14516
Prostate Cancer in Elderly Patients
Pedro Recabal, Chung-Han Lee, and Dana E. Rathkopf
INTRODUCTION
Prostate cancer (PCa) is the most common malignancy among men, and the second leading cause of cancer death in the United States (1); the American Cancer Society estimates more than 180,000 new cases and more than 26,000 deaths in 2016. The disease burden is expected to increase along with aging of the population. More than half of men diagnosed with PCa (2) meet the conventional definition of elderly person (≥65 years old) (3); for this chapter, elder will be defined as greater than 70 years old. The prevalence of incidental PCa increases with age (59% in autopsies of men ≥ 79) (4). Elders are more likely than younger patients to present with high-risk features, including higher Gleason grade (5,6), and metastatic disease; these patients account for more than half of all PCa deaths (7). A study (8) estimated that around one-third of avoidable cancer deaths would be diagnosed in elders. However, current evidence-based recommendations suggest that prostate-specific antigen (PSA) screening should end at age 70, unless a man is very healthy and has a higher than average PSA, and at 75 for all men (9).
LIFE EXPECTANCY AND HEALTH STATUS ASSESSMENT
For therapeutic decisions, the survival benefit of treatments must be weighed against the patient’s life expectancy, for which a predictive model has been validated for patients with localized PCa, and is available online (10). Age by itself is a poor predictor of survival (11), and comorbidity is a stronger predictor of noncancer mortality than age (12). The current International Society of Geriatric Oncology (SIOG) guidelines recommend individualized management of PCa patients who are 70 or more years old, stratifying health status into three groups (healthy, vulnerable, and frail) based on comorbidities, dependence, nutritional status, and cognitive and physical functions (13) using the G-8 screening tool (see Chapter 14 and Figure 16.1).
146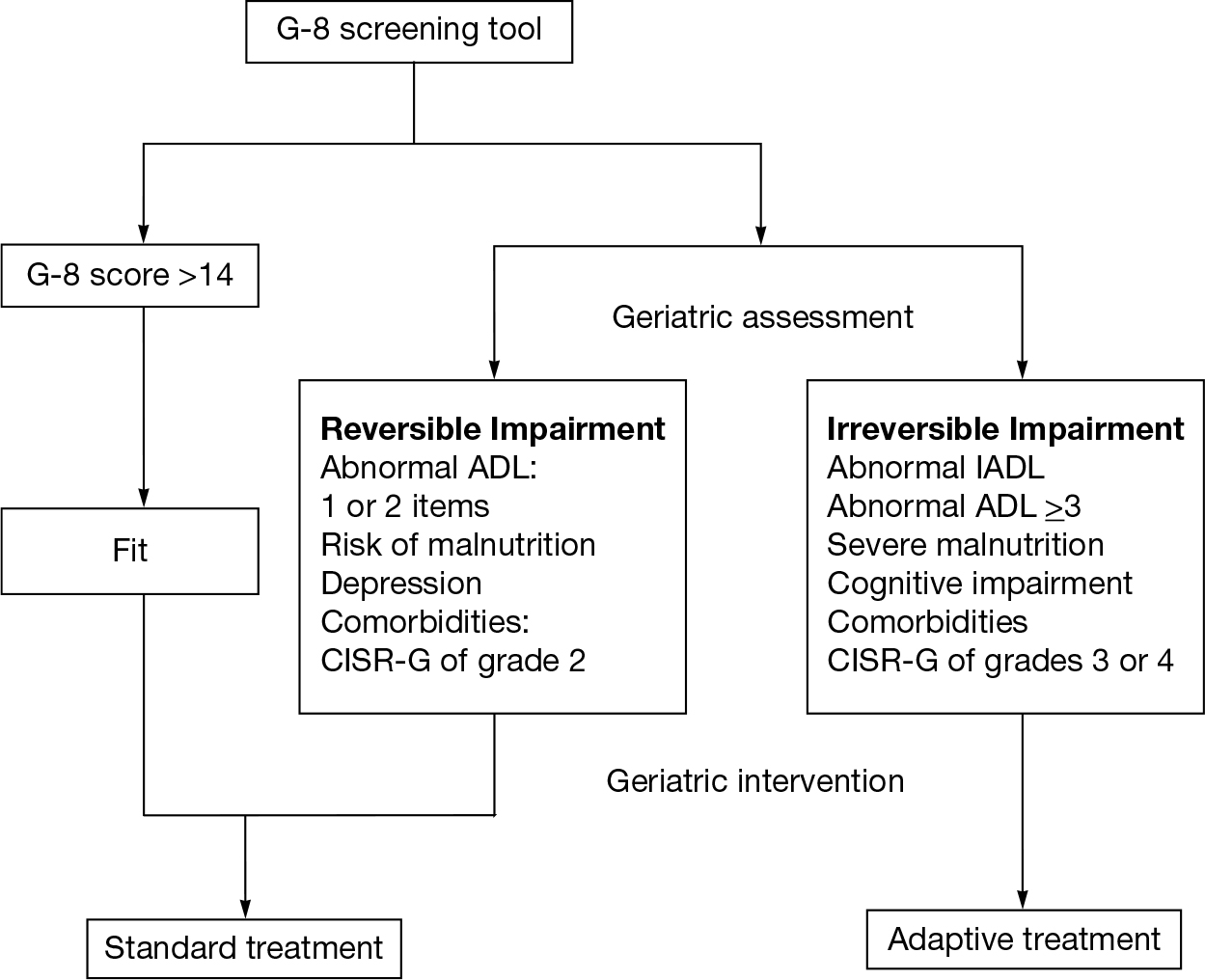
FIGURE 16.1 SIOG guidelines for treatment of PCa according to health status.
ADL, activities of daily living; IADL, instrumental activities of daily living; CISR-G, cumulative illness score rating-geriatrics; SIOG, International Society of Geriatric Oncology.
CLINICAL STATES OF THE DISEASE
The long natural history of PCa and evolving therapeutic alternatives have been the rationale for developing a model that partitions the disease continuum into dynamic clinical states characterized by the status of the primary tumor, presence of metastases, prior and current treatment, and testosterone levels (Figure 16.2). This model aids in assessing the prognosis at different clinical states—information not accounted for in nomograms or traditional staging systems (14).
DISEASE RISK STRATIFICATION
The TNM staging system, along with the Gleason score and the PSA, are used in combination to assess the risk of progression, recurrence, and cancer-related death. Different classification systems are available. These cluster patients according to cutoffs for each variable. The National Comprehensive Cancer Network (NCCN) risk groups (15) and criteria are presented in Table 16.1. Several nomograms have also been published which provide a better estimation of an individual’s risk. However, the risk estimation provided by these tools may not be generalizable. For example, the accuracy of a nomogram predicting recurrence after surgery may vary from institution to institution.
147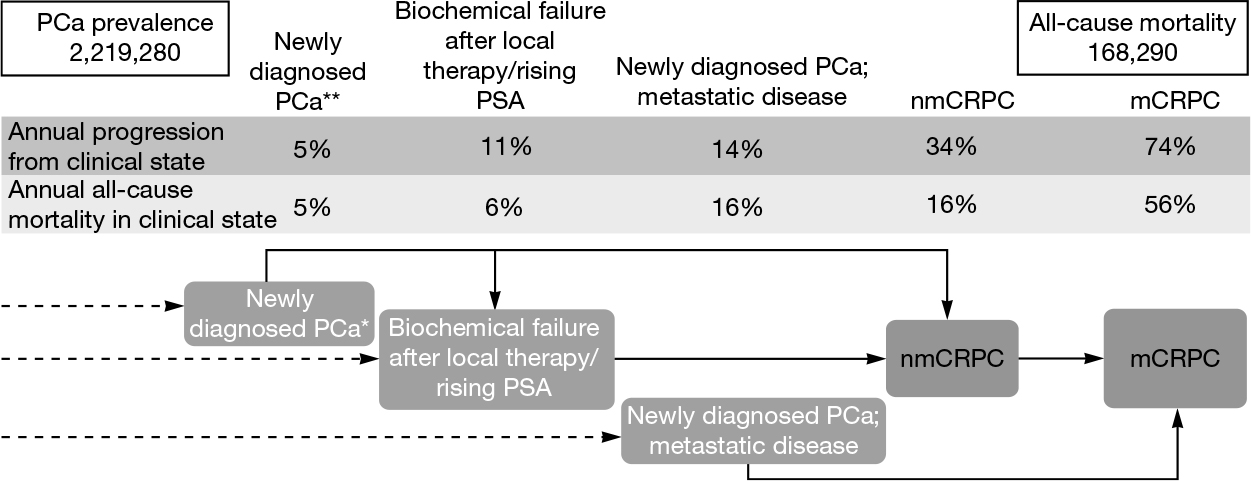
FIGURE 16.2 Clinical states of disease.
mCRPC, metastatic castration-resistant prostate cancer; nmCRPC, non metastatic, castration-resistant prostate cancer; PCa, prostate cancer; PSA, prostate-specific antigen.
*Weighted averages of patients diagnosed with localized, locally advanced, and metastatic disease.
**Localized disease or locally advanced disease.
TREATMENT: NEWLY DIAGNOSED LOCALIZED/LOCALLY ADVANCED PCa
Treatment decisions in older PCa patients should consider the risk of cancer and noncancer mortality, the side effect profile of treatments, and patient preference. Elders classified as healthy (G-8 score > 14) and vulnerable patients with reversible causes of impairment may benefit from treatment of the primary tumor according to cancer risk stratification; in contrast, frail patients should be spared from curative treatments, and expectant management is preferred, especially for low-risk tumors and men with a short life expectancy.
For elders selecting radical prostatectomy (RP), comorbidity is a better predictor of morbidity and mortality than age; healthy men have low operative mortality (0.66% in men between 70 and 79 years old [17]) and good long-term overall survival (18). Most men can recover urinary and sexual function after surgery; however, age has a negative impact on functional recovery. In a large retrospective study, 1-year continence rates for age groups less than 65, 65 to 70, 70 to 75, and greater than 75 were 93.2%, 90.8%, 86.0%, and 86.5%, and 1-year potency rates were 59.3%, 46.9%, 44.4%, and 31.3% (19). In men 70 years of age or older, minimally invasive RP is feasible, and has been associated with decreased rates of transfusions, postoperative complications, and anastomotic stricture compared to the open approach (20).
Radiotherapy (RT) is also a viable treatment for older healthy or vulnerable patients with localized intermediate or high-risk tumors (21). Randomized control trials (RCTs) demonstrate a survival benefit of RT plus androgen-deprivation therapy (ADT) over ADT alone (22); however, elderly populations are underrepresented in those trials. In a cohort study, the combination of RT and ADT was associated with reduced cause-specific and all-cause mortality in older men compared to ADT alone (23). In contrast, for men older than 70, adjuvant radiation after RP may have a detrimental effect (24).
TABLE 16.1 NCCN Cancer Risk Stratification and Treatment Options for Newly Diagnosed PCa
148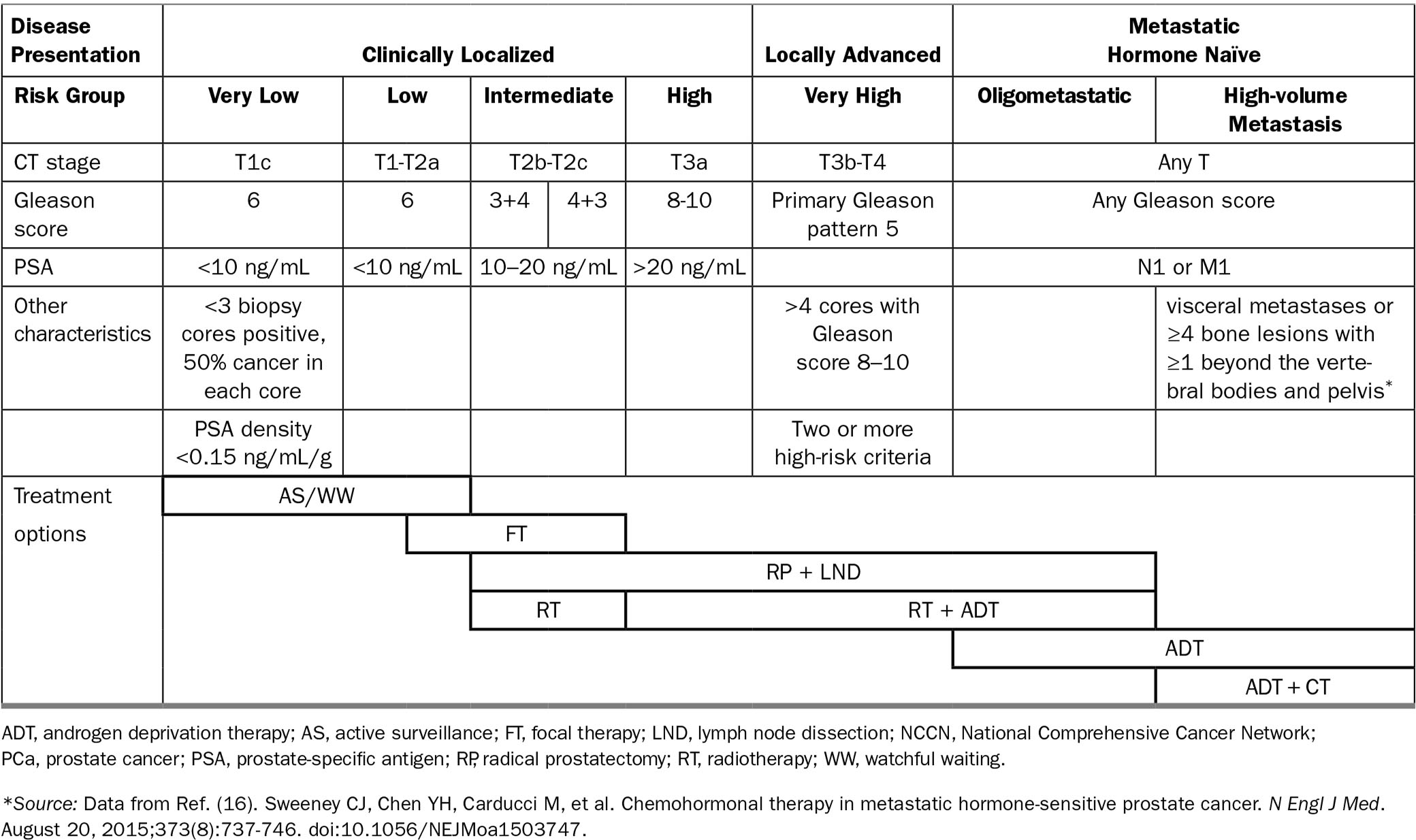
149ADT has been used as primary therapy in unfit patients with nonmetastatic high-risk PCa. In this group (T0-4 N0-2 M0), immediate ADT provided a small but significant benefit in overall survival (but not in symptom-free or cancer-specific survival) compared to deferred ADT (25). This benefit should be weighed against the side effects of ADT, which can include decreased bone mineral density; metabolic changes such as weight gain, decreased muscle mass, and increased insulin resistance; decreased libido and sexual dysfunction; hot flashes; gynecomastia; reduced testicle size; anemia; and fatigue (26).
RISING PSA (BIOCHEMICAL FAILURE AFTER LOCAL THERAPY)
Treatment decisions should be preceded by a health status assessment in older men with a rising PSA (27). Early salvage RT can improve survival, although the benefit for older patients is unclear, and age is an independent predictor of long-term grade 3 urinary toxicity. Early ADT at biochemical recurrence has no proven benefit in survival. It should be reserved for those at highest risk of progression (PSA-DT < 6–12 months; Gleason score > 7, and long life expectancy) (28).
METASTATIC NONCASTRATE PCa
ADT has been the mainstay of treatment in this setting for decades, despite the lack of RCTs (21). Level 1b evidence supports the role of ADT to palliate symptoms and improve quality of life (QoL). Complete androgen blockade provides a small survival benefit but increases toxicity (28). Seven phase III trials testing continuous versus intermittent ADT have reached inconclusive results (29–31).
Recent studies have found that chemo-hormonal treatment with six cycles of docetaxel combined with continuous ADT is associated with overall and progression-free survival advantage in castration-sensitive metastatic patients (especially for men with “high-volume disease,” defined as visceral metastases or ≥4 bone lesions with ≥1 beyond the vertebral bodies and pelvis) (16). This benefit may extend to healthy older patients who are fit for chemotherapy (30).
METASTATIC CASTRATION-RESISTANT PCa (mCRPC)
Several new agents that may affect survival have been approved for treatment of mCRPC in recent years. The survival benefit of taxane chemotherapy appears to be independent of patient age, with acceptable toxicity (32–34). In the elderly, the next-generation androgen-targeted agents abiraterone (35) and enzalutamide (36) 150have also been shown to improve survival. Additional agents have been approved based on a survival benefit for patients with mCRPC (although studies in elderly patients are ongoing), including immunotherapy (Sipuleucel-T) and radiopharmaceuticals (radium-223) (37). The optimal sequence for these newly approved agents and appropriate patient selection remains uncertain at present (38).
PALLIATIVE CARE
Palliative care should be offered to patients with advanced PCa concurrently with disease-directed treatment. Palliative care teams can collaborate with cancer clinicians to manage complex pain, fatigue, depression, anxiety, caregiver distress, and existential crisis (39).
TAKE HOME POINTS
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree