Introduction
Perioperative management of a patient with Cushing’s syndrome involves reconciling the physiologic implications of excess cortisol with the complex changes occurring in the perioperative period. Successful surgical intervention for Cushing’s syndrome fully involves the selection and optimization of surgical candidates to receive appropriate operations in a timely manner with meticulous perioperative management. This includes data-proven strategies for prevention and early detection of known common complications as well as timely identification of potential complications with directed intervention. This process is often aided by the recognition of prespecified postoperative milestones.
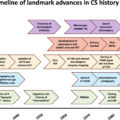
This chapter will first explore the variety of techniques utilized for the surgical management of Cushing’s syndrome. This will include all elements with the exception of ACTH-dependent pituitary-based Cushing’s syndrome (Cushing’s disease) as this is discussed in the previous chapter ( Fig. 14.1 ) .
Outline of possible diagnoses emanating from a clinical diagnosis of Cushing’s syndrome.
The perioperative physiologic considerations will then be addressed including preoperative workup and preparation, potential complications, and recovery timeline goals as well as strategies for prevention of the known common complications in surgery for Cushing’s syndrome.
Surgical approach—ACTH-independent Cushing’s syndrome
It follows that the surgical approach to Cushing’s syndrome pathology depends on the etiology responsible for hypercortisolemia. In general, for an adrenal source, total adrenalectomy is the procedure of choice in these patients. However, the stratification of ACTH-independent Cushing’s syndrome can impact surgical management. This includes the decision on bilateral versus unilateral adrenalectomy for specific instances of bilateral pathology. This section will discuss the spectrum of ACTH-independent Cushing’s syndrome and decision-making regarding the surgical approach.
Benign disease—overview
The pathology of benign adrenal lesions causing ACTH-independent hypercortisolemia includes adenoma and macro- or micronodular hyperplasia involving unilateral or bilateral adrenal glands. The severity of Cushing’s syndrome varies from mild endogenous hypercortisolism or subclinical Cushing’s syndrome (discussed separately) to severe disease with multiorgan consequences. For unilateral adenomas, unilateral adrenalectomy is almost always curative and therefore is the procedure of choice. Minimally invasive adrenalectomy is the most common approach because of less postoperative pain and faster recovery [ ]. Minimally invasive adrenalectomy can be done via anterior transperitoneal laparoscopic or robotic approach or retroperitoneal endoscopic approach. Unilateral macronodular and micronodular adrenal hyperplasia are similarly addressed surgically with minimally invasive unilateral adrenalectomy after a thorough workup.
After biochemical workup confirms ACTH-independent Cushing’s syndrome, CT adrenal imaging will usually create a paradigm that dictates surgical management based on the above-referenced stratification ( Fig. 14.2 ). This stratification of ACTH-independent Cushing’s syndrome is further outline in the following.
Surgical candidates with ACTH-independent Cushing’s syndrome.
Benign disease—primary bilateral macronodular adrenal hyperplasia
Primary bilateral macronodular adrenal hyperplasia (PBMAH) is characterized by multiple adrenal nodules with a size at least >1 cm with variable biochemical cortisol production. A highly heterogenous disease, the clinical presentation can range from asymptomatic subclinical Cushing’s syndrome to severe Cushing physiology with multiorgan dysfunction. The heterogeneity of PBMAH creates difficulty in managing incidental diagnoses from unrelated abdominal imaging. This entity is further subdivided into isolated and inherited forms. The inherited form of PBMAH can be derived from multiple endocrine neoplasia type 1 (MEN1), familial adenomatous polyposis (FAP), or hereditary leiomyomatosis and renal cell cancer (HLRCC, more rarely) [ ]. McCune–Albright syndrome (MAS) is another inherited form of adrenal hyperplasia but will be discussed later. Isolated PBMAH can be sporadic or familial, with sporadic cases linked to altered G protein–coupled receptor (GPCR) expression. Familial cases have recently been elucidated with the identification of an ARMC5 germline mutation responsible for approximately 20%–25% of PBMAH cases [ ].
There has been recent debate regarding the indications for unilateral versus bilateral adrenalectomy in the management of PBMAH. While bilateral adrenalectomy has historically been the gold standard for the management of bilateral macronodular adrenal hyperplasia, surgeons have recently begun to employ unilateral adrenalectomy to remove the larger adrenal gland as a successful alternative in select cases, sparing patients from lifelong steroid replacement therapy. A recent review of 71 patients highlighted a high success rate of unilateral adrenalectomy, with nearly 95% remission and only 19% of patients with recurrent Cushing’s syndrome after a median follow-up of 75 months [ ]. Because none of the patients with the size of a remaining adrenal gland smaller than 3.5 cm developed persistent Cushing’s syndrome following unilateral adrenalectomy, the results support unilateral adrenalectomy, especially when there is a size discrepancy between adrenal glands. Although the rate of persistent Cushing’s syndrome following unilateral adrenalectomy for PBMAH was 20% in patients with a contralateral gland larger than 3.5 cm, the probability of initial remission was still high (80%) and thus should be considered in select patients. It must be emphasized that these cases should be discussed in multidisciplinary fashion, considering both patient preferences and other comorbid factors that may influence the need for future interventions ( Table 14.1 ) .
Table 14.1
Summary of outcomes from studies of unilateral adrenalectomy for PBMAH [ ].
| Source | Patients | Remission after surgery (%) | Recurrence (%) | Subsequent bilateral adrenalectomy (%) | Persistent adrenal insufficiency (%) | Mean/median follow-up (months) |
|---|---|---|---|---|---|---|
| Sheikh-Ahmad et al. ECED, 2020 | 71 | 67 (94) | 13 (19) | 10 (14) | Only general adrenal insufficiency reported | 75 |
| Osswald et al. JCEM, 2019 | 25 | 21 (84) | 3 (14) | 3 (12) | 1 (5) | 50 |
| Debillon et al. JCEM, 2015 | 15 | 15 (100%) | 2 (13) | 1 (7) | 5 (33) | 60 |
| Li and Yang et al. Exp Ther Med, 2015 | 15 | 15 (100%) | 3 (20) | 3 (20) | Not specified | Not specified |
Benign disease—micronodular adrenal hyperplasia
There are two variants of micronodular bilateral adrenal hyperplasia (MiBAH): primary pigmented nodular adrenocortical disease (PPNAD) and isolated micronodular adrenocortical disease (i-MAD). PPNAD will be discussed primarily in the following sections on Carney’s complex. MiBAH, characterized by the lack of pigmented micronodules (<1 cm), can be a difficult pathology to diagnose and may warrant conservative medical management depending on the severity of Cushing’s symptoms. Due to the bilateral nature and less incidence of lateral dominance (compared to PBMAH), surgical intervention almost always necessitates bilateral adrenalectomy. However, in some mild cases with asymmetry, unilateral adrenalectomy can be considered [ ].
Benign disease—McCune–Albright syndrome
McCune–Albright syndrome (MAS), also referred to as fibrous dysplasia, is a rare and broad disorder encompassing a constellation of clinical findings that may include hyperpigmented skin macules, variable hyperfunctional endocrinopathy, and bony fibrous dysplasia [ ]. MAS is caused by a somatic mutation in GNAS that subsequently leads to mosaic downstream activation of various related GCPR pathways [ ]. The mosaic phenotype of MAS leads a broad range of heterogenous clinical presentations. Adrenal hyperplasia and resultant hypercortisolism can be present in MAS, reportedly occurring in less than 5% of patients [ , ]. Patients with MAS commonly exhibit a pathognomonic paradoxical response to Liddle’s test. A natural history NIH cohort of MAS patients demonstrated Cushing’s syndrome in 7.1% of patients. In a broadened cohort, 12/19 MAS patients with hypercortisolemia underwent adrenalectomy with 6/19 demonstrating spontaneous resolution [ ]. Guidelines state that spontaneous resolution of hypercortisolemia may occur in up to one-third of patients and close observation may be reasonable in mild/stable cases [ ].
For patients with MAS, bilateral adrenalectomy (preferred laparoscopic) will often be offered due to severe clinical features and a paucity of indications for unilateral adrenalectomy. Unilateral adrenalectomy may be offered in select cases of unilateral disease but with concerns of persistent or recurrent disease These patients will often be directed into operative management after failed attempts at medical management or due to severe clinical phenotype. In these patients, concomitant cardiac and hepatic dysfunction (as a result of prolonged hypercortisolemia) may complicate a perioperative course of adrenalectomy.
Benign disease—Carney’s complex
Carney’s complex is a rare, primarily inherited syndrome that comprises a constellation of clinical findings including multiple skin abnormalities and endocrinopathies [ , ]. An autosomal dominant inherited condition, most cases (∼70%) carry pathogenic germline PRKAR1A mutations, which encodes for the type 1 alpha regulatory subunit of protein kinase A [ ]. Despite this, there remains significant genetic heterogeneity. Primary pigmented nodular adrenocortical disease remains the most common etiology of Cushing’s syndrome in these patients, observed in approximately 50% of patients. However, it should be noted that these patients may also present with sex hormone or thyroid tumors. Of particular importance in the surgical workup of a Carney’s patient is the concomitant incidence of cardiac myxoma that may preclude aggressive cardiology investigation, stratification, and possibly intervention for these cases. In fact, the most common causes of death in patients with Carney’s complex include complications from cardiac myxomas (emboli, cardiomyopathy, arrhythmia, surgical complications) [ ].
Similar to MAS, patients with Carney’s complex are almost exclusively offered bilateral adrenalectomy due to bilateral pathology. In a review of 34 patients with either micronodular hyperplasia and/or Carney’s complex at the NIH, 30/31 (97%) of patients undergoing bilateral adrenalectomy were biochemically cured and none of the 3 patients undergoing unilateral adrenalectomy were biochemically cured [ ]. 18/34 (53%) of these patients represented Carney’s complex and demonstrated primary pigmented nodular adrenocortical disease. It should also be noted that in rare cases patients may have persistent biochemical Cushing’s syndrome after bilateral adrenalectomy due to residual microscopic disease [ ]. The NIH cohort experience further emphasizes the need for cardiac risk stratification and workup in Carney’s complex patients due to the 53% incidence of myxoma with three patients delaying adrenalectomy to treat myxomas.
Benign disease—sporadic adenoma
Sporadic unilateral adenoma warrants appropriate workup and additional imaging as dictated earlier in this textbook. Once a confirmed diagnosis of unilateral functioning adenoma with hypercortisolemia is confirmed, preparations can be made for operative intervention. The intricacies of diagnosis for a single unilateral sporadic adenoma will not be detailed in this chapter as it is discussed extensively in earlier chapters. It should be noted (discussed further below) that mild endogenous hypercortisolemia (previously subclinical Cushing’s syndrome) is indeed the most common associated endocrine abnormality in adrenal incidentalomas [ ].
Benign disease—mild endogenous hypercortisolemia (previously subclinical Cushing’s syndrome)
The indications and timing for surgical intervention in mild endogenous hypercortisolemia depend largely on cardiovascular risk stratification factors that may derive benefit from unilateral adrenalectomy. While there are several randomized studies that conclude a benefit in patients undergoing unilateral adrenalectomy for mild endogenous hypercortisolemia, these studies have numerous limitations including nonstandardized medical management comparison arms (conservative, nonsurgical) and unknown long-term outcomes including quality of life [ , ]. For example, one randomized controlled trial observed no development of over clinical Cushing’s syndrome in their conservative management arm over a mean follow-up of 7.7 years, however, simultaneously noting vague clinical worsening of diabetes and blood pressure [ ]. Additionally, three of the patients randomized to conservative management did crossover into surgical management due to adrenal lesion size increase, bringing to the forefront the importance of continued monitoring and surveillance in these patients even if medical management is the consensus. In a broad sense, young patients with worsening morbidity attributable to excess cortisol (including hypertension, hyperglycemia, dyslipidemia, and osteoporosis) should be considered for adrenalectomy. The American Association of Endocrine Surgeons Guidelines for Adrenalectomy recommend unilateral adrenalectomy in patients with mild endogenous hypercortisolemia secondary to a unilateral adenoma [ ]. In fact, 30%–35% of patients with adrenal incidentaloma may also harbor a diagnosis of mild endogenous hypercortisolemia and the opportunity for improvement of cardiometabolic comorbidities obviates the consideration of unilateral adrenalectomy [ , ].
Malignant disease—adrenocortical cancer
The most common malignant source of ACTH-Independent Cushing’s syndrome is adrenocortical carcinoma (ACC). This cancer is incredibly aggressive and typically carries a dismal prognosis when it is not confined to the adrenal gland. Approximately 60% of patients diagnosed with ACC present with Cushing’s syndrome [ ]. Because an R0 or negative-margin (gross and microscopic) resection is the only possibility of cure, these cases are traditionally approached with an open technique that involves meticulous dissection that prevents tumor capsule rupture. However, advances in minimally invasive techniques have allowed for this approach in select cases for smaller tumors, typically <6 cm. The following section will highlight data that both supports and refutes the indications for open versus minimally invasive adrenalectomy for ACC. Unfortunately, there have been no randomized-controlled trials to investigate this question, and therefore, the bulk of this review is focused on retrospective experiences.
One retrospective review from France focused solely on 49 patients with localized ACC (stage 1–2) and concluded no difference in overall survival but a statistically significant difference in 3-year disease-free survival (89.7% vs. 73.3%, P = .020) [ ]. An NCDB analysis of 1483 patients in the United States undergoing adrenalectomy for ACC from 2010–17 revealed no difference in overall survival on univariable analysis and surprisingly a survival benefit on multivariable analysis for the minimally invasive surgery (MIS) group (HR 0.83, 95% CI 0.70–0.99) [ ]. Interestingly, the comparable survival persisted even across large tumor sizes >6 cm and >10 cm. However, the retrospective limitations of this study are evident in the discrepancies between cohorts; the open surgery cohort had a statistically significant higher incidence of clinically stage IV disease (38.9% vs. 22.4%, P < .01) and lymphovascular invasion (56.6% vs. 47.9%, P < .01). A separate retrospective analysis of 201 patients from 13 tertiary care referral centers demonstrated no differences in rates of intraoperative tumor rupture or margin-negative (R0) resection as well as similar overall and disease-free survival [ ]. These studies as well as others are summarized in Table 14.2 .
Table 14.2
Summary of open adrenalectomy (OA) versus laparoscopic adrenalectomy (LA) cohort analyses [ ].
| Source | Number of patients | OA/LA-OS/RFS difference | Notable comments/conclusions |
|---|---|---|---|
|
Gaillard et al. J Clin Med, 2023
France |
49 |
OA 3 year DFS 89.7%
LA 3 year DFS 73.3% ( P = .020) |
Localized (stage 1–2 ACC) only |
|
NCDB. Delman et al. J Surg Res, 2022
The United States |
1483 | No difference |
Database analysis
Size not predictive |
|
Lee et al. J Gastrointest Surg, 2017
The United States |
201 | No difference | OA favored for local invasion or enlarged lymph nodes |
It should be noted that guidelines allow for an MIS approach as long as it does not impact the certainty of margin-negative resection [ ]. Guidelines also permit adrenalectomy in cases with systemic disease when these sites are eligible for resection or local therapy and performance status is optimal. Neoadjuvant systemic therapy should only be considered in cases where R0 resection is not initially feasible.
Surgical approach—ACTH-dependent
The workup of ACTH-dependent Cushing’s syndrome has been addressed in earlier chapters. A brief summary will be highlighted:
After biochemical endogenous hypercortisolemia is confirmed, an elevated late afternoon ACTH may indicate ACTH-dependent Cushing’s syndrome and prompt MRI evaluation for pituitary lesions. If late afternoon ACTH is low or borderline, a corticotropin-releasing hormone (CRH) stimulation test is indicated. A positive CRH stimulation test will then usually prompt MRI evaluation for pituitary lesions. A high-dose dexamethasone suppression test (HDDST) with ACTH may also be performed in conjunction with MRI to further solidify the diagnosis of ACTH-dependent Cushing’s syndrome. If there is no definitive pituitary pathology on imaging, inferior petrosal sinus sampling (IPSS) is indicated to confirm the presence or absence of a responsible pituitary lesion. If IPSS is negative, whole-body CT imaging is indicated to attempt localization of an ectopic source of ACTH.
Preoperative considerations will depend on whether the ACTH-producing lesion is identified and the extent of surgery required to remove the source of ACTH production. The primary goal of the surgery is to completely remove ACTH-producing lesions. However, it is not uncommon that bilateral adrenalectomy is required in patients with persistent Cushing’s syndrome following the initial surgical intervention or in patients with ectopic ACTH-producing lesions that cannot be identified.
ACTH-dependent Cushing’s syndrome—pituitary lesions
Surgical management of pituitary macro- and microadenoma causing Cushing’s disease typically involves transsphenoidal resection or radiation. Gamma knife radiosurgery has also emerged as a promising technique for pituitary-based Cushing’s syndrome; this will be discussed in the chapter on Cushing’s disease specifically [ ]. However, bilateral adrenalectomy is indicated in patients with persistent disease following initial intervention who are not candidates for reoperation. These refractory cases should be considered for bilateral adrenalectomy only after thorough multidisciplinary review. Bilateral adrenalectomy will be discussed later.
ACTH-dependent Cushing’s syndrome—ectopic ACTH-producing lesions
Ectopic-ACTH-producing tumors can arise from any tumors with neuroendocrine cells, most commonly in the lung or gastroenteropancreatic tumors (GEP-NETs). Other sources include medullary thyroid cancer, thymic carcinoids, pheochromocytomas, and paragangliomas. Bronchial carcinoids represent the most common ectopic source of ACTH (ranging from 20% to 40%) but can sometimes be difficult to visualize or localize on standard imaging [ ]. On occasion, there are occult ectopic ACTH-producing neuroendocrine tumors that cannot be localized and therefore preclude a definitive surgical intervention until the tumor is identified. For the purposes of this chapter, surgical intervention for primary ectopic ACTH-producing tumors will not be discussed.
In general, surgical resection of ectopic sources of ACTH is the procedure of choice for these patients when localization is achievable and the site is resectable for cure. For these cases, it is important to clearly define and localize the tumor by CT or MRI with or without somatostatin analog-based imaging studies (such as 68Ga-DOTATATE) preoperatively. Regional lymphadenectomy is of particular importance in bronchial carcinoids and neuroendocrine tumors of the lung as these tumors have a propensity to metastasize to lymph nodes and proper lymphadenectomy has been shown to influence survival [ ]. Because the prognosis of patients with uncontrolled Cushing’s syndrome is poor, cytoreduction of metastatic ACTH-producing tumors can be considered in select patients to aid in the control of hypercortisolemia.
It should also be noted that bilateral adrenalectomy can be employed for patients with tumors of unknown origin (occult ectopic ACTH) to treat the sequelae of resultant Cushing’s syndrome. Bilateral adrenalectomy may also be employed when primary surgical resection of the suspected source of ectopic ACTH fails to achieve biochemical cure. A recent retrospective review spanning from 1995–2017 of 48 patients undergoing bilateral adrenalectomy for ectopic ACTH secretion reported a 100% biochemical cure rate with an equal component of unknown and known origin [ ]. Bilateral adrenalectomy may also be utilized in cases where cortisol-lowering or steroidogenesis-inhibiting agents are relatively or absolutely contraindicated. An example of this includes patients eligible for tyrosine kinase inhibitors (for treatment of metastatic malignancy) who cannot proceed with cortisol-lowering agents interfering with drug metabolism.
Bilateral adrenalectomy carries a high success rate (biochemical cure) in ACTH-dependent cases with acceptable surgical morbidity and mortality, often leading to rapid improvement of Cushing physiology [ ]. It should be noted that these cases do have elevated morbidity/mortality when compared with adrenally sourced hypercortisolemia due to the higher instances of severe Cushing’s syndrome and therefore drastically worsened physiologic risk stratification [ ].
Techniques—unilateral versus bilateral adrenalectomy versus partial adrenalectomy
The decision of whether to proceed with unilateral versus bilateral adrenalectomy predominantly relates to instances of bilateral adrenal pathology or ectopic ACTH secretion. The differences in operative technique between unilateral and bilateral adrenalectomy relate primarily to patient positioning, allowing for simultaneous access to the retroperitoneum on either side. The retroperitoneal endoscopic approach carries a distinct advantage in regard to bilateral adrenalectomy as positioning is the same for either side. Partial adrenalectomy is an organ-preserving approach to adrenal surgery that has emerged over the previous decades in an attempt to minimize postoperative exogenous steroid dependence.
Unilateral adrenalectomy
The specific goals of unilateral adrenalectomy involve the safe removal of left or right adrenal parenchymal tissue with meticulous hemostasis and identification of critical structures as indicated by prior discussion. Classically a “vein-first” approach is employed, which allows for control of venous blood supply followed by control of arterial blood supply and completion of adrenal dissection free from the retroperitoneal bed.
For right adrenalectomy, anatomical considerations include the superior pole of the right kidney, the inferior vena cava, and the right lobe of the liver. Blood supply derives from a dominant right adrenal artery directly off the aorta and the right adrenal vein directly off the inferior vena cava. The right adrenal gland does have an extensive vascular network with many small arterial branches often emanating from the inferior phrenic artery and right renal artery.
For left adrenalectomy, anatomical considerations include the superior pole of the left kidney, the aorta, the left renal vein, and the pancreatic tail. Blood supply derives from the left adrenal artery directly off the aorta while the left adrenal vein usually branches off the left renal vein. The left adrenal gland similarly has an extensive vascular network with many small arterial branches often emanating from the inferior phrenic artery and left renal artery.
Bilateral adrenalectomy
The specific goals of bilateral adrenalectomy involve the safe removal of all adrenal parenchymal tissue for the indications discussed previously. The bilateral adrenalectomy can be performed in transperitoneal open, transperitoneal laparoscopic (usually requiring patient repositioning), or retroperitoneal endoscopic fashions. As mentioned previously, the retroperitoneal endoscopic approach carries with it the advantage of avoiding patient repositioning. The bilateral adrenalectomy simply involves the aforementioned indications for unilateral adrenalectomy on both sides.
Partial adrenalectomy
Partial adrenalectomy has emerged in recent decades as an alternative approach for patients with multiple endocrine pathologies where there is a benefit of the organ-preserving technique. These include hereditary pheochromocytoma syndromes with a low risk for metastasis such as von Hippel–Lindau disease (VHL) and multiple endocrine neoplasia type 2 (MEN2) [ ]. This operative approach should only be considered at institutions with appropriate experience regarding partial adrenalectomy as well as after a thorough multidisciplinary review.
Techniques—MIS adrenalectomy
As in other realms of surgery, the transition from open-to-laparoscopic surgery has been swift in the management of adrenal pathology. MIS adrenalectomy carries with it significantly reduced morbidity, hospital length-of-stay, and analgesic requirements [ , ]. In fact, the AAES guidelines agree upon MIS adrenalectomy (whether it be laparoscopic or robotic) as the “gold-standard” for small benign adrenal pathology. We have previously discussed the points and counterpoints for the employment of MIS adrenalectomy in cases of ACC; however, most modern endocrine surgeons will agree that in benign cases the MIS adrenalectomy is the standard.
Laparoscopic adrenalectomy—transabdominal lateral approach
The laparoscopic adrenalectomy has become the most utilized operative approach to remove an adrenal gland and has evolved into a very safe procedure with improvements in laparoscopic techniques. This approach carries many advantages beyond the scope of improved cosmesis with smaller incisions. These advantages include earlier return of bowel function, rapid postoperative return to ambulation and physical activity, less pain and need for postoperative narcotics, and lower risk and incidence of wound infection and hernia (which carries profound implications in Cushing’s syndrome patients, to be discussed later in this chapter).
The intricacies of surgical techniques will not be addressed in detail. Patients are positioned in a “modified semilateral” position with the operating table tilted to elevate the laterality of the operation. Because this positioning is similar to lateral decubitus, some surgeons may avoid this approach in Cushing’s patients with overt osteoporosis and/or a history of pelvic fractures [ ].
Retroperitoneal endoscopic adrenalectomy
Since its debut in the surgical literature in 1996 by Walz et al., the retroperitoneal approach to laparoscopic adrenalectomy has been adopted by numerous endocrine surgeons and urologists in the surgical management of Cushing’s syndrome [ ]. This operative approach carries with it the advantage of avoiding mobilization of intraabdominal/peritoneal compartment organs. By exclusively accessing the retroperitoneum and creating a false surgical space with insufflation, surgeons can avoid the morbidity of an abdominal operation in patients. This approach can greatly benefit the surgical management of patients with a heavy abdominal surgical history where concern for adhesiolysis and related complications may play a role. Additionally (and as mentioned previously), this approach can also make the bilateral adrenalectomy easier by eliminating the need for patient repositioning required in the laparoscopic transabdominal lateral approach. It should be noted that surgical management with retroperitoneal access requires meticulous operative expertise of the surgeon as well as refined patient selection. In fact, many reviews of retroperitoneal endoscopic adrenalectomy (RPA) in the literature will note the difficulty in this approach for obese patients [ ]. This derives from the combination of increased retroperitoneal fat content as well as poor ventilatory capacity with prone positioning [ ]. It is for this reason that RPA may not be employed as often for overt Cushing’s syndrome as these patients may be clinically obese due to the severity of their physiology.
For retroperitoneal endoscopic approach, the patient is placed in prone positioning with access to the retroperitoneum just under the 12th rip on the indicated side. A working space is created with an inflatable balloon within the blunt trocar combined with a blunt dissection using an endoscope. This space is then insufflated, commonly at a higher pressure than the intraperitoneal cavity, to further refine the operating boundaries.
Robotic adrenalectomy
The robotic platform can be incorporated for adrenalectomy with either the transabdominal lateral approach or retroperitoneal access. Of particular relevance for surgeons, the robotic platform may aid in refining visualization and instrument articulation for the more meticulous partial adrenalectomy [ ]. Studies have demonstrated the equivalence of robotic adrenalectomy in terms of morbidity and operative time, when considering a learning curve [ ]. Of course, as with all other subspecialities, an employment of the robotic platform should account for the financial ramifications of robotic surgery when deciding on how to proceed with patient surgical management from an institutional perspective.
Techniques—open adrenalectomy
As discussed earlier, open adrenalectomy is most often employed in cases of ACC where there is significant attention paid to avoiding tumor capsule rupture/violation. These cases are usually addressed via an upper midline laparotomy but can also be accessed nontraditionally with subcostal transverse incisions (left or right).
The intricacies of open adrenalectomy closely mirror the considerations involved earlier in the overview of unilateral versus bilateral versus partial adrenalectomy. While open adrenalectomy is rare in the modern surgical experience for the management of benign adrenal disease, there should never be a question of sacrificing morbidity for patient safety.
Complications—preoperative considerations
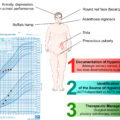
The management of CS patients in the preoperative setting is important for optimization prior to surgical intervention. The primary preoperative factors to consider for optimization prior to operative intervention include electrolyte levels (most importantly hypokalemia), hyperglycemia and sequelae of diabetes, cardiovascular sequelae such as hypertension and cardiac dynamics, nutritional deficiencies and osteoporosis, infectious risk, and thromboembolic risk. When considering all of these factors, optimization and appropriate awareness can mitigate the catastrophic surgical complications well-known to perioperative Cushing’s patients including perioperative thromboembolism, postoperative infection, ventilator-associated pneumonia, and wound complications including fascial dehiscence and hernias. When surgery is less urgent and can be planned in a “semielective” fashion, patients can benefit from a 3–6 month period of prehabilitation with nutritional optimization and physical therapy to prepare for the stresses of surgery ( Table 14.3 ) .
Table 14.3
Outlined preoperative clinical factors for consideration with proposed interventions and possible consequences of neglect.
| Preoperative clinical sequelae | Optimization measures | Potential complications |
|---|---|---|
| Electrolyte abnormalities | Hypokalemia—sprinolactone/eplerenone/amiloride, potassium supplementation | Arrhythmia, wound complications/poor healing |
| Hyperglycemia, diabetes | Target HgbA1C, diabetes medications, diet/exercise | Wound complications/poor healing |
| Hypertension, cardiac function | Antihypertensives, echocardiography for risk stratification, possible cardiac drugs for improvement | Arrhythmia, perioperative MI |
| Nutritional deficiency and osteoporosis | Prehabilitation, diet/exercise, vitamin supplementation | Wound complications/poor healing, fractures |
Preoperative control of hypercortisolemic sequelae—steroid blockade
Preoperative anticortisol treatments in patients with Cushing’s syndrome such as metyrapone (first-line), ketoconazole, mifepristone, etomidate, mitotane, and others ( Fig. 14.3 ) are usually reserved for patients whose operative intervention is delayed and/or patients requiring medical therapy for symptom control and/or optimization of comorbidities due to severe Cushing’s syndrome [ ]. This may also be the case in patients who are refractory after initial unsuccessful intervention. While some retrospective analyses have concluded a decreased risk of bleeding and possibly other perioperative complications in patients receiving medical antisteroidogenic therapies prior to operative intervention [ ], this possible beneficial impact has never been definitively proven. In fact, a European cohort analyses concluded no definitive improvement for adrenally based Cushing’s syndrome when comparing patients receiving medical management prior to operative intervention [ ].
Medication list of most common anticortisol therapies used in the treatment of Cushing’s syndrome.
Adapted from Miller BS, Auchus RJ. Evaluation and treatment of patients with hypercortisolism: a review. JAMA Surg 2020;155(12):1152–1159. 10.1001/jamasurg.2020.3280.
In general, patients receiving anticortisol medications should continue the treatments until the morning of surgery and then discontinue them after surgery. All decisions regarding tapering and/or discontinuation of antisteroidogenic medications should be discussed extensively with the managing endocrinologist as well as a multi-disciplinary team.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree