Cutaneous Manifestations of Diabetes Mellitus
Mark H. Lowitt
Jeffrey S. Dover
Patients with diabetes mellitus commonly suffer from a wide variety of cutaneous maladies. Estimates of the frequency of skin disease in people with diabetes range from 30% overall (1) to 71% of patients with type 1 diabetes mellitus (2). While several skin conditions are specific to diabetes, most of them also occur in individuals without diabetes. The clinical manifestations and complications of skin disease are frequently more severe in the setting of diabetes. The pathophysiology of most cutaneous disorders in the patient with diabetes remains poorly understood. Recent advances in genetic research may soon answer some longstanding questions. For example, results from a recent study indicate that the G82S polymorphism of the RAGE gene (receptor for advanced glycosylation end products) is associated with microangiopathic skin conditions in patients with type 2 diabetes (3). In this chapter, we describe the characteristic skin conditions associated with diabetes, incorporating discussion of clinical presentation, pathology, pathogenesis, and treatment (Table 62.1).
TABLE 62.1. Skin Manifestations of Diabetes | |||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
CUTANEOUS INFECTIONS IN DIABETES
Candidiasis
Candidal infections of mucosal membranes, genitalia, and nails are more prevalent in patients with poorly controlled diabetes than in the population without diabetes (4,5,6,7). Women are more prone to these infections than are men (5). Although yeast infections may be the initial presentation of diabetes, they generally occur in patients already known to have diabetes (6). In individuals
with diabetes, the ratio of epidermal glucose to blood glucose is higher than in persons without diabetes (8), a situation that may produce an environment that is more favorable for the growth of yeast and fungi (9).
with diabetes, the ratio of epidermal glucose to blood glucose is higher than in persons without diabetes (8), a situation that may produce an environment that is more favorable for the growth of yeast and fungi (9).
ORAL MUCOSAL CANDIDIASIS
Distinctive forms of oral mucosal candidiasis include thrush (curdlike white colonies) over intra-oral surfaces; atrophic candidiasis, which manifests as bright red atrophy of the hard palate or tongue; sore mouth; and angular cheilitis (perlèche), which presents as superficial or deep erosions along the labial commissures. Antifungal creams or troches usually are required to eradicate infection.
CANDIDAL PARONYCHIA
The frequency of candidal paronychia, the inflammation surrounding the nail that is caused by a candidal infection, is increased in persons with diabetes. In one study of 250 women with diabetes, 9.6% had clinical evidence of candidal paronychia as compared with 3.4% of 500 controls without diabetes (10). Typical candidal paronychia begins with erythema, swelling, and pain at the lateral nail fold, leading to its separation from the nail margin. The proximal nail fold then becomes involved, and the cuticle may separate from the nail plate (11). Purulent drainage may be mistaken for bacterial infection. Candidal paronychia is characterized by intermittent exacerbations with episodic indolent periods, a course that may result in a rippled appearance of the nail plate. Patients with occupations in which the hands are frequently immersed in water are more prone to this condition (12). Practical treatment includes keeping the hands as dry as possible. The wearing of cotton gloves under rubber or vinyl gloves can protect hands during dishwashing. Topical antifungal solutions usually are adequate for treatment; however, patients with refractory cases may require oral therapy with an imidazole.
CANDIDAL VULVITIS
Candidal vulvovaginitis is characterized by pruritus, vulvar erythema, and occasionally fissuring and pustules (11). Glycosuria may promote growth of Candida species. Certain case studies have shown that the severity of pruritus is proportional to the degree of glycosuria and hyperglycemia (13). Broad-spectrum antibiotics, birth-control pills, and topical corticosteroids are also common causes of candidal vulvovaginitis. Antifungal vaginal suppositories or oral imidazole antifungal agents are necessary to eradicate infection. Non-Candida albicans species of Candida are now being recognized as pathogens in vulvovaginitis, particularly in women with diabetes. These organisms are often resistant to standard oral antifungal agents (13). Oral nystatin solution may be beneficial in reducing the reservoir of Candida organisms in the gastrointestinal tract.
CANDIDAL BALANITIS
Men with candidal balanitis present with diffuse or focal erythema of the glans penis, often in association with pain or pruritus. White discharge in association with erosions or pustules is commonly seen, particularly under the foreskin of uncircumcised patients. Men with balanitis and phimosis, especially if middle-aged or elderly, should be evaluated for diabetes. In one retrospective study, among 100 men requiring circumcision for phimosis, 35% had diabetes. For most of these patients, diabetes had not been diagnosed previously (7). Administration of topical or oral imidazoles is the treatment of choice. If phimosis is present, a cotton-tipped applicator may be used to gently introduce the anticandidal preparation underneath the foreskin (11).
Dermatophyte Infections
Tinea pedis (athlete’s foot) is more prevalent in persons with diabetes than in the general population (4). Although innocuous in most people, tinea can create fissures and portals of entry that may lead to severe bacterial infections in those with diabetes. Web spaces are pruritic, scaly, erythematous, and macerated. Vesicles and pustules may also be present. A “moccasin” distribution of scaling extending from the soles onto the lateral feet is another variant of tinea pedis. Long-standing tinea pedis may involve the nails, which develop a thick, yellowish brown roughened nail plate and subungual debris. Potassium hydroxide (KOH) scraping identifies branching, septate hyphae (12). Patients who have tinea involving their hands or fingernails may inoculate other body sites as well.
Topical antifungal agents are the mainstays of treatment for these infections. For severe fungal infections of skin, hair, or nails, administration of an oral imidazole (such as itraconazole) or an allylamine antifungal agent (such as terbinafine) is the treatment of choice. Blood counts and liver function tests must be monitored during treatment, and drug interactions between
imidazole antibiotics and other drugs metabolized by the hepatic cytochrome P450 system must be anticipated. Preventive foot care, including careful drying, wearing of cotton socks, and wearing of sandals in public shower facilities may help prevent infection. Topical ciclopirox administered as a nail lacquer is a welcome recent therapeutic option.
imidazole antibiotics and other drugs metabolized by the hepatic cytochrome P450 system must be anticipated. Preventive foot care, including careful drying, wearing of cotton socks, and wearing of sandals in public shower facilities may help prevent infection. Topical ciclopirox administered as a nail lacquer is a welcome recent therapeutic option.
Phycomycetes Infections
The phycomycetes are a group of fungi found on decaying vegetation and foods with a high sugar content. Although rarely pathogenic, the genera most commonly involved in human infection include Rhizopus, Mucor, and Absidia (14). In the clinical setting, all diseases caused by these organisms are referred to as mucormycosis.
Several major clinical syndromes, involving the rhinocerebral, thoracic, gastrointestinal and cutaneous systems are described (15). Of these, rhinocerebral disease is most closely associated with diabetes. The fungal organism colonizes the nasal turbinates and sinuses but can invade the orbit and brain, particularly in the setting of diabetic ketoacidosis. Characteristically black crusty or purulent material is present on the turbinates, septa, or palate, and mucormycosis may initially be mistaken for bacterial sinusitis. Cutaneous mucormycosis occurs when the organism colonizes burned or skin-grafted tissues or nonhealing leg ulcers in patients with diabetes (16).
The diagnosis can be confirmed by skin biopsy or a KOH tissue preparation of sputum or skin, which may demonstrate the large, broad, nonseptate hyphae characteristic of phycomycetes (14). Debridement of necrotic tissue, intravenous administration of amphotericin B, correction of acid-base imbalance, and control of hyperglycemia are the necessary components of treatment (17). The more invasive forms of disease frequently are fatal.
Bacterial Infections
Although patients with well-controlled diabetes do not have increased susceptibility to most bacterial infections, evidence suggests that patients with poorly controlled diabetes may develop more frequent or severe bacterial infections than experienced by the rest of the population (18). Several authors have described abnormalities in leukocyte function associated with diabetes, including diminished chemotaxis (19), depressed phagocytosis (20,21), decreased bacteriocidal activity, (22,23,24,25), decreased leukocyte migration (25), and an altered early granulocyte phase of the local cellular inflammatory response (26).
Bacterial infections of the lower extremities are a particular risk for patients with diabetes who have vascular disease or neuropathy (18). Careful foot care and early treatment of dermatophytosis are essential in minimizing potential bacterial portals of entry. A recent review of predisposing factors in hospitalized patients with cellulitis identified diabetes, prior history of cellulitis, edema, dry feet, and evidence of tinea pedis as the most important risk factors (27) (also see Chapter 66).
MALIGNANT EXTERNAL OTITIS
Malignant external otitis is a severe necrotizing bacterial infection that occurs almost exclusively in patients with diabetes (28). Beginning in the external auditory canal, the infection invades local soft tissues and ultimately leads to osteomyelitis of the temporal bone, occasionally progressing to fatal meningitis. Pseudomonas aeruginosa is the causative agent in virtually all cases. Symptoms can include unremitting ear pain, purulent otic discharge, and cranial nerve palsies. A tender ear and mastoid area with granulation tissue or polyps in the external auditory canal are typical physical findings. More than 20% of patients have bilateral disease (29). Extended therapy with antipseudomonal agents is necessary, and surgical debridement may also be required.
ERYTHRASMA
Erythrasma is a superficial infection usually affecting intertriginous areas of the skin that is caused by Corynebacterium minutissimum. Carriage of fluorescent diphtheroids on clinically normal skin is more common in patients with poorly controlled diabetes than in persons without diabetes. Obesity may be an additional contributing factor, especially in those with adult-onset diabetes (30).
Reddish brown, slightly scaly patches typically occur in the intertriginous areas such as the axillae, groin, and web spaces and can be pruritic or asymptomatic. Erythrasma is commonly limited to the toe webs, manifesting with white discoloration and maceration. Frequently, the diagnosis is missed, and not infrequently toe-web involvement is misdiagnosed as tinea pedis. The characteristic coral red fluorescence using a Wood light is diagnostic. The red glow seen is from a porphyrinlike substance produced by the bacteria (31). Gram stain or culture of affected skin scrapings shows gram-positive rods and threadlike filaments traversing the stratum corneum (32). Topical imidazole antifungal agents, topical antibiotics, and systemic erythromycin or tetracycline each provide effective treatment for the disorder. Fusidic acid in a topical preparation is a recent addition to the therapeutic armamentarium (33).
NEUROPATHIC AND ISCHEMIC DIABETIC SKIN DISEASES
Diabetic Polyneuropathy
Diabetic neuropathy is usually bilateral and more severe in the lower extremities than in the upper extremities (34). Dry shiny skin and ulceration on pressure points, particularly on structurally deformed feet, are common. Skin hypoesthesia is one of the most important signs of polyneuritis (34). Lack of thermal sensitivity may lead to burns, calluses, and subsequent ulceration (35). Abnormalities of motor function are much less obvious than sensory defects. Paralysis of the intrinsic muscles of the feet, the most common motor alteration, results in atrophy of the interosseus muscles, which is demonstrated by the “fan sign”—the inability to separate the toes. Such alteration in motor alteration causes the foot to adopt unusual positions, resulting in claw toe or hammer toe deformities or the Charcot joint. In the latter, the tarsal and metatarsal bones are gradually destroyed and joint spaces are obliterated (34,36). As autonomic neuropathy develops and progresses, sweat glands become inactivated. Patients with diabetes may exhibit a compensatory hyperhidrosis of the trunk or face in response to anhidrosis of the lower body (37). Dry and thickened skin of the anhidrotic diabetic foot is prone to fissures, which may serve as portals of entry for bacteria and fungi. Unperceived chronic pressure to the toenails results in thickening, abnormal curvature, and hypertrophy of the nail plate. Ingrown toenails may lead to secondary bacterial infection (12).
Peripheral Vascular Disease
Peripheral vascular disease (PVD) in patients with diabetes can be diffuse and early in onset. The predilection for atherosclerosis and microangiopathy plays a role in the vessel occlusion that occurs in diabetes (38). Some authors consider that microangiopathy plays a part in diabetic vascular disease, while others
disagree (38,39). The skin of the lower extremities in patients with diabetes who have PVD is thin, smooth, cold, and often mottled in the dependent position (40,41). Hair is either sparse or absent. Other signs of PVD include pallor or cyanosis with elevation, dependent rubor, and delayed capillary refill.
disagree (38,39). The skin of the lower extremities in patients with diabetes who have PVD is thin, smooth, cold, and often mottled in the dependent position (40,41). Hair is either sparse or absent. Other signs of PVD include pallor or cyanosis with elevation, dependent rubor, and delayed capillary refill.
Diabetic Ulcers
Among the 16 million patients with diabetes in the United States, 1,200 amputations are performed every week (42). This shocking figure indicates the grave importance of identification and treatment of the myriad of causes of cutaneous ulceration in patients with diabetes.
Neuropathic ulcers develop most frequently in areas of high pressure and repeated trauma such as the toes, heels, and metatarsal heads (35) (Fig. 62.1; see also color plate). The patient may present with pain, paresthesia, or anesthesia of the legs and feet. Only occasionally is the ulcer completely asymptomatic in presentation (36).
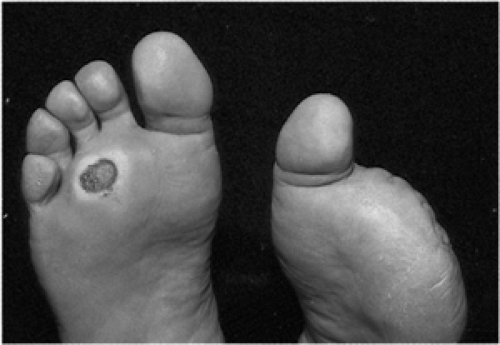 Figure 62.1. (See also color plate.) Acute neuropathic ulcer in a 34-year-old man with diabetes who has had previous amputations for osteomyelitis secondary to ulceration. |
Venous ulcers tend to occur on the medial malleoli in association with superficial varicosities and yellow-brown discoloration of the skin and are seldom painful. Rest and elevation of the leg ease venous ulcers by controlling edema and reducing venous hypertension.
Arterial ulcers are typically painful (except when accompanied by neuropathy) and occur more distally, on the tips of the toes and on the heel (41). Ulcers of arterial origin may respond to control of hypertension and diabetes as well as to increased exercise, which promotes collateral circulation. Cessation of smoking can be critical. Aggressive treatment of infection, administration of antiplatelet agents or anticoagulants, and surgical revascularization are important treatment modalities in severe cases. Advances in wound healing offer new effective treatments. A recent series of ten patients with diabetic ulcers demonstrated complete healing in seven of ten with the use of sharp debridement and application of a bilayered human-skin equivalent (a construct of live human keratinocytes on a type I collagen dermal matrix, both derived from human fetal foreskin) (42).
A recent study examined the proliferative capacity and ultramicroscopic appearance of fibroblasts taken from leg ulcers of patients with diabetes as compared with fibroblasts taken from nonulcerated skin of patients with diabetes and from control subjects without diabetes (43). Fibroblasts from cutaneous ulcers in individuals with diabetes were found to proliferate at a significantly slower rate than fibroblasts from either control group. Electron microscopic evaluation of fibroblasts from lesional and nonlesional skin in patients with diabetes revealed a large dilated endoplasmic reticulum, a lack of microtubular structures, and multiple lamellar and vesicular bodies. These abnormalities were not seen in the fibroblasts from the control subjects.
DISORDERS OF COLLAGEN
Necrobiosis Lipoidica
Necrobiosis lipoidica is an unusual skin disorder that is strongly associated with diabetes mellitus. The entity was first described in 1929 (44) and was named necrobiosis lipoidica diabeticorum (NLD) in 1932 (45). Because of the significant minority of cases of this condition that are associated with diabetes, most investigators now choose to call this condition simply necrobiosis lipoidica (NL).
Typical lesions of NL occur on the pretibial skin as irregular ovoid plaques with a violaceous indurated periphery and a yellow central atrophic area (Figs. 62.2 and 62.3; see also color plate). Overlying superficial telangiectasia and scattered hyperkeratotic plugs often are noted (46). The lesions can start as small firm reddish brown papules, which slowly enlarge. They are usually multiple and bilateral, with ulceration seen in approximately 35%. Ulcers often are secondary to minor trauma or are iatrogenically induced by intralesional injection of corticosteroids. It is curious that the ulcers only rarely lead to infection. Patients may complain of pruritus, dysesthesia, or pain at the site of lesions. More frequently, however, the lesions of NL are asymptomatic, and it is the cosmetic disability that is of greatest concern to the patient. Although most commonly found on the lower legs (85% of cases involve only the legs), other locations include the hands, fingers, forearms, face, and scalp (47). Lesions in these locations are often annular, erythematous, or brown and can coalesce to form larger serpiginous plaques, which demonstrate little or no atrophy.
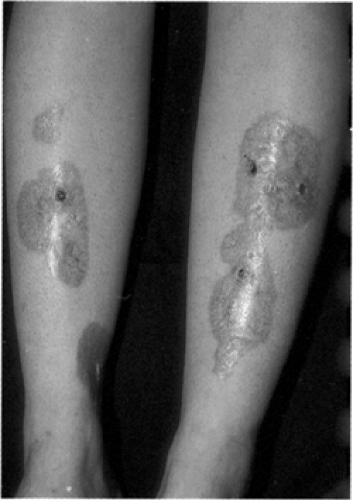 Figure 62.2. (See also color plate.) Necrobiosis lipoidica in a 24-year-old patient with type 1 diabetes. |
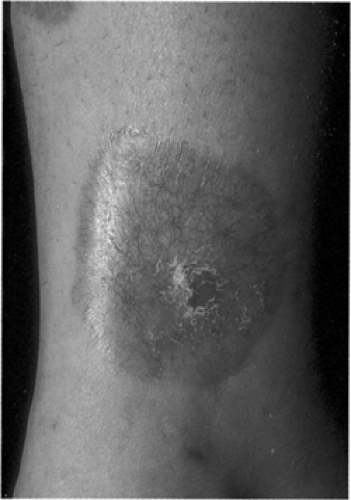 Figure 62.3. (See also color plate.) Necrobiosis lipoidica in a 24-year-old patient with type 1 diabetes. |
Muller and Winkelmann’s classic papers in 1966 (47,48) demonstrate the close association between NL and diabetes. Of their 171 patients with NL, 65% had diabetes. Of the remainder available for study, 42% (8 of 19) demonstrated abnormal glucose tolerance with abnormal oral glucose tolerance tests or cortisone glucose tolerance tests. Among those with normal glucose tolerance, 55% had family histories of glucose intolerance. Despite the high prevalence of diabetes in patients with NL, it is relatively uncommon; the reported prevalence being 3 per
1,000 patients with diabetes. The female-to-male ratio is 3:1, with an average age of onset of 30 years in patients with diabetes. Fifty percent of patients with NL demonstrate other diabetes-related end-organ damage (47).
1,000 patients with diabetes. The female-to-male ratio is 3:1, with an average age of onset of 30 years in patients with diabetes. Fifty percent of patients with NL demonstrate other diabetes-related end-organ damage (47).
Despite extensive investigation, the etiology of NL remains largely unknown. Evidence suggests that NL and glycemic control are not related (49) and that genetic factors do not play a large role (50). Vascular etiologies have been entertained; however, vascular changes are absent in one third of biopsy samples. A link with diabetic microangiopathy has been suggested, yet the caliber of the affected vessels in NL is usually larger than the caliber of vessels affected in diabetes. The abnormal collagen found in NL has invited speculation on a direct etiologic relationship of NL to the collagen itself, as a result of accelerated aging of collagen in diabetes, (51) abnormal collagen cross-linking, or overhydrated collagen produced in response to osmotic effects generated by the end products from the aldose reductase (polyol) pathway (52). Abnormal leukocyte mobility has also been implicated (53). Immune-complex disease has been considered as a cause of NL. Ullman and Dahl (54) reported deposition of C3, fibrinogen, and immunoglobulin around dermal blood vessels in nine of 12 patients with NL, but subsequent reports only partially confirm these results. A review of NL addresses these theories in greater detail (55).
Clinically, early NL can be difficult to distinguish from granuloma annulare. As the lesions enlarge, however, NL becomes more distinct, with the epidermal change, atrophy, and yellow color seen developing. Other conditions included in the differential diagnosis for NL are listed in Table 62.2.
TABLE 62.2. Clinical Differential Diagnosis for Necrobiosis Lipoidica | ||||||
|---|---|---|---|---|---|---|
|
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








