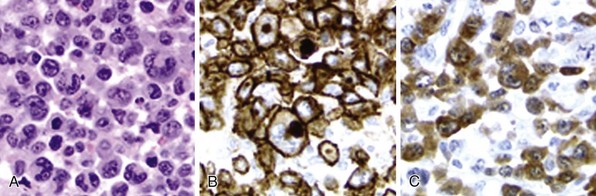
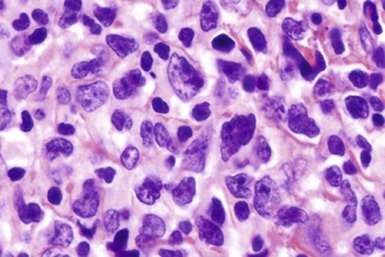
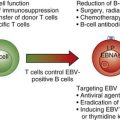
Chapter 37 T-Cell Lymphomas
Treatment of Peripheral T-Cell Lymphoma
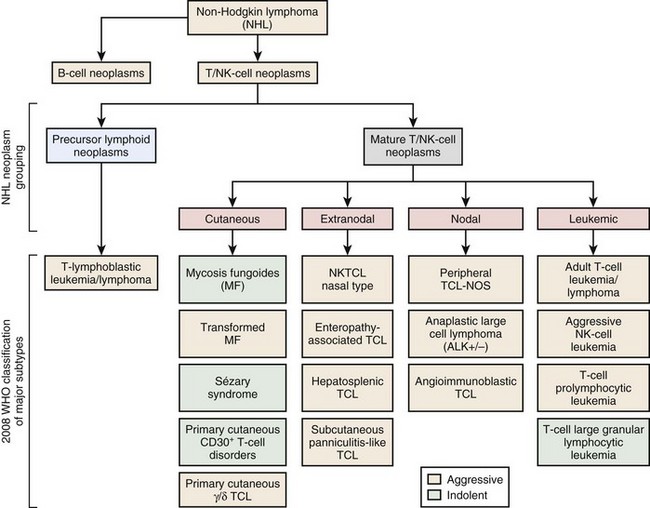
Figure 37-1 WHO CLASSIFICATION OF THE MATURE T-CELL NEOPLASMS.
NK, Natural killer; NOS, not otherwise specified; TCL, T-cell lymphoma.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree