Subclinical Infection as a Cause of Premature Labor
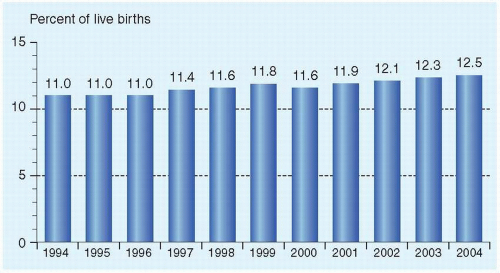
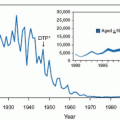
Preterm birth, with its subsequent morbidity and mortality, is the leading perinatal problem in the United States. In 2004, the rate of preterm birth was 12.5% (up from 11.0% in 1994) (Fig. 18.1). Preterm infants (defined as infants born before the 37th week of gestation) account for 70% of all perinatal deaths and half of long-term neurologic morbidity.
Evidence from many sources links preterm birth with symptomatic infections. Untreated bacteriuria in pregnancy results in acute pyelonephritis in 20% to 40% of bacteriuric patients, and pyelonephritis has a seriously increased risk of fetal morbidity and mortality. In the preantibiotic era, many systemic maternal infections, such as pneumonia, often led to premature birth. In the last decade, great interest has been generated by subclinical infection as a cause of premature labor, and much new information has been presented, as categorized in Table 18.1. Additional exciting information has suggested that subclinical infection is responsible not only for preterm birth but also for many serious neonatal sequelae, including periventricular leukomalacia, cerebral palsy, respiratory distress syndrome, and even bronchopulmonary dysplasia and necrotizing enterocolitis.
THE INFECTION-INFLAMMATION HYPOTHESIS
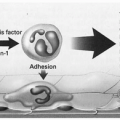
The hypothesis linking subclinical infection and premature birth has been developing for over 30 years. Microbes or microbial toxins such as endotoxin (lipopolysaccharide) enter the uterine cavity during pregnancy, primarily by the ascending route from the lower genital tract but, on occasion, by the bloodborne route from a nongenital focus. A complex interaction occurs between the microbes or their products, most likely in the decidua or possibly in the membranes. This interaction, probably mediated through toll-like receptors and intracellular transcription factors, leads to inflammatory responses, which, in turn, lead to cervical ripening, myometrial contractions, membrane rupture, and preterm birth (Fig. 18.2). We now critique the evidence.
HISTOLOGIC CHORIOAMNIONITIS IS INCREASED IN PRETERM BIRTH
One of the most consistent observations linking subclinical infection and preterm birth is the increased likelihood of histologic chorioamnionitis in cases of preterm birth. Most cases of histologic chorioamnionitis are caused by infection. In preterm membranes, there is a very strong association between positive membrane cultures and degree of membrane infiltration. When birth weight was greater than 3,000 g, the percentage of placentas showing histologic chorioamnionitis was less than 20%; when birth weight was less than 1,500 g, the percentage was 60% to 70% (Fig. 18.3).
CLINICAL INFECTION IS INCREASED AFTER PRETERM BIRTH
Another consistent observation is that clinically evident infection is increased in both neonates and mothers after preterm birth. For example, sepsis and meningitis are increased threefold to 10-fold in preterm infants. Less widely recognized is the increase in maternal infection after preterm birth. In a study of 8,400 term and 1,250 preterm births at the University of Colorado, clinical chorioamnionitis, postpartum endometritis, and neonatal infection were all significantly increased among preterm pregnancies, even after correction for the presence of premature rupture of the membranes (PROM). These observations are consistent with the concept that subclinical infection was the cause of the preterm birth and that the infection became clinically evident during or shortly after birth. An alternative explanation is that the preterm infant simply is more susceptible to infection developing after delivery.
THERE ARE ASSOCIATIONS OF PRETERM BIRTH OR PREMATURE RUPTURE OF THE MEMBRANES WITH VARIOUS MATERNAL LOWER GENITAL TRACT ISOLATES OR INFECTIONS
Several surveys have found statistically significant associations between lower genital infection and adverse pregnancy outcome. Early studies reported that Ureaplasma urealyticum in the lower genital tract was associated with low birth weight (LBW) infants and that in couples with histories of pregnancy wastage and with positive genital or urinary cultures for genital mycoplasmas, doxycycline or erythromycin treatment reduced the pregnancy loss.
However, better-designed studies reported no associations of U. urealyticum with any adverse pregnancy outcome in a large collaborative study. This study included more than 4,500 patients and tested for lower genital tract microbes including bacteria, mycoplasmas, and chlamydia. The analysis
shown in Table 18.2 excludes patients with either group B streptococci (GBS) or chlamydia, confounding organisms that have been implicated in preterm birth. Interestingly, U. urealyticum in the lower genital tract is not associated with LBW/preterm pregnancies, but this organism is one of the most commonly isolated in the amniotic fluid of women in preterm labor.
shown in Table 18.2 excludes patients with either group B streptococci (GBS) or chlamydia, confounding organisms that have been implicated in preterm birth. Interestingly, U. urealyticum in the lower genital tract is not associated with LBW/preterm pregnancies, but this organism is one of the most commonly isolated in the amniotic fluid of women in preterm labor.
In early studies of lower genital infection with Chlamydia trachomatis, results regarding adverse pregnancy outcome has been mixed. In one study, for example, pregnant women with endocervical C. trachomatis infection at 24 weeks were significantly more likely to deliver prematurely, but those infected at 28 weeks were not (Fig. 18.4).
An association between maternal GBS colonization and premature birth has not been found consistently. In a large National Institutes of Health (NIH)-sponsored investigation of approximately 13,000 women, pregnancy outcome was investigated for three groups of women: those without GBS, those with high-density lower genital tract colonization, and those with low-density colonization. Compared with the 10,295 noncolonized women, those with heavy GBS colonization had a small but significant increase in risk for LBW (odds ratio [OR], 1.2; 95% confidence interval [CI], 1.01 to 1.5). There was no significant increase in other adverse outcomes, including preterm birth, for heavily colonized women. Women with light colonization were not at an increased risk for any adverse outcomes. In a randomized treatment trial of erythromycin versus placebo in GBS-colonized women, erythromycin use was not shown to be effective in prolonging gestation or increasing birth weight.
TABLE 18.1 ▪ EVIDENCE FOR SUBCLINICAL INFECTION AS A CAUSE OF PRETERM BIRTH | ||
|---|---|---|
|
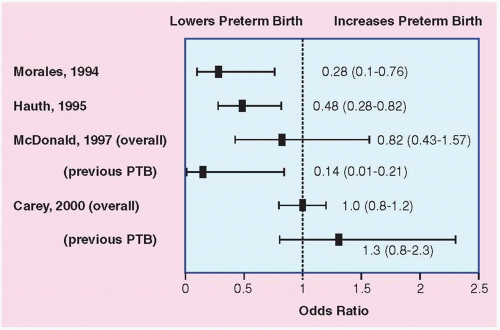
Several studies found an increased odds radio for preterm birth among women with T. vaginalis infection (Fig. 18.4). In the large Vaginal Infections and Prematurity Study, lower genital tract carriage of T. vaginalis at midpregnancy was significantly associated with preterm LBW. Preterm LBW occurred in 7.1% of women with T. vaginalis compared with 4.5% of women without T. vaginalis (OR, 1.6; 95% CI, 1.3 to 1.9).
Bacterial vaginosis (BV), a vaginal condition characterized by a predominance of anaerobes, has consistently been associated with preterm birth. Overall, patients with BV have an approximately twofold to threefold increase in spontaneous preterm delivery (Fig. 18.4). It is unclear whether BV causes preterm delivery by leading to intrauterine subclinical infection or whether BV acts locally in the lower genital tract infections, as it is associated with increased concentrations of elastase, mucinase, and sialidase.
Untreated pyelonephritis has been associated with an approximately 24% risk of preterm delivery, and asymptomatic bacteriuria is associated with a 60% higher rate in LBW (95% CI, 1.4 to 1.9) and a 90% higher rate of preterm delivery (95% CI, 1.3 to 2.9). Untreated gonococcal infection of the lower genital tract also has been associated with preterm delivery.
ORGANISMS ARE FOUND IN AMNIOTIC FLUID/MEMBRANES/DECIDUA OF PATIENTS IN PREMATURE LABOR
When amniotic fluid is obtained by amniocentesis from asymptomatic women in premature labor, the range of positive cultures is 3% to 24%, an eightfold difference. Part of these widely ranging results may be explained by patient selection. For example, it is possible that investigators reporting a low culture positivity rate (3% to 4%) performed amniocentesis on patients in “prodromal” or “false” premature labor. Women in preterm labor and with bacteria or Candidia albicans in the amniotic fluid are less likely to respond to tocolytics and more likely to deliver promptly.
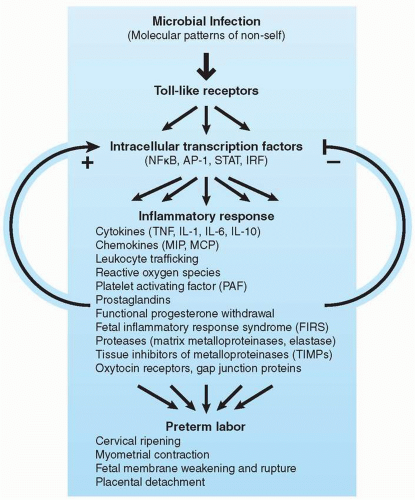 FIGURE 18.2 Simplified model of the pathophysiology of infection-induced preterm labor triggered by activation of the maternal and fetal innate immune systems. The proposed signaling cascade likely involves a high degree of positive and negative feedback looping, multiple redundant pathways, and interaction between the maternal and fetal components (depicted schematically in the figure by arrows; however, it should be noted that these interactions occur in several directions and on several levels simultaneously). NF-κB, nuclear factor κB; AP-1, activator protein 1; STAT, signal transducers and activators of transcription; IRF, interferon regulatory factor; TNF, tumor necrosis factor; IL, interleukin; MIP, macrophage inflammatory protein; MCP, macrophage/monocyte chemoattractant protein. (From Hirsch E, Wang H. The molecular pathophysiology of bacterially induced preterm labor: insights from the murine model. J Soc Gynecol Investig 2005;12(3):145-155, with permission.) |
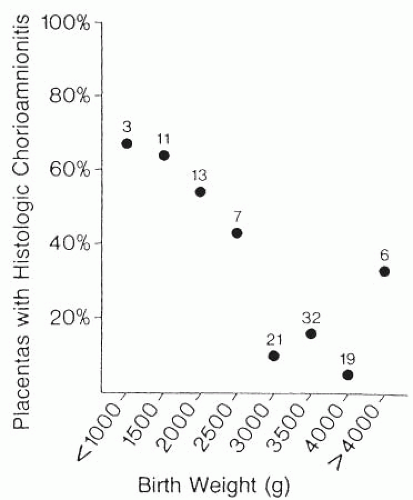 FIGURE 18.3 Placentas with histologic chorioamnionitis by gestational age. (From Hillier SL, Martius J, Krohn M. A case controlled study of chorioamnionitis infection and histologic chorioamnionitis. N Engl J Med 1988;3198:972-978, with permission.) |
The most likely route for bacteria to enter the amniotic fluid of patients in preterm labor is an ascending path through the vagina and cervix. It also is possible to speculate that bacteria may enter the uterine cavity hematogenously through spread via the placenta. Potential sources of organisms for hematogenous spread include bacteremia from renal or periodontal disease.
Among women in spontaneous preterm labor with intact membranes, genital mycoplasmas and anaerobic organisms, as well as Gardnerella vaginalis (the so-called BV organisms), are those organisms most commonly found in the amniotic fluid. Sexually transmitted organisms, such as Neisseria gonorrhoeae and C. trachomatis, are rarely found in the amniotic fluid, and GBS and Escherichia coli are found only occasionally. When organisms from the lower genital tract ascend into the uterine cavity, it is most likely that the initial site is in the choriodecidual space. In some women, bacteria cross the intact membranes into the amniotic fluid; in some cases, the fetus becomes infected.
TABLE 18.2 ▪ UREAPLASMA UREALYTICUM AND ADVERSE PREGNANCY OUTCOME (N = 4,576) | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||
Women in preterm labor with the highest likelihood of having a positive culture of the amniotic fluid are those patients in very early preterm labor. It may be speculated that intrauterine infection occurs early in pregnancy—or even preceded the pregnancy—and may remain without clinical detection for months. Evidence to support such chronic infection arises from studies showing positive cultures for ureaplasmas from amniotic fluid taken for routine chromosome analysis at midpregnancy. In addition, high fetal fibronectin concentrations in the lower genital tract at 24 weeks (possibly suggestive of an upper genital tract infection) have been associated with development of chorioamnionitis almost 2 months later.
THERE ARE “MARKERS” OF INFECTION IN PREMATURE LABOR
As noted earlier, when premature labor is induced by infection, the primary site of infection is not the amniotic fluid but the decidua or membranes. Thus, looking for organisms in amniotic fluid may be an insensitive diagnostic technique. Accordingly, studies have sought potentially more sensitive markers of infection. These markers have been identified both in women presenting with signs and symptoms of preterm labor as well as in asymptomatic patients (usually at risk for preterm labor) during prenatal care. As shown in Table 18.3, these specimens have been obtained from the amniotic fluid, the vagina and cervix, and the serum. Despite the variety of these markers, relatively few are clinically useful. Among patients in preterm labor, a low amniotic fluid glucose correlates well with the likelihood of a positive culture. Observing bacteria or white cells on a Gram stain also is useful, as is absolute white blood count in the amniotic fluid. Among the cytokines, an elevated amniotic fluid interleukin (IL)-6 level probably is the most sensitive marker for infection but is not yet widely available for clinical use.
It has not been established that the results of amniotic fluid testing in patients in preterm labor improve the outcome of pregnancy. Thus, we use amniocentesis selectively among patients in preterm labor. Examples of such cases are patients who may have subtle signs and symptoms of infection, patients who do not respond well to tocolytics, and patients with recurrent preterm labor.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree