Cost and Cost Effectiveness Considerations
Bruce E. Hillner
Cancer care costs have been growing faster than the overall rates of health care, especially over the last 20 years. The combination of the 2008 worldwide recession and the recent U.S. debate over healthcare reform has focused attention on the rising costs of new cancer therapies. U.S. costs for breast cancer care were estimated to exceed $16 billion in 2010 (1) and are the highest overall of any cancer type.
The spiraling costs of new cancer therapies are increasingly recognized by cancer professional organizations such as ASCO (2)and ACS, but what to do about them is another hurdle without consensus. Since 2008, the greatest source of discussion focusing on breast cancer costs is likely to have been around the FDA. decision to revoke the approval of bevacizumab. Those discussions were usually a mix of concern about the bevacizumab’s costs (∽$88,000 per year of treatment) and/or an implicit cost-effectiveness analysis (“it’s not worth it”) given the uncertainties of any overall benefit (3).
In this chapter, I will review the literature published since 2008 that addresses the costs and cost-effectiveness of breast cancer assessment and treatments for the initial care, adjuvant therapies, and advanced/recurrent disease as well as highlight where these reports by inference give guidance to quality of care indicators.
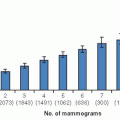
For this review, I searched Medline for publications from January 2008 to late 2012 with the following search characteristics: breast neoplasm/economics, an abstract (except for editorials), English languages, and excluded reports addressing prevention, strategies for high-risk women, and screening (principally mammography). The individual report abstracts were reviewed for relevance to U.S. clinical practice, thereby excluding many reports from limited resource countries. Table 91-1 lists the categories used to structure this review.
COST ANALYSIS VOCABULARY
Before jumping into the review, because most readers of this book are not knowledgeable about the methodology used in these reports, the following brief section discusses the key terms as a platform for the chapter.
There are five common categories of cost studies used in assessing healthcare. The first category, cost of illness studies, are studies that describe the financial burden or consequences of illness but do not make any comparison to other conditions. A cost comparison report explicitly addresses the relative costs of two (or more) approaches or populations. A comparison report will specify what cost elements and time frame are being compared and defers to the reader to make any inferences.
Cost minimization reports either explicitly or implicitly assume no difference in the benefit (or harm) incurred between two different approaches. By comparing the costs incurred, the lowest cost strategy will be, by definition, preferred as it has the lowest cost. The recent American Board of Internal Medicine Foundation Choosing Wisely® (www.choosing-wisely.org) campaign fits the definition of cost minimization. Each specialty was asked to identify five tests or procedures commonly used whose common use and clinical value are not supported by the literature. The ASCO list for cancer included the following breast cancer topic: avoid advanced imaging in staging early breast cancer as well as in the surveillance of asymptomatic individuals’ post-initial treatment (4).
Cost-effectiveness analyses (CEA) and cost-utility analyses (CUA) estimate the additional cost per unit of benefit associated with the use of a given intervention as compared to the alternative. The intervention of interest can be of any type: prevention, screening, diagnosis, treatment, or symptom control. CEAs consider either a specific health effect
(e.g., disease free survival) without assigning a specific value to it or use years of life. A CUA is a specific type of cost-effectiveness analysis that combines mortality and morbidity into a single multi-dimensional measure called a quality adjusted life year (QALY).
(e.g., disease free survival) without assigning a specific value to it or use years of life. A CUA is a specific type of cost-effectiveness analysis that combines mortality and morbidity into a single multi-dimensional measure called a quality adjusted life year (QALY).
TABLE 91-1 Review Categories of Costs of Breast Cancer Therapy Literature | ||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||
The result of a CEA is usually expressed as a ratio of the difference in cost between the two competing strategies divided by the difference in benefit. A CEA is of most interest when the new intervention is more effective and more costly than the reasonable alternative strategy. The lower the ratio of the additional cost of the new intervention to gain the additional benefit, the more appealing to society as a whole it is to add the intervention to current medical approaches.
For the interested reader, several sources of standards and checklists for best practice reporting of CEA studies are available (5, 6 and 7). In this chapter, three recurring concerns will be pointed out: —i) In cost minimization studies, was it appropriate to assume the two strategies are equally effective? ii) Were the data used to guide the efficacy estimates accurate, representative, and credible? and iii) Was the default strategy representative of current patterns of care in a particular location and during those years?
LIFETIME COSTS
Accurate, current estimates of the cost of illness incurred for breast cancer treatment are hard to identify. In the United States data sources have shifted from single centers to claims analyses have been predominantly of three different groups—patients insured by Medicaid (poor and relatively young), patients insured by self-insured employers (wealthier, young) and the elderly (usually with a linkage of Medicare claims with registry data from SEER).
TABLE 91-2 NCI 2010 Estimates of U.S. Costs for Breast Cancer Care | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||
Mariotto and colleagues from the NCI (1) have recently updated their national projection of costs for 2010 and projected to 2020 associated with care for 15 different cancer types including breast cancer (1). Table 91-2 shows their projections for breast cancer. The higher costs for women younger than 65 years were based on studies from integrated managed care organizations (in the 1990s) that showed costs for younger patients (of all cancer types) were 20% higher in the initial year and 50% higher in the last year of life than for Medicare beneficiaries. In my opinion, especially for younger women, the initial management costs are likely a substantial under-estimation as the type and frequency of more-expensive adjuvant chemotherapies are more common in younger women. In either case, their first year costs (local surgical therapy, axillary staging/dissection, and adjuvant therapies) were estimated at $25,000 to $30,000, continuing care after the first year at $2,200, and for those developing recurrent disease, the average costs in the last 12 months of life were $63,000 to $94,000.
The challenges in doing cost-of-illness studies in breast cancer were addressed in two excellent reviews (8, 9). The review (n = 29) reports of U.S. costs of illness studies published between 1984 and 2007 covered a broad range ($20,000 to $100,000 per lifetime) (8), were predominantly from an insurer/third party payer perspective, predominantly of early stage disease, and did not include many, or most, of the expensive innovations of the last decade (e.g., IMRT, CSFs, 3rd generation chemotherapy). These limitations, or under-estimations, from either inaccurate or outdated sources have an often important hidden impact when added to most economic evaluations because these need to project the future. Fortunately, there have been recent reports of new, large, detailed analyses of U.S. metastatic disease costs that will be discussed later.
DIAGNOSTIC PROCEDURES
The increased use of stereotaxic core biopsy has reduced the rates of open surgical biopsy throughout the world. Between 2000 and 2005, in the Netherlands, surgical biopsies were reduced from 54% to 10% of the total diagnostic costs associated with screen detected breast cancers (10). A Swedish audit of fine-needle aspiration (FNA) cytology compared to core needle biopsy (CNB) found a difference; FNA had lower rates of definitive diagnosis, more additional needle biopsy, or surgical biopsy prior to definitive surgery (11). An Austrian budget impact audit of CNB compared to open surgical biopsy found a 30% total cost reduction per lesion and obviated the need for open surgical procedure in 60% of women (12).
For these non-palpable lesions, the current standard of wire localization (WL) to guide surgical excision appears to be evolving. WL is unfortunately associated with positive margins due to migration. Radioactive seed localization that permits equal or superior localization and is associated with lower re-excision may be another major cost-reduction tool if the results from experienced centers using it become generalizable (13). To date, no cost consequence studies of radioactive seeds were identified.
Ongoing uncertainty persists concerning the role of breast MRI in the initial staging. The cost of a dedicated breast MRI is high and the downstream costs of pursuing false-positives are well known. Changes in primary surgical treatment were noted in 8% to 33% of women in a recent meta-analysis, yet without randomized, prospective assessments the benefit value cannot be determined (14).
STAGING THE AXILLA
The rapid acceptance of the use of axillary sentinel lymph node detection to guide managing axillary lymphadenectomy was reinforced by several cost-minimization reports from around the world. The French reported a prospective national, multi-institutional, cost comparative analysis found an ˜12% reduction in costs (15) while a large, single center, Italian series found cost savings of 10% to 26%. The potential role of axillary ultrasound in obviating the need for SLN for patients with bulky enlarged nodes has intuitive appeal but prospective, comparative studies are needed (see Chapter 38) (16).
RADIATION THERAPY
Alternatives to the traditional 6-week course of whole breast radiation following breast-conserving surgery have obvious cost of care implications with uncertain relative cost-effectiveness. The first alternative is more expensive with uncertain reductions in local control. Intensitymodulated radiation therapy (IMRT) has been rapidly adopted in the United States, increasing from <1% of women in 2001 to 11.2% in 2005 among the U.S. elderly, with a doubling of costs compared to whole breast radiation (Medicare payments $15,230 for IMRT vs. $7,180 for whole breast) (17).
Shorter courses using hypo-fractionation, accelerated partial breast, or targeted intra-operative radiotherapy are all less costly. Hypo-fractionated whole-breast radiotherapy (WBR) (over 3 weeks) in Australia was estimated to reduce total treatment costs by 24% (18). The major uncertainty is whether these shorter courses lead to inferior outcomes in terms of local recurrences. Another factor is whether patient selection is appropriate. The economic consequences of accelerated partial-breast irradiation (PBI) as an alternative to WBR or IMRT are still uncertain, especially in light of a recent report stating that about one-half of Medicare patients receiving PBI did not receive the level of care recommended by the American Society for Radiation Oncology consensus guidelines (19).
PROGNOSTIC AND PREDICTIVE FACTORS
The challenges associated with obtaining Medicare coverage for innovative diagnostics (e.g., molecular imaging, genomic profiling, circulating tumor cells) are well illustrated by the many year battle prior to recent Medicare coverage of Oncotype Dx and Mammaprint. Fifteen reports were found addressing the cost consequences of genomic predictions. As reviewed in detail in Chapter 48, the current patient subgroups for which these tests should be used are cancer that are ER+ with axillary node negative and possibly one-three node positive women eligible for chemotherapy. Pricing in 2012 for Oncotype Dx and Mammaprint are almost identical at $4,200.
The economic consequences have been assessed by hypothetical decision models using Markov chains to estimate projected costs from the societal perspective in Canada, Europe, Israel, Japan, and the United States Hornberger et al compared and summarized these economic assessments in a 2012 review (20). In sum, each gene profiler is superior to authoritative guidelines, Adjuvant!, or current practice. We are unaware of any direct comparisons of Oncotype Dx to Mammaprint. The incremental cost-effectiveness ratios, or cost-savings results, varied with the pre-test recurrence risk (node-negative, positive, or both), the costs of chemotherapy used (especially dose-dense), time horizon, and comparator (Adjuvant! was the second best). For example, in a U.S. managed care setting, Oncotype showed costs savings and the incremental costs were <$30,000/QALY. These assays are the first of their kind and suggest that the best care is less care.
ADJUVANT THERAPY—ENDOCRINE THERAPY
Several reports addressed the question of adherence to hormonal aromatase inhibitors (AI) in U.S. women using administrative pharmacy benefit claims. Neuget et al. assessed mail order prescription records from January 1, 2007, to December 31, 2008 for women older than 50 years (21). They used two distinct, useful definitions—“non-persistence” was defined as a prescription supply gap of more than 45 days without subsequent refill and “non-adherence” was a medication possession ratio of less than 80% of eligible days. This study of 8,100 women age 50 to 64 years and 14,000 age >65 found non-persistence in 21% and 24% and nonadherence in 11% and 9% by age group, respectively. As expected, higher co-payment amounts were associated with both types of gaps. A similar type of analysis that assessed the first year following an initial claim for an AI found non-adherence (using the same definition as Neuget) was 23% over the year in 13,600 commercially insured women. Co-payments >$30 per prescription, younger age, and higher co-morbidity was associated with non-adherence (22). In a one-state Medicaid cohort (n = 1,538) adherence was even lower (58% at one year) and more troubling was that hormonal therapy of any kind was not promptly initiated within one-year (68%) in an analysis of linked pharmacy claims and state cancer registry (23).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree