Summary of Key Points
- •
Size counts: From less than or equal to 1-cm to less than or equal to 5-cm tumor size, every centimeter has prognostic impact and separates into different T categories, and tumors greater than 5 cm but less than 7 cm are now T3; and those greater than 7 cm are now T4.
- •
Distance to carina does not count: Tumors with endobronchial location less than 2 cm from the carina have similar outcomes to those greater than 2 cm from the carina.
- •
N status remains largely the same.
- •
An oligometastasis is now classified as M1b.
- •
Descriptors for same lobe as well as other lobe intrapulmonary nodules as metastases or synchronous primary lesions are delineated.
- •
Part-solid adenocarcinoma size will be defined by the size of the solid component on computed tomography and of the invasive component on microscopic examination.
The tumor, node, and metastasis (TNM) classification of malignant tumors is promulgated by the Union for International Cancer Control and the American Joint Committee on Cancer. The TNM classification undergoes periodical revisions based on the reports from the National TNM Committees and on the annual assessment of original articles. For the latest two editions of the TNM classification of lung cancer (7th edition of 2009 and 8th edition of 2016), the revisions were based on two international databases collected by the International Association for the Study of Lung Cancer (IASLC). These databases were stored, managed, and analyzed by Cancer Research And Biostatistics, a not-for-profit data center based in Seattle, WA, USA, in collaboration with the members of the IASLC Staging and Prognostic Factors Committee. This chapter presents the process of revision leading to the eighth edition of the TNM classification of lung cancer, the innovations introduced, and their clinical implications. The eighth edition of the TNM classification of malignant tumors will be enacted on January 1, 2017.
The International Association for the Study of Lung Cancer Database for the Eighth Edition
For the second consecutive time, the IASLC registered data from around 100,000 patients with lung cancer. For this revision, the period of diagnosis was from 1999 to 2010. Data originated in 35 different databases in 16 countries of 5 continents. Their geographical origin and number of patients are as follows: Europe, 46,560; Asia, 41,705; North America, 4660; Australia, 1593; and South America, 190, for a total of 94,708 patients. After exclusions, 77,156 patients met the requirements for analysis: 70,967 with nonsmall cell lung cancer (NSCLC) and 6189 with SCLC. Table 25.1 shows the type of databases contributing to the IASLC Staging Project and the nature of the data. Most data were retrospective, that is, contributors around the world already were registering data on patients with lung cancer and submitted their databases to the IASLC. These databases contained the minimum information regarding TNM descriptors, but some of them lacked the necessary detail needed for deeper analyses. By contrast, data registered through the electronic data capture online system are smaller in numbers but richer in detail and were useful, for example, to analyze the descriptors of the M component because they contained the information on number and location of metastases. Table 25.2 shows the type of treatment undergone by the registered patients with SCLC and NSCLC. The proportion of patients treated with tumor resection either alone or in combination with chemotherapy and/or radiotherapy is higher in this database than in the one used for the seventh edition. This is due to the fact that databases of clinical trials, usually collected for advanced disease, were not submitted. However, surgical registries have complete information on the descriptors of the anatomic extent of the disease and are readily available for analysis. Despite this lack of patients with advanced lung cancer, all findings potentially leading to recommendations for changes were validated in the populations of patients with clinically and pathologically staged tumors, except those of the M component that were exclusively based on clinically staged tumors.
| Type of Database | Retrospective | Prospective (EDC) | Total |
|---|---|---|---|
| Consortium | 41,548 | 2089 | 43,637 |
| Registry | 26,122 | 26,122 | |
| Surgical series | 5373 | 592 | 5965 |
| Institutional series | 1185 | 1185 | |
| Institutional registries | 208 | 208 | |
| Unknown | 39 | 39 | |
| Total | 73,251 | 3905 | 77,156 |
| Treatment Modality | % |
|---|---|
| Surgery alone | 57.7 |
| Chemotherapy and surgery | 21.1 |
| Radiotherapy and surgery | 1.5 |
| Trimodality | 4.4 |
| Chemotherapy alone | 9.3 |
| Chemotherapy and radiotherapy | 4.7 |
| Radiotherapy alone | 1.5 |
Innovations in the T, N, and M Descriptors
T Descriptors
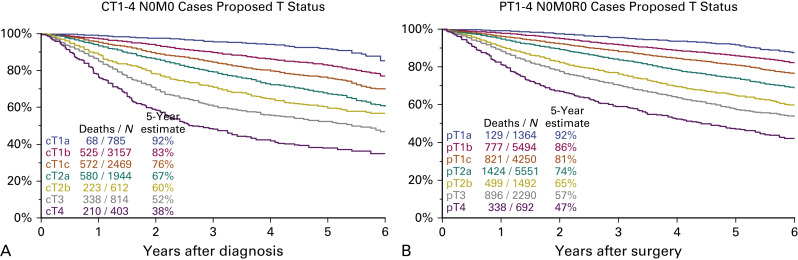
The T component of the classification is complex to analyze because it has many descriptors: tumor size, endobronchial location, atelectasis/pneumonitis, and invasion of various anatomic structures surrounding the lung. For their analyses, the prognostic impact of each descriptor was analyzed individually in five different populations of patients: three with pathologically staged tumors (pT1–4 N0 M0 completely resected [R0]; pT1–4 any N M0 R0; and pT1–4 any N M0 any R, i.e., including resections with microscopic [R1] and macroscopic [R2] evidence of residual tumor) and two with clinically staged tumors (cT1–4 N0 M0 and cT1–4 any N M0). Additional univariate and multivariate analyses were performed after adjustment by histopathologic type, geographical region of origin, age, and sex. These analyses generated multiple survival curves that were closely analyzed to see if the different descriptors were properly assigned to their T category. The results of these analyses can be summarized as follows:
- •
The 3-cm landmark still separates T1 from T2 tumors.
- •
Tumor size, analyzed at 1-cm intervals, has more prognostic impact than previously shown in past editions of the TNM classification. From less than or equal to 1-cm to less than or equal to 5-cm tumor size, every centimeter counts and separates different T categories.
- •
The prognosis of tumors greater than 5 cm to less than or equal to 7 cm was similar to that of T3 tumors.
- •
The prognosis of tumors greater than 7 cm was similar to that of T4 tumors.
- •
The prognosis of tumors with endobronchial location less than 2 cm from the carina (a T3 descriptor in the 7th edition) was found to be similar to that of their T2 counterparts (endobronchial location >2 cm from the carina).
- •
The prognosis of tumors with total atelectasis/pneumonitis (a T3 descriptor in the 7th edition) was found to have a T2 prognosis, similar to that of tumors with partial atelectasis/pneumonitis.
- •
The prognosis of tumors with invasion of the diaphragm (a T3 descriptor in the 7th edition) was found to be similar to that of T4 tumors.
- •
The invasion of the mediastinal pleura was rarely used as a unique descriptor.
Based on the aforementioned findings, the recommendations for changes in the T categories are as follows:
- •
Subdivide T1 into three new subcategories: T1a (≤1 cm), T1b (>1 cm but ≤2 cm), and T1c (>2 cm but ≤3 cm).
- •
Subdivide T2 into two new subcategories: T2a (>3 cm but ≤4 cm) and T2b (>4 cm but ≤5 cm).
- •
Reclassify tumors greater than 5 cm but less than or equal to 7 cm as T3.
- •
Reclassify tumors greater than 7 cm as T4.
- •
Reclassify tumors with endobronchial location less than or equal to 2 cm from the carina, but without involvement of the carina, as T2.
- •
Reclassify tumors with total atelectasis/pneumonitis as T2.
- •
Reclassify tumors with invasion of the diaphragm as T4.
- •
Delete the invasion of the mediastinal pleura as a descriptor.
When survival is analyzed according to these new T descriptors, survival curves separate well and do not cross over. All survival differences are significant and there is a clear difference between T3 and T4 that was not seen in the seventh edition ( Fig. 25.1 ).
Visceral pleural invasion, defined as the invasion of its elastic layer, is well assigned to the T2 category ( Fig. 25.2 ). There are survival differences between PL1 and PL2, but these are only identifiable at pathologic staging, so they cannot be used to modify the present T2 descriptor based on the extent of visceral pleural invasion. However, they are useful to refine postoperative prognosis in those patients with resected tumors in which visceral pleural invasion is identified. Because visceral pleura involvement impacts prognosis, the use of elastic stains is emphasized again in the eighth edition of the TNM classification of lung cancer, if it is not evident on hematoxylin and eosin stains.
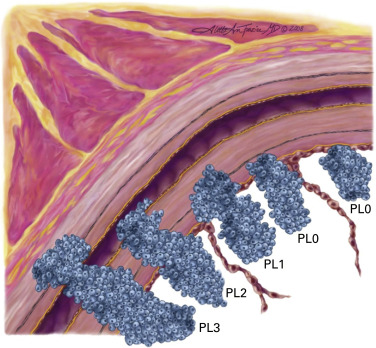
(Copyright 2008 Aletta Ann Frazier, MD.)
(Reprinted with permission from Travis WD, Brambilla E, Rami-Porta R, et al. Visceral pleural invasion: pathologic criteria and use of elastic stains. Proposal for the 7th edition of the TNM classification for lung cancer. J Thorac Oncol. 2008;3:1384–1390.)
N Descriptors
The N descriptors were analyzed in the population of patients with clinically and pathologically staged tumors. In both populations, the present N descriptors (N0, N1, N2, and N3) separate tumors with statistically significant prognosis ( Fig. 25.3 ). Therefore, there is no need to suggest any modification in the N component.
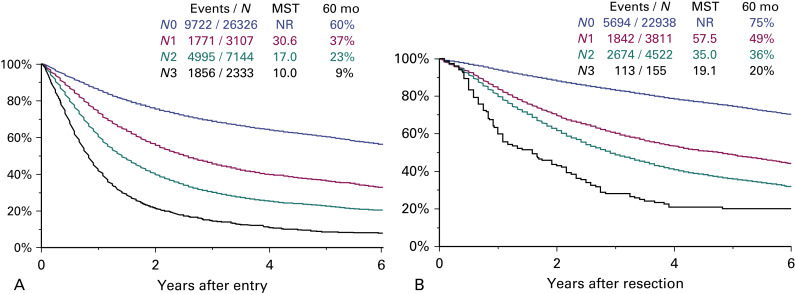
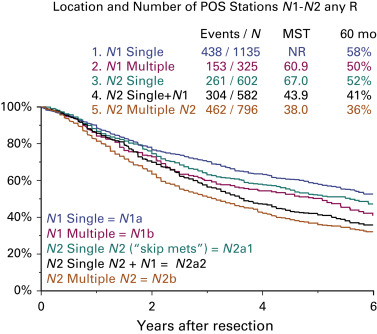
Exploratory analyses on the quantification of nodal disease also were performed. Survival was analyzed according to the number of nodal stations involved in the population of patients with pathologically staged tumors and adequate information on the nodal stations explored. There are five groups of tumors according to the number of nodal stations involved:
- •
N1a: single-station N1
- •
N1b: multiple-station N1
- •
N2a1: single-station N2 without N1 disease (skip metastasis)
- •
N2a2: single-station N2 with N1 disease
- •
N2b: multiple-station N2.
Fig. 25.4 shows the survival of patients with pathologically staged tumors and nodal disease classified according to the number of nodal stations involved. All survival differences are statistically significant except those between N1b (multiple-station N1) and N2a1 (single-station N2 without N1). When these analyses are performed in the clinical staging setting, these differences cannot be replicated. This is the reason why this suggested subclassification cannot be used to modify the N descriptors, because, in principle, clinical and pathologic descriptors must be identical. However, this subclassification is clinically relevant as it may be used to refine postoperative prognosis for those patients with resected lung cancers harboring nodal disease.
The IASLC regional lymph node map proposed in 2009 was the result of a multidisciplinary and international consensus ( Fig. 25.5 ). No map or anatomic scheme is perfect, and this map is not perfect either. One advantage of this map over the others previously proposed is that this one has clear definitions for each nodal station that can be recognized by radiologists, endoscopists, and thoracic surgeons performing mediastinoscopy and systematic nodal dissection at the time of lung resection ( Table 25.3 ). The IASLC Staging and Prognostic Factors Committee recommends the use of this map for nodal labeling and the prospective collection of data to examine its potential limitations and suggest modifications in the future.
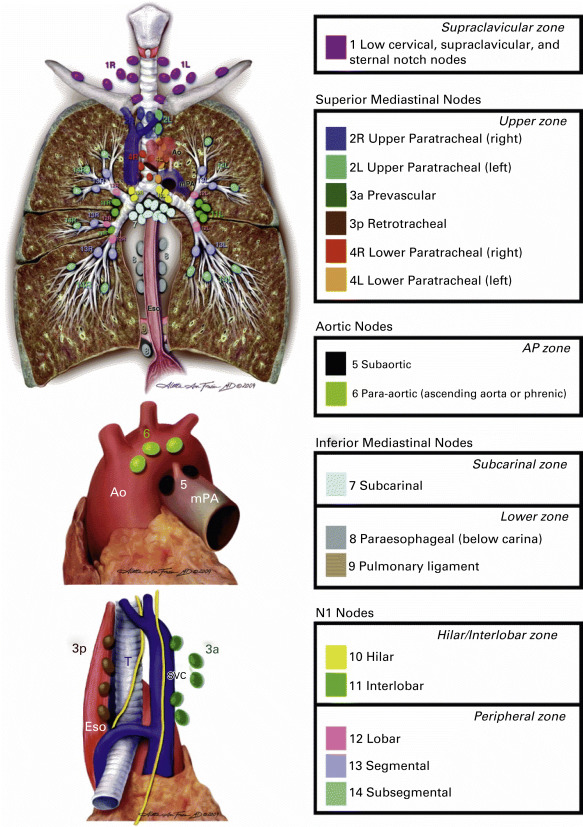
| Lymph Node Station No. (#) | Anatomic Limits |
|---|---|
| Supraclavicular Zone | |
| #1: Low cervical, supraclavicular, and sternal notch nodes |
|
| Upper Zone | |
| #2: Upper paratracheal nodes |
|
| #3 Prevascular and retrotracheal nodes |
|
| #4: Lower paratracheal nodes |
|
| Aortopulmonary Zone | |
| #5: Subaortic (aortopulmonary window) |
|
| #6: Para-aortic nodes (ascending aorta or phrenic) |
|
| Subcarinal Zone | |
| #7: Subcarinal nodes |
|
| Lower Zone | |
| #8: Paraesophageal nodes (below carina) |
|
| #9: Pulmonary ligament nodes |
|
| Hilar/Interlobar Zone | |
| #10: Hilar nodes |
|
| #11: Interlobar nodes |
|
| Peripheral Zone | |
| #12: Lobar nodes |
|
| #13: Segmental nodes |
|
| #14: Subsegmental nodes |
|
M Descriptors
The electronically captured data, that is, data registered prospectively online, were used for the revision of the M descriptors because they had adequate detail for the planned analyses. Patients with resected metastases were excluded from these analyses. Regarding the M1a descriptors, those defined in the seventh edition (metastasis within the pleural space: contralateral separate tumor nodules, pleural and pericardial nodules, and malignant pleural and pericardial effusions) could be validated with the database of the eighth edition. Fig. 25.6A shows how similar the survival curves for all these descriptors are. Therefore, there is no need to modify them. For extrathoracic metastases (M1b), it was found that patients with single extrathoracic metastasis had similar survival to that of those with M1a tumors, but significantly worse than those with multiple extrathoracic metastases. The survival of those with multiple extrathoracic metastases in one organ and that of those with multiple extrathoracic metastases in several organs was similar. These analyses showed that the number of extrathoracic metastases had more prognostic impact than their location ( Fig. 25.6B ). According to these results, the recommendations for changes were the following:
- •
Keep the M1a descriptors as they are
- •
Redefine M1b to code tumors with a single extrathoracic metastasis
- •
Create a new category, M1c, to code tumors with multiple extrathoracic metastases either in one or in several organs.
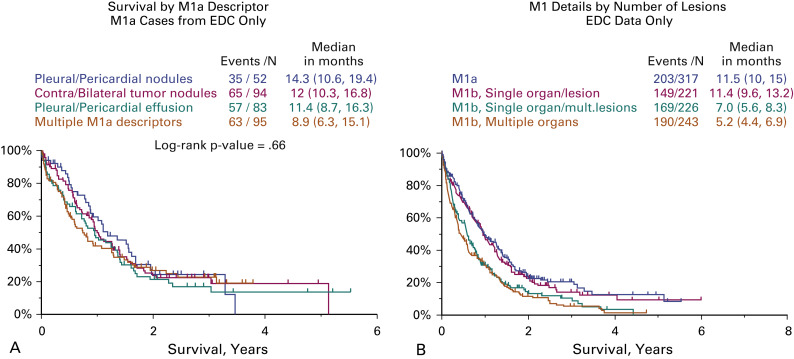
M1a and M1b tumors have similar prognoses, but it makes sense to code them differently as they represent different anatomic forms of metastatic disease and have a different diagnostic and therapeutic approach.
Stage Grouping
Table 25.4 shows the definitions of the T, N, and M descriptors for the eighth edition of the TNM classification of lung cancer. For the stage grouping, several models were discussed. Table 25.5 shows the model eventually chosen because it is the one that best separates tumors with different prognoses. Stage IA is divided into stages IA1, IA2, and IA3 to accommodate the new T1a, T1b, and T1b tumors with no nodal involvement and no metastasis. Stages IB and IIA will now group T2a and T2b tumors with no spread beyond the lung, respectively. All N1M0 tumors will be grouped in stage IIB, together with T3 N0 M0, except for T3–T4 N1 M0 tumors that are grouped in stage IIIA. Similarly, all N2M0 tumors are stage IIIA, except for T3–T4 N2 M0 tumors that are in stage IIIB, together with all N3M0 tumors, except for T3–T4 N3 M0 for which a new stage IIIC was created. Finally, stage IV is divided into stage IVA to group M1a and M1b tumors, and stage IVB to include M1c tumors. Fig. 25.7 shows the survival of patients according to clinical and pathologic stages, respectively. The expected worsening in survival is observed as tumor stage increases. All differences are statistically different except that of clinical stages IIIC and IVA. However, it makes sense to separate these tumors into two different stages as they represent different types of anatomic spread of the tumor: locoregional for those in stage IIIC and metastatic for those in stage IVA.