(1)
Medical Sciences Division Northern Ontario School of Medicine West Campus, Lakehead University, Thunder Bay, Ontario, Canada
Key Topics
Molecular pathology of endocrine cancers.
Circulating thyroid cancer (ThyCa) epigenetic, genetic, and transcript biomarkers.
Serum proteins (antithyroglobulin antibodies) as ThyCa biomarkers.
Circulating ThyCa cells.
Circulating endothelial cells as ThyCa biomarkers.
Circulating pheochromocytoma (PCC) and paraganglioma (PGL) biomarkers.
Key Points
The most common malignancy of the endocrine system is ThyCa, followed by those of the adrenal gland. ThyCa mortality is globally low; however, this remains high in the developing parts of the world.
Circulating thyroid cancer biomarkers of clinical relevance include ctDNA detection by gene methylation and mutations (e.g., BRAF V600E ), as well as transcripts and antibodies to thyroglobulin (TG). Circulating miRNA, proteomic discoveries, circulating ThyCa, and endothelial cells are emerging biomarkers.
There is paucity of circulating biomarkers for adrenal cortical tumors, but PCC and PGL are managed by monitoring the levels of serum catecholamines and granins.
17.1 Introduction
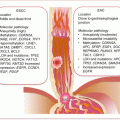
The endocrine system is a multi-tissue and organ system that regulates almost all body functions through secretions (hormones) that are spread throughout the body via the vasculature. They are anatomically organized into connective-tissue delineated cells arranged in clumps, cords, or follicles, as well as diffused cells in organs such as the kidney, heart, gastrointestinal system, and reproductive organs. In regard to malignant transformation, the common malignancies are associated with the well-organized tissues, especially the thyroid and adrenal glands, with low incidence cancers among the remaining endocrine structures.
Neoplastic transformation of thyroid follicular cells is the most common malignancy of the endocrine system. Although not as common as other epithelial cancers, the estimated 2012 global ThyCa incidence was 68,179, with 56.5 % occurring in the less developed world. In the same year, the estimated mortality was 12,626, but unlike the incidence distribution, the deaths in the less developed world (71.1 %) more than doubled those of the developed world. Thus, while in the US 64,300 new cases are expected in 2016, the projected mortality is only 1,980. Most thyroid nodules are benign follicular adenomas. Only ~5 % of thyroid tumors are malignant, mainly carcinomas of follicular cells. ThyCas are three times more prevalent in women than men, and the global incidence has been on the rise over the past few decades, probably as a result of improved early detection of possibly nonprogressive tumors.
ThyCas are histologically classified into three main groups, namely well-differentiated thyroid carcinomas (papillary and follicular carcinomas), poorly differentiated thyroid carcinomas, and undifferentiated thyroid carcinomas or anaplastic ThyCas. Parafollicular or C cell tumors are referred to as medullary ThyCas. Well-differentiated ThyCas have good prognosis, with 10-year survival of 80–95 %. However, recurrences can occur 30 years after diagnosis even in those supposedly deemed to have been cured. Biomarkers that can be used for noninvasive monitoring of recurrence will thus be useful for follow-up surveillance. Anaplastic ThyCa is the most aggressive subtype, being responsible for >30 % of all ThyCa-related deaths. Biomarkers for the early detection and treatment monitoring of ATC have thus been explored.
Tumors of the adrenal medulla and cortex are also rare. Many (80–85 %) chromaffin cell tumors arise from the adrenal medulla and are referred to as pheochromocytomas. The remaining tumors originate in the extra-adrenal sympathetic and parasympathetic nervous system and constitute the paragangliomas. These tumors are rare with incidence of 2–5 per million per year. They are mostly benign, with malignancy occurring in 10–15 % of all cases. Thus, the overall survival is good at ~89 %, but when malignant, the survival outcome is dismal at 20–70 %. It is therefore imperative to establish malignancy at the earliest possible time during the diagnostic work-up.
Carcinomas from the adrenal cortex are equally rare, with estimated annual incidences of 0.7–2 per million. They are more common in children <5 and adults between ages 40–50. Because of inefficient therapies, complete surgical tumor resection remains the mainstay of curative treatment. Thus, recurrences are common, occurring in 60–80 % of cases. Patients who develop recurrent disease and those initially diagnosed with advanced stage IV disease have 5-year survival rate of <5 %. Indeed, those unsuitable for resection only survive 3–9 months. There is thus a needed understanding of the molecular pathology and development of effective therapies for adrenal cortical tumors. Noninvasive biomarker assays for early detection and disease management are of importance as well. However, the rarity of these tumors may pose as a challenge to the development of effective novel therapies, but he effort is worthwhile.
17.2 Screening Recommendations for Endocrine Cancer
Because of the low incidence and prevalence of endocrine cancers, screening is not recommended for the general population at moderate to low risk. However, certain risk factors elevate the probability of people developing certain types of cancers. For example, the risk factors for ThyCa include gender, age, hereditary factors, radiation exposure, and low iodine intake. Female gender is associated with elevated ThyCa risk and early age at diagnosis (40–50 years compared to 60–70 years in men), while radiation exposure to the head and neck region, especially during childhood and adolescence, increases the risk. Family history of ThyCa, genetic risk factors such as inherited RET mutations, and diet low in iodine are all associated with elevated risks above the general population. Therefore, people with these risk factors are periodically examined for early detection of ThyCas. The surveillance work-up includes physical examination of the thyroid and cervical lymph nodes, imaging especially by diagnostic ultrasound and serum measurement of thyroid-stimulating hormone. Normal or elevated TSH carries very low risk for cancer; however, low levels require radionuclide thyroid scan. Again a “hot” or autonomous nodule carries low risk for cancer.
17.3 Molecular Pathology of Endocrine Cancers
The molecular pathology of cancers of the thyroid and adrenal glands will be presented followed by synopsis of other endocrine glands and tissues.
17.3.1 Molecular Pathology of ThyCa
The thyroid gland is composed primarily of the thyroxin-secreting follicular cells. In addition, there are scattered parafollicular or “C” cells that secrete calcitonin. As a result, the majority of ThyCas originate from these two cell types. ThyCas are heterogeneous in nature because any cell type in the gland can give rise to tumors. Thus, both thyroid epithelial and non-epithelial cells are susceptible to transformation. However, almost all thyroid tumors are of epithelial cell origin. Based on the cell of origin, primary thyroid tumors fall into two categories. Many (>95 %) ThyCas are of follicular cell origin, with just 2–5 % emanating from parafollicular cells, and rarely from other non-epithelial cell types in the thyroid. Tumors of follicular cell origin can be benign (follicular adenomas and oncocytomas) or malignant. The malignant cancers are further subclassified into papillary ThyCa (PTC), follicular ThyCa (FTC), poorly differentiated ThyCa, and undifferentiated or anaplastic ThyCa (ATC). Papillary ThyCa, FTC, benign thyroid tumors, as well as the normal follicular cells can acquire genetic lesions and progress to the poorly differentiated and anaplastic carcinomas. Noteworthy, there are several other minor variants of ThyCas such as epithelial tumors of uncertain origin, mixed follicular and C cell tumors, as well as the other subtypes of non-epithelial cell origin.
The molecular pathology of ThyCa is well elucidated. Mutations in genes that primarily control the MAPK pathway are strongly implicated in thyroid carcinogenesis. Mutually exclusive mutations in BRAF, RAS, and RET/PTC rearrangement underlie the vast majority (>70 %) of PTC. Similarly, >70 % of FTC is characterized by mutually exclusive mutations in RAS and rearrangement mutations in PAX8/PPARγ. While ATCs harbor some of these mutations (e.g., ~25 % have BRAF mutations), they are characterized by additional mutations involved in the WNT/β-catenin, PI3K, and cell cycle pathways. Activating mutations in CTNNB1 that lead to WNT/β-catenin signaling are found in 25–60 % of ATC. Similarly, loss of function in PTEN tumor suppressor leading to PI3K pathway activation is present in up to 16 % of ATCs. Over 50 % of ATCs harbor TP53 mutations, while mutations in the promoter region of TERT are demonstrated in 30–50 % of these tumors. TERT mutations are frequently found in tumors with RAS or BRAF mutations.
17.3.1.1 BRAF Mutations in ThyCa
Activating mutations in BRAF are demonstrable in about 35–70 % of PTCs. Whereas mutations have been described at codons 598, 599, 600, and 601, the BRAF valine substitution with glutamic acid at nucleotide position 1799 of codon 600 is the most common, occurring in up to 90 % of PTCs. This mutation is more common in the tall cell variant of PTC and is associated with aggressive disease and relapse, and hence is a predictor of poor prognosis. PTC originating on the background of radiation exposure may harbor AKAP9/BRAF rearrangements.
17.3.1.2 RAS Mutations in ThyCa
The RAS genes (KRAS, NRAS, and HRAS) are mutated in ThyCa. While KRAS, NRAS, and HRAS codon 61 mutations have been reported, KRAS codon 12 and 13 mutations are the most common. These mutations are present in 20–40 % of adenomas, in 10–15 % of PTCs (especially the follicular subtype), and in 40–50 % of FTCs. However, they are detected in ~35 % of poorly differentiated thyroid carcinoma and up to 50 % of ATCs.
17.3.1.3 PIK3CA Mutations in ThyCa
Mutations in this gene are present in 25–45 % of ThyCas. The mutations, which are commonly in exons 9 and 20, are rare in PTC (just up to 5 %) but more common in poorly differentiated and anaplastic cancers. Moreover, the PIK3CA genomic locus, 3q26.3, is amplified in ~40 % of ATCs.
17.3.1.4 PAX8/PPARγ Rearrangement in ThyCa
Translocation between chromosomes 2 and 3 t(2;3)(q13;p25) gives rise to the fusion of the thyroid-specific paired domain transcription factor, PAX8, with PPARγ giving rise to the PAX8/PPARγ rearrangement. This causes overexpression of PPARγ in ThyCas. This gene rearrangement, which tends to be exclusive of RAS mutation, is common in FTC, with a frequency of 30–40 %, but is associated with favorable prognosis. These genomic rearrangements are also found in a small fraction of benign thyroid tumors. About 2–10 % of follicular adenomas and ~5 % of oncocytomas harbor them. Rarely (<5 %), the PAX8/PPARγ rearrangement has been reported in the follicular subtype of PTC.
17.3.1.5 RET/PTC Rearrangement in ThyCa
The fusion of the 3′ end of RET tyrosine kinase receptor to the 5′ ends of a number of genes creates the RET/PTC rearrangements. RET/PTC1 and RET/PTC3 are intra-chromosome 10 fusions, while the rest including RET/PTC2 are fusions of genes between chromosomes. However, all these fusion genes retain the tyrosine kinase activity of RET and hence activate the MAPK pathway important in thyroid carcinogenesis. These rearrangements are demonstrable in 10–20 % of sporadic adult PTCs but are clustered more in patients with a history of radiation exposure where as many as 50–80 % will have them. In sporadic non-radiation-mediated PTC, RET/PTC1 (frequency of 50–70 %) and RET/PTC3 (frequency of 20–30 %) are the most common.
17.3.2 Molecular Pathology of Adrenal Cortical Cancer
Adrenal cortical tumors (ACTs) are diseases of epigenetics and genetics. While the molecular pathology is not fully unraveled, some progress is made in the alterations of genes important in the development and progression of this disease. Specific gene methylations, miRNA deregulation, mutations in specific genes, and deranged growth factor signaling have all been demonstrated in adrenocortical cancers (ACCs).
17.3.2.1 Epigenetics of ACTs
DNA methylation and miRNA deregulation underlie ACCs. ACCs demonstrate global genomic hypomethylation compared with benign adrenal tumors and normal adrenal tissues. However, promoter hypermethylation at CpG islands occurs in a number of genes including NTNG2, KIRREL, KCTD12, SYNGR1, GATA4, CDKN2A, DLEC1, PYCARD, and SCGB3A1. Of interest, promoter methylation at 11p15, the IGF2 and H19 imprinted gene locus, is found in ACCs.
There are differential miRNA expressions between ACC and adrenal cortical adenomas (ACAs) and normal adrenal tissues. MiR-139-5p, miR-184, miR-210, miR-483-5p, and miR-503 are upregulated, while miR-139-3p, miR-195, miR-335, and miR-675 are downregulated in ACC compared to control samples. Of importance, miR-483-5p and miR-195 are reproducible. MiR-483-5p may control IGF2 overexpression and the levels are potential diagnostic and prognostic biomarkers. The potential of circulating miR-483-5p and miR-195 in management of patients with ACC has been demonstrated.
17.3.2.2 Chromosomal Changes in ACTs
Alterations in chromosomal regions that harbor genes mutated or underexpressed in ACC characterize these tumors as well. LOH and CGH studies indicate increased frequency of chromosomal losses and genetic lesions in sporadic ACC, and the frequencies are much higher in these cancers compared to adenomas. The regions of chromosomal losses include 2p16, 11q13, 11p15, 17p13, and 17q22–24. These loci house important genes, such as TP53 (17p13) involved in the pathogenesis of ACC.
17.3.2.3 TP53 Mutations in ACTs
Germline mutations in TP53 characterize the vast majority (50–80 %) of childhood ACCs, while somatic mutational inactivation is found in 20–35 % of sporadic adult ACCs. In sporadic adult ACC, hotspot exon 5–8 mutations occur in 20–27 % of the cases. LOH at the TP53 locus occurs in ~80 % of ACCs. However, these do not match TP53 mutations or loss of function, suggestive of other pertinent genes in this locus. Candidate genes include ALOX15B and ACADVL, which have decreased expression in ACCs. The importance of TP53 mutations in ACC is testified by the disproportionately high incidence of ACCs in southern Brazil. Here, the recurrent TP53 germline mutation, p.R337H, accounts for the majority of childhood cases. Approximately, 80% of all sporadic ACCs harbor this mutation in southern Brazil.
17.3.2.4 Growth Factor Signaling Pathways in ACTs
Growth factor signaling, primarily through the PI3K and RAS-MAPK pathways, is demonstrated in many ACCs. Abnormal expressions of VEGF, TGFα, TGFβ1, βFGF, and cytokines characterize ACC. VEGF and its receptor, VEGFR2, is overexpressed in 70–80 % of ACCs. Indeed, circulating VEGF levels in patients with ACC fall following tumor removal. Over 90 % of ACCs overexpress EGFR, and FGFR1 and FGFR4 levels are high in ACTs.
17.3.2.5 IGF2 Alterations in ACTs
The IGF2 locus, 11p15, shows LOH in ~80 % of ACCs. This locus also contains H19 and CDKN1C/p57kip2. Abnormal imprinting, such as duplication of the paternal allele and loss of the maternal allele (which houses the H19 gene), leads to overexpression of IGF2. Together with other regulatory modes, as many as 80–90 % of ACCs overexpress the IGF2 gene.
17.3.2.6 SF1 Alterations in ACTs
The amplification and overexpression of steroidogenic factor 1 (SF1) occurs in a substantial number of both childhood and adult ACCs. However, these abnormalities are more frequent in pediatric than adult ACCs. Overexpression of SF1, which occurs in some tumors without amplification, suggests alternative regulatory pathways of SF1 overexpression in ACCs.
17.3.2.7 WNT/β-Catenin Pathway Alterations in ACTs
Intracellular accumulation of β-catenin and WNT/β-catenin pathway activation occurs in both benign (38 %) and malignant (>70 %) ACTs. While not too clear, somatic mutations in β-catenin, which occur in ~27 % of ACA and in 31 % of ACC, may partly account for the lack of β-catenin degradation.
17.3.2.8 MC2R Alterations in ACTs
LOH and hence inactivation of the ACTH receptor (MC2R) is associated with a subset of ACTs. However, in patients with functional tumors, MC2R shows a rather upregulated expression. The loss of MC2R causes decreases in mineralocorticoid and glucocorticoid production as well as adrenocortical atrophy.
17.3.3 Molecular Pathology of Pheochromocytoma and Paraganglioma
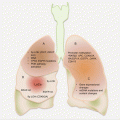
The vast majority (up to 85 %) of chromaffin cell tumors arise from the adrenal medullary chromaffin cells (referred to as pheochromocytomas—PCCs). The remainder is derived from extra-adrenal sympathetic nervous system of the chest, abdomen, and pelvis as well as the head and neck parasympathetic nervous tissues (known as paragangliomas—PGLs). Whereas tumors from sympathetic tissues are endocrinologically active, those from the parasympathetic system are silent and hence cannot be detected using the standard biochemical assays targeting circulating catecholamine.
Although identical, the molecular pathology of PCCs and PGLs is very diverse. Germline and somatic gene mutations account for ~52 % of all cases. Germline heritable mutations in ten genes account for ~35 % of all cases. Thus, PCC and PGL syndromes are accounted for by mutations in the von Hippel Lindau (VHL), multiple endocrine neoplasia 2 (MEN2/RET), the four succinate dehydrogenase genes (SDHA, SDHB, SHDC, and SDHD), neurofibromatosis type 1 (NF1), SDH complex assembly factor 2 (SDHAF2), transmembrane protein 127 (TMEM127), and MYC-associated factor X (MAX). Other pertinent features of these mutations are the fact that SDHB mutations carry a risk for malignant transformation, while alterations in RET, TMEM127, and SDH genes are associated with multifocal tumors. Somatic gene mutations in VHL, MAX, RET, NF1, SDH, as well as HIF2α belies 17 % of sporadic PCCs and PGLs.
17.3.4 Molecular Pathology of MENs
Multiple endocrine neoplasias (MENs) are a conglomerate of tumors that arise from the unrelated neuroendocrine cells that secrete most of the nonsteroidal hormones of the body, primarily amine precursor uptake and decarboxylation (APUD) cells. These diseases are inherited in an autosomal dominant fashion. The multiple endocrine neoplasia syndromes involve genes important in the regulation of neuroendocrine functions, and hence abnormalities lead to multiple neuroendocrine tissue involvement. There are three types based on genetic abnormalities and primary tissues involved. These are MENI, MENIIA, and MENIIB.
17.3.4.1 MENI
MENI is a rare disease, with an incidence of 0.25 % and prevalence of 0.02 to 0.2 per thousand. Despite its rarity, penetrance is very high. This disease is characterized by hyperplasia or neoplasm of endocrine pancreas, parathyroid, and pituitary glands. These abnormalities lead to hyperparathyroidism in 90 % of the patients, pancreatic islet cell tumors, mostly islet cell gastrinoma, which occurs in ~60 % of the cases, and pituitary adenomas that are found in ~40 % of the patients. Molecularly, MENI is characterized by mutations, mostly nonsense mutation (~80 %) in the MEN1 gene located on chromosome 11q13 that encodes a 610 amino acid protein called menin. While its functions are not fully elucidated, menin interacts with multiple transcriptional regulators that control cellular proliferation.
17.3.4.2 MENIIA/B
MENII is an autosomal dominant disease with high degree of penetrance. Based on the structures mostly involved, MENII is further divided into MENIIA and MENIIB. All MENIIA patients will have calcitonin-secreting parafollicular or C cell hyperplasia or medullary ThyCa (MTC); 50 % will present with pheochromocytoma and 20–30 % will demonstrate hyperparathyroidism. Similarly, almost all MENIIB patients will have MTC and pheochromocytomas but rarely have hyperparathyroidism. Additionally, MENIIB patients may demonstrate other gastrointestinal neuroendocrinopathies such as ganglioneuromatosis of the gastrointestinal tract. Molecular pathologically, mutations in the RET tyrosine kinase proto-oncogene, which is expressed by tissues of neural crest origin, are implicated in MENII syndromes. All MENIIA patients have RET mutations, but a few MENIIB patients lack these mutations. Additionally, LOH at chromosomes 1p, 3p, 17p, and 22q are associated with MENII.
17.4 Circulating ThyCa Biomarkers
ThyCa biomarkers of clinical relevance are detection of BRAF V600E mutations in ctDNA and measurement of the circulating levels of thyroglobulin (TG or Tg) transcripts and antibodies. New members under investigation include circulating miRNA, circulating ThyCa cells, and circulating endothelial cells.
17.4.1 Circulating Cell-Free Nucleic Acid as ThyCa Biomarkers
Data on ccfDNA content or levels for ThyCa detection are lacking; however, studies of the epigenome and genome alterations in ccfDNA suggest their potential in ThyCa management. Methylation and loss of function of genes involved with iodine metabolism (SLC5A8, SLC26A4) occur in ThyCa, and SLC5A8 hypermethylation is associated with BRAF V600E mutation, the most common genetic alteration in PTC. Because of these associations, the levels of ccfDNA as well as alterations in these genes were assayed in ccfDNA of ThyCas of all histologic types. CcfDNA levels highly discriminated cancer from healthy controls and correlated with various histologic subtypes of ThyCa. A panel consisting of ccfDNA, methylation of SLC5A8 and SLC26A4, and BRAF V600E mutations demonstrated some diagnostic potential [1].
Similar to melanoma, a majority of PTC harbors BRAF V600E oncogene mutations. While this mutation was absent in benign adenomas, follicular neoplasms or carcinoma, and thyroid lymphoma, 35.7 % of PTCs were positive for this mutation of which three were detected in serum samples as well, and these were associated with lymph node metastasis [2]. In contrast to the above low frequency of detection, Kim et al. found the mutation frequency to be 68.1 % in PTC tissue samples, but only three patients with lymph node and lung metastasis had detectable circulating BRAF V600E mutations [3]. Pupilli et al., however, measured the percentage of BRAF V600E in ccfDNA, which was significantly different between patients and controls, as well as different histologic categories [4]. Patients with PTC histology had higher percentage of the mutation than those with benign histology. As a marker of tumor removal, the levels fell after surgery. Used as a diagnostic, circulating levels of BRAF V600E achieved an AUROCC of 0.797 at a sensitivity of 80 %, specificity of 65 %, PPV of 33 %, and NPV of 80 %.
17.4.2 Circulating ThyCa Coding RNA Biomarkers
Primarily TG mRNA has been investigated for its utility in diagnosis and recurrence monitoring. Recurrences of ThyCa are monitored by serum TG immunoassays and 131I whole-body scans. But antibodies can hinder the sensitivity of the immunoassay. Hence, RT-PCR-based transcript measurements have been explored for such clinical utility. Thyroid-specific transcripts targeting TG, TPO, TSHR, NIS, and PDS are known biomarkers of recurrence or presence of residual cancer after surgery. These transcripts in circulation were measured and compared to the performance of serum TG protein levels. Overall, serum TG protein assay was superior. TG levels were higher in serum samples from cancer patients and much higher in those with residual tissue or metastatic disease during thyroid hormone stimulation test (THST) or recombinant human thyrotropin (rhTSH) stimulation. Patients with relapse had increased serum TG levels compared to disease-free patients, and THST and rhTSH stimulations had 81.4 % and 90.9 % accuracy, respectively. The transcripts had lower sensitivities and specificities, but a panel of TPO and TSHR transcripts may be specific for detection of relapse [5]. In another study, circulating TSHR mRNA measured in comparison to TG mRNA as well as serum TG protein levels revealed a sensitivity of 100 % and specificity at 98 % for TSHR mRNA in detecting differentiated ThyCa. Additionally, TSHR mRNA had 95 % correlation with TG mRNA and protein levels. The detection in preoperative patients indicates its possible utility in ThyCa detection in people with undiagnosed thyroid nodules [6]. Circulating TSHR mRNA for preoperative diagnosis of ThyCa in patients with thyroid nodules was compared to FNA cytology and to serum TG levels and/or whole-body 131I scan. TSHR mRNA levels were significantly increased in cancer patients compared to controls. At an established cutoff, circulating TSHR mRNA correctly detected ThyCa at a sensitivity of 72 % and specificity of 82.5 %. TSHR mRNA and ultrasound features of follicular lesions correctly classified all follicular cancers and potentially could have saved 31 % of patients with benign lesions from unnecessary surgery. Postoperative day 1 levels of TSRH, as a measure of circulating cancer cells, were predictive of disease status. Elevated levels were indicative of residual or metastatic disease [7]. TSHR mRNA in circulation has also been explored as an adjunctive predictive biomarker to monitor ThyCa recurrence. TSHR mRNA was correlated with TG immunoassay, imaging analysis, and disease status. Overall, TSHR mRNA was better than TG and agreed with whole-body scan detection of recurrent disease in 79 % of cases. TSHR mRNA also predicted disease status in 77 % of patients. In conjunction with TG immunoassay, this panel achieved a sensitivity of 90 % and specificity of 94 % in detecting cancer recurrences. Note however that this group comprised mainly papillary-type ThyCa patients (91 %) [8].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree