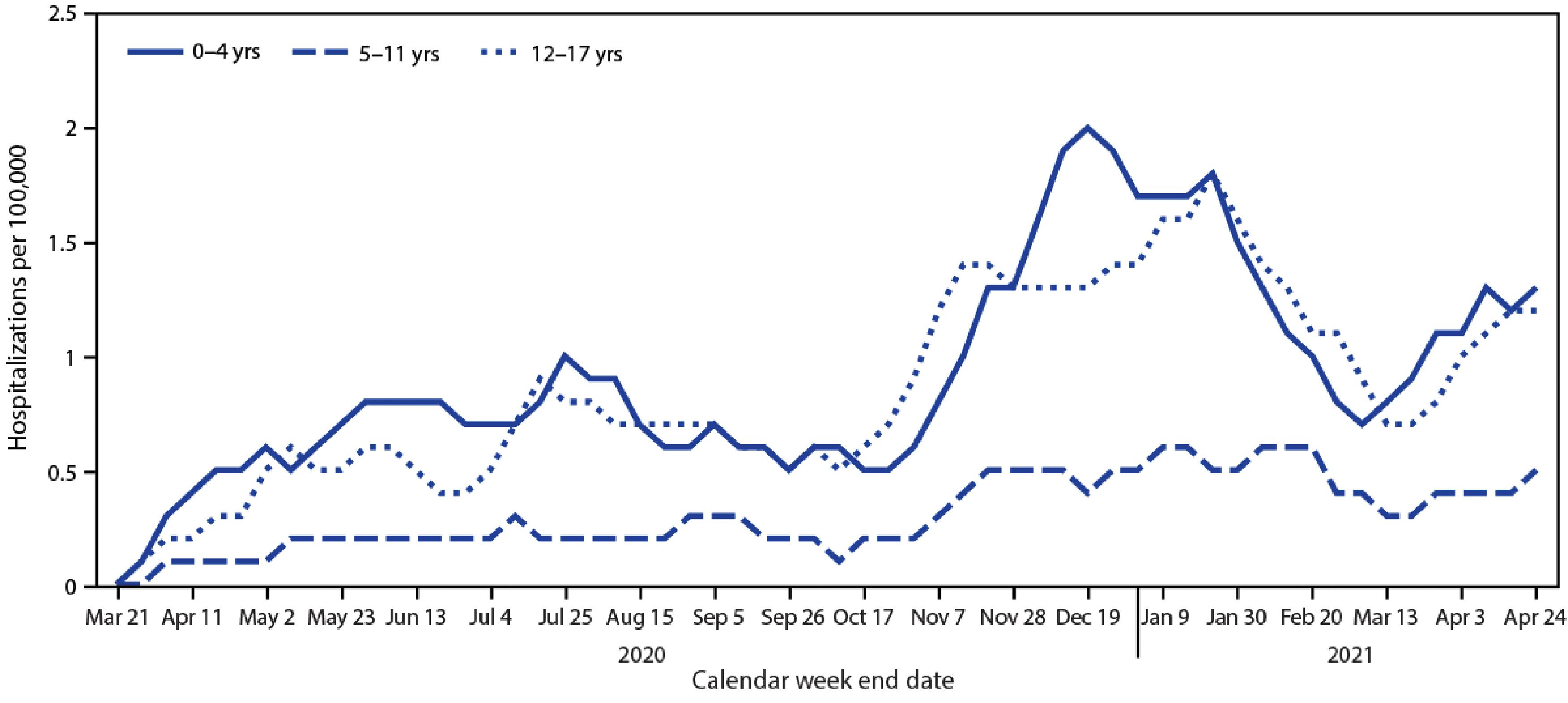
Hospitalization and mortality rates for SARS-CoV-2 have been consistently lower among children than adults. Despite variability in testing and reporting of SARS-CoV-2 infections and deaths, reports from most parts of the world show that pediatric cases account for 1% to 2% of infections documented. As of September 16, 2021, the United States reported over 5.5 million pediatric SARS-CoV-2 infections, representing 15% of all cases, 1% to 4% of total hospitalizations and less than 1% of all COVID-19 deaths. Children 5 to 11 years had lower hospitalization rates compared with younger and older children ( Fig. 14.1 ). Among 121 people younger than 21 years of age who died from SARS-CoV-2 in the United States between February and July 2020, 10% were infants younger than 1 year and 70% were between the ages of 10 and 20 years.
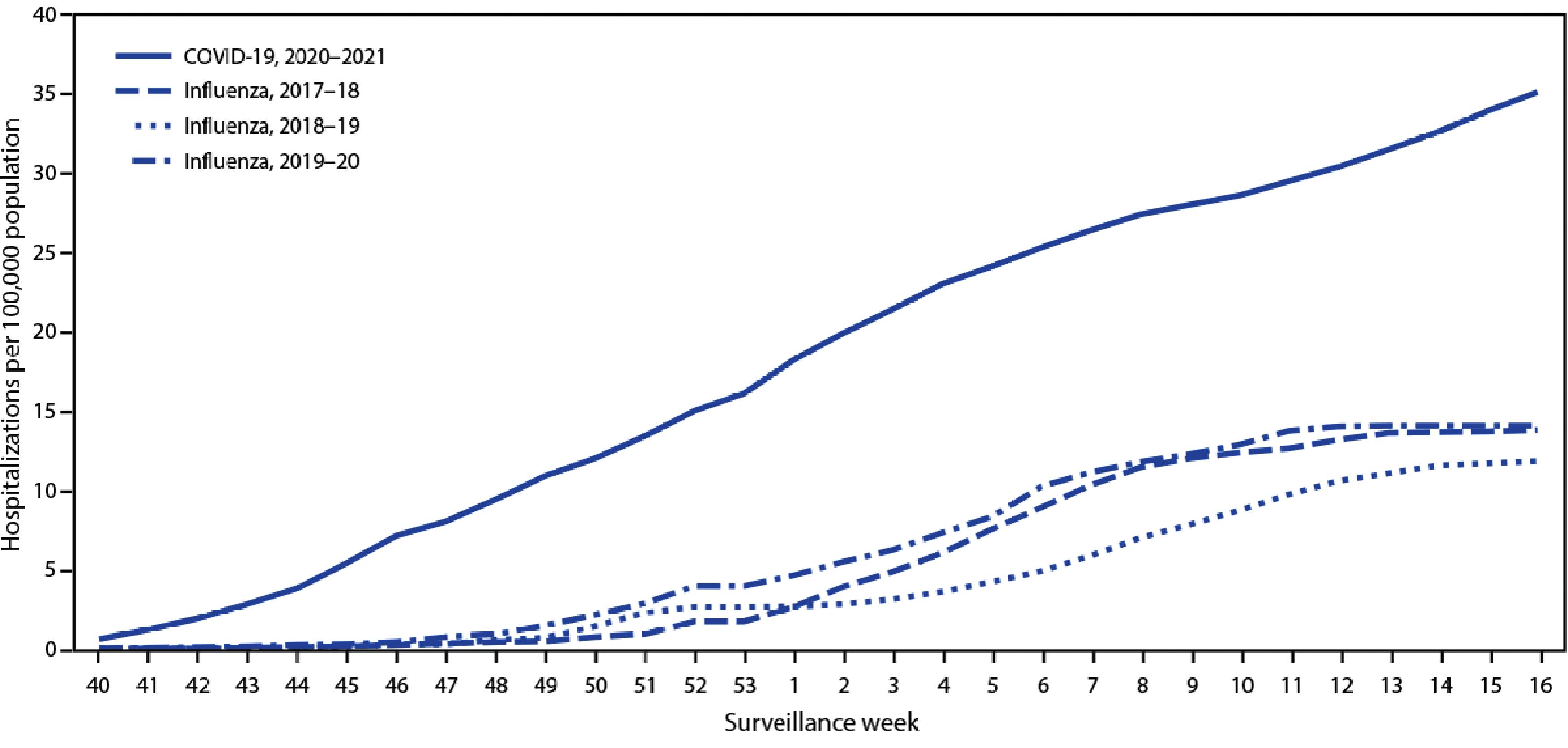
Although hospital admissions for COVID-19 among children and adolescents are rare, it is important to consider that COVID-19 has caused more pediatric hospitalizations than influenza. Among US adolescents, COVID-19–associated hospitalizations between October 2020 and April 2021 were almost three times higher than influenza-associated hospitalizations from three previous influenza seasons ( Fig. 14.2 ). In children, as in adults, SARS-CoV-2 infection and hospitalization rates and mortality vary by race and ethnicity. Hispanic, Black, or American Indian/Alaskan Native persons composed 75% of a US cohort of persons younger than 21 years who died from COVID. ,
In the summer of 2021, the B.1.617.2 (delta) variant of SARS-CoV-2 emerged and spread in the United States. This fourth surge of SARS-CoV-2 coincided with off-season circulation of RSV and return to in-person school for children after summer vacation, resulting in high numbers of pediatric SARS-CoV-2 infections and hospitalizations in the United States. , The delta variant appears to be more transmissible than SARS-CoV-2 variants that circulated earlier in the pandemic. Although the delta variant has caused increased numbers of pediatric COVID infections, thus far it does not seem to cause more severe disease than earlier variants. Hospitalization rates and outcomes of children infected with delta variant have not been different from those seen with earlier variants. ,
Susceptibility of Children to SARS-CoV-2 Infection
SARS-CoV-2 is transmitted person to person by respiratory droplets generated during coughing, sneezing, and speaking and from contact with fomites. Unlike for SARS-CoV-1, asymptomatic and presymptomatic individuals infected with SARS-CoV-2 can transmit the virus, which has led to challenges controlling viral spread during the pandemic. Transmissibility of SARS-CoV-2 appears to be greatest 2 days before until 3 days after symptom onset.
The significantly higher burden of COVID-19 infection and severe disease in adults suggests that children may be less susceptible to SARS-CoV-2 infection than adults. This hypothesis has biological plausibility because children have been found to have fewer copies of angiotensin-converting enzyme-2 (ACE2), the receptor for SARS-CoV-2, in their nasal epithelium. ACE2 gene expression increases with age, potentially protecting the youngest children, who tend to be the least symptomatic from COVID-19, from acquiring infection. Another explanation is that unlike adults, children have frequent and recent exposure to endemic human coronaviruses, and thus may have some cross-protective antibodies against SARS-CoV-2, preventing the hyperinflammation associated with severe COVID-19.
It is unclear what role children have played in the spread of SARS-CoV-2. Unlike other respiratory viruses such as influenza, which is commonly spread by children to others in the community, SARS-CoV-2 transmission was initially thought to be driven by adult infections, which were so much more prevalent than pediatric infections. However, SARS-CoV-2 viral loads in respiratory secretions and stool have been high in children. Some reports suggest prolonged periods of viral shedding among children, although it is controversial whether children routinely have higher viral loads than adults. A comparison of reverse transcription polymerase chain reaction (RT-PCR) cycle threshold values from respiratory specimens of children in California between April and August 2020 found that cycle threshold values were lower (and therefore viral loads were higher) in children younger than 5 years and in symptomatic versus asymptomatic children. Similarly, in a retrospective evaluation of nine US institutions performing pediatric SARS-CoV-2 testing between March and July 2020, viral loads were 10-fold lower in specimens from asymptomatic compared with symptomatic children. In a single-center study of 145 specimens, conducted in Chicago, viral loads from respiratory specimens were highest in children younger than 5 years of age and viral loads were similar between children older than 5 years and adults. However, other studies reported that viral loads in respiratory specimens did not differ by age. The discrepancies between these studies may be due to differences in testing practices (e.g., preferential testing of symptomatic vs. asymptomatic individuals), differences in timing of testing, inconsistencies in classifying symptoms, and differences in the assays used.
Although early reports did not suggest that children contributed significantly to SARS-CoV-2 spread, larger studies conducted later in the pandemic suggest children can efficiently transmit SARS-CoV-2, including within households. A systematic review and meta-analysis of 87 studies representing over 1 million households across 30 countries reported a household secondary attack rate of SARS-CoV-2 of 18.9%. In a large cohort study conducted in Ontario, Canada of 6280 households with pediatric index SARS-CoV-2 cases, 27% of households experienced secondary transmission. Furthermore, within these households, children younger than 3 years had the highest odds of transmitting SARS-CoV-2 to household contacts compared with older children. This may be because young children have higher viral loads in their nasopharynx, are less commonly symptomatic, and have more frequent physical contact with household members compared with older children.
Clinical Manifestations of SARS-CoV-2 Infection in Infants and Children
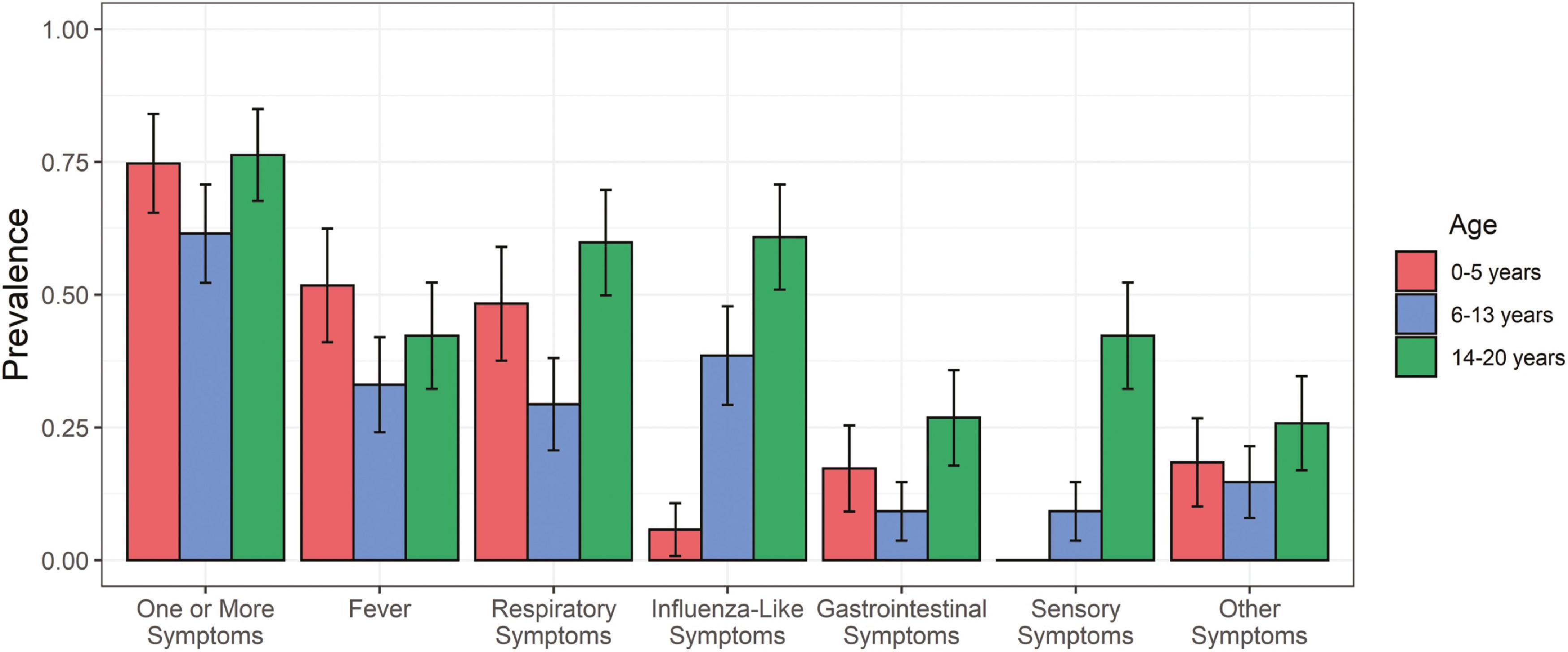
As observed with the other human coronaviruses, the clinical presentation of SARS-CoV-2 is milder in young children than in adolescents and adults. It is estimated that approximately 15% to 35% of infected children can be asymptomatic. When children do exhibit symptoms, they range from mild fever, with or without respiratory symptoms (cough, rhinorrhea, congestion, sore throat), gastrointestinal symptoms (nausea, vomiting, diarrhea), and other systemic symptoms (headache, myalgias, fatigue), to acute respiratory distress syndrome and respiratory failure and death. An early report comparing 291 children to over 10,000 adults suggested that children experience fever, cough, or shortness of breath less commonly than do adults. In general, it is difficult to distinguish SARS-CoV-2 infection from other viral illnesses based on symptoms alone. SARS-CoV-2 infection also has been associated with extrarespiratory symptoms, including neurological manifestations such as loss of taste and/or smell; cerebrovascular accidents and seizures; Guillain-Barré syndrome; cutaneous findings, including chilblain-like (pernio) lesion, erythema multiforme, and urticaria ; conjunctivitis ; myocarditis , ; and liver and renal dysfunction. , In addition, thrombotic complications are commonly seen in COVID-19 patients with critical illness. The constellation of symptoms appears to vary by age of the child, with adolescents having more respiratory, influenza-like, and sensory symptoms than younger children ( Fig. 14.3 ). ,
Children with asymptomatic or mild COVID-19 may have mild lymphopenia and thrombocytopenia. Those with moderate to severe symptoms are more likely to have laboratory abnormalities, including lymphopenia, thrombocytopenia, coagulopathy, and elevated inflammatory markers such as C-reactive protein and procalcitonin. A wide spectrum of abnormalities can be seen on chest radiographs among children with COVID-19 pneumonia, including unilateral or bilateral infiltrates, often with peripheral and lower lung zone predominance, peribronchial thickening or opacities, ground-glass opacities, and consolidations. To date, bacterial superinfection has been seen in only a small proportion of hospitalized adults with COVID-19. , This is likely also true in hospitalized children, although pediatric data are still emerging.
Duration of symptoms is generally short, with symptom resolution occurring in 45% by 5 days and in 94% by 1 month in an assessment of 1000 infected children. Postacute sequelae of SARS-CoV-2 infection (PASC), commonly referred to as long-COVID, has been reported in many adults after SARS-CoV-2 infection but appears to be rare among children. Among 366 adults in California who had a positive SARS-CoV-2 infection between April and December 2020, 128 (35%) endorsed ongoing symptoms 2 months later. In contrast, in a large prospective cohort study of SARS-CoV-2–infected children in the United Kingdom in which parents reported data about their symptomatic children using a mobile application, median illness duration in positive children was 6 days and only 25 of 1379 (1.8%) experienced symptoms for at least 56 days. However, emerging data suggest that PASC may be more common in children than previously reported and warrants further study.
Risk Factors for Hospitalization and Severe COVID-19 in Children
Risk factors for severe COVID-19 disease and hospitalization in children and adolescents are incompletely understood. Early descriptions of severe disease in children were single-center studies with small sample sizes and identified older age, comorbid conditions, and elevated CRP as risk factors for severe COVID-19. , As the pandemic has continued and more children have become infected or hospitalized, larger studies have similarly demonstrated that underlying medical conditions are associated with hospitalization for COVID-19 in children. However, it is unclear whether the increased hospitalization rate among children with comorbidities is due to lower threshold for admission or more severe COVID-19 disease.
Numerous studies have identified specific comorbid conditions as risk factors for severe COVID or hospitalization in children. In a cross-sectional study across 46 pediatric intensive care units (ICUs) conducted in North America between March and April 2020, 40 of 48 (83%) children admitted to ICUs had preexisting medical conditions ( Table 14.1 ). Likewise, in a retrospective case series of 112 persons younger than 21 years reported to the Centers for Disease Control and Prevention (CDC) with suspected SARS-CoV-2–associated deaths, 86% had at least one underlying condition, most commonly obesity, asthma, or developmental disorders. In a retrospective single-center cohort study of the first 1000 SARS-CoV-2–infected children in a health system in the US Southeast, comorbid conditions, Black race, Hispanic ethnicity, and dyspnea and vomiting at presentation were associated with pediatric hospitalization. An analysis using a large US health care claims database included over 43,000 children younger than 18 years with emergency department (ED) or inpatient encounters for COVID-19 and demonstrated that the risk for hospitalization was greatest in children with complex chronic diseases, specifically type 1 diabetes, obesity, and congenital cardiac or circulatory diseases, with adjusted risk ratios of 4.6, 3.07, and 2.12, respectively. Hospitalization was also associated with infants younger than 1 year with history of prematurity. Although this was a very large study, it relied on diagnostic codes and had no adjudication of the primary reason for admission and thus may have misclassified and overestimated COVID-19 hospitalizations.
| Comorbid conditions |
| Obesity |
| Type 1 diabetes mellitus |
| Chronic respiratory diseases |
| Medical technology dependence (e.g., tracheostomy) |
| Congenital cardiac or circulatory diseases |
| Neurological/neuromuscular disorders |
| Immunocompromising condition |
| Sickle cell anemia |
| Black race or Hispanic ethnicity |
| Dyspnea, hypoxia, or vomiting at presentation |
Additionally, an analysis using the Coronavirus Diseases 2019 Associated Hospitalization Surveillance Network (COVID-NET) found that among children, hospitalization rates were lowest in children aged 5 to 11 years and higher in the 0 to 4 and 12 to 17 age groups. , Among 376 adolescents hospitalized between January and March 2021, obesity (defined as body mass index [BMI] ≥95th percentile for age and sex); chronic lung disease; neurological disorders; chronic metabolic disease, including diabetes; immunocompromised condition; blood disorder, including sickle cell anemia; and cardiovascular disease were more commonly seen in those hospitalized for COVID-19 than among those hospitalized for other reasons. In a multicenter evaluation of hospitalized children with COVID from eight sites in three states in the Northeastern United States, obesity and hypoxia on admission were predictive of severe respiratory disease. Antoon et al. conducted a retrospective cohort study of pediatric patients with a primary diagnosis of COVID-19 and discharged from the ED or inpatient units of 45 free-standing US hospitals. They observed that 20% of encounters resulted in hospital admission and that factors associated with ICU admission among hospitalized patients were obesity, older age, and preexisting conditions, including cardiovascular disease, obesity, type 2 diabetes mellitus, and pulmonary, neurological, or neuromuscular conditions. Importantly, in several studies, Black and Hispanic or other non-White race were associated with higher hospitalization rates and disease severity in children, demonstrating that health disparities have affected children as well as adults during this pandemic. , , ,
Multisystem Inflammatory Syndrome in Children
A rare but serious consequence of SARS-CoV-2 infection first identified in children in the United Kingdom and Italy in April 2020 is multisystem inflammatory syndrome in children (MIS-C). , A similar syndrome has been identified in adults, called MIS-A. MIS-C typically manifests 2 to 6 weeks after a COVID-19 infection or exposure. Therefore MIS-C hospitalizations typically peak weeks after COVID-19 surges. Notably, about two-thirds of MIS-C cases in the United States have occurred in Hispanic or Black children. By the end of August 2021, in the United States there had been 4661 cases of MIS-C reported and 41 MIS-C–associated deaths.
MIS-C appears to be a delayed immunological response to SARS-CoV-2 infection and has features resembling Kawasaki disease and toxic shock syndrome, with evidence of inflammation in multiple organ systems. It is unclear why MIS-C disproportionately occurs in children and young adults compared to older patients. In May 2020, the CDC created a case definition of MIS-C and made it a reportable disease. The case definition consists of a patient younger than 21 years with fever, laboratory evidence of inflammation, and evidence of clinically severe illness requiring hospitalization, with multisystem organ involvement (cardiovascular, dermatological, gastrointestinal, hematological, neurological, renal, or respiratory) who has tested positive for SARS-CoV-2 or had exposure to COVID-19. Clinical manifestations and laboratory abnormalities associated with MISC vary by age ( Fig. 14.4 ). More than 95% of children with MIS-C have a positive SARS-CoV-2 PCR or antibody test at time of diagnosis.
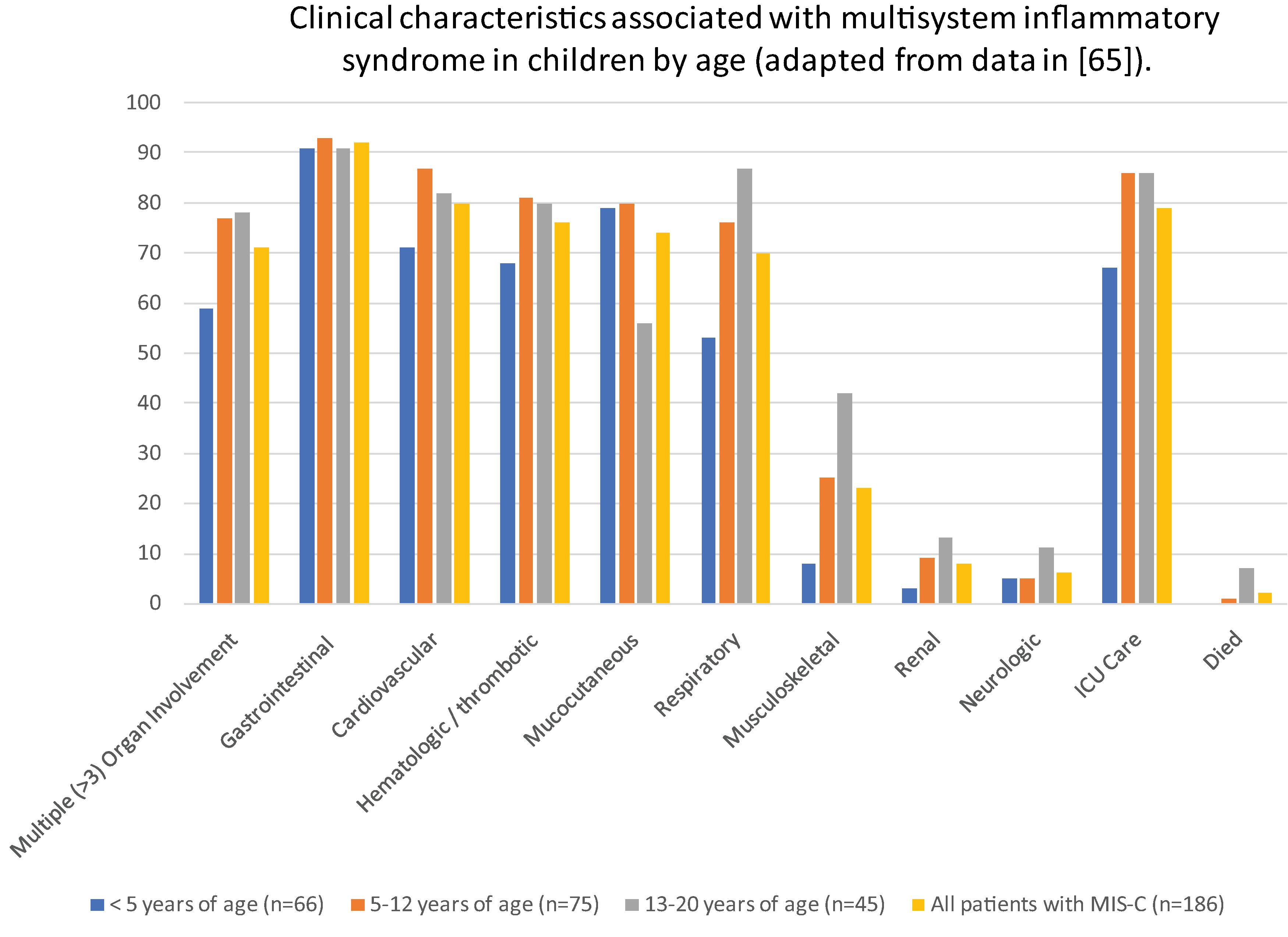
In a large cross-sectional cohort study including 1733 patients with MIS-C, the most common symptoms in addition to fever were abdominal pain (65%), vomiting (64%), and rash (56%), diarrhea (53%), and conjunctivitis (54%). Respiratory symptoms were less common. Half of patients had hypotension, slightly over a third developed shock, 58% were admitted to intensive care, and 1.4% died. Cardiac dysfunction was reported in 31% and was more common in adolescents and young adults than in younger children.
Recently the CDC identified three classes of MIS-C patients with differing illness manifestations. Class 1 patients had a median age of 9 years, the highest number of involved organs, and did not have manifestations that overlapped with acute COVID-19 or Kawasaki disease. Class 2 patients had a median age of 10 years and had respiratory system involvement that overlapped with manifestations of acute COVID-19. Class 3 patients were younger, with a median age of 6 years and had the highest prevalence of rash and mucocutaneous lesions and coronary artery dilation or aneurysm that overlapped with complete Kawasaki disease. Treatment with intravenous immunoglobulin (IVIG), steroids, or other immunomodulatory agents, such as the IL-1 receptor anatagonist anakinra, have shown favorable outcomes. In a follow-up study of 68 patients with MIS-C in the United Kingdom, nearly 90% of children had normal echocardiograms at 1 year after hospitalization, with no significant short- or long-term sequelae.
Neonatal SARS-CoV-2 Infection
Neonatal SARS-CoV-2 infections are rare, but when they do occur, they are generally acquired postnatally from infected adult caregivers and associated with good outcomes. Early in the pandemic, pregnant women with SARS-CoV-2 infection underwent cesarean section, were separated from their infants, and were advised not to breastfeed because of concerns for perinatally acquired infection. Since then, many studies have demonstrated that severe SARS-CoV-2 infection acquired in the neonatal period is unlikely when caregivers adhere to appropriate masking and hand hygiene. A population-based active surveillance study in the United Kingdom found that neonatal inpatient admission for SARS-CoV-2 infection occurred in only 5.6 per 10,000 livebirths. Additionally, SARS-CoV-2–infected neonates are generally asymptomatic or have relatively mild symptoms. Accordingly, guidance from the WHO and many countries is to avoid separation of mother and baby. , For additional details, see Chapter 13 .
SARS-CoV-2 Infections in Immunocompromised Children
The impact of SARS-CoV-2 infection on immunocompromised children is variable and depends on the extent of immunosuppression. Some studies report mild symptoms with SARS-CoV-2, and the United Kingdom’s National Institute of Health and Care Excellence (NICE) has stated that COVID-19 disease is not worse in immunocompromised children compared with immunocompetent children. However, other studies report poor outcomes after SARS-COV-2 infection in immunocompromised children. Severe COVID-19 has been reported in children receiving intensive chemotherapy for leukemia and CAR T-cell therapy and in patients with hypogammaglobulinemia and lymphopenia. Mortality after SARS-CoV-2 infection in hematopoietic stem cell transplant recipients is lower for pediatric than adult patients. A study using the Global Registry of COVID-19 in Childhood Cancer, a global cohort of children with cancer or prior hematopoietic stem cell transplant who had laboratory-confirmed SARS-CoV-2 infection between April 2020 and February 2021 followed 1319 patients for 30 days. Investigators found that almost 20% of subjects had severe or critical infection, over half had modifications to cancer treatment, and 3.8% died from COVID-19 infection. Lymphopenia, neutropenia, and residence in a low- or middle-income country were associated with severe or critical illness. It is known that patients receiving active chemotherapy or recent hematopoietic stem cell transplantation can shed viable SARS-CoV-2 for prolonged periods and for at least 2 months after infection in one case series.
Pediatric patients with cancer in remission or solid organ transplant recipients have greater risk for severe COVID-19 than the general pediatric population but overall have done well after SARS-CoV-2 infection, likely because they have sufficiently functional immune responses targeting the virus. In a case series of 25 pediatric kidney and liver transplant recipients in Israel who developed SARS-CoV-2 infection, 84% developed mild disease and 96% developed detectable antibodies at a median of 7 weeks after acute infection. In a retrospective cohort study of 26 pediatric solid organ transplant recipients with a positive SARS-CoV-2 test between April and July 2020 at five US transplant centers, almost all had mild disease and few required hospitalization. Among 24 pediatric kidney transplant recipients, over two-thirds were managed as outpatients, 8 were hospitalized, and, among these, 2 required ICU-level care. None had respiratory failure or allograft loss or died. Pediatric patients receiving other immunosuppressive agents for treatment of rheumatological or inflammatory conditions do not appear to have increased risk for severe COVID.
Treatment of Acute COVID-19 in Children
The safety and efficacy of treatments for acute COVID-19 have been evaluated almost exclusively in adult patients. Very few of these treatment trials included or reported data from pediatric participants. Expert panels have developed treatment guidelines for COVID-19 that, in the absence of pediatric-specific data, have been applied to the care of children with SARS-CoV-2 infection. These therapies are summarized in Table 14.2 . Benefits of most therapies are greatest if given early in the course of COVID-19 disease. Because there is a substantial inflammatory component to COVID-19 pathological processes, immunomodulatory therapies have been used to treat severe disease in hospitalized adults and children. Use of these therapies in children are reviewed in this guidance document. To date, tolerability and safety of these therapies has been relatively unproblematic in the pediatric population, but future studies of SARS-CoV-2 therapeutics should include children.