Keywords
amenorrhoea, BRCA genes, chemotherapy, DNA double-strand breaks, ovarian damage
4.1
Introduction
Chemotherapy has considerably improved survival of many cancer types; however, a patient’s quality of life is influenced by long-term complications of chemotherapeutic regimens. These complications include cardiac abnormalities, secondary malignancies, renal and hepatic impairment, and gonadal dysfunction. Gonadal dysfunction may result in an early menopause and infertility in reproductive-age patients, which is associated with poorer quality of life. Chemotherapy-induced gonadotoxicity is not limited to cancer patients but also includes patients with rheumatologic diseases, aplastic anaemia and systemic lupus erythematosus. Therefore, patients diagnosed with these conditions are also at risk of premature ovarian insufficiency (POI) after treatment.
4.2
Risk Factors for Premature Ovarian Insufficiency After Chemotherapy
Most long-term follow-up studies evaluating ovarian toxicity after adjuvant chemotherapy are retrospective and have used the menstrual pattern as the only surrogate marker. The extent of gonadotoxic injury following chemotherapy is influenced by the age of the patient at the time of treatment, as well as the type, dose and duration of chemotherapy, and the clinical degree of ovarian dysfunction, which varies from transient amenorrhoea to true menopause, may reflect the magnitude of gonadotoxicity.
4.2.1
Age
Age-related differences are most likely due to a reduction of the ovarian reserve with ageing, which increases the risk of developing POI post-chemotherapy. Females less than 40 years of age exposed to chemotherapy agents have a 22–61% risk of developing amenorrhoea, and this rate increases to 66–95% in females older than 40 years. Resumption of menstruation was higher in patients younger than 40 years compared to those older than 40 years. In the National Surgical Adjuvant Breast and Bowel Project (NSABP) B-30 study, 708 patients diagnosed with breast cancer were treated with four cycles of doxorubicin and cyclophosphamide (AC) and four cycles of docetaxel. The resumption of menses occurred within 24 months in 45.3% of patients aged under age 40 years, 10.9% of those aged 40–50 and in 3.2% of patients aged over 50 years.
4.2.2
Type of Chemotherapy
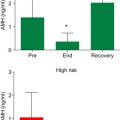
The type of chemotherapy is a strong determinant of POI ( Figure 4.1 ). Alkylating agents carry the highest risk because these drugs are not cell cycle specific, meaning that both resting and developing follicles can be damaged. Cyclophosphamide is the most commonly utilized agent in this group; it is frequently used to treat a wide range of cancers including lymphomas, leukaemia, neuroblastoma, retinoblastoma and breast carcinoma. Other clinical uses for cyclophosphamide include immunosuppressive therapy following organ transplants or as a treatment for connective tissue disorders. Cyclophosphamide leads to major disruptions in nucleic acid function and inhibits DNA and protein synthesis. Its use is associated with DNA crosslinking in granulosa cells, stromal fibrosis and capillary changes in the female reproductive system. Gonadal toxicity with cyclophosphamide alone or in combination with other chemotherapeutics has been documented. The authors reported previously that even a single dose of cyclophosphamide resulted in a drastic reduction in human primordial follicle reserve in a xenograft model. Boumpas et al. compared the effects of short (seven doses) and long (>15 doses) courses of cyclophosphamide treatment in 39 females aged under age 40 years diagnosed with systemic lupus erythematosus to evaluate the effect of number of doses of cyclophosphamide on menstruation and the incidence of sustained amenorrhoea was found to be higher in the long-course group (12.5 vs. 39%, p=0.07).
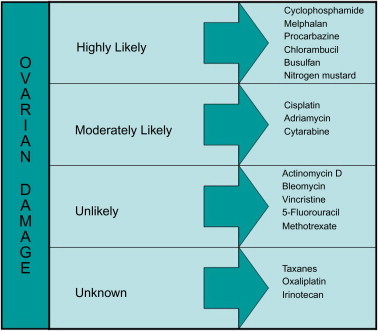
Doxorubicin, a topoisomerase inhibitor antracyclin antibiotic, is widely integrated in a variety of cancer regimens, and acts by intercalating DNA. It is one of the most effective drugs for the treatment of solid tumours such as breast cancer. DNA damage and apoptotic cell death in human granulosa cells and primordial follicles, as well as a reduction in stromal blood flow, has been shown after in vivo administration of doxorubicin. Depending on intensity, duration, and combination with other chemotherapy agents, doxorubicin treatment may result in POI in cancer patients.
Taxanes are relatively new chemotherapeutic agents that act on the cytoskeleton to disrupt microtubule function. They are commonly used to consolidate doxorubicin- and cyclophosphamide-based treatment in breast cancer patients. Laboratory studies of taxane-induced ovarian toxicity are limited and conflicting. Tarumi et al. suggested that the ovarian toxicity of paclitaxel is mild and does not involve the primordial follicles; however, in another study in rats, primordial follicle counts decreased following the administration of paclitaxel. Recently, Lopes et al. demonstrated that docetaxel directly impairs the early stages of ovarian follicle development, but it has no direct effect on the primordial follicle reserve in mice. Though the data are limited, clinically, taxanes do not appear to have a strong ovary-damaging effect.
Cycle-specific agents – such as 5-fluorouracil, methotrexate and vinca alkaloids – appear to affect only the developing and mature follicles and hence cause transient amenorrhoea without damaging primordial follicles.
4.2.3
Combined Treatment Protocols
The assessment of ovarian function after chemotherapeutic agent combinations in the treatment of different cancer types is, in most studies, based on the menstrual pattern. However, comparisons of chemotherapy-induced amenorrhoea rates and resumption of menstruation exhibit substantial variability due to differences in mean age, treatment dose, and follow-up duration.
4.2.3.1
Breast Cancer
Breast cancer is the most frequent cancer diagnosed in females of reproductive age. Because breast cancer in young females presents with a high prevalence of ductal infiltration, most of those patients are likely to undergo adjuvant systemic chemotherapy. Although a cyclophosphamide, methotrexate and 5-fluorouracil regimen (CMF) is rarely used today in breast cancer, in a detailed analysis of the published studies with classic CMF, the incidence of amenorrhoea ranged from 21–71% in females younger than 40 years of age and 40–100% in older females. The actual risk of POI associated with taxanes, which have been incorporated recently in the adjuvant setting for breast cancer, remains controversial. With AC-based regimens, the incidence of amenorrhoea ranged from 15–93% with or without taxanes. A study by Tham et al. included 191 premenopausal females with breast cancer and the rate of chemotherapy-induced amenorrhoea in patients who received AC followed by taxanes was significantly higher compared with AC alone (64 vs. 55%, p=0.05). In addition, chemotherapy-induced amenorrhoea rates were higher in older than younger females (82 vs. 55%, p=0.004). However, Davis et al . found no difference in amenorrhoea rates between females receiving AC-based chemotherapy with or without consolidating docetaxel in review of 159 premenopausal females. The results of another study comparing a similar regimen, 5-fluoruracil, epirubicin and cyclophosphamide (FEC) with or without docetaxel, were consistent with the findings of Davis et al. In this study, 154 premenopausal patients were included and 84 treated with six cycles of FEC and 70 with three cycles of FEC plus docataxel. The incidence of chemotherapy-induced amenorrhoea at the end of chemotherapy was similar in the two groups (93% vs. 92.8%).
Resumption of menstruation can occur early or be delayed 1–2 years from the initiation of chemotherapy-related amenorrhoea, depending on the type of treatment. Fornier et al. investigated long-term amenorrhoea lasting >12 months in 166 females with breast carcinoma after adjuvant AC- and taxane-based chemotherapy. In this study 25 patients (15%) developed long-term amenorrhoea, and 141 patients (85%) resumed menstruation. In a large prospective study investigating the maintenance of menstrual bleeding in 466 patients with breast cancer that received AC, AC-T or CMF, females receiving CMF were less likely to menstruate after 6 months of follow-up compared to AC and AC-T regimens (23% with CMF vs. 68% with AC vs. 57% with AC-T, p=0.002). Berliere et al. compared resumption of menses after FEC vs. FEC plus docetaxel; more patients with consolidating docataxel recovered menses in the year following the end of chemotherapy (35.5 vs. 23.7%, p=0.01).
4.2.3.2
Hodgkin Lymphoma
Hodgkin lymphoma (HL) is a relatively rare cancer with an incidence of 2–3 per 100,000 females. In HL, treatment protocols combining doxorubicin, bleomycin, vinblastine and dacarbazine (ABVD), without alkylating agents, rarely result in POI; however, treatment protocols containing alkylating agents, especially procarbazine and cyclophosphamide, induce POI more often, varying from 20–85% depending on the protocol. Whitehead et al. investigated the effect of combination chemotherapy on ovarian function in females treated for HL. Ovarian function was studied in 44 adult females who previously received mechlorethamine, vincristine, procarbazine and prednisone (MOPP) for HL. The median age at treatment was 23 years, and the length of time between completion of treatment and study ranged from 6 months to 10 years (median, 30 months). Seventeen females (38%) developed amenorrhoea and 10 (22.7%) developed oligomenorrhoea. In another study by Schilsky et al. , persistent amenorrhoea occurred in 11 of 24 patients (46%) treated with the MOPP protocol and the time from diagnosis to amenorrhoea was significantly shorter in older patients (p=0.001). The German Hodgkin Lymphoma Study Group analysed 405 females with HL younger than 40 years of age who were given dose-escalated BEACOPP (bleomycin, etoposide, doxorubicin, cyclophosphamide, vincristine, procarbazine and prednisolone), ABVD, or standard BEACOPP. After a median follow-up of 3.2 years, 51.4% of the females receiving eight cycles of dose-escalated BEACOPP had continuous amenorrhoea. Amenorrhoea was significantly more frequent after dose-escalated BEACOPP compared with ABVD or standard BEACOPP (p=0.006). More females over the age of 30 treated for advanced-stage HL with four cycles of alternating COPP/ABVD or eight cycles of standard BEACOPP or eight cycles of dose-escalated BEACOPP experienced amenorrhoea compared with females younger than 30 years (66.7 vs. 40% with COPP/ABVD, 53.3 vs. 18.2% with standard BEACOPP, 95 vs. 51.4% with dose-escalated BEACOPP).
4.2.3.3
Non-Hodgkin Lymphoma
The incidence of Non-Hodgkin lymphoma (NHL) in females has been reported as 7–9 per 100,000. Most treatment regimens for NHL include alkylating agents. Ellis et al. evaluated a cohort of 36 females younger than 40 years of age with aggressive NHL treated with mostly cyclophosphamide, adriamycin, oncovine and prednisone (CHOP). During treatment, 18 patients (50%) had amenorrhoea, six (17%) had irregular menstrual cycles and 12 (33%) continued their regular cycles. All but two females resumed menses in the first complete remission. Dann et al. analysed 13 females with NHL less than 40 years of age, treated with four cycles of intensified CHOP (cyclophosphamide 2000–3000 mg/m 2 per cycle, doxorubicin 50 mg/m 2 , vincristine 1.4 mg/m 2 , prednisone 100 mg/day) with a median follow-up of 70 months and reported that 12 of 13 patients (92%) experienced recovery of ovarian function.
4.2.3.4
Leukaemia
Leukaemia is the most commonly diagnosed cancer in children, and acute lymphoblastic leukaemia (ALL) accounts for 80% of all leukaemias. Modern chemotherapy protocols of childhood ALL or acute myeloid leukaemia are unlikely to affect ovarian function in many patients due to the absence or lower dose of alkylating agents when haematopoietic stem cell transplantation (HSCT) is not required. However, HSCT requires preconditioning regimens such as alkylating agents with or without total body irradiation, resulting in infertility and POI.
4.2.3.5
Role of Gonadotropin-Releasing Hormone Analogues for the Preservation of Ovarian Function
In several studies, it was shown that when gonadotropin-releasing hormone (GnRH) analogues were given as adjuvant therapy to protect the ovaries from the deleterious effect of chemotherapy, the reported rates of amenorrhoea were lower compared with chemotherapy alone. However, no study has demonstrated a beneficial effect regarding fertility recovery. Since the current data indicate that infertility is increased after chemotherapy, even if menstrual cycles are resumed, further research is required to investigate the role of GnRH agonist administration in fertility preservation. This topic is covered in detail in Chapter 8 .
4.3
How do Chemotherapeutics Damage the Ovary?
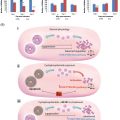
Chemotherapy-induced ovarian damage is associated with a decrease in the follicular pool, impaired blood vasculature and stromal fibrosis. The existing molecular and in vivo evidence show that chemotherapy treatments reduce the ovarian reserve by directly damaging DNA in human primordial follicle oocytes. Based on molecular studies, both cyclophosphamide and doxorubicin affect primordial follicle reserve, which shortens the reproductive lifespan. Both agents are toxic against both the primordial follicle population and those that are in growth phase. Both cyclophosphamide and doxorubicin induce double-strand DNA breaks (DSBs) in oocytes and granulosa cells, which likely triggers a germ cell-specific p53 family member transcription factor TAP-63-mediated apoptotic death. However, there is some indirect evidence that some oocytes may avoid apoptotic cell death when the ataxia telangiectasia mutated (ATM)-mediated DNA DSB repair pathway is activated, thus repairing damaged DNA ( Figure 4.2 ).
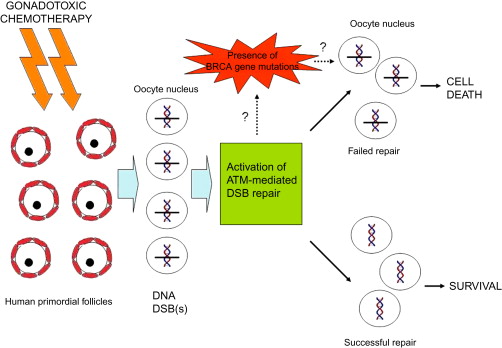
Others have suggested alternative mechanisms based on work in rodents. One idea put forward was that chemotherapy induced massive activation of primordial follicles (“burn out”) via the PI3K/PTEN/Akt pathway, in mice. However, this theory does not explain the increase in malformations of offspring when mouse primordial follicles were exposed to cyclophosphamide, which we have shown to cause DNA DSBs in human primordial follicle oocytes. In addition, it was shown that chemotherapy-induced DNA breaks in primordial follicles can even have transgenerational effects. If the main cause of follicle death was due to the activation of primordial follicles and hence the later impact of chemotherapeutic agents on the more sensitive, now growing follicles, one would not expect to see transgenerational effects or the late malformations that have been reported. While a transient increase in follicle growth initiation may contribute to follicle loss, as has been reported in rodents when ovarian reserve was acutely reduced by unilateral oophorectomy, it is not known if this reaches proportions to explain the significant ovarian damage caused by chemotherapeutic agents in humans.
Granulosa cells are somatic cells that surround the oocyte and proliferate during follicle maturation, and may be another potential target of chemotherapeutic agents. Raz et al. incubated human ovarian cortical slices from premenopausal females with medium containing cyclophosphamide for 2–48 h, and found damage in granulosa cell nuclei and follicular basement membranes. Morgan et al. reported that doxorubicin preferentially damaged the granulosa cells, whereas cisplatin showed more oocyte-specific damage in mouse ovaries. The authors showed previously in a model of human fetal ovary pieces xenografted into severe combined immunodeficient mice that all or nearly all apoptotic follicles had an apoptotic oocyte with or without the pregranulosa cell layer that were also positive after cyclophosphamide treatment. Since granulosa cells can be replenished, apoptosis of the oocyte may be considered more lethal to ovarian reserve.
Ovarian stromal tissue has also been shown to be vulnerable to chemotherapy in human studies. Meirow et al. investigated stromal damage in human ovaries after exposure to chemotherapy in cancer patients. In this study of 35 cancer patients (mean age; 28.7 years), 17 were previously exposed to chemotherapy and 18 were not. After histopathological evaluation, focal cortical fibrosis and blood vessel damage were detected in ovaries of patients previously exposed to chemotherapy. Marcello et al. also reported severe signs of stromal fibrosis and capillary changes in ovarian biopsy specimens of 10 girls who underwent treatment for ALL. The authors previously assessed ovarian samples from 26 patients who were undergoing ovarian tissue cryopreservation for fertility preservation to detect stromal function via measuring in vitro oestradiol levels, and found that regardless of whether they include an alkylating agent, most chemotherapy regimens can have detrimental effects on ovarian stromal function. The authors have also shown in both in vivo xenograft and in vitro organ culture models that gonadotoxic treatments cause microvascular damage, resulting in tissue hypoxia. This may further contribute to the complex damage imparted by cancer treatments on ovarian function.
It has been shown that the ceramide-induced death pathway plays a role in the apoptotic death of mouse oocytes in response to radiation- and chemotherapy-induced damage. Sphingosine-1-phosphate (S1P) is a naturally occurring byproduct of that pathway that can negate the apoptotic consequences of the activation. Numerous studies in rodent models have confirmed that the exogenous administration of S1P protects oocytes against chemotherapy- and radiotherapy-induced death. The authors recently showed that S1P can block the human apoptotic follicle death induced by doxorubicin and cyclophosphamide in a human ovarian xenograft model. Moreover, the research indicates that S1P does not interfere with the effectiveness of chemotherapy while protecting human ovarian reserve against chemotherapy.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree