Introduction
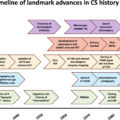
Cushing’s syndrome (CS) was first discovered as a neurosurgical condition, and its history is linked to the birth of neurosurgery as a surgical specialty. In 1912, Harvey Cushing documented the first case of hypercortisolism [ , ]. While the patient’s symptoms were originally thought to be a result of a polyglandular disease. However, an incidental craniotomy due to elevated intracranial pressure demonstrated that the pituitary plays an integral role in CS [ ]. Twenty years later, Cushing published 12 cases of “pituitary basophilism,” discussing the presence of “the pituitary lesion (the basophil adenoma)” and how it is likely the etiology of hypercortisolism in these patients [ , ]. Nevertheless, physicians often debated about the relative importance of the pituitary and the adrenal glands to hypercortisolism until 1950, when it was established that cushingoid symptoms could be caused both by the adrenal glands and the pituitary. This was also when ectopic causes of hypercortisolism were identified [ ].
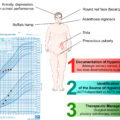
Adrenocorticotrophic hormone (ACTH)–secreting adenomas of the pituitary gland, now known as Cushing’s disease (alternatively as Cushing’s disease [CD]; Fig. 13.1 ) is the predominant cause (80%–85% of cases) of hypercortisolism [ ]. While this disease affects females and males in a 1:1 ratio in children, CD is more prevalent in females than males in adulthood (5:1 vs. 3:1) [ ]. Prevalence of CD is 39.1 per 1,000,000 with an incidence rate of 1.2–2.4 per 1,000,000 per annum [ , ]. CD patients, as those with other causes of CS, present as adults and children, with symptoms including obesity, hypertension, facial plethora, diabetes, dorsal fat pads, moon facies, amenorrhea, and cognitive/psychiatric deficits [ , , ].
Trends in PubMed publications with “Cushing’s disease” and “Cushing’s disease”.
Diagnosis and preoperative considerations
To detect corticotroph-derived pituitary adenomas, biochemical and imaging studies have significantly improved the time to diagnosis, thereby improving surgical outcomes. After ruling out sources of exogenous glucocorticoid exposure, 24-hour urine-free cortisol, low-dose dexamethasone testing (1 mg overnight or 2 mg over 48 hours), and late-night salivary cortisol laboratory tests confirm CS via endogenous cortisol production [ , ]. If elevated, plasma adrenocorticotropic hormone (ACTH) levels help establish ACTH dependency. If plasma ACTH levels are suppressed (less than 10 pg/mL), it is consistent with ACTH-independent causes of CS, including adrenal hyperplasia, adenomas, and carcinomas [ , , ]. Conversely, if plasma ACTH levels are inappropriately elevated, a presumptive diagnosis of ACTH-dependent CS is made. Ovine corticotropin-releasing hormone (oCRH) stimulation [ ] or desmopressin (DDAVP) stimulation test [ ] and high-dose (8 mg) dexamethasone suppression test are used to confirm phenotypic similarity of the ACTH source to pituitary corticotrophs [ , , ]. However, there is overlap between the pituitary and ectopic tumor responses to stimulation and suppression, resulting in false positives and negatives [ , ].
Preoperative imaging
If a putative pituitary source of ACTH is determined by the aforementioned diagnostic regime, the next task is localization of the corticotroph adenoma within the pituitary gland. Detecting the causal adenomas in CD with preoperative imaging is of utmost importance to patient outcomes. With adenomas detected in preoperative imaging, the surgical remission rates improve from 50% [ , , ] to about 90% of higher [ , , ]. This task is complicated by the small size (90% are smaller than 1 cm across), and the high incidence (15%–20%) of incidental pituitary adenomas in the general population [ ]. Since its first use to detect pituitary adenomas [ ], magnetic resonance imaging (MRI) has become a gold standard for localizing corticotroph adenomas in CD [ ]. Current options for MR imaging of pituitary adenomas include dynamic contrast imaging of the pituitary gland [ ] that results in rapid, lower-resolution images (approximately 2–3 mm thick slices) in rapid sequence through the thickness of the gland. This allows for detection of pituitary adenomas as hypo-enhancing regions due to slower and lower gadolinium contrast enhancement [ ]. An alternative strategy comprises high-resolution (∼1 mm thickness slices) obtained very early after gadolinium contrast injection. These sequences comprise either 3D gradient recall (GRE) [ , ] or volume interpolated GRE-VIBE [ ] MRI sequences. More recently, high strength MRIs up to 7 T have been used to detect pituitary adenomas causing CD; however, the utility and increase in detection accuracy remains limited [ ]. In addition to detection, contrast-enhanced MRI provides additional information about size, location, intratumoral heterogeneity, and cavernous sinus invasion [ ] by the adenoma. All of the aforementioned contrasted MRI sequences are similar to T1-weighed sequences. Adding T2-weighted sequences can provide the clinicians with additional information including cystic changes, tumor-normal interface, and cavernous sinus invasion [ ]. However, despite best available imaging, current clinical series still report up to 40% rate of failure in detecting CD adenomas [ , ].
MRI-negative Cushing’s disease
MRIs depend on the differential contrast enhancement (between the normal gland and the adenoma) to highlight the tumor, a property designated as contrast to noise ratio (CNR) [ ]. Dynamic and GRE MRI sequences attempt to increase the CNR by using the differential distribution of gadolinium contrast between the normal pituitary and adenomas. In CD, the small size of adenomas adds another layer of difficulty that is tackled by increasing the spatial resolution of MRI images. Image resolution may be improved by either using a stronger MRI magnet [ , ], or by increasing the time spent in the scanner by the patient [ ]. However, even with the best optimization, some microadenomas may remain MRI invisible. In this context, the size of the microadenoma may not be the only determinant of MRI visibility. There is a significant overlap in the sizes of the microadenomas that were detected, versus the MRI invisible adenomas [ ] ( Fig. 13.2 ). These findings suggest that strategies that depend solely on CNR may not resolve all currently MRI invisible adenomas, and that complementary strategies for localization of CD adenomas are needed.
Relative sizes of pituitary microadenomas that were MRI negative versus the ones that were detected on MRI imaging.
From Jagannathan J, et al. Outcome of using the histological pseudocapsule as a surgical capsule in Cushing disease. J Neurosurg 2009;111:531–539.
Inferior petrosal sinus sampling
Bilateral inferior petrosal sinus sampling (BIPSS) is a well-established and recommended [ , ] technique. BIPSS depends on the detection of higher levels of ACTH in the petrosal sinuses in close proximity, and downstream of the pituitary adenoma. As originally devised [ , ], venous catheters are inserted and parked in the bilateral inferior petrosal sinuses. Serial blood sampling then helps identify trends in ACTH levels following stimulation with secretagogues—ovine CRH [ ] or more recently, DDAVP [ ]. BIPSS is recommended for patients with biochemical testing suggesting ACTH-dependent hypercortisolism, and either a negative MRI or MRI demonstrating adenomas smaller than 6 mm [ ]. BIPSS, in the current usage, has up to 95% diagnostic accuracy [ ] in identifying the pituitary as the source of the ACTH (central to peripheral gradient for ACTH). However, the value of BIPSS in localizing the adenoma within the pituitary gland has been debated over the decades [ , ]. Although a lateralization accuracy is up to 69% with BIPSS, this accuracy is not associated with the remission rates from CD following surgery [ ]. Often, a nondiagnostic BIPSS result is due to unequal or inadequate venous drainage on one (or both) inferior petrosal sinuses. Strategies to overcome such drainage deficits include on-the-fly modification of sampling sites using angiography [ ], or by using prolactin as an indicator for successful catheterization of the inferior petrosal sinus [ ]. Prolactin levels indicate whether the inferior petrosal sinus has been adequately catheterized, and the ratio of prolactin is used to normalize the ACTH levels. Such a strategy can help rule out the possibility of ectopic Cushing’s syndrome with negative MRI effectively [ ]. Most case series report that BIPSS is a safe procedure with a very low rate of complications; however, the complications can be serious including brainstem venous infarction, leading to permanent neurological deficits [ , , ].
PET imaging
Corticotropinomas can be detected by radioactive 18 F tagged fluorodeoxy glucose ( 18 F-FDG) positron emission tomography (PET) in 50% of patients [ ]. Prior work by us suggests that secretagogue stimulation with corticotropin-releasing hormone (CRH) leads to increased 18 F-FDG uptake, and improved PET detection of pituitary microadenomas, including a portion of tumors that are invisible on MRI [ ]. 18 F-FDG-PET imaging could provide a complementary imaging modality to improve visualization of ACTH-secreting microadenomas and surgical planning. Accurate localization of pituitary adenomas in patients with Cushing’s disease is strongly correlated with improved remission rates and fewer adverse events during after surgery [ , ]. Modern imaging techniques, including dynamic and volumetric MRI, cannot detect ACTH-secreting adenomas in 20%–50% of these patients [ , , , , , ]. Since adenomas in patients with negative MRI findings are identified less frequently during surgery, these patients undergo more extensive exploration of the normal pituitary gland with more invasive resection techniques such as hemihypophysectomy or subtotal hypophysectomy [ , , , , ]. To improve detection rates of these small tumors, radioactive 18 F-FDG-PET has been used to demonstrate increased glucose uptake in nonfunctioning and hormone-secreting tumors with minimal uptake in the normal pituitary gland [ ]. This helps to differentiate small tumors from normal surrounding pituitary tissue. Uptake within the sella is specific but not sensitive for pituitary [ , ]. In large observational cohorts of whole-body 18 F-FDG-PET studies, incidental sellar 18 F-FDG uptake was found in less than 1% of cases and was highly specific for the presence of pituitary adenomas [ ]. We have previously demonstrated that 18 F-FDG PET imaging can detect tumors as small as 3 mm in size as well as 40% of tumors that are not detected on MRI. Using in vitro and ex vivo modeling, we have also demonstrated that secretagogue stimulation (with oCRH) leads to delayed glucose uptake (2–4 hours) through increased glucose transporter 1 (GLUT1) transcription and membrane translocation in human ACTH-secreting adenomas [ , , ]. Secretagogue stimulation leads to increased 18 F-FDG uptake and increased rate of tumor detection in other neuroendocrine adenomas [ , ]. We applied this paradigm of secretagogue stimulated 18 F-FDG-PET study to pituitary adenomas secreting ACTH. In patients who underwent PET-imaging after coadministration of oCRH and 18 F-FDG, we demonstrated a significant increase in the SUVmax in ACTH-secreting tumors and no change in 18 F-FDG uptake characteristics within normal pituitary gland. These findings suggest a selective effect of secretagogue stimulation on adenoma over normal glandular tissue. Combined with a larger effect of SUVmean (over SUVmax) on adenoma detection rate, this selective effect suggests that secretagogue stimulation underlies the increased rate of detection (44%–55%) of ACTH-secreting adenomas on PET images by blinded neuroradiologists. A salient finding of our prior investigation was that two of the five (40%) tumors undetected on MRI were subsequently discovered on PET imaging. In one of these instances, adenoma detection occurred only after oCRH stimulation, suggesting a role for FDG-PET imaging following secretagogue stimulation in MRI-negative cases. We have now begun a clinical trial utilizing DDAVP as a secretagogue prior to 18 F-FDG PET imaging, since oCRH is no longer available in North America. This translates into a clear clinical role for secretagogue stimulation on PET detection of MRI invisible tumors. While the majority of prior papers have investigated the use of 18 F-FDG in PET imaging of pituitary adenomas, there are several other radioligands that are being developed.
Somatostatin receptor and amino acid PET
DOTATATE and DOTATOC are ligands tagged with 68 Gallium ( 68 Ga) which bind to subtypes of the somatostatin receptor (SSTR). These radioligands are commonly used to detect and stage neuroendocrine tumors, particularly those in the lungs, gastrointestinal tract, and pancreas [ ]. There are a number of SSTRs within the pituitary gland, with differing levels of expression in healthy tissue and disease states (including CD), thereby providing a potential target for imaging MR-negative corticotropinomas [ ].
Methionine is an essential amino acid, and its cellular uptake can be measured through a 11 C tag ( 11 C-MET). Koulouri et al. had promising MET-PET results by identifying de novo and recurrent adenomas in 15 out of 18 patients with Cushing’s disease (83%), four of which were negative on SPGR MRI [ ]. Regarding sensitivity and specificity, more recent studies by Feng et al. and Berkmann et al. demonstrated MET-PET to have sensitivities ranging from 89% to 100% and specificities ranging from 89% to 100% [ , ]. Despite the promise of MET-PET for CD detection, it is important to acknowledge normal tissue also demonstrates physiological uptake of 11 C-MET and that anatomical variations, vascular disorders, and inflammation can affect the image interpretation, resulting in potential false positives and negatives [ ].
Among the various radiotracers tested, 11 C-MET has a higher detection rate compared with 18 F-FDG (87% vs. 49%), while 18 F-fluoroethyl- l -tyrosine, 68 Ga-DOTA-TATE, and 68 Ga-DOTA-CRH have up to 100% detection rate; however, these three findings were based on single studies [ ]. In an initial study, use of 68 Ga-DOTA-CRH PET/CT resulted in detection of all 24 cases of CD, of which four were MRI negative, while also delineating CS secondary to ectopic ACTH secretion [ ].
In using a ratio of FDG-PET SUVmax over 68 Ga-DOTA-TATE SUVmax, Wang and colleagues developed an imaging methodology that resulted in 89% sensitivity and 96% specificity [ ]. There is promise for utilizing PET molecular imaging to aid in both diagnosis and localization of Cushing’s adenomas.
Surgical techniques
The first record of successful pituitary surgery was in 1893 under Sir Victor Horsley and Richard Caton using a transcranial approach [ , ]. However, due to increased mortality, Dr. Schloffer’s transsphenoidal approach, founded in 1907, gained popularity, giving rise to infranasal and gingival approaches [ ]. Harvey Cushing developed the sublabial transnasal route, by creating a frontal flap and elevating the frontal lobe [ ]. While this fell out of favor, transcranial routes, including through the cavernous sinus and the suprasellar space, rose to prominence, until the transsphenoidal approach made a resurgence under Drs. Gerard Guiot and Jules Hardy using intraoperative radiofluoroscopy to enhance visualization of the nasal passage [ ]. Currently, there are a number of approaches being used by surgeons when approaching pituitary gland pathology ( Fig. 13.3 ).
Surgical approaches for Cushing’s disease. A. Although, theoretically, transcranial approaches are available for pituitary masses, the transsphenoidal approach is now the standard of care for patients needing surgery for CD. B. Microscopic sublabial approach, originally devised by Harvey Cushing, remains the approach of choice at our institution for microadenomas underlying CD. C. Microscopic endonasal approach utilizes the advantages of B, but, is limited by the narrower surgical field afforded with a mononarial approach. D and E. Currently, the most popular forms of TSS, endonasal endoscopics surgeries can be performed via a single nostril (D) or with both nostrils (E).
Microscopic approach
Dr. Hardy reintroduced the transsphenoidal sinus approach (TSS) in the 1970s, he concurrently introduced the idea of microsurgery, and was the first person to use an operative microscope during pituitary surgery [ , , ]. Since its usage, microsurgery has evolved greatly since Cushing’s efforts, including the development of a mononarial and sublabial approach [ ]. Mason et al. [ ] and Kouri et al. [ ] describe a modified transsphenoidal approach that allows for wide bone exposure of the anterior surface of the of sella, removal of the posterior planum sphenoidale, and access to the parasellar and clival regions. This transsphenoidal approach was further expanded by Couldwell and Weiss by using an asymmetric retractor with alternating positions for greater lateral visualization [ ]. At the NIH Clinical Center, Dr. Edward H. Oldfield adapted the sublabial approach to also include sublabial, transseptal, and bilateral approach to the sella, allowing for improved dissection of pituitary microadenomas [ , , ]. This is the surgical approach that we still use for all pituitary microadenoma surgeries for CD.
For the sublabial approach, the first step is to create a horizontal sublabial incision, about 1 cm above the gingivobuccal sulcus, and extending between the alae nasalis through the premaxillary periosteum. The mucosa lining the left pyriform aperture and anterior edge of the nasal septum are elevated with blunt dissection to create a lateral submucosal tunnel. Next, a left medial submucosal tunnel is created within the subperichondrial space. This tunnel is extended to up to the boney septum. The maxillary floor and posterior bony septum are then separated from the quadrangular cartilage and pushed laterally to the right. This caused the right-sided mucosa to be elevated from the boney septum and vomer. The vomer is removed and stored till the end of the case. To introduce the microscope, a Hardy retractor is inserted and expanded until the sphenoid sinus ostia are identified submucosally. The anterior wall of the sphenoid sinus comprising the sphenoid antrum and sphenoid keel is identified and removed. The sphenoid sinus is entered, and excess bone is removed with a high-speed drill to expose the sellar floor. To open and remove the sellar floor, a drill and Kerrisons are used to remove the mucus from the sellar floor. Lastly, the dura is incised, using a cruciate incision, and the tumor is excised using the pseudocapsular method [ ].
Similarly, for the mononarial approach, the nasal mucosa is unilaterally separated from the cartilaginous and osseous nasal septae with a medial nasal incision, creating a submucosal tunnel. Once the microscope is guided through the tunnel, the operation follows the similar procedure as the sublabial approach [ ].
Endoscopic approach
In the late 1970s, the endoscope was introduced by Bushe and Halves [ ]. However, this technique did not garner attention for pituitary adenoma resection until the 1990s by Jankowski et al. [ ] and otolaryngologists who preferred transsphenoidal endoscopic sinus surgery compared with the traditional open route due to improved cranial sinus visualization. It currently has a mainstream following due to faster postoperative recovery period, larger window for inspection, and lack of external incisions [ , ]. Instead of a skin incision, an incision is made immediately anterior to the vomer using a direct endonasal approach [ ]. Additionally, nasal-septal flaps, fascia lata, fat, and fibrin glue augmentation are used to assist in the closure and to prevent CSF leaks, especially for macroadenoma resections [ ]. Intraoperative nasal packing is often used for endoscopic surgery with mixed opinion among the surgical community, but a common consensus is that it can help with nasal mucosa healing and should be used after extensive nasal dissection [ ].
Selecting an approach
Much of the clinical literature has focused on comparing the outcomes and techniques between the pituitary approaches (sublabial, endonasal, microscopic, and/or endoscopic) [ ]. Certainly, there are arguments to be made for each approach depending on the familiarity and experience of the surgeon ( Fig. 13.3 ). Often, the surgeon’s training, technical skills, and comfort with a given approach determine the surgical practice. Regardless of the technical aspects of the surgical approaches, an experienced surgeon can deliver favorable outcomes [ ]. Factors that influence likelihood of endocrinologic remission include the presence of preoperative imaging findings (i.e., a visible adenoma), dural invasion of the adenoma, and adenoma size. These factors inform the surgical plan as discussed in the following.
Selective adenectomy and pseudocapsular dissection
Following the initial approach to the sella, surgeons can then use clearly defined strategies to remove the pituitary adenoma while preserving the normal gland (selective adenectomy). A strategy that us and others have adopted successfully to perform selective adenectomies hinges on identifying the “pseudocapsule” encompassing the pituitary adenoma. The pseudocapsule is the layer of compressed normal gland and distinct from the capsule of the pituitary gland. As an adenoma grows, the adjacent gland is compressed, and multiple layers of compressed reticulin form a pseudocapsule with higher tensile strength than the adenoma tissue. Costello first described this pseudocapsule in 1936 on pituitary adenomas found incidentally during autopsy [ ]. The pseudocapsule develops irrespective of the histological subtype of pituitary tumor [ ]. The capsule covering the pituitary gland, despite its translucent nature, is a barrier against adenoma invasion across the envelope of the pseudocapsule and into the surrounding anterior lobe [ ]. During surgery, we develop a plane between the pseudocapsule and normal gland and then to carry this plane circumferentially around the tumor, removing the tumor en bloc when possible [ ]. The pseudocapsule technique was later adapted to remove pituitary macroadenomas as well [ ].
Surgery for MR-negative CD
Preoperative adenoma identification on MR imaging is associated with higher odds of finding the adenoma at surgery (18-fold higher) and postresection biochemical remission (4-fold) [ ]. When an adenoma is seen on MR imaging, it directs surgical exploration to the adenoma location within the pituitary (86% correlation between MR imaging and surgical findings) [ ]. IPSS can be performed with bilateral simultaneous samples drawn from each inferior petrosal sinus, which permits direct comparison of ACTH concentrations from the left and right petrosal sinuses. This technique can be used to predict lateralization of the microadenoma with a 69% positive predictive value [ ]. An MR imaging-invisible adenoma often can be found in CD patients by systemic exploration of the pituitary gland by a series of incisions carried deeper in stages [ , ]. However, if a pituitary adenoma or pseudocapsule is not found, depending upon the results of the IPSS study, the surgeon may opt to perform a partial or total hypophysectomy. The reticulin capsule with adenomas appears to form as adenomas grow from 1 to 3 mm in diameter. The absence of a reticulin capsule with very small tumors may contribute to the challenges during surgical exploration of the pituitary [ ].
Dural invasion
Overall, the reported incidence of dural invasion is between 28% and 38% in pediatric and adult CD patients, respectively [ ]. The medial wall of the cavernous sinus is a frequent nidus for recurrence in CD and Oldfield advocated for its removal [ , , ]. Pituitary adenomas with lateral cavernous sinus dural invasion only have a 22% detection accuracy on MRI and are less likely to lead to remission after the index surgery. Microscopic dural invasion (without gross cavernous sinus invasion) remains undetected in 100% of patients [ ]. Given the numerous critical vascular and neurologic structures that course through the cavernous sinus, the region was previously thought to be surgically inaccessible.
The lateral dura is particularly susceptible to invasion by pituitary tumors because of a “route of least resistance” provided by veins that penetrate the lateral surface of the capsule to enter the medial wall of the cavernous sinus (MWCS) ( Fig. 13.4 ). Thus, most dural invasion by pituitary tumors is based on anatomic, rather than previously proposed genetic factors. Dural invasion occurs frequently with tumors that reach the pituitary capsule and is the most common basis of recurrent CD [ ]. When patients have invasion, it is usually limited to the MWCS, which can be surgically addressed by removing the patch of the wall involved with the tumor [ ]. Wide exposure of the anterior and inferior sella dura, including the medial dural wall of the CS, is critical to assess for sites of dural invasion. Some patients have free tumor in the cavernous sinus. In these patients, surgery is rarely curative, and we have advocated for early stereotactic radiation despite biochemical signs of early remission [ ].
Relationship of the presenting adenoma to the adjacent structures. A. Adenomas that are entirely within the pituitary parenchyma will often have a robust, circumferential pseudocapsule. The outcomes following resection of such adenomas have the best endocrinological outcomes. B. With lateral, intraparenchymal adenomas, the pseudocapsule may be breached during surgery, making the chances of tumor spillage and recurrence a distinct possibility. C. Adenomas that are adherent to the medial wall of the cavernous sinus need resection of the involved medial wall. D. Even with the adenoma invading the medial wall of cavernous sinus, durable endocrinological remission may be achieved with a complete resection of both (medial and lateral) leaflet of the medial wall of the cavernous sinus. E. With frank invasion, and presence of free tumor in the cavernous sinus, adjuvant radiation is strongly recommended following the initial surgery.
Intraoperative frozen section pathology
For patients undergoing TSS for CD, particularly with equivocal or small microadenomas on MRI, we refrain from calling for a frozen section pathology diagnosis of suspicious surgical lesions. Although there have been studies that support the use of frozen section diagnosis for suspected microadenomas [ ], we have found that processing for frozen pathology exhausts minuscule pathological tissues needed for confirmatory diagnosis. Frozen section processing may result in a number of specimens not being available for confirmatory diagnosis with paraffin-embedded permanent pathology [ ]. Therefore, we reserve the entirety of the surgical specimen for permanent pathology.
Intraoperative imaging
Several technological advances, including intraoperative MRIs [ , ], high-resolution ultrasound (USG), and endosphenoidal surface coils (ESCs) [ ], have shown efficacy in gaining further confirmation of tumor location and resection intraoperatively. Intraoperative MRIs are specifically used to determine the extent of resection, including in cases involving cavernous sinus and suprasellar invasion [ ]. However, MRIs are not infallible, thereby requiring other forms of imaging, including USG. USG is also able to account for the false negatives and positives from MRI by identifying 77% of microadenomas in a single study [ ]. Lastly, we are currently working on and studying, given the difficulty and monetary cost of increasing traditional MRI intensity past 3 T intraoperatively [ ]. The apparatuses operate by placing the coil within the sphenoid sinus during sublabial TSS.
Endocrine outcomes
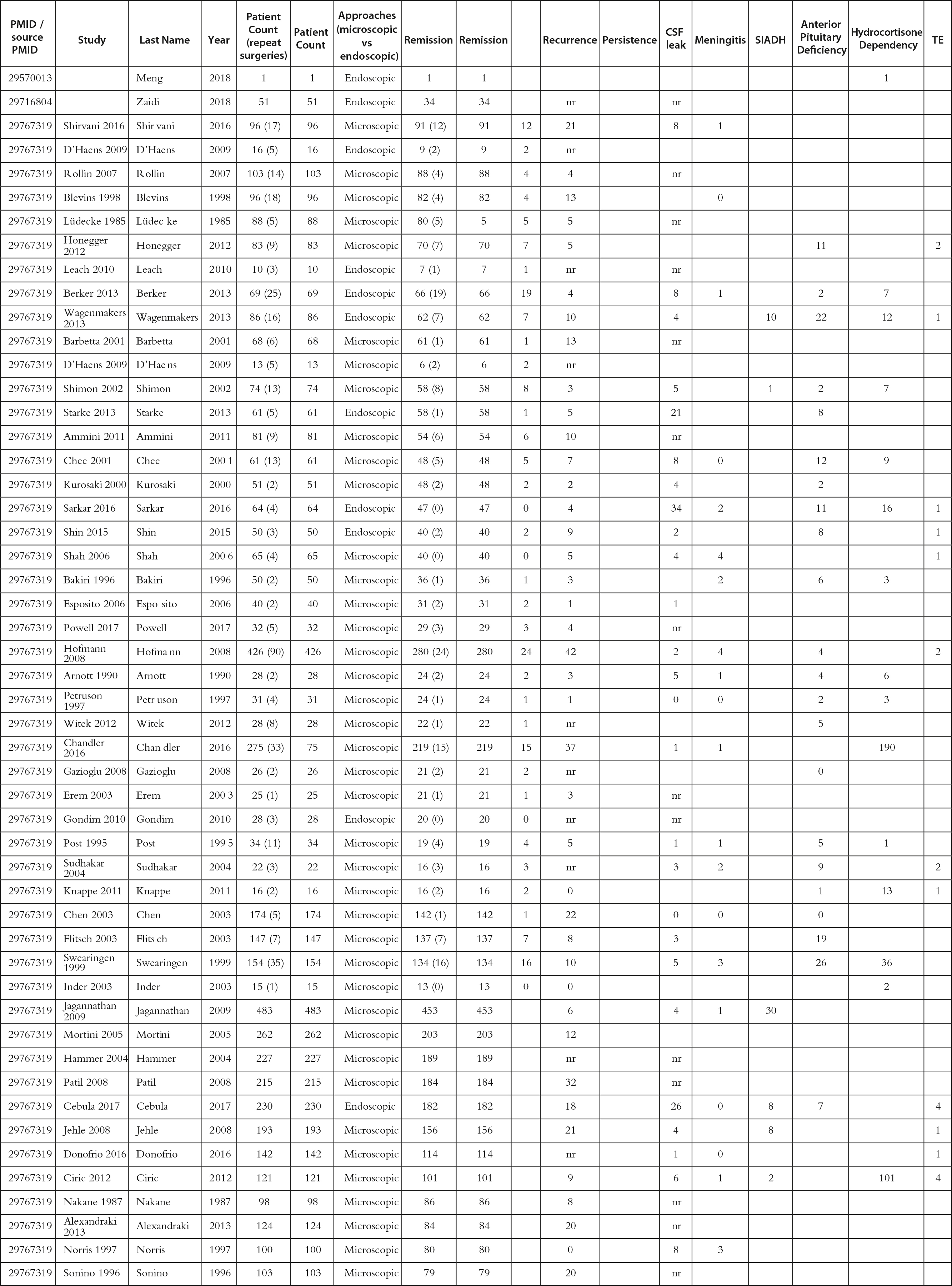
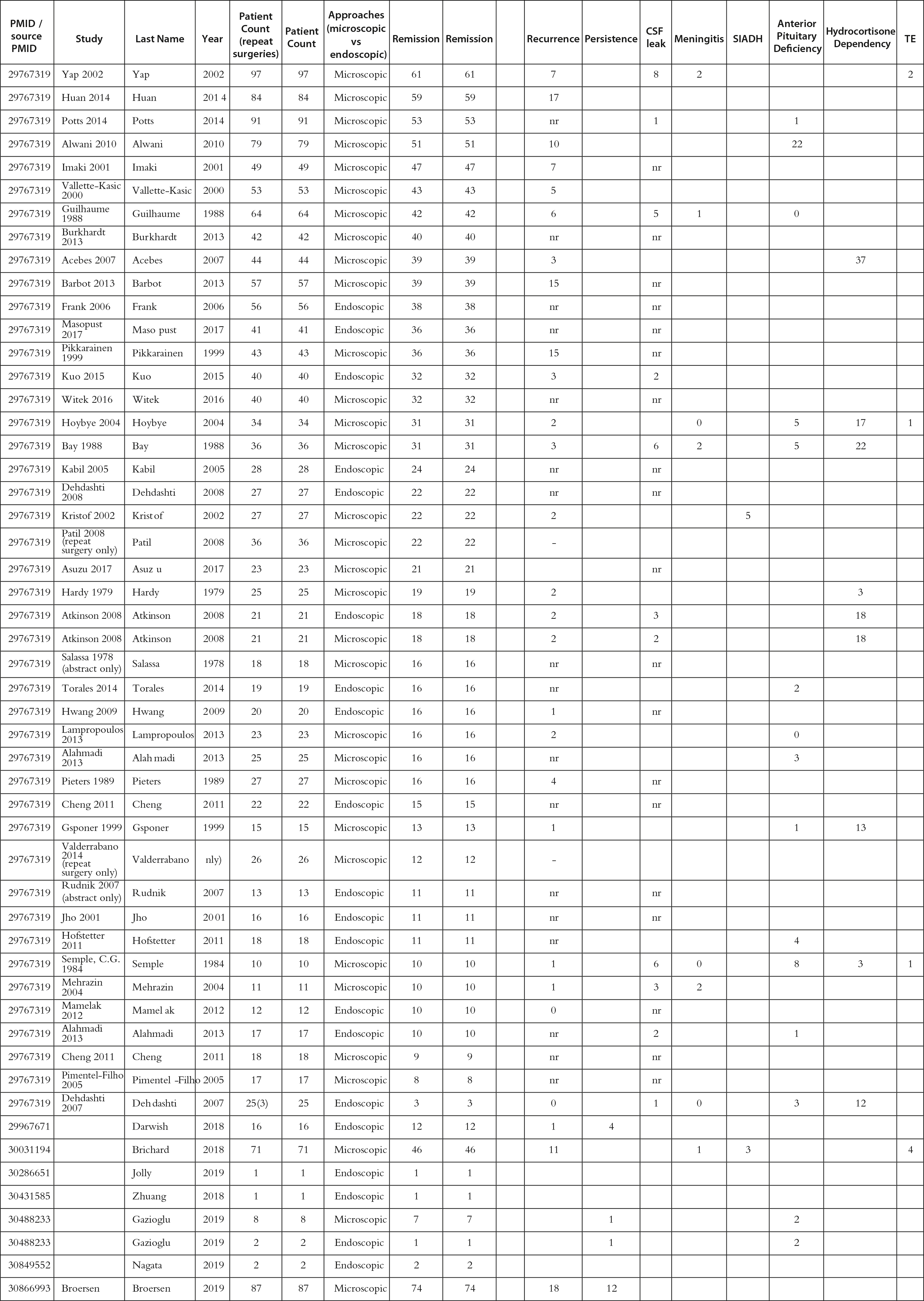
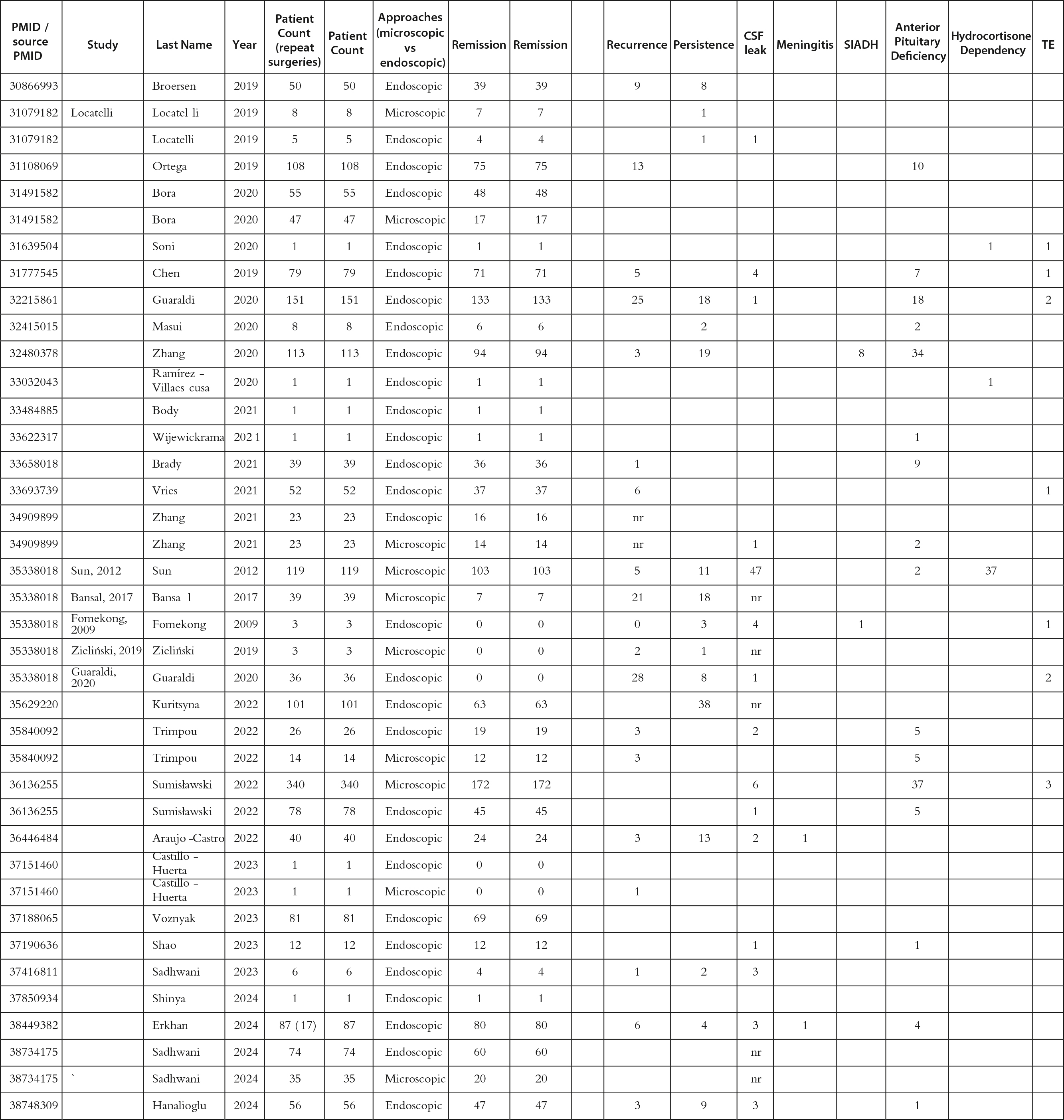
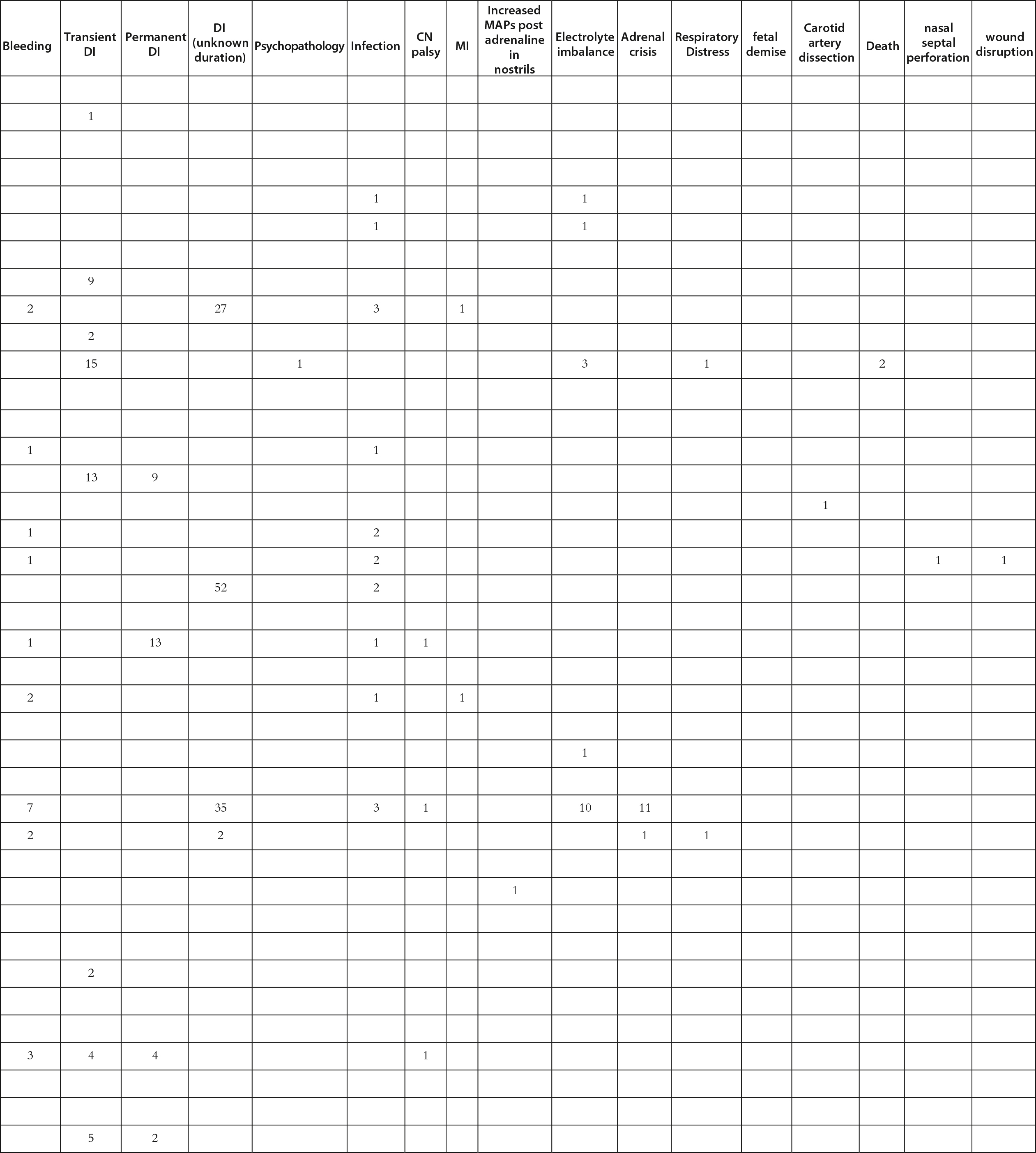
In our institution, reflexive perioperative corticosteroid replacement is withheld, and hypocortisolism is denoted as nadir serum cortisol of less than 5 mg/dL [ ] during the postoperative period. Early hypocortisolism is strongly linked to the determination of endocrinological remission [ , ]. Remission rates following surgery for CD vary widely with the anatomical details of the presenting adenoma. We have found that in the ideal case scenario of a small, central microadenoma, with a detectable and preserved pseudocapsule, immediate remission is as high as 95% [ ]. There exists a wide range of reported remission rates following TSS in the literature, ranging from 42% to 96% [ ] ( Fig. 13.5 ).
Summary statistics for Table 13.1 .
In the literature, there appears to be no significant differences in endocrine outcomes between the endoscopic and microscopic approaches [ , ]. A 2018 meta-analysis [ ] viewed 97 articles and 5711 patients who underwent the microscopic approach and 984 who underwent the endoscopic approach. While overall remission rates were approximately the same (80.5 vs. 79.2%), microscopic transsphenoidal surgery had a lower rate of CSF leaks (4 vs. 12.9%) and higher remission rates for repeat resections (55.7 vs. 42.6%). On the other hand, the endoscopic approach had higher rates of remission for patients with macroadenomas (76.3 vs. 59.9%). In our experience, using an exclusively sublabial microscopic approach for microadenoma surgery, the remission rates are between 89% and 94%, which is comparable with academic institutions that solely focus on the endoscopic approach [ , , , ].
Complications of TSS
Morbidity and mortality for operative CD are 2%–10% and 2%, respectively [ , , ]. Mild symptoms in the perioperative period include headaches, anosmia, fatigue, and sinusitis. Moderate complications that may require a repeat surgery include a CSF leak (0%–13.1%). Endocrinological abnormalities, such as transient and/or permanent DI (0.7%–2.3% vs. 1%–3.5%), SIADH, and hyponatremia, can be monitored for fluid intake and DDVAP administration if necessary. Severe complications include internal carotid artery and nerve injuries, stroke (0%–7.9% for vascular incidents), and death. Catastrophic complications are rarer and require careful consideration of surrounding vasculature during the procedure [ ]. For this book chapter, we performed a literature review for manuscripts discussing surgery for CD ( Table 13.1 ) by adding recent reports to the excellent meta-analysis in 2018 by Broerson et al. [ ]. Among 8752 patients who underwent pituitary surgery with any approach, up to 77.1% of patients had clinical remission. However, among the patients who required a repeat surgery, only 44.1% achieved remission. The most common complication noted was anterior pituitary dysfunction (9.9%), followed by SIADH (5.4%), permanent DI (4.2%), and CSF leak (4.1%). Catastrophic complications were low, with meningitis at 1.1% and bleeding at 2.5%. These data suggest that TSS for CD remains a safe and effective first-line treatment in the modern era.
Table 13.1
Manuscripts discussing surgery for Cushing’s disease reviewed for this work.
|

|
|

|
|
|
Postoperative management
Detection of remission
Detecting remission in the postoperative period requires a thorough understanding of the postoperative hormone dynamics [ ]. This allows clinicians to use absolute levels of cortisol [ ], or the results of hormone testing to detect early signs of remission following surgery [ , ]. Markers for remission include postoperative cortisol (less than 2 mg/dL morning serum and less than 20 mg/day urine free) and ACTH levels (less than 5 pg/mL) and are typically lowered within the first week of the surgery [ , ]. There are also multiple short-term metrics that can be used to predict long-term remission. For morning serum cortisol, a level of less than 1 mg/dL on day 3–5 off exogenous steroids has a 96% positive predictive value of remissions [ ]. In studying the postoperative hormone dynamics, our group has learned from early experiences in intraoperative HPA axis investigations [ ] to devise a theoretical framework for interpreting postoperative hormone levels [ ]. We first realized that the surgical stress of TSS (especially when the patient is waking up from anesthesia) is a valid “intrinsic” stress test. The level of stress induced by surgery is similar to the preoperative oCRH or DDAVP stimulation tests, offering us a direct comparator. When we compare the cortisol (or ACTH) levels in the postoperative period with the maximal hormone stimulation results, we find a robust and reliable method to detect signs of early remission. We have named this strategy as normalized early postoperative (NEPV) cortisol/ACTH to predict remission [ ]. With NEPV for cortisol at −12, we predicted early remission with 100% sensitivity. Recently, we showed that NEPV ACTH was a highly reliable marker for remission at awakening from surgery, while NEPV cortisol remained a powerful predictor for remission at 6 hours [ ]. Similarly, we have found that if we examined only patients with signs of early remission (cortisol nadir < 5 μg/dL) [ ], the slope of fall of cortisol was predictive of long-term, durable remission. Specifically, if serum cortisol fell below 2 μg/dL within the first 21 hours after surgery, it was a reliable indicator (100% sensitivity) of long-term durable remission from CD [ ]. Lastly, we focused on plasma ACTH level variations [ ] in health and disease. It was known that ACTH level variability is mildly suppressed in CD. However, the variability between the measurements remains large and is a major reason why single plasma ACTH measurements are not useful as predictors of remission. We first found that ACTH variability was restored with remission. Next, we found that the diurnal range of plasma ACTH measurements was also restored with remission. These two markers offer clinicians reliable indications for remission following TSS for CD. Lastly, we found that a greater than 50% reduction in plasma ACTH predicted remission (100% sensitivity and specificity at the evening—1800 hours—measurement on postoperative day 2) [ ].
Early reoperation
Early reoperation for a missed adenoma may be planned, especially if the postoperative hormone dynamics are well documented. In addition, a thorough examination of the pathological specimen by an experienced neuropathologist is helpful in deciding on an early reoperation. Complete exhaustion of the pathological block is recommended with serial sectioning and staining for hematoxylin/eosin, reticulin, and ACTH. If the hormone and pathology data suggest a missed adenoma, an early reoperation during the same hospitalization can achieve remission in more than 90% of first surgical failures [ , ].
Role for hypophysectomy
During reoperation, a strategy to maximize the chances of remission is needed, particularly in instances where the index case was MRI negative. In these cases, distinct adenomas are identified less often during TSS [ ], leading to a more extensive exploration of the pituitary gland or additional tissue resection without tumor identification [ , ]. Although hemihypophysectomy [ , ] and total hypophysectomy do not improve remission rates in general [ , , ], if an adenoma is discovered in the resected specimen, remission rates are high [ ]. In the recent years, we have adopted a cautious, stepwise approach in instances of nonremission where histopathology is also negative. We have performed early reoperations, followed by a more extensive exploration of the remaining pituitary gland with serial, vertical incisions. If a putative adenoma is not found with this approach, we then proceed to a 75% hypophysectomy. The remaining normal gland is adjacent to the pituitary stalk. This approach has led a to 90% remission rate in these challenging cases. The hormone function remains intact in 50% of these patients, suggesting that the remnant quarter of the pituitary gland is adequate to maintain hormonal homeostasis (unpublished data).
Remote monitoring
Post-op follow-up
After remission status is determined, glucocorticoid replacement at supraphysiologic doses (40–60 mg/day) should be given to counteract the adrenal insufficiency (serum cortisol less than 5 mg/dL) 72 hours postoperatively for a stable patient. In an unstable patient, such as one undergoing an adrenal crisis or who has had a complicated surgery, replacement should be provided immediately. Additionally, serum sodium, urine osmolarity, and specific gravity should be monitored every 6–12 hours with strict control of patient intake and output. 1–2 weeks postoperatively, free T4 should be checked, with a more thorough assessment of the thyroid, gonadal, and growth hormone pituitary axes in the proceeding months [ , ].
Detection of recurrence
Under the commonly used transsphenoidal technique, CD recurs in approximately 14% of patients after their index surgery, with the average time to recurrence between 15 and 50 months. The rate of recurrence increases if the patient initially is diagnosed with a macroadenoma (greater than 1 cm in diameter) [ ]. The current long-term recommendation for postoperative CD is to have a follow-up within 8 weeks if the patient is in remission and is placed on glucocorticoid replacement and within 1 year of the index surgery. If the patient has a recurrent or persistent adenoma, they are to follow up within 4 weeks, and within 8–24 weeks post radiotherapy and stable status on adjuvant medical therapy [ ].
Recurrence should be tested periodically using 24-hour cortisol free urine, late-night salivary cortisol, and/or a dexamethasone stress test. Without histological confirmation, IPSS also allows for monitoring and/or surgical planning [ ]. If a delayed recurrence (months to years after initial remission) is detected, MR imaging is indicated. If an adenoma is found on imaging, it should be evaluated for resection. If resection is not possible or if no adenoma was found on imaging but the clinical symptoms are reminiscent of the initial presentation, adjuvant therapy can be considered. However, with an identifiable or suspicious adenoma on MRI, repeat surgery should be considered [ ].
Treatment of recurrence
The rate of endocrinological remission is lower for repeat surgeries for CD. Given these findings, adjuvant therapy is considered at the time of remission to provide a definitive treatment of CD. Adjuvant therapy comprises medical treatment or radiotherapy. Medical treatment includes steroidogenesis inhibitors, corticotroph-directed agents, and glucocorticoid receptor antagonists [ ]. Medical therapy is usually started prior to radiotherapy for hormonal control. Radiosurgery has grown to include stereotactic radiosurgery for targeted and localized therapy [ ]. One disadvantage of this treatment is the loss of pituitary cells and function (20%–40% over 10 years), but an advantage for patients is that the therapy can be delivered in one session. Lastly, for persistent and recurrent CD, bilateral adrenalectomy can be useful to eliminate the terminal receptor for cortisol production, with 95% success in refractory cases [ ].
Conclusion
Surgical resection of the offending ACTH-secreting adenoma remains the treatment of first choice for CD. TSS, either microscopic or endoscopic, is the surgical approach of choice for most surgeons when treating CD. The endocrinological outcomes are influenced heavily by the ability to visualize adenomas, to remove the adenoma along with its pseudocapsule, and invasion into adjacent anatomical structures. Severe, life-threatening complications of TSS remain rare. Postoperative management following TSS includes hormonal measurements to detect early signs of remission. In the modern era, TSS is a remarkably safe surgical procedure with high rates of endocrinological remission.
Funding
This project was supported by the Intramural Research Program of the National Institute for Neurological Diseases and Stroke.
References
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree