Introduction
Prior to the 1990s, cytotoxic chemotherapy was used uniformly for all malignancies. This resulted in heterogeneous responses even within a specific tumor type. As insight was gained about the role of driver mutations (key alterations in the oncogenic addiction pathways of malignant cells), the concept of targeted therapies was born. By inhibiting driver pathways for carcinogenesis, these targeted agents were successful in managing cancers that did not have robust responses to cytotoxic chemotherapy. Imatinib, a tyrosine kinase inhibitor targeting the BCR-ABL1 fusion gene seen in chronic myeloid leukemia, and trastuzumab for human epidermal growth factor receptor 2 (HER2)-positive breast cancer, were among the earliest targeted agents discovered. The success of these agents heralded an era of targeted therapies that are now being used in a broad spectrum of solid tumors and hematologic malignancies. With genomic sequencing being used more frequently than in the past, newer therapeutic targets are being discovered. With these discoveries come targeted drug development that will lead to many more targeted therapies in the coming years.
There are some limitations to molecular-targeted therapies. In malignancies that develop in the presence of carcinogen or environmental exposures, often multiple coexisting mutations are observed. In such malignancies, targeting a single pathway either leads to a complete lack of response or development of early resistance by a variety of escape pathways. Some examples of such cancers are non–small-cell lung cancer (NSCLC) related to smoking or melanoma as a result of exposure to ultraviolet (UV) radiation. Even in malignancies that are dependent on a driver mutation for oncogenesis, tumor cells eventually develop resistance to targeted agents via alternate pathways.
In the current era, molecular-targeted therapies play an important role in the treatment of multiple tumor types. Preclinical work has shown that these agents demonstrate clinical efficacy when combined with other therapeutic modalities such as immunotherapy. Ongoing clinical trials are exploring the utility of combining these molecular-targeted therapies with immunotherapies; therefore it is quite likely that the clinical application of targeted agents will continue to rise in the foreseeable future.
As targeted therapies are being widely used, it is imperative that prescribing clinicians are aware of the potential for toxicity and monitor patients closely while on therapy. In this chapter, we will discuss pulmonary toxicities of targeted therapies. Similar to other drugs, lung toxicity with targeted therapy may be idiosyncratic, or may be more predictable when related to cumulative dosing of the drug. By having a low-degree of suspicion in patients on therapy with drugs that have the potential for pulmonary toxicity and early intervention, morbidity and mortality can be significantly reduced. In the next section, we describe the various targeted therapies listed by class and their specific pulmonary toxicities including the pathogenesis, risk factors, and clinical manifestations.
Agents and Mechanism of Action
EPIDERMAL GROWTH FACTOR RECEPTOR (EGFR)TYROSINE KINASE INHIBITORS
- ■
Driver mutations in the EGFR gene are seen in close to 15% of patients with NSCLC in the United States and in over 60% in Asian populations, possibly due to the lower number of smokers. , Patients with EGFR mutations have a better prognosis than other NSCLC patients and usually have favorable responses to therapy with EGFR tyrosine kinase inhibitors (TKIs). The EGFR TKIs that are currently being used in clinical practice include first-generation reversible TKIs, erlotinib and gefitinib, the second-generation irreversible TKI, afatinib, and the third-generation TKI, osimertinib. Since the results of the FLAURA trial, which compared outcomes with first-line osimertinib versus erlotinib or gefitinib demonstrated superior overall survival, progression-free survival, tolerability, reduction in intracranial metastases, and activity against the T790M resistance-mutated lung cancer, osimertinib has become the agent of choice for frontline therapy for many practitioners.
- ■
Incidence: In a meta-analysis of 15 trials studying a total of 2201 patients treated with first- and second-generation EGFR TKIs, the most frequent etiology of mortality related to toxicity of the drug was pneumonitis (11 deaths, comprising 65% of the total reported deaths). Overall, the incidence of interstitial lung disease (ILD) with EGFR TKIs is less than 5%, but the associated mortality can range from 0.6% with osimertinib to as high as 31%, which has been reported with gefitinib. ,
- ■
Risk factors: Risk factors for pulmonary toxicity include preexisting pulmonary disease, smoking, and radiation exposure. A higher incidence of gefitinib-induced ILD was noted in men (6.6%) compared with women (3.3%) in an analysis of over 1900 Japanese patients treated with the agent. Patients usually become symptomatic in the first few months of treatment.
- ■
Pathophysiology: EGFR pathways are essential for the turnover and repair of the alveolar wall. They are expressed on type II pneumocytes. By inhibiting the EGFR pathway and impairing the repair mechanism, EGFR TKIs can not only induce alveolar damage themselves, but also increase susceptibility to other injury mediated by infections, radiation, or other drugs. , ,
Specific EGFR–Targeting Agents
- ■
Gefitinib: The incidence of gefitinib-associated ILD has been reported to be slightly different in postmarketing experience in the United States (0.3%) versus Asian populations (2%). Patients usually present with dyspnea, with or without cough, and low-grade fever, with a median onset of symptoms between 24 and 42 days. Endo et al. performed a multi-institutional analysis of the various radiographic manifestations of ILD and classified them into four major patterns: (1) nonspecific ground-glass opacities, (2) multifocal airspace consolidations, (3) patchy ground-glass opacities with septal thickening, and (4) extensive ground-glass opacities or consolidations with traction bronchiectasis ( Fig. 14.1 ). The majority of the patients had areas of ground-glass opacities, or a pattern of extensive parenchymal involvement (fourth pattern), which reflects diffuse alveolar damage. The fourth pattern was associated with the highest mortality. The histological findings on pathology include interstitial pneumonitis or fibrosis and other less common findings such as diffuse alveolar damage, organizing pneumonia, hypersensitivity, or eosinophilic pneumonitis.
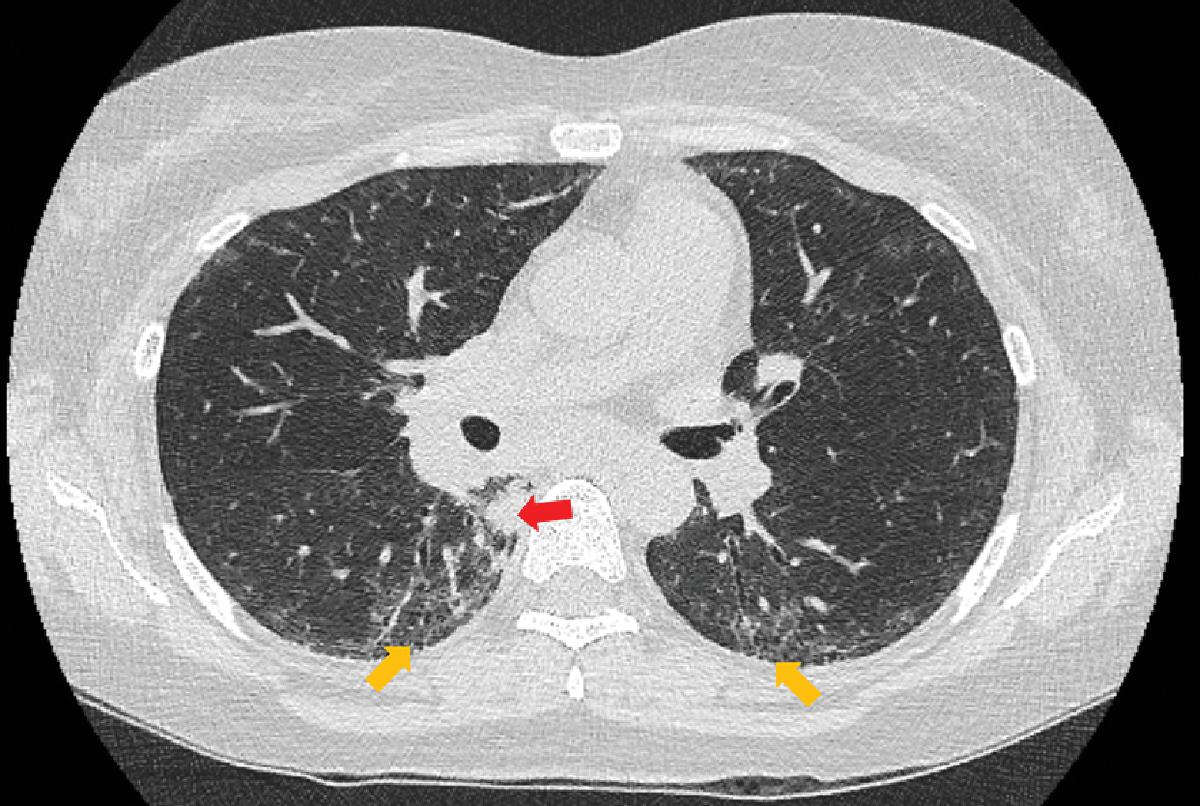
Fig. 14.1
Basilar traction bronchiectasis (yellow arrows) with rare ground glass opacities consistent with a nonspecific interstitial pneumonia pattern in a patient treated with gefitinib for non–small-cell lung cancer with an epidermal growth factor receptor (EGFR). The red arrow points to the residual tumor.
- ■
Erlotinib: The overall incidence of ILD with erlotinib is approximately 1.1% ( Fig. 14.2 ). In an analysis of 9907 Japanese patients treated with erlotinib in the phase IV POLARSTAR surveillance study, the reported incidence was 3.4% to 5.1%. Risk factors for ILD include preexisting pulmonary fibrosis or lung disease, radiation, and use of other drugs with potential for pulmonary toxicity, such as gemcitabine. Clinical presentation and imaging findings are similar to gefitinib-induced lung disease.
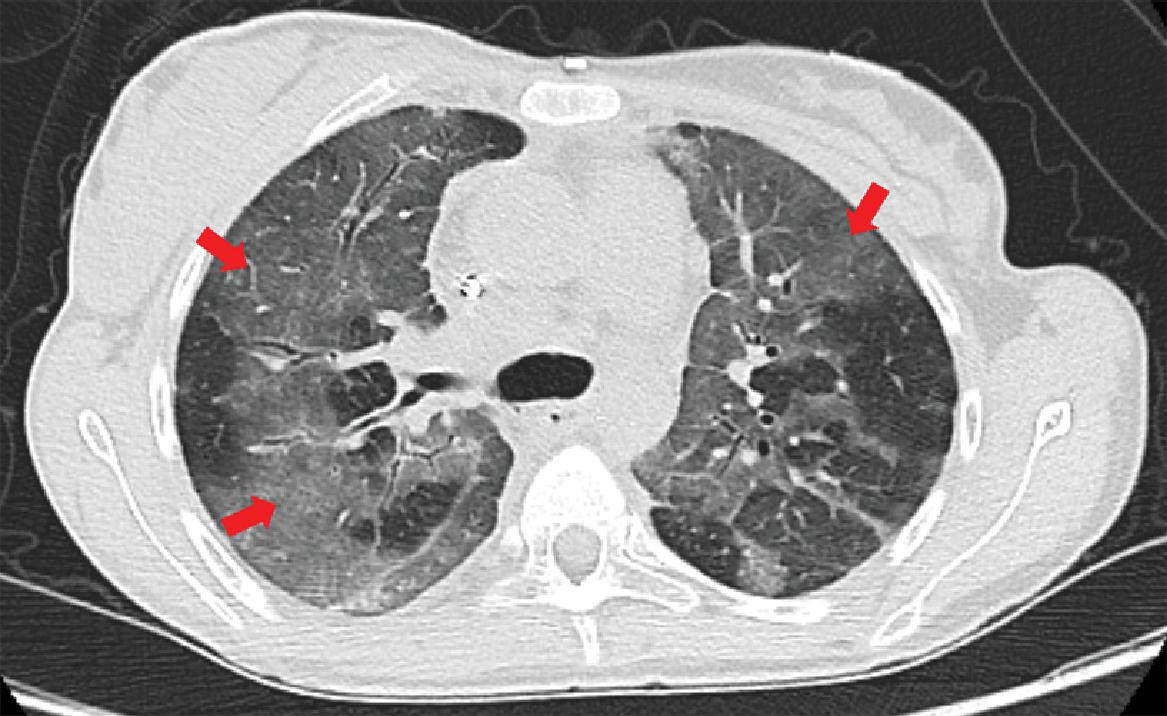
Fig. 14.2
Diffuse ground glass opacities (red arrows) in a patient with Stage IV epidermal growth factor receptor (EGFR)-mutated non–small-cell lung cancer treated with erlotinib.
- ■
Afatinib: Afatinib is an irreversible ErbB family TKI. Three out of 230 patients in the LUX-Lung 3 trial developed ILD. One out of 242 patients in the LUX-Lung 6 trial had fatal ILD secondary to afatinib.
- ■
Osimertinib: The overall incidence of pneumonitis has been reported to be around 3.5%. Osimertinib is an irreversible EGFR-TKI that selectively inhibits EGFR sensitizing mutations, as well as the resistance mutation, T790M, but has lesser activity against wild-type EGFR, which could be a potential explanation for the lower grade 3 and 4 adverse events noted in comparison with erlotinib or gefitinib. The clinical manifestations appear to be similar to other EGFR TKIs. A unique pulmonary manifestation of osimertinib, which has been described in literature, is transient asymptomatic pulmonary opacities that occur at a median time of 8.7 weeks (range 1.6–43 weeks) into therapy and last for a median duration of 6 weeks (range 1–11 weeks). Patients with these asymptomatic transient opacities may be continued on therapy with close monitoring.
Anaplastic Lymphoma Kinase (ALK) Inhibitors
- ■
The identification of the role ALK rearrangements in 2% to 7% of patients with NSCLC in 2007 led to the development of ALK inhibitors to target the EML4-ALK fusion oncogene. The ALK inhibitors that have been used in clinical practice since 2011 include crizotinib (a multitargeted small molecule TKI) and second-generation agents including ceritinib, alectinib, and brigatinib. Although crizotinib was the first targeted agent to be approved, in the global ALEX study, alectinib demonstrated superior efficacy in terms of progression-free survival and central nervous system (CNS) progression and had lower toxicity in comparison to crizotinib. Although the overall incidence of pneumonitis with ALK inhibitors is low, many of these can be severe and life threatening. Among patients treated with the earlier ALK inhibitors crizotinib and ceritinib, 1% to 4% develop pneumonitis. Alectinib has been associated with a lower incidence of pneumonitis (0.4%). Brigatinib has a higher incidence of pulmonary toxicity, with 3.7% of patients in the 90 mg group and 9.1% of patients in the 180 mg after a 90-mg lead-in group reported in the ALTA trial. Pneumonitis developed in 6.4% of patients within 9 days of initiation of brigatinib-therapy, with a median onset of 2 days. Therefore patients who are initiated on brigatinib should be monitored very closely for respiratory symptoms in the first few weeks of therapy. ,
BCR-ABL1 Tyrosine Kinase Inhibitors
- ■
Imatinib: Imatinib is a TKI that inhibits BCR-ABL, the constitutive abnormal gene product of the Philadelphia chromosome, in addition to other targets in such as platelet-derived growth factor (PDGFR), stem cell factor, and c-Kit. Imatinib has clinical activity in chronic myelogenous leukemia (CML), Philadelphia chromosome positive acute lymphoblastic leukemia (ALL), certain hypereosinophilic syndromes, and gastrointestinal stromal tumors (GISTs). A variety of pulmonary toxicities have been associated with the use of this drug including pleural effusions, interstitial pneumonitis, hypersensitivity pneumonitis, and eosinophilic pneumonia.
- ■
The most common pulmonary complication of imatinib is secondary to the associated fluid retention, which can lead to pleural effusions and pulmonary edema. In patients with CML, 1.3% of newly diagnosed patients and 2% to 6% of other CML patients on imatinib develop severe fluid retention. The incidence of severe fluid retention is higher in patients with GIST at 9% to 13.1%.
- ■
The median time to onset of interstitial pneumonitis was 49 days (range 10–282 days) in the largest case series of 27 patients with imatinib-induced ILD. Notably, there was no clear correlation between the dose or duration of therapy and development of pneumonitis. Among these patients, 41% had preexisting pulmonary disease, suggesting a predisposition in this patient population. Imaging findings in these patients revealed a hypersensitivity pattern (30%) ( Fig. 14.3A – C ), interstitial pneumonitis (26%), and cryptogenic organizing pneumonia (15%) ( Fig. 14.4 ), a peribronchovascular bundle pattern (15%), and a nodular pattern in 11% of patients. Transbronchial lung biopsies were performed in five patients which demonstrated varying degrees of inflammatory changes and fibrosis. Peripheral eosinophilia was noted in 5 of 27 patients.
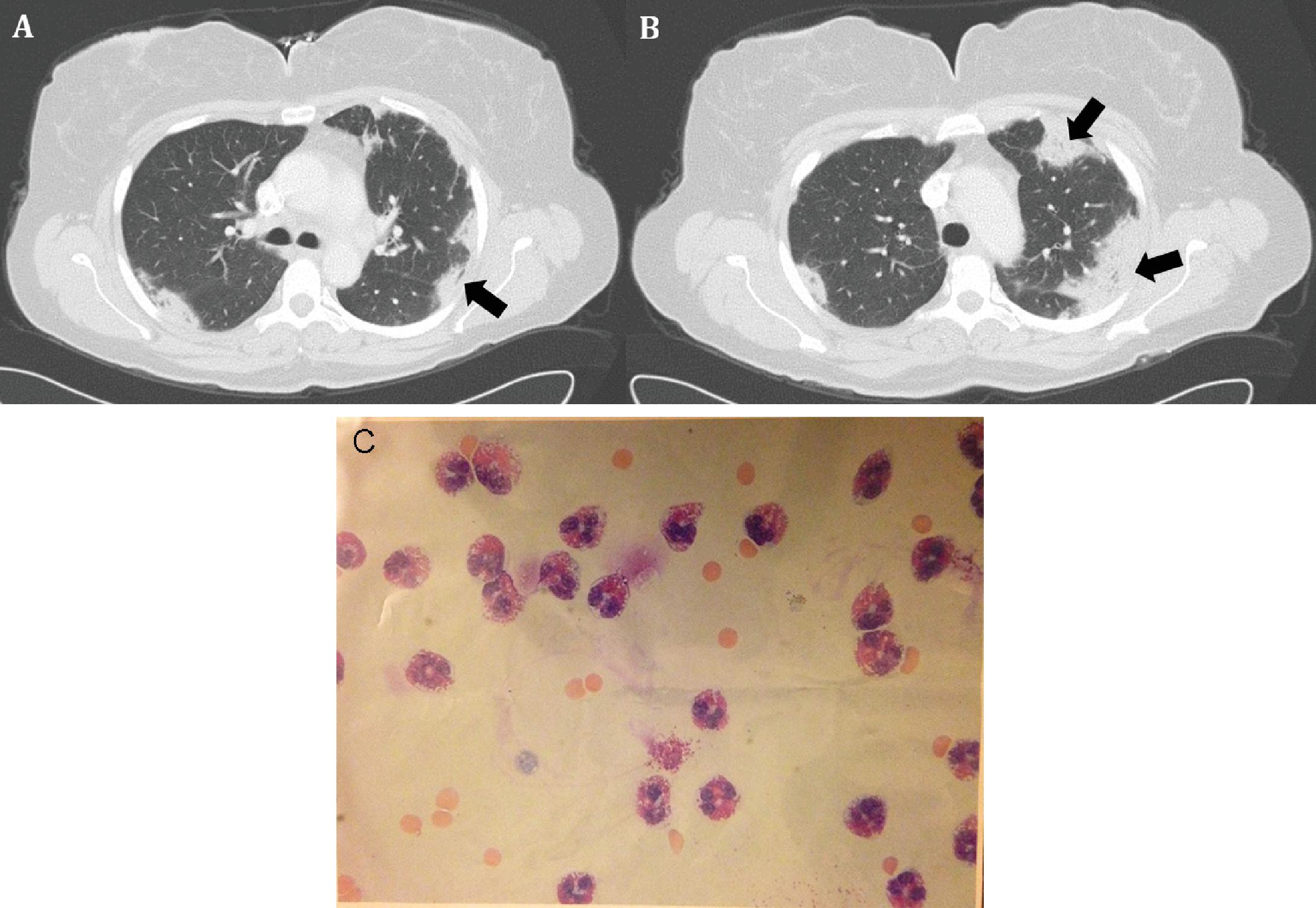
Fig. 14.3
(A, B) Dense peripheral consolidations (black arrows) in a patient treated with imatinib. Bronchoalveolar lavage showed 40% eosinophils on differential counts consistent with a diagnosis of chronic eosinophilic pneumonia. (C) Forty percent eosinophils noted on the smear of the bronchoalveolar lavage obtained from the patient consistent with a diagnosis of chronic eosinophilic pneumonia.
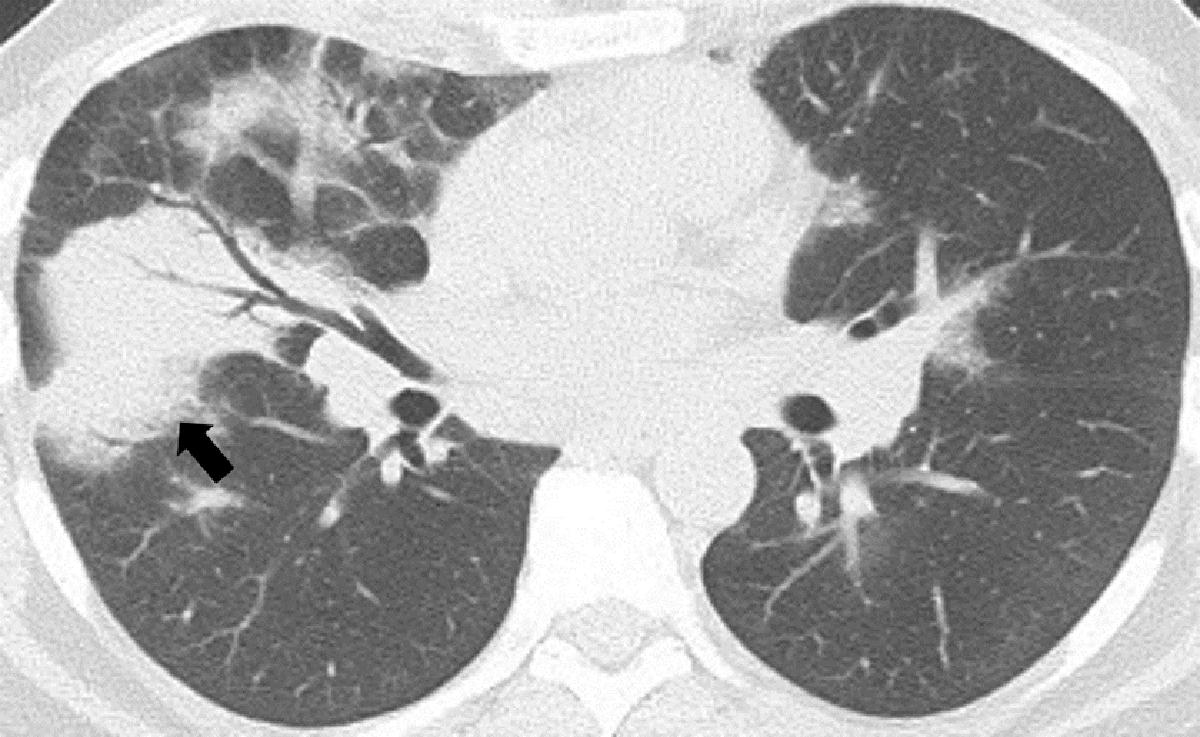
Fig. 14.4
Dense consolidations (black arrow) on computerized tomogram of a patient who was treated with imatinib. Transbronchial biopsy findings were consistent with a diagnosis of organizing pneumonia.
- ■
- ■
Dasatinib: Dasatinib is a second-generation BCR-ABL TKI that binds to both active and inactive conformations of the ABL gene and is 325 times more potent than imatinib in inhibiting the growth of BCR/ABL cells in vitro. It is predominantly used in the treatment of CML. It has the highest incidence of reported pulmonary adverse events.
- ■
The most common pulmonary toxicity reported with dasatinib is the development of pleural effusions. The median time to onset of pleural effusions is 11 months (range3.6–18.6 months). In a 5-year analysis of patients from the DASISION trial comparing dasatinib to imatinib, the overall incidence of pleural effusions was 28% in the dasatinib group versus 0.8% in the imatinib group. Patients who were older than65 years had a higher incidence of pleural effusions. Some reports suggest that the rate of recurrent pleural effusions can be as high as 15%. Other risk factors that have been associated with the development of pleural effusions include prior pulmonary disease in patients treated with higher doses (140 mg).
- ■
Pulmonary hypertension has also been noted with dasatinib in an estimated 5% of treated patients, however, because all reported patients did not have confirmatory right heart catheterizations, the exact incidence is unknown. Based on data from the French Pulmonary Hypertension registry, pulmonary hypertension occurs after 8 to 48 months of exposure. Patients in this registry developed precapillary pulmonary hypertension. Evidence suggests that receptors of tyrosine kinases (RTKs) such as PDGFR, fibroblast growth factor 2, c-KIT, c-Src, and epidermal growth factor, play a role in the pathogenesis of pulmonary hypertension. As an example, the src tyrosine kinase pathway is essential for the activation of K + channels in the pulmonary smooth muscle cells, which results in muscle relaxation. TKIs that inhibit this pathway result in pulmonary vasoconstriction and vascular remodeling over time, resulting in pulmonary hypertension. Unlike imatinib and nilotinib, dasatinib is a potent inhibitor of RTKs that may be responsible for the higher incidence of pulmonary hypertension observed with the drug. Whereas in some patients pulmonary hypertension may be reversible, most patients do not recover completely despite discontinuation of the drug.
- ■
The other less common pulmonary toxicity of dasatinib is pneumonitis, which is usually reversible with interruption of therapy.
- ■
- ■
Bosutinib: Up to 8% of patients treated with bosutinib can develop pleural effusions. Additionally, a case report of worsening preexisting pulmonary hypertension while on bosutinib has been described.
- ■
Nilotinib: Pulmonary toxicity is rare and is seen in less than 1% of treated patients.
- ■
Ponatinib: For patients with CML who develop the T315I resistance mutation, ponatinib is the drug of choice. Pleural effusions can be seen in 1% of treated patients. Other pulmonary toxicities are rare. A case report of a patient who had been treated with dasatinib in the past and developed pulmonary hypertension while on treatment with ponatinib has been described.
Vascular Endothelial Growth Factor (VEGF) Inhibitors
- ■
Sunitinib, sorafenib, and pazopanib: Sunitinib, sorafenib, and pazopanib are oral multikinase inhibitors that are used in the therapy of renal cell cancer, hepatocellular carcinoma, thyroid cancer, and GIST. Similar to other antiangiogenic agents, pulmonary hemorrhage can be seen with sunitinib. In clinical trials, up to 26% of patients who received sunitinib complained of dyspnea, but only 6% had grade 3 or higher toxicity. Other respiratory symptoms include flu-like symptoms or upper respiratory tract infections and nasopharyngitis.
- ■
Dyspnea, cough, and upper respiratory tract symptoms are less common with sorafenib. In patients who develop sorafenib-induced lung injury (incidence <1%, peak time to onset within 2–4 weeks), the mortality is 41% in those with a diffuse alveolar damage pattern on imaging.
- ■
A unique pulmonary toxicity observed in patients treated with pazopanib is the development of pneumothoraces, which have been reported in 3% to 14% of treated patients. Patients with lung metastases with pleural or subpleural involvement seem to have a higher incidence of pneumothoraces, possibly due to the formation of pleural fistulas, as a result of tumor necrosis. ,
- ■
- ■
Bevacizumab: Bevacizumab is a recombinant monoclonal antibody against VEGF, which is used in the treatment of patients with NSCLC, colorectal cancer, and glioblastoma, among other malignancies. In patients with lung cancer, bevacizumab use can lead to central tumor cavitation in 14% to 25% of cases. , In patients with lung cancer of squamous histology, a high incidence of pulmonary hemorrhage and hemoptysis (4 out of 13) was noted in a phase II clinical trial, and subsequently, the use of bevacizumab for squamous NSCLC has been discouraged. Pulmonary hemorrhage is uncommon with bevacizumab in other tumor types. Another unique side effect of bevacizumab, which occurs in patients who have been exposed to radiation in the past, or are receiving concurrent chemoradiation, is the development of a tracheoesophageal fistula. Because bevacizumab has been associated with an increased risk of venous thromboembolism, pulmonary embolism should be considered in the differential diagnosis of a patient presenting with a new onset dyspnea and hypoxia.
Human Epidermal Growth Factor Receptor 2(HER2)-Targeting Antibodies
- ■
Trastuzumab: Trastuzumab is used in the treatment of HER2-positive breast cancers and gastric malignancies that express HER2. Similar to other monoclonal antibodies, infusion reactions are seen in 20% to 40% of patients receiving the drug. These reactions often present with pulmonary symptoms such as hoarseness and shortness of breath. A higher incidence of infusion reactions may be seen when trastuzumab is given in combination with other chemotherapeutic agents. In a postmarketing analysis, 0.3% of patients who received the drug had severe infusion reactions with an anaphylactoid presentation.
- ■
Other rare but potentially fatal pulmonary toxicities reported with trastuzumab include interstitial pneumonitis, , organizing pneumonia, pleural effusions, noncardiogenic pulmonary edema, acute respiratory distress syndrome, and pulmonary fibrosis. Bronchoalveolar lavage in these patients may reveal a predominantly eosinophilic or neutrophilic fluid. In addition to preexisting ILD, patients with extensive lung metastases are also at an increased risk.
- ■
- ■
Ado-trastuzumab emtansine (T-DM1): Ado-trastuzumab emtansine is an antibody–drug conjugate of trastuzumab that is used in advanced breast cancer patients who have previously received trastuzumab. Infusion reactions are much less frequent than with trastuzumab and are usually very mild. Cases of ILD have been reported in 0.8% of patients treated with T-DM1 and may be severe enough to lead to acute respiratory distress syndrome.
- ■
Pertuzumab: Hypersensitivity reactions during infusions are seen in 13% of patients, however, most of them are very mild with less than 1% being grade 3 or higher.
CD20-Targeting Agents
- ■
Rituximab: Rituximab is a murine human chimeric monoclonal antibody targeting the CD20 molecule on the surface of B lymphocytes. By binding to CD20, it activates antibody-dependent cellular cytotoxicity and complement-dependent cytotoxicity and is therefore used in the treatment of chronic lymphocytic leukemia (CLL), non-Hodgkin lymphoma, and CNS lymphoma, in addition to many other nonmalignant conditions that require immunosuppression, such as autoimmune hemolytic anemia and rheumatoid arthritis, among others.
- ■
Infusion reactions occur with the first infusion of this drug in over 50% of patients, however, incidence of severe reactions accompanied by bronchospasm or hypotension are seen in less than 10% of patients. These usually occur within 2 hours of infusion initiation. Some patients may also develop pulmonary infiltrates and acute respiratory distress syndrome with rituximab infusions. These reactions are secondary to cytokines and tumor necrosis factor α that are released as a result of the cytotoxic effect on B lymphocytes by rituximab. Therefore patients with CLL, mantle cell lymphoma, or malignancies with a high tumor burden are more likely to develop a reaction to rituximab. In these patients, using a fractionated regimen of rituximab may be safer. Infusion reactions tend to be less severe with subsequent administrations of the drug.
- ■
Interstitial pneumonitis is rare but has been reported in patients who received chemotherapeutic regimens containing rituximab for lymphomas. ,
- ■
EGFR-Targeting Antibodies
- ■
Cetuximab: Cetuximab is a recombinant chimeric monoclonal antibody, which binds to the EGFR and inhibits binding with EGFR ligands, thereby activating the receptor-associated kinases. Cetuximab is predominantly used in KRAS wild-type colorectal cancer and head and neck cancer. Cetuximab has been associated with severe infusion reactions, which may be associated with rapid onset of airway obstructive symptoms such as bronchospasm and hoarseness of voice. These infusion reactions have been reported in 2% to 5% of patients receiving the drug and may occur despite premedication with antihistamines. ILD is a rare but potentially fatal adverse effect of cetuximab. Four out of 1570 patients included in the original clinical trials testing this drug developed ILD, out of which one was fatal. Older patients or those with prior lung disease appear to be at an increased risk.
- ■
Panitumumab: Panitumumab is a recombinant human monoclonal antibody targeting the EGFR with clinical application in colorectal cancer. Because it does not have a murine component and lacks galactose 1,3-alpha galactose on the Fab fragment, infusion reactions are less frequent (1%) than with cetuximab. Pulmonary fibrosis has been reported in less than 1% of patients, however, it can be fatal. In a patient with preexisting pulmonary fibrosis, there was significant progression of fibrosis, leading to death after four doses of panitumumab. Therefore patients with preexisting lung disease have, in general, been excluded from clinical trials.
Mitogen-Activated Protein Kinase (MEK) Inhibitors
- ■
Trametinib: Trametinib is a reversible inhibitor of the mitogen-activated extracellular kinases (MEK) 1 and 2. It is used in combination with BRAF inhibitors for patients with melanoma whose tumors harbor the BRAF V600 mutation. In the MEKINIST and METRIC clinical trials, the reported incidence of interstitial lung disease was 2% and 2.4%, respectively, with a median time to symptom onset of 5.3 months. Permanent discontinuation of the drug is recommended once ILD develops because there is insufficient data regarding the reversibility of lung damage with this drug.
Polyadenosine Diphosphate-Ribose Polymerase (PARP) Inhibitors
- ■
Olaparib, niraparib, and rucaparib: PARP inhibitors are used in the management of patients with BRCA-mutated breast cancer. Pneumonitis has been observed in less than 1% of patients treated with olaparib, but some were associated with fatalities. Niraparib has been associated with nasopharyngitis in 23% of patients, dyspnea in 20%, and cough in 16% of patients. Rucaparib can also have similar respiratory manifestations as those seen with niraparib.
Cyclin-Dependent Kinase (CDK) 4/6 Inhibitors
- ■
Abemaciclib: Abemaciclib is used in the treatment of hormone-positive, HER2-negative advanced breast cancer. In the MONARCH 2 trial, up to 5% of patients who received a combination of abemaciclib and fulvestrant developed venous thromboembolism. In addition, cough has been reported in up to 19% of patients receiving this drug in the MONARCH 1 trial, a single-arm, open-label study of abemaciclib in 132 women.
Phosphatidylinositol-3-Kinase (PI3K) Inhibitors
- ■
Idelalisib: Idelalisib inhibits the delta isoform of phosphatidylinositol 3-kinase, a molecule that is highly expressed in malignant B lymphocytes and thus has clinical applications in CLL, small lymphocytic lymphoma, and follicular lymphoma. Severe and fatal pneumonitis have been reported in 4% of treated patients, therefore the US Food and Drug Administration (FDA) suggests close monitoring for pulmonary symptoms and thorough evaluation of patients with a decrease in oxygen saturation of over 5%. Other manifestations that have been reported include pneumonia (25%), cough (29%), and dyspnea (17%).
- ■
Copanlisib: Copanlisib is used in refractory follicular lymphoma and has also been associated with severe pneumonitis. In addition, up to 17% of patients develop infections with this drug; pneumonia is the most commonly reported. In particular, these patients are at an increased risk of developing pneumocystis jiroveci pneumonia (PJP), and thus it is recommended to initiate PJP prophylaxis prior to treatment in at-risk populations.
mTOR Inhibitors
- ■
Everolimus: Everolimus has been used in the treatment of advanced hormone receptor-positive, HER2-negative breast cancer, advanced renal cell carcinoma, and neuroendocrine tumors. Among patients treated with this drug, 8% to 14% were reported to develop pneumonitis, most of which were low grade. , As with most other drugs, the presence of baseline lung disease predisposes patients to pulmonary toxicity. Bronchoalveolar lavage in these patients have revealed either lymphocytic or eosinophilic samples. The most common radiographic findings in these patients are focal consolidations at the lung bases and ground-glass opacities.
- ■
Temsirolimus: Temsirolimus is used in patients with advanced renal cell carcinoma and endometrial cancer and has been reported to cause pneumonitis in up to 5% of patients; however, pulmonary involvement is often mild and may be asymptomatic in close to half of affected patients.
Pharmacological Approaches for the Management of Pulmonary Toxicities
- ■
As described previously, although most targeted therapies have a low incidence of pulmonary toxicities, some of these are potentially fatal and therefore warrant close monitoring and a low-degree of clinical suspicion for such adverse events. If a patient develops dyspnea, cough, hypoxia, or other respiratory symptoms on treatment, it is advisable to perform a thorough workup to determine the etiology of the dyspnea, including chest radiography, tests to rule out infections, cardiovascular etiologies, and venous thromboembolism. On occasion, invasive testing with bronchoscopy and bronchoalveolar lavage with or without transbronchial lung biopsies may provide crucial information to support a diagnosis of therapy-induced pulmonary toxicity.
- ■
Discontinuation of drug: If drug-induced pulmonary toxicity is suspected, the suspected culprit drug should be discontinued. In a very small fraction of patients, such as those with asymptomatic or mild symptoms, as is seen commonly in everolimus-associated pneumonitis, the drug may be continued with close monitoring. In continuation of this example, the FDA label for everolimus states that for patients who develop moderate to severe symptoms while on the standard dose of 10 mg daily, the drug may be withheld temporarily but may be reintroduced at a lower dose of 5 mg daily, if clinically indicated, after the resolution of symptoms. In some drug-related lung disease, discontinuation of the drug may lead to the reversal of pulmonary symptoms, whereas others might require further pharmacological interventions with glucocorticoids.
- ■
Glucocorticoids: Glucocorticoids form the mainstay of therapy for drug-induced pneumonitis. The optimal dose of steroids depends on the severity of lung involvement and symptoms and degree of hypoxia. Once respiratory symptoms improve, steroids should be tapered gradually over an extended time (at least 4 weeks). Some patients may require a longer taper of corticosteroids, and the regimen should be tailored to each individual patient’s clinical progress. Some patients may have a rapidly deteriorating course despite the use of corticosteroids. ILD associated with EGFR inhibitors such as gefitinib and erlotinib may be resistant to steroids in a fraction of patients.
- ■
Alternative dosing: Some situations will allow for alternative dosing schedules. As an example, for dasatinib-associated pleural effusions, an alternative dosing strategy of 100 mg daily instead of 70 mg twice a day has been associated with a reduced risk of further pleural effusions without affecting the efficacy of the drug. Note that for some indications, 140 mg once daily or 180 mg once daily may be appropriate. Diuretics and glucocorticoids may be used for the management of pleural effusions in this population. Endothelin receptor antagonists and calcium channel blockers have been used in the treatment of pulmonary hypertension related to dasatinib, however, their exact role in altering the clinical course of pulmonary arterial hypertension from dasatinib is unknown.
- ■
Bronchospastic reactions: For bronchospasm related to a hypersensitivity reaction, beta-2 agonists and steroids should be promptly administered. If the reaction occurs during an infusion, the infusion should be halted. For monoclonal antibodies associated with a high incidence of infusion reactions, premedication with acetaminophen and diphenhydramine should be used prophylactically prior to infusion. Prophylactic steroids prior to rituximab infusions do not reduce the incidence of severe infusion reactions. Fractionated doses of rituximab may also be used to reduce the risk of infusion reactions in at-risk patients. Epinephrine should be administered in any patients experiencing anaphylaxis.
Rechallenge
- ■
For most drug-related pulmonary toxicities, permanent discontinuation of the offending drug is recommended. In the case of certain drugs such as imatinib, a reduced dose has been reintroduced successfully without recurrence of ILD in a few patients. On the other hand, certain toxicities, such as pulmonary arterial hypertension with dasatinib, are an absolute contraindication for rechallenge with the drug. Some authors, however, report successful use of a more selective kinase such as nilotinib after dasatinib-induced pulmonary hypertension. In patients with other lung toxicities from dasatinib, rechallenge with a lower dose of dasatinib may be an option in the absence of alternative therapies. For patients with gefitinib-associated pneumonitis, erlotinib has been used without any major lung adverse events. , For patients who develop infusion reactions with rituximab, the likelihood of subsequent reactions is lower. For mild reactions, a lower infusion rate is usually well tolerated. However, patients with recurrent infusion reactions should be premedicated with steroids (hydrocortisone 100 mg or dexamethasone 10-12 mg) along with acetaminophen and diphenhydramine. Some patients may require desensitization protocols if they continue to have infusion-reactions with rituximab.
Nonpharmacological Approaches for Management of Pulmonary Toxicities
- ■
Optimal patient selection is important in order to decrease the risk of pulmonary toxicities. For example, pazopanib should be avoided in patients with extensive pulmonary or pleural metastases, and bevacizumab should be avoided in patients with squamous cell lung cancer. Patients who have received radiation should be carefully evaluated for the risk of subsequent pulmonary toxicities. In addition, caution is advised in patients with preexisting pulmonary disease, as they are at increased risk of lung toxicity with treatment.
- ■
Supportive care and symptom management play important roles in the management of pulmonary toxicities. Oxygen and ventilatory support may be prescribed as indicated. For patients with pleural effusions, such as those seen with dasatinib, therapeutic thoracentesis or even pleurodesis may be required if recurrent. Patients with bleeding secondary to antiangiogenic agents should be supported with blood products as needed. In cases with severe bleeding, bronchoscopic interventions for bleeding, bronchial arterial embolization, radiation, or surgery may be required.
References
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree