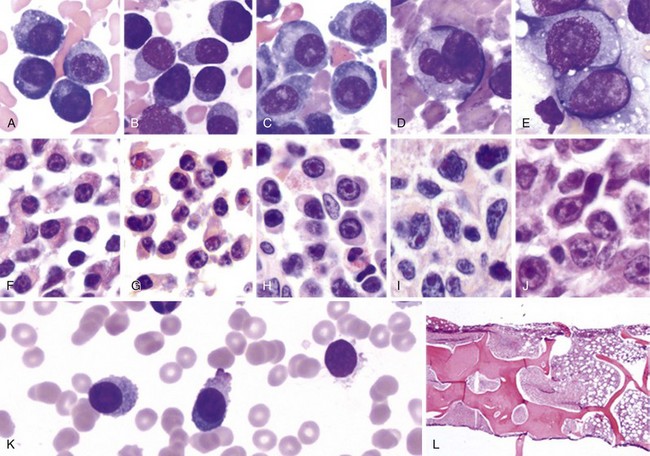
Chapter 38 Plasma Cell Neoplasms
Treatment of Relapsed Multiple Myeloma
Ensure that the patient has a relapse requiring intervention. The conversion from a negative immune-fixation analysis to a positive study result is not an indication to initiate salvage therapy. A clear clinical relapse with new or increased evidence of end-organ damage or a substantial or aggressive biochemical relapse as defined by criteria for progressive disease are indications for the initiation of treatment.
Selection of treatment depends on prior therapy received.
Table 38-1 Thalidomide Regimens in Relapsed or Refractory Multiple Myeloma
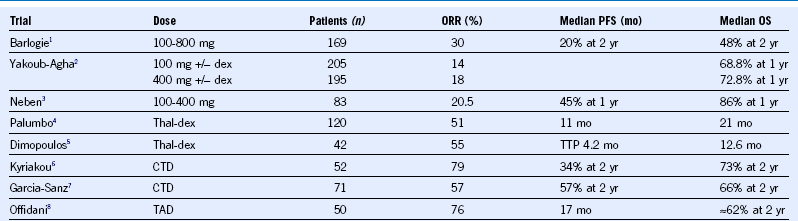
CTD, Cyclophosphamide, thalidomide, and dexamethasone dex, dexamethasone; ORR, overall response rate; OS, overall survival; PFS, progression-free survival; TAD, thalidomide, adriamycin, and dexamethasone; Thal, thalidomide; TTP, time to progression.
Table 38-2 Lenalidomide Regimens in Relapsed or Refractory Multiple Myeloma
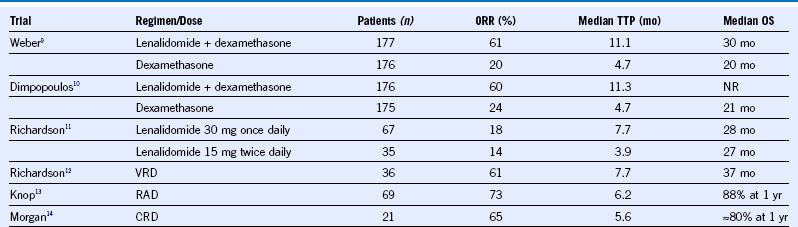
CRD, Carfilzomib, lenalidomide, dexamethasone; NR, not reported; ORR, overall response rate; OS, overall survival; RAD, lenalidomide, adriamycin, dexamethasone; TTP, time to progression; VRD, bortezomib, lenalidomide, dexamethasone.
Table 38-3 Bortezomib Regimens in Relapsed or Refractory Multiple Myeloma
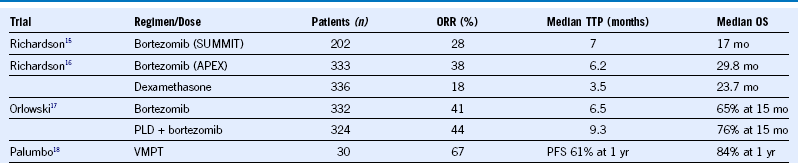
ORR, Overall response rate; OS, overall survival; PFS, progression-free survival; PLD, Liposomal doxorubicin; TTP, time to progression; VMPT, bortezomib, melphalan, prednisone, thalidomide.
Treatment of Newly Diagnosed Multiple Myeloma
Confirm that the patient has symptomatic myeloma. Patients with MGUS or SMM should not be treated outside of a clinical trial.
Perform risk stratification using ISS staging, FISH, or cytogenetics, and assessment of renal function and measurement of LDH level to develop long-term plan based on estimate of prognosis. Although initial treatment is not affected by risk category, later consideration of more aggressive consolidation or use of allogeneic SCT in eligible patients can be considered in high-risk younger patients.
Induction: Both transplant-eligible and ineligible patients can be treated with a triple-drug regimen (bortezomib and dexamethasone with lenalidomide, with thalidomide or cyclophosphamide, or cyclophosphamide, thalidomide, dexamethasone) for 3 to 6 cycles before transplant or 9 to 12 months for transplant-ineligible patients. For transplant-ineligible patients, additional options include a two-drug regimen (dexamethasone with lenalidomide, with bortezomib, or with thalidomide) or melphalan containing regimen such as melphalan with prednisone in combination with any of the newer agents, MPT, MPV, or MPR.
Drug Selection should be influenced depending on patient characteristics.
Transplant-eligible patients can undergo a single autologous transplant with melphalan with a second autologous transplant being optional depending on the overall response or on the protocol in which the patient is participating.
Maintenance: After transplant or optimal induction therapy (to best response), patients can receive maintenance with thalidomide, lenalidomide, or bortezomib. For high-risk patients, both bortezomib and lenalidomide or thalidomide combination can be used. Selection can be influenced based on their induction regimen.
Improving complete response rates is a key goal of current trials, but patients can live a long time with residual paraprotein.
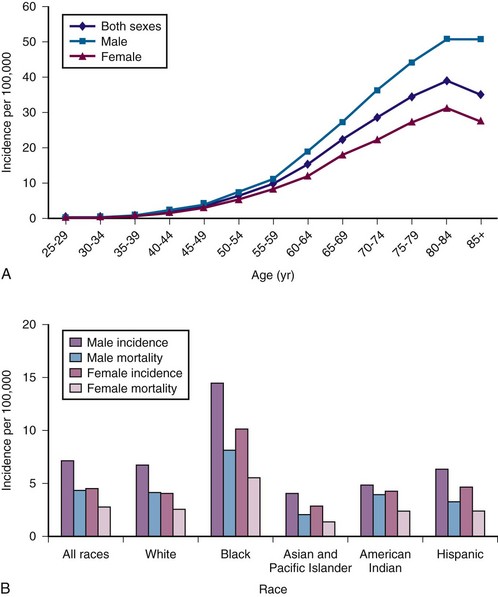
Figure 38-1 A, Multiple myeloma (MM) average annual age- and gender-specific incidence per 100,000 population in the United States in 2009. B, MM average annual race-specific incidence per 100,000 population in the United States in 2009. An increase in incidence is noted with advancing age, men are affected more than women, and a higher incidence is observed in blacks than whites.
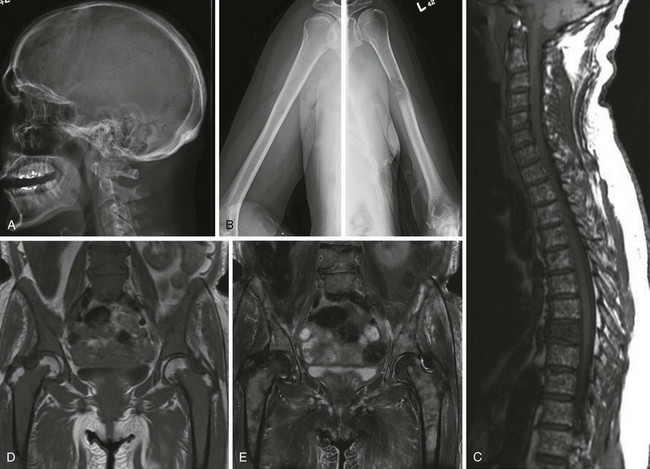
Figure 38-3 A and B, Typical skeletal changes on radiography. Example of “punched-out” lytic lesions in the skull and humerus. C to E, Magnetic resonance imaging pattern in multiple myeloma in the spine and pelvis showing diffuse involvement with focal lesions.
(Courtesy Dr. Nikhil Ramaiya, MD.)
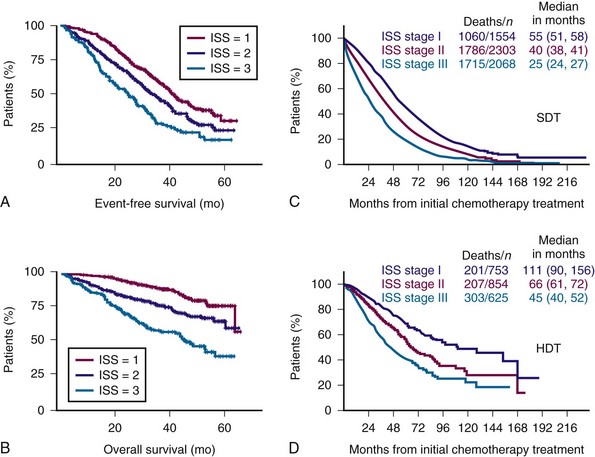
Figure 38-4 International Staging System (ISS) predicts both event-free survival (A) and overall survival (B) and overall survival with both standard-dose therapy (SDT) (C) and high-dose therapy (HDT) (D).
(A and B from Avet-Loiseau H, Attal M, Moreau P, et al: Genetic abnormalities and survival in multiple myeloma: The experience of the Intergroupe Francophone du Myélome. Blood 109:3489, 2007; C and D from Greipp PR, San Miguel J, Durie BG, et al: International staging system for multiple myeloma. J Clin Oncol 23:3412, 2005.)
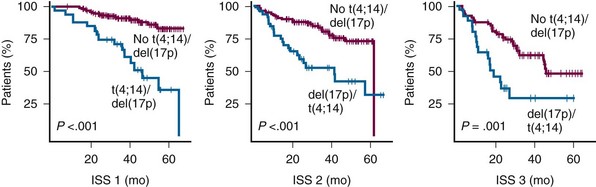
Figure 38-5 KAPLAN-MEIER ESTIMATES OF SURVIVAL ACCORDING TO THE INTERNATIONAL STAGING SYSTEM (ISS) STAGES AND t(4;14) OR del(17p).
(From Avet-Loiseau H, Attal M, Moreau P, et al: Genetic abnormalities and survival in multiple myeloma: The experience of the Intergroupe Francophone du Myélome. Blood 109:3489, 2007.)
Table 38-4 Phenotypic Characterization of Plasma Cells
| Adhesion Molecule | Normal Plasma Cells | Multiple Myeloma Cells |
|---|---|---|
| CD138 | + | + |
| CD19 | + | − |
| CD28 | − | − |
| CD38 | + | + |
| CD40 | + | + |
| CD45 | + | −* |
| CD27 | − | + |