INTRODUCTION
SUMMARY
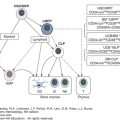
Hairy cell leukemia (HCL) is an uncommon form of adult chronic B-cell leukemia. Whereas the cell of origin is uncertain, at diagnosis the characteristic leukemic cells are found in the marrow, the blood, and the spleen. Patients present with fatigue, infections, and many have splenomegaly. They are often pancytopenic, or may have isolated cytopenias, and usually have monocytopenia. The leukemic cell in classic hairy cell leukemia (HCL-c) has a characteristic immunophenotypic profile (CD11c+, CD19+, CD20+[bright], CD22+, CD25+, CD103+, and CD123+ and CD27−). A variant of hairy cell leukemia (HCL-v), which occurs less frequently, has been identified as a separate entity. A genetic mutation, BRAF V600E, has been identified in the majority of patients with the classic form of this disease, but is not present in the variant. This mutation is also present in the hematopoietic stem cells of patients with HCL-c.
HCL is characterized by impaired marrow function and immunity leading to a high incidence of infectious complications. Both pentostatin and cladribine are effective in achieving durable complete remissions. Long-term studies demonstrate prolonged survival of patients, but the disease-free survival curves do not plateau suggesting that the disease is not cured but subject to relapse. Survival has been markedly improved with the introduction of purine nucleoside analogues and is estimated to be 90 percent at 5-year followup. When patients relapse, high-quality remissions can be achieved with salvage therapy.
Acronyms and Abbreviations
HCL, hairy cell leukemia; HCL-c, classic HCL; HCL-v, variant of HCL; IHC, immunohistochemical stains; IL-2, interleukin-2; MRD, minimal residual disease; SEER, Surveillance, Epidemiology, and End Results Program; SIR, standardized incidence ratio; TGF, transforming growth factor; TRAP, tartrate-resistant acid phosphatase; WHO, World Health Organization.
DEFINITION AND HISTORY
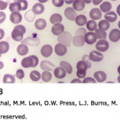
Cases of malignant diseases involving marrow that probably represented examples of hairy cell leukemia (HCL) were reported through the first half of the 20th century and designated by such terms as “lymphoid fibrosis.” They had features characteristic of HCL including marrow replacement by mononuclear cells, marrow fibrosis, splenomegaly and anemia and thrombocytopenia. In 1958, Bouruncle, Wiseman, and Doan described this constellation of findings in a group of patients.1 At that time there was no means to characterize the immunophenotype of malignant lymphoid cells and they called the disease “leukemic reticuloendotheliosis.” In 1966, Schrek and Donnelly, described the distinctive feature of cytoplasmic projections that were evident on the blood cells in two cases of this disorder. They called these “hairy” cells.1A The designation HCL has become universally accepted as the name of this lymphocytic neoplasm, characterized by infiltration of the marrow by malignant B-lymphocytes of a specific immunophenotype (see “Laboratory Features” below), often accompanied by reticular fibrosis, splenomegaly, anemia, thrombocytopenia, neutropenia, monocytopenia, and usually pancytopenia. Occasionally, there is an elevated total white cell count because of the abundance of malignant B-lymphocytes in the blood. Splenectomy was the sole therapeutic approach until the early observations of responses secondary to α-interferon and the purine analogues.2,3 Whereas splenectomy improved the blood counts, the impact of this intervention was temporary, resulting in improvement of symptoms related to splenic sequestration. Most standard chemotherapy was ineffective and poorly tolerated. In 1984, Quesada described the benefits of daily α-interferon in achieving responses in seven patients with progressive HCL.2 Three patients achieved a complete response, with the remaining four having partial responses. Extensive description of the effects of α-interferon on marrow showed improvement in granulopoiesis associated with increases in the number of both circulating granulocytes and platelets. While this approach was heralded as a major achievement in treating this disease, other opportunities emerged in the same time frame.
Grever and colleagues demonstrated that low-dose pentostatin was effective in achieving responses in patients with far-advanced low-grade B-cell malignancy.4,5 Spiers reported the initial response of HCL to pentostatin in a limited number of patients,3 which was followed by a larger study with a complete remission rate of 59 percent.6 Kraut and colleagues subsequently showed that low-dose, less-intense, intermittent pentostatin produced a higher complete remission rate (approximately 89 percent) in patients with HCL.7 Others confirmed these findings,8 and a large prospective randomized trial of pentostatin versus α-interferon solidified that frontline therapy should be based upon a purine analogue.9 Piro and colleagues reported that cladribine produced complete responses in 11 of 12 patients with this disease.10 Numerous trials with cladribine confirmed that a single course of therapy was equally capable of inducing a high percentage of long-term complete responses.11,12 However, the initial trials with cladribine excluded patients with an active infection.10,11 Several studies using cladribine aimed at optimizing the remission rate and attempting to reduce myelosuppression showed that either 5 to 7 days of intravenous administration or subcutaneous injection produced responses, but these alternate approaches did not consistently reduce the risks for febrile neutropenia.13,14,15 In contrast, the reported frequency of febrile neutropenia was less in a study using intermittent administration of pentostatin9 compared to those reported with cladribine.10 Consequently, either purine nucleoside analogue is now used to induce remission.
In patients with an active infection, it is advisable to control the infection before initiating immunosuppressive chemotherapy. However, if this is not possible then administration of either pentostatin alone or following α-interferon may be effective in controlling the underlying disease.9,16 If α-interferon is initially used to improve the neutrophil count and control the leukemia, the subsequent use of pentostatin to achieve a complete remission is not compromised. Therefore, some investigators have initially treated patients with HCL complicated by infection with α-interferon, which was followed by pentostatin in an effort to reduce infectious complications.16 While the use of filgrastim does reduce the degree and the duration of neutropenia, it does not reduce the number of febrile episodes in patients being treated with cladribine, in comparison to historical controls.17 However, it may be helpful in treating serious infections in neutropenic patients with HCL who are in a precarious medical condition. The optimal management of actively infected patients with HCL requiring treatment is still challenging.
The remarkable advances in diagnosis and therapeutics with purine analogues over the past 25 years have clearly changed the natural history of this disease.18 Patients may now lead a near-normal life, despite the likelihood of intermittent relapses requiring retreatment.19,20 Although the risk for serious bacterial infection is greatest during initial therapy, there are some delayed risks associated with impaired recovery of T-lymphocyte cell numbers and function following administration of purine analogues.19,21
In patients who achieve a complete morphologic remission, the presence of minimal residual disease (MRD) has been repeatedly shown by immunohistochemical staining of the marrow.19 Some patients in hematologic remission with MRD may live normal lives, and do not require retreatment unless there is deterioration in their blood counts.22,23 Consequently, further research to define the optimal therapeutic approach and timing for evaluation of response is needed.
The excellent results achieved with purine nucleoside induction associated with high percentages of complete remission have contributed to the improvement in overall survival for patients with this disease.20 Using Surveillance, Epidemiology, and End Results Program (SEER) data after 1984, extensive analysis over the past 3 decades shows a progressive improvement in patient survival with HCL. Despite these truly remarkable results, at least 40 percent of patients will relapse.19 Many of these patients will be successfully retreated, but the failure of the disease-free survival curve to flatten attests to the fact that this disease has been controlled but not cured. Strategies to predict who will be prone to relapse will enable new treatments to be risk-stratified. Furthermore, new agents are being developed to successfully treat patients who have developed resistant disease.19,24,25
EPIDEMIOLOGY
HCL is a rare chronic B-cell lymphoid malignancy accounting for approximately 2 percent of adult leukemias. The estimated annual incidence in the United States is approximately 3.3 persons per million person-years in the United States.24 The mean age at diagnosis is 55 years, but there is a wide range in age of onset.20 Patients may present in their 20s and 30s, and a report on 88 patients who were diagnosed at age 40 or younger showed that these patients do well long-term.26 Younger patients respond better to therapy, but may relapse and require retreatment in order to experience long-term survival benefits.9,19 There is an unexplained male predominance with this disease with a ratio of 4:1 males to females. There is also an unexplained racial difference with more than 90 percent of patients being white.20
There is a bimodal presentation in age raising the possibility of different etiologies in the younger and older patient groups.27 The search for causative relationships in the older subset of patients has included extensive investigation of environmental and occupational exposures.28 There is a suggestion of increased cases associated with farming and in jobs with extensive exposure to insecticides. Extensive investigation of the risk for secondary malignancies identified an increased overall risk (standardized incidence ratio [SIR] 1.24) in a large population-based study according to SEER data from 1973 through 2000.29 In this study of more than 3000 patients with HCL, three separate malignancies were identified as being of particular concern (SIR 6.61 Hodgkin lymphoma; SIR 5.03 non-Hodgkin lymphoma; and SIR 3.56 thyroid cancer). Whether or not these second malignancies are related to an immune deficit from the leukemia or a result of the therapy for the leukemia is unknown.
ETIOLOGY AND PATHOGENESIS
HCL represents a clonal population of leukemic cells predominantly infiltrating the marrow, the spleen, and the liver. These cells are characterized as mature activated memory B cells based upon their immunophenotypic profile.30 The neoplastic cells in classic HCL (HCL-c) have hypermutated immunoglobulin genes in approximately 90 percent of the cases.31 In those patients who have unmutated immunoglobulin genes, the disease appears to be more aggressive, as is the case in chronic lymphocytic leukemia. Leukemic cells use a variety of VH gene families. In those patients showing a VH4–34 gene, the clinical course has also been noted to be less favorable.32,33 Molecular parameters may have prognostic value. For example, patients with a mutation in p53 have been less responsive to purine analogue therapy. Therefore, characterization of the molecular and genetic profile of the leukemic cells may elucidate the cell of origin and have prognostic value with respect to clinical outcome and responsiveness to standard therapy.
The identification of a mutation in BRAF V600E in almost 100 percent of patients with HCL-c has had a major impact on classification of the previously defined subsets of this disease.34 The leukemic cells from patients with the variant of HCL (HCL-v) express BRAF wild-type. This genetic difference confirms that these two entities are clearly unrelated with a completely separate clinical course and therapeutic responsiveness. It is interesting that the small subset of patients with an immunophenotype consistent with HCL-c, yet using VH4–34 rearrangement, appear to be BRAF wild-type, further suggesting that there are multiple unique clonal patterns with distinct clinical courses.35 Despite immunophenotypic features consistent with HCL-c, the lack of expression of BRAF V600E identifies a clinical course unlike the highly responsive form of the leukemia.
The presence of BRAF V600E mutation appears to activate the MEK-ERK signaling pathway responsible for leukemic cell survival and proliferation. Downstream activation of pERK correlates with presence of this mutational pathway.36 Hematopoietic stem cells expressing this mutation have been identified in the marrow of murine models of the disease,37 as well as in the marrow of patients with HCL.37 BRAF V600E in marrow stem cells may explain the impairment in normal hematopoiesis that occurs in HCL-c. Furthermore, cytokines secreted by the leukemic cells may be responsible for the areas of hypocellularity observed in some patients with HCL-c.38,39 Alternatively, impaired hematopoiesis may result from inadequate growth factor production.40 Pancytopenia may also result either from extensive marrow infiltration with leukemia or fibrosis induced by the overproduction of transforming growth factor (TGF)-β by leukemic cells.41
CLINICAL FEATURES
The most common presenting symptoms are weakness and fatigue, which occur in 50 percent of patients with HCL.42 Although many patients also have an enlarged spleen, the gradual onset of symptoms is described as fullness in left abdomen, early satiety, and discomfort. Initially, Bouroncle reported that splenomegaly was found in 96 percent of patients.1 However, more recently the percentage with a markedly enlarged spleen may be less at diagnosis because of earlier detection of the disease. Patients may present with a history of increased infections, and approximately 17 percent have an active infection at the time of diagnosis.1 Bleeding manifestations are also noted in patients with severe thrombocytopenia. Patients may present with few symptoms, but an abnormal laboratory report suggesting a hematologic disorder may emerge during a routine health examination.
Infection has been the leading cause of death in patients with HCL, and accounted for 55 percent of the fatalities in a large longitudinal review of 725 patients in an Italian series.43 In general, the infections occur as a result of granulocytopenia, monocytopenia, and impaired function of immune effector cells. Approximately 30 percent of patients are found to have a documented source of infection, but an equal number of suspected infections cannot be documented microbiologically.44 Fever in this patient population should prompt a search for infection. It has been estimated that 48 percent of infections are caused by pyogenic organisms including Staphylococcus aureus, Pseudomonas aeruginosa, Streptococcus pneumoniae, Escherichia coli, Klebsiella pneumonia, and Legionella pneumophilia.21 Multiple other organisms have been identified as a source of infection including Aspergillus, Candida, Blastomyces, Histoplasma, Cryptococcus, Toxoplasmosis, Pneumocystis jiroveci, and atypical mycobacteria.
While the majority of infections occur before effective treatment has been initiated, the additional risk of infection as a complication of treatment exists both immediately following therapy and for many months thereafter.21 The purine nucleoside analogues that are used as a backbone of induction therapy can produce profound and prolonged myelosuppression. Because of the extensive marrow involvement with leukemia, the myeloid reserve is severely compromised at the initiation of therapy. Following effective therapy, the granulocytes gradually recover, but the purine nucleoside analogues usually induce a prolonged period of reduction in lymphoid cells, thus opportunistic infections resulting from compromised lymphocyte function may also emerge in the posttreatment period.45 Once the patient has achieved a complete remission, the risks for infection become progressively less as the hematologic parameters improve. Full recovery of lymphocyte function following purine analogue therapy, however, may require several years.46,47
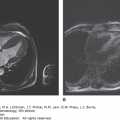
Unusual symptoms related to HCL may include bone pain and autoimmune complications.48,49 Bone pain may be the result of lytic disease that can involve the spine, the femur, and other skeletal sites. Lytic bone disease can occur at any time during the course of the disease. Both magnetic resonance imaging (MRI) and computed tomography (CT) scans have been helpful in identifying these lesions even when plain films of involved areas are normal. Biopsies of bone lesions have confirmed the presence of hairy cells, and these manifestations may respond to effective treatment of the leukemia. Many of the patients with bone lesions respond to systemic treatment of the disease, but others require additional localized irradiation.
Diverse autoimmune findings in patients represent other unusual complications.49,50 Patients may complain of migratory inflammatory episodes involving the joints and tenosynovial tissues. These painful inflammatory episodes are usually self-limited and resolve spontaneously but may be recurrent. Vasculitic skin lesions and erythema nodosum have been reported.51 Autoimmune hemolytic anemia and thrombocytopenia have also been observed.52,53,54 The autoimmune phenomena are not related to tumor burden, and may be present at initial diagnosis or occur anytime throughout the course of the disease. Finally, patients have also presented with paraneoplastic neurological syndromes.55
LABORATORY FEATURES
Patients with HCL reviewed in a large Italian series had pancytopenia (77 percent) reflecting impaired hematopoiesis due to marrow infiltration and splenic sequestration.21,43 Approximately 28.4 percent of patients had a hemoglobin less than 8.5 g/dL with 14.9 percent requiring a blood transfusion, 39 percent of patients had an absolute neutropenia (neutrophils <500/μ
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree