Fig. 14.1
Proliferative endometrium. The glands are round to tubular with pseudostratified nuclei and mitotic figures
14.3.1.2 Sixteen Day Endometrium
Sixteen day endometrium is characterized by glands with cells displaying subnuclear vacuoles combined with abundant mitotic figures (Fig. 14.2). The overall gland architecture is the simple tubular glands of proliferative endometrium and, although mitoses are prominent, they are not as numerous as during the proliferative phase. In sixteen day endometrium, the presence of numerous mitoses prevents one from confirming that ovulation has occurred [11].
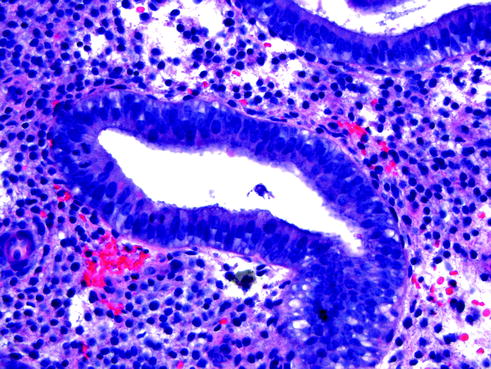

Fig. 14.2
Sixteen day endometrium. The glands are similar to proliferative endometrium and contain scattered subnuclear vacuoles
14.3.1.3 Early (Vacuole Phase) Secretory Endometrium
Entry into the vacuole phase of the cycle suggests that ovulation has occurred. Glands with cells showing uniform subnuclear vacuoles and rare mitotic figures warrant the diagnosis of day seventeen secretory endometrium (Fig. 14.3a). In day eighteen secretory endometrium, the glandular epithelial cells have both sub- and supranuclear vacuoles where the nuclei have migrated toward the gland lumen and now occupy the center of the cell (Fig. 14.3b). By day nineteen, the nuclei have nearly all returned to the base of the cell. There are abundant intraluminal secretions and the glands begin to show secretory exhaustion with low columnar to cuboidal cells, loss of vacuoles, and a lack of mitotic activity [12].
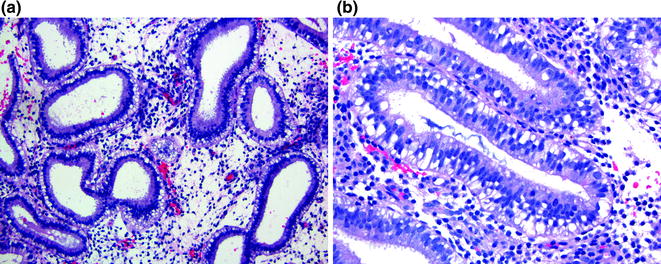

Fig. 14.3
Early (vacuole phase) secretory endometrium. a Day 17 has uniform prominent subnuclear vacuoles. b Day 18 has both sub-and supranuclear vacuoles
14.3.1.4 Mid (Secretory Exhaustion Phase) Secretory Endometrium
The loss of secretory vacuoles signals the end of the early secretory phase and the beginning of the mid phase. From this point forward, the focus in assigning dates to endometrial samples will be on stromal changes. Day twenty secretory endometrium exhibits maximal intraluminal secretions with only very rare residual subnuclear vacuoles (Fig. 14.4a). The stroma beneath the endometrial surface becomes more compact and can mimic predecidualized stroma. However, the stromal cells still have dark nuclei and a high nuclear to cytoplasmic ratio, unlike predecidualized cells. Day twenty-one secretory endometrium shows increased stromal edema, which peaks at day twenty-two. The stromal cells may appear widely spaced and their high nuclear to cytoplasmic ratios make them appear to have “naked nuclei.” There are no conspicuous predecidual changes (Fig. 14.4b).
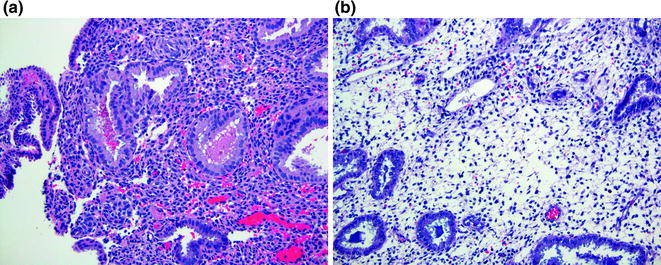

Fig. 14.4
Mid (secretory exhaustion phase) secretory endometrium. a Day 20 has rare subnuclear vacuoles and exhibits maximal intraluminal secretion. b Day 22 shows prominent endometrial stromal edema that has an appearance of “naked nuclei”
14.3.1.5 Late (Predecidual Phase) Secretory Endometrium
There are three well-defined components of predecidual change. Initially, there are aggregates of stromal cells surrounding spiral arterioles. As the changes develop, the stromal cells acquire distinct cell borders and slightly basophilic cytoplasm. Finally, the chromatin texture becomes much finer and the nuclei take on much less basophilia. For day twenty-three secretory endometrium, the predecidual cells are found surrounding individual spiral arterioles (Fig. 14.5a). By day twenty-four, the aggregates of predecidua bridge multiple vessels. Day twenty-five secretory endometrium is characterized by predecidua in thin aggregates below the endometrial surface (Fig. 14.5b). A thick subsurface band of predecidua is present by day twenty-six, accompanied by the presence of endometrial stromal granulocytes. Day twenty-seven endometrium shows abundant predecidua expanding downward from the endometrial surface, with increased numbers of granulocytes [11] (Fig. 14.5c).
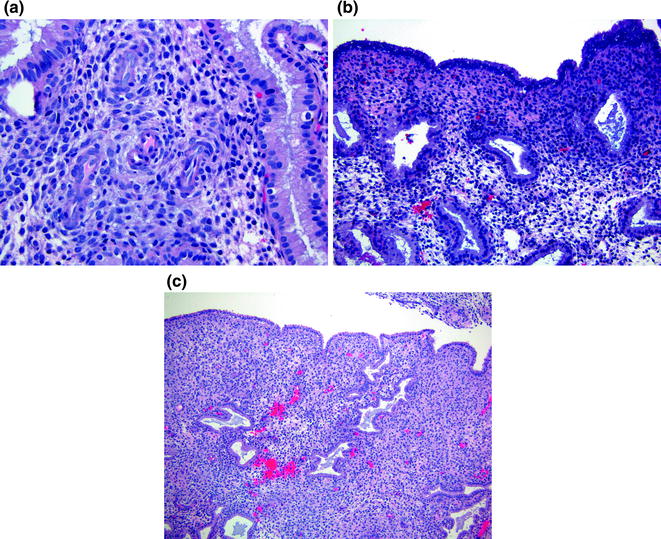

Fig. 14.5
Late (predecidual phase) secretory endometrium. a Day 23 has spiral arterioles surrounded by predecidual cells. b Day 25 contains a thin layer of predecidua underneath the surface. c Day 27 shows abundant predecidua expanding downward from the endometrial surface, with increased numbers of granulocytes
14.3.1.6 Menstrual (Breakdown) Endometrium
The menstrual phase is defined by the presence of discrete aggregates of condensed predecidual cells (stromal breakdown), admixed with inflammatory cells and blood [13]. Interspersed between these fragments of condensed stroma are glands showing secretory exhaustion (Fig. 14.6). The presence of both components (stromal breakdown and secretory exhausted glands) is evidence that breakdown has occurred.
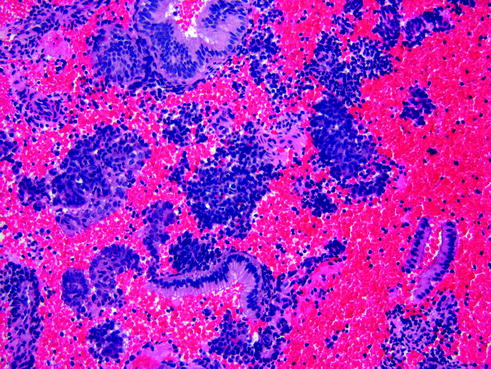

Fig. 14.6
Menstral (breakdown) endometrium. Diffuse stromal breakdown admixed with inflammatory cells, blood and associated exhausted secretory glands
14.3.2 Dysfunctional Uterine Bleeding
A number of conditions may lead to dysfunctional uterine bleeding, which can be thought of as bleeding resulting from alterations in the normal cycling pattern of the endometrium. The most common causes include anovulation (typically seen in patients in their fifth decade), chronic endometritis, endometrial polyps, and submucosal leiomyomata [11].
14.3.3 Anovulation
Anovulatory cycles are very common and these changes can lead to multiple histologic patterns depending on the presence or absence of elevated estrogen levels. Persistent estrogen stimulation leads to the formation of cystically dilated endometrial glands with an irregular distribution (Fig. 14.7a). Stromal breakdown may be patchy due to ischemia from focal fibrin thrombi in spiral arterioles. In the absence of elevated estrogen, the cystic, dilated glands are not prominent, but other features of anovulation including tubal metaplasia (Fig. 14.7b), patchy breakdown due to fibrin thrombi, and surface repair are present. Another pattern seen when the proliferative phase is followed by abrupt loss of estrogen (absence of a persistent ovarian follicle) is that of uniform tubal glands with diffuse stromal breakdown. This can be distinguished from normal ovulatory breakdown by the absence of predecidual changes in stroma and the lack of secretory changes in the glands in the case of anovulation [14].
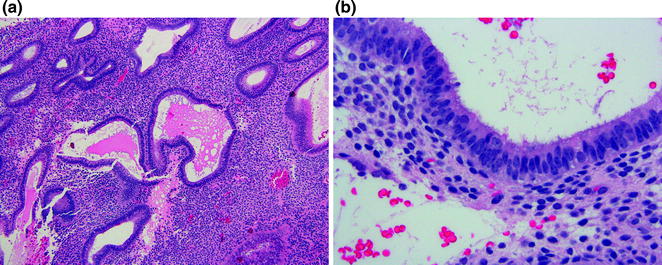

Fig. 14.7
Anovulatory endometrium. a Cystically dilated glands. b Tubal metaplasia
14.3.4 Benign Endometrial Hyperplasia
Benign endometrial hyperplasia (or hyperplasia without atypia) typically occurs in perimenopausal women with a presenting symptom of abnormal uterine bleeding. It is a condition that results from unopposed estrogen that may be caused by multiple factors including chronic anovulation, absence of progesterone, androgen to estrogen conversion within adipose tissue secondary to obesity, polycystic ovarian syndrome (PCOS), estrogen-secreting ovarian tumors (e.g., granulosa cell tumor), or exogenous estrogen (e.g., Tamoxifen therapy). Women with excess estrogen carry a 3–4 fold increased risk of developing endometrial carcinoma and 10-fold increased risk after a decade of exposure [15]. Women with abnormal uterine bleeding usually will undergo endometrial biopsy or curettage to rule out the possibility of uterine cancer [16]. Histologically, the samples contain endometrial glands that vary in size and shape with an associated increase in gland to stromal ratio; however, there is no significant cytologic demarcation from the background endometrium [17, 18].
14.3.5 Endometrial Intraepithelial Neoplasia
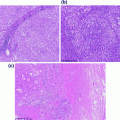
Endometrial intraepithelial neoplasia (EIN) (or atypical hyperplasia) commonly affects perimenopausal to postmenopausal women. Similar to benign endometrial hyperplasia, this condition is developed secondary to excess endogenous or exogenous estrogen, causing abnormal uterine bleeding as the presenting clinical symptom. Much evidence supports EIN as the precursor lesion to endometrioid type of endometrial cancer [18–22]. In addition, ~25–35 % of patients with EIN will also develop concurrent or subsequent endometrial carcinoma [19, 21]. The diagnostic criteria for EIN include: (1) an area where glandular content exceeds that of stroma (glands/stroma > 1), (2) nuclear and/or cytoplasmic features of epithelial cells differ between architecturally abnormal glands and normal background glands, (3) maximum linear dimension exceeds 1 mm, and (4) excluding benign mimics (e.g., endometrial polyps or anovulatory endometrium) and adenocarcinoma (Fig. 14.8). All of these criteria must be satisfied in order to make the diagnosis of EIN.
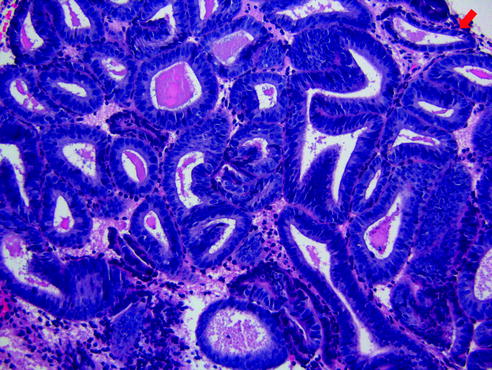

Fig. 14.8
Endometrial intraepithelial neoplasia. There is increased gland to stromal ratio that exceeds 1 mm in linear dimension. In addition, the nuclear cytology differs from the normal background glands (highlighted by red arrow)
14.3.6 Endometrial Carcinoma
Endometrial carcinoma is the most common malignant tumor in the female genital tract with an age-adjusted incident rate of 24.6 per 100,000 women per year in the United States (US) [23]. Patients often present with abnormal uterine bleeding that initiates endometrial sampling [16]. Based on clinical and pathologic information, endometrial carcinomas have been divided into type 1 and type 2 [24, 25]. Type 1 cancers are usually low-grade tumors associated with estrogen overexposure, whereas type 2 cancers are aggressive high-grade tumors which occur independently of estrogen stimulation.
14.3.6.1 Endometrioid Adenocarcinoma
This tumor is the prototypic type 1 cancer. It is the most commonly encountered cancer in the endometrium, affecting predominantly perimenopausal to postmenopausal women with a mean age around 60 years [26]. The most common presenting symptom is abnormal uterine bleeding where endometrial thickening can sometimes be visualized by transvaginal ultrasound imaging. As discussed earlier, EIN is the precursor lesion for the endometrioid type of endometrial adenocarcinoma [18–22]; therefore, they share similar risk factors. Genetically, individuals with Lynch syndrome or Cowden syndrome also have higher risk of developing endometrioid adenocarcinoma [27–29].
Histologically, endometrioid adenocarcinoma has glands that are similar to benign endometrium. The columnar lining cells have a stratified or pseudostratified appearance and the glands are crowded with complex maze-like or cribriform architecture. Gland fusion and solid areas develop as the tumor becomes less differentiated. The grading of endometrioid adenocarcinoma is based on the amount of solid growth pattern (non-squamoid) in these cancers. Grade 1 tumors have <5 % solid component (Fig. 14.9a), grade 2 tumors have solid areas between 5 and 50 % (Fig. 14.9b), and grade 3 tumors have >50 % solid growth pattern (Fig. 14.9c). The tumor nuclei typically demonstrate a low to moderate amount of cytologic atypia in grade 1–2 tumors. The presence of severe cytologic atypia in >50 % of the tumor is associated with more aggressive clinical behavior and justifies increasing the tumor by one grade in architecturally grade 1 or 2 tumors [30]. Squamous, villoglandular and secretory differentiations can also be seen in endometrioid adenocarcinomas.
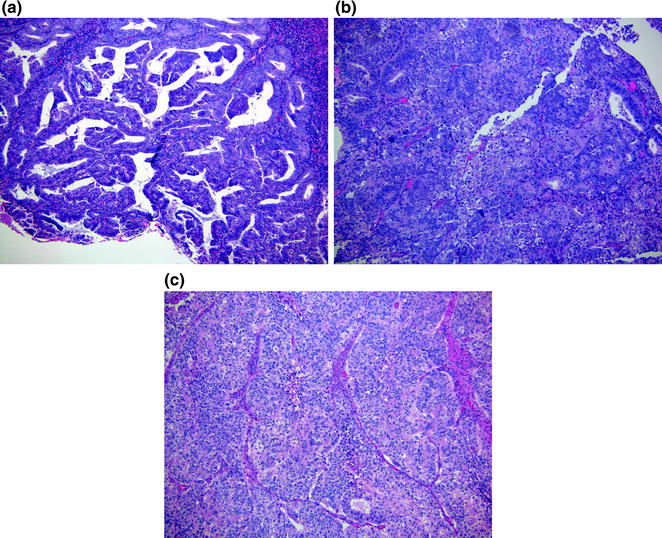

Fig. 14.9
Endometrial adenocarcinoma. a Grade 1 tumor showing glands with complex architecture. b Grade 2 tumor showing glands similar to grade 1; however, >5 % of solid areas can be seen. c Grade 3 tumor with mostly solid pattern (>50 %)
In biopsy/curettage material, when the lesion is present in both endometrial and endocervical samples, it can be difficult to decipher the primary site of origin based on histomorphology alone. However, it is important to make this distinction as endometrial and endocervical cancers have different clinical and surgical managements. Identification of precursor lesions (EIN or endocervical adenocarcinoma in situ) may assist in this scenario. However, when absent, immunohistochemistry with a panel of antibodies including ER, vimentin, monoclonal CEA and p16 can be helpful in making this distinction. Endometrial cancers typically are positive for ER and vimentin while endocervical lesions are positive for p16 and monoclonal CEA. On the other hand, it should be noted that high-grade endometrial cancers can occasionally stain diffusely positive for p16, therefore, the staining should be interpreted with caution and in conjunction with histomorphology.
14.3.6.2 Mucinous Adenocarcinoma
This tumor is defined by >50 % of neoplastic cells containing cytoplasmic mucin. It is an uncommon lesion, found in <10 % of all endometrial cancers [31]. Clinically, these tumors are usually associated with low-stage disease, although 50 % of cases can develop deep myometrial invasion [31]. Some experts consider mucinous adenocarcinoma as a variant of endometrioid adenocarcinoma with mucinous differentiation, as these tumors behave similarly and the distinction poses no clinical significance. However, this entity is listed separately under the 2014 World Health Organization classification of endometrial tumors [32]. Histologically, the neoplastic glands are lined by mucin-rich cells with minimal stratification (Fig. 14.10). The mucin is producing cells that are positive for mucicarmine and CEA. There is a mild to moderate cytologic atypia.
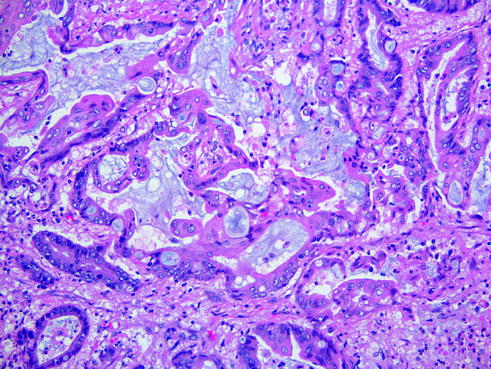

Fig. 14.10
Mucinous adenocarcinoma. There are abundant mucin pools seen within the tumor and the tumor cells contain cytoplasmic mucin
14.3.6.3 Serous Adenocarcinoma
Serous carcinoma is one of the most aggressive endometrial carcinomas and is considered a type 2 tumor. Women with this disease are generally postmenopausal and older than patients with type 1 tumors. Estrogen excess is less commonly associated with this tumor. Histologically, the tumor consists of complex papillary and/or glandular architecture where neoplastic cells contain pleomorphic high-grade nuclei and macronucleoli (Fig. 14.11a). Mitoses are readily identified. Papillary tuftings and irregular slit-like spaces are other morphologic features seen in serous carcinoma (Fig. 14.11a). By definition, serous carcinoma is a high-grade tumor; therefore, grading is not required. Serous carcinoma has an aggressive clinical behavior and is often associated with deep myometrial and lymphovascular invasion. Even in the absence of myometrial invasion, it is not uncommon to find extrauterine metastasis.
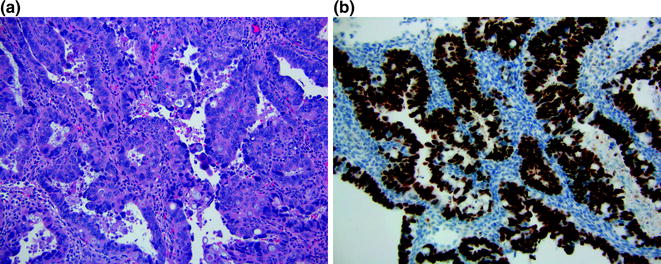

Fig. 14.11
Serous adenocarcinoma. a The tumor contains papillary/glandular architecture with high-grade nuclei and mitoses. b This tumor harbors p53 mutation, which is demonstrated by p53 immunostain
Serous carcinoma and grade 3 endometrioid adenocarcinoma can have overlapping morphologic features. Immunohistochemistry may help in differentiating these two different entities. Aberrant p53 expression (either diffuse positivity or complete absence) is typically seen in serous carcinoma (Fig. 14.11b). However, it is important to note that some grade 3 endometrioid adenocarcinomas can harbor p53 mutations, making this immunostain unhelpful in this situation. Fortunately, this distinction is not critically important for clinicians as both of these tumors often present with high stage disease that requires similar aggressive clinical management.
14.3.6.4 Clear Cell Adenocarcinoma
Clear cell carcinoma is a rare type 2 tumor that is encountered in <5 % of all endometrial cancers. Histologically, these tumors have papillary, tubulocystic and solid growth patterns (Fig. 14.12a). Hobnail cells with clear cytoplasm are the hallmark characteristic of this tumor (Fig. 14.12b). Occasionally, the cytoplasm may appear eosinophilic rather than clear. Severe nuclear atypia is always present. Similar to serous carcinoma, this tumor is by definition a high-grade neoplasm; therefore, grading is unnecessary.
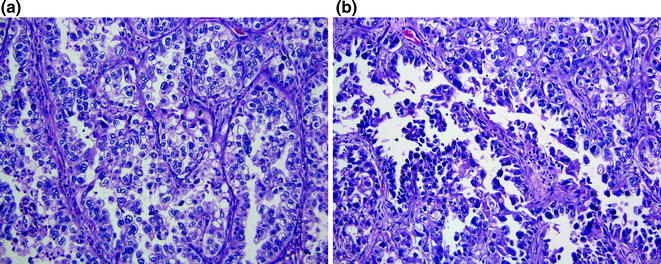

Fig. 14.12
Clear cell adenocarcinoma. a As its name implies, the tumor cell contains clear cytoplasm. b Hobnail cells with high nuclear grade can be appreciated
14.3.6.5 Carcinosarcoma
Carcinosarcoma (or malignant mixed Mϋllerian tumor) is a highly aggressive malignant neoplasm that consists of both malignant epithelial and mesenchymal components. It affects postmenopausal women and represents <5 % of malignant uterine tumors. Carcinosarcomas have been associated with tamoxifen or exogenous estrogen therapies [33, 34]. The majority of women with carcinosarcoma present as high-stage disease with tumor extending beyond the uterus. Clinically, the tumor may present as a polypoid mass that protrudes through the cervical os. Histologically, the epithelial component more commonly consists of endometrioid or serous adenocarcinomas (Fig. 14.13); however, other types of epithelial malignancy, such as clear cell carcinoma, can also be identified. The malignant mesenchymal component is typically composed of non-specific high-grade sarcoma; however, heterologous elements such as benign cartilage or rhabdomyosarcoma can be present. Recent studies have suggested that carcinosarcomas are of epithelial in origin with transitions to mesenchymal differentiation [35–37].
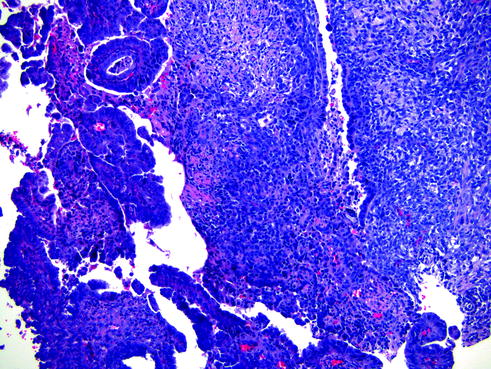

Fig. 14.13
Carcinosarcoma. This tumor has a mixture of high-grade malignant epithelial (left) and mesenchymal (right) components
14.3.6.6 Less Common Tumors
There are other less commonly seen uterine neoplasms, such as neuroendocrine tumors, undifferentiated carcinoma, leiomyosarcoma, adenosarcoma and endometrial stromal sarcoma. However, for the scope of this chapter, these rare entities will not be discussed.
14.3.7 Spread and Staging
Staging of endometrial cancers is based on the International Federation of Gynecology and Obstetrics (FIGO) and the American Joint Committee on Cancer (AJCC) staging systems, which are similar. These systems take into account the depth of the myometrial invasion and the involvement of cervical stroma, uterine serosa, adnexa, vagina, parametrium, bladder, or bowel mucosa [38]. Lymphatic spread initially involves the pelvic lymph nodes, eventually reaching the para-aortic lymph nodes. However, a subset of endometrial cancers do metastasize directly to the para-aortic lymph nodes.
14.3.8 Genetics and Genomics
As mentioned previously, endometrial carcinoma have been divided into type 1 and 2 tumors. Type 1 tumors are typically associated with microsatellite instability (MSI), PTEN and KRAS mutations, and β-catenin nuclear accumulation, while type 2 tumors are associated with p53 abnormalities and loss of heterozygosity at different loci.
14.3.8.1 Familial Syndromes
It has been estimated that about 5 % of all newly diagnosed endometrial cancers can be attributed to inherited familial cancer syndromes [39]. Of such familial syndromes, Lynch syndrome accounts for the majority of inherited endometrial cancers and is also characterized by a high risk of colorectal tumors [40–43]. This familial cancer syndrome results from germline mutations in the mismatch repair (MMR) genes MLH1, MSH2, MSH6 and PMS2 [40–43]. The cumulative lifetime risk (typically calculated by age 75) of developing endometrial cancer in MMR gene mutation carriers ranges from 20 to 70 % and depends on the mutated gene, with patients carrying MSH6 mutations having the highest risk (up to 71 % by age 75) [44]. On average, the age of diagnosis of endometrial cancer in Lynch syndrome is ~2 decades younger than in sporadic cases [44]. Histologically, most Lynch syndrome cancers are endometrioid tumors. In addition to a personal and/or familial history of colorectal and endometrial tumors, or early age of diagnosis and endometrioid histology, another hallmark of Lynch syndrome cancers is the presence of MSI, which can be diagnosed using microsatellite testing or MMR protein immunohistochemistry. Although MSI and MMR testing can aid in the identification of Lynch Syndrome-associated cases, they are not completely diagnostic since ~10–15 % of all endometrial cancers have a sporadic/non-germline-related MSI phenotype that is caused by methylation of the MLH1 gene promoter [45].
A small fraction of inherited endometrial cancer cases can also be seen in Cowden syndrome and in the polymerase proofreading-associated polyposis (PPAP) syndrome [46]. Cowden syndrome is caused by very rare inherited PTEN gene mutations and has a cumulative lifetime risk of developing endometrial cancer that ranges from 19 to 28 % [47–50]. Women with Cowden syndrome also have a high risk of developing breast (up to 50 % by age 70) and thyroid (up to 10 % by age 70) cancers [47, 49–52]. Therefore, a family or personal history of breast and thyroid tumors could help in establishing a diagnosis of Cowden syndrome [48]. We recently described the PPAP syndrome, a condition that is associated with tumors of the colon and endometrium (a syndromic association also seen in Lynch syndrome, see above), which results from rare mutations in the polymerase epsilon (POLE) and delta (POLD1) genes [53]. Endometrial cancers in PPAP have been described in only a handful of patients, who typically develop early onset disease (age 35–55) and synchronic or metachronic colorectal tumors and cancers [53–55].
In summary, endometrial cancer cases can be found in three familial syndromes (Lynch, Cowden and PPAP) that are characterized by an early onset and a higher risk of other malignant conditions such as colorectal, breast or thyroid cancers. The use of clinical, pathological, and molecular information can be informative and identify such patients who will benefit from genetic counseling and testing, which can help them minimize the risk of developing additional malignancies through early detection.
14.3.8.2 Low-Penetrance Risk Variants
The genetic basis of endometrial cancer has been recently investigated at the population level using genome-wide associations (GWA) and candidate gene studies. Two genomic regions, near the HIF1B and TERT genes, have been identified by these population-based studies and harbor several independent endometrial cancer risk variants [56–58]. Unlike very rare causal mutations that result in familial syndromes, these low-penetrance HIF1B and TERT variants are very common in the population and are not necessarily causal of endometrial cancer on their own. However, the identification of low-penetrance variants is important because they highlight potentially important pathways, such as hypoxia and telomere biology in endometrial carcinogenesis. Furthermore, these variants can be used in the future, in combination with other clinical, genetic, and environmental information, to improve risk prediction models for early cancer detection [58, 59].
14.3.8.3 Somatic Genetics
Like most other tumors, endometrial cancers are characterized by unique patterns of genomic instability as well as by the presence of mutations in oncogenes and tumor suppressor genes. Several studies have found that endometrial tumors harbor somatic mutations in well-established cancer genes such as PTEN, FGFR2, ARID1A, CTNNB1, PIK3CA, PIK3R1, and KRAS [60, 61]. Also, as mentioned above, the MSI tumor phenotype, a pattern of genomic instability that involves mutations in di-, tri-and tetra-nucleotide repeats, is found ~15–30 % of all endometrial cancers [44, 45]. Our knowledge of endometrial cancer genomics was greatly enhanced by the recent molecular analysis carried out by The Cancer Genome Atlas (TCGA) study [62], which performed a multi-omic characterization of 373 endometrial tumors that included 307 endometrioid, 66 serous and 13 mixed histology cancers. These studies led to the identification of four major endometrial cancer subtypes including: (1) near-diploid and ultra-mutated tumors with somatic POLE gene mutations, (2) near-diploid tumors with MSI, (3) genomically stable/microsatellite stable tumors and (4) genomically instable/serous-like tumors. The patterns of mutations and genomic instability in these four tumor subtypes appeared to be unique and provided an alternative classification to that based on histological features [62].
Near-diploid/POLE mutant tumors represented about 8 % of all endometrial cancer analyzed by the TCGA. All of these tumors had endometrioid histology and were characterized by the presence of mutations in the proofreading domain (exons 9 and 13) of the POLE gene, a region that was also found to be mutated in the germline DNA of patients with PPAP syndrome (see above) [46]. The lack of proofreading capacity of POLE (a DNA polymerase) is likely to explain why these tumors are characterized by an extremely high mutation rate (~232 mutations per megabase, the highest of all human tumors) and why they are characterized by a mutation signature that involves mostly G:C > T:A transversions [46]. Interestingly, the TCGA study, as well as more recent studies, have provided evidence suggesting that endometrial tumors with POLE mutations have a relatively good prognosis, independent of other clinical and histological variables, particularly for high-grade tumors [53]. A recent study by Church et al. [53], which assessed the prognostic effect of POLE mutations in 1416 patients, including 788 from two clinical trials, suggested that POLE mutations could be used as a novel biomarker to identify endometrial cancer patients with a good prognosis that could benefit from less intensive treatments.
The second group defined by the TCGA study, which represented ~28 % of all tumors, included endometrial cancers with the MSI phenotype. Consistent with previous studies, MSI endometrial tumors have a predominant endometrioid histology and MLH1 silencing [62]. The third group of TCGA tumors represented about 40 % of all samples and included low-grade endometrioid tumors that had a low mutation rate, microsatellite stability, a low proportion of copy number changes and a high frequency of β-catenin (CTNNB1) mutations. Lastly, a genomically instable/serous-like tumor group was also identified by the TCGA study and was characterized by extensive copy number variation and a very high rate of TP53 mutations (which were rare in the other three subtypes). Interestingly, this was a histologically heterogeneous group which included all serous cancers as well as about 25 % of all high-grade endometrioid tumors analyzed in the study. The latter finding was significant as it suggested that such a molecular subtype could benefit from the therapies used for serous tumors, which typically involves chemotherapy rather than adjuvant radiotherapy [62].
In summary, recent advances in the genomic characterization of endometrial tumors have identified four major subtypes that offer an alternative to histological classification. The most clinically relevant findings in this regard include the identification of the POLE mutation group, representing ~8 % of all tumors, that have a particularly good prognosis and that could benefit from less aggressive treatment and the discovery of a particularly aggressive type of endometrioid tumors that will benefit from the chemotherapeutic approaches used in serous tumors [62]. Furthermore, a plethora of therapeutic targets is now available and will offer unique opportunities for the future development of molecularly guided treatments. While these observations need to be tested under rigorous clinical trials, they represent a unique opportunity for the establishment of personalized medicine in endometrial cancer in the coming years.
14.4 The Ovary
14.4.1 Histology of the Normal Ovary
The ovary is lined by a single layer of low columnar to cuboidal cells which have been come to be known as “ovarian surface epithelium” although in reality it is a specialized mesothelium (Fig. 14.14). This surface layer commonly invaginates to form cortical (or epithelial) inclusion cysts which have lost their surface connections. Occasionally, invaginations of transitional-type epithelium may be encountered. These are known as Walthard nests (or rests).
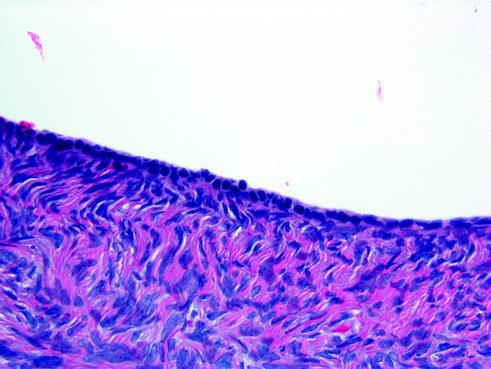

Fig. 14.14
Ovarian surface epithelium. The ovarian surface is lined by a single layer of low columnar to cuboidal cells
As discussed above, the primary oocytes with their surrounding specialized stromal (granulosa) cells are known as primordial follicles (Fig. 14.15). As the oocyte and its surrounding cells become larger, the primordial follicle transitions into a primary follicle. As the surrounding granulosa cells proliferate, a strongly eosinophilic band forms around the oocyte—the zona pellucida. Simultaneously, through inductive effects, the cortical stromal cells immediately surrounding the granulosa cells develop into the theca interna and externa. The oocyte takes on an eccentric position within the follicle structure, giving rise to an antral (or Graafian) follicle. During reproductive years, one follicle will become dominant during each ovulatory cycle and will continue to grow into a dominant follicle. Immediately following the luteal surge (peak level of luteinizing hormone) ovulation occurs, and the oocyte is liberated into the peritoneal cavity. Following ovulation, the granulosa cells—in response to progestin stimulation—become enlarged with pale eosinophilic cytoplasm. If pregnancy occurs, the granulosa cells continue to enlarge and accumulate eosinophilic material in the cytoplasm, forming a corpus luteum of pregnancy (Fig. 14.16). At the conclusion of the pregnancy, the corpus luteum undergoes regression and hyalinization to form a persistent structure: the corpus albicans [63].
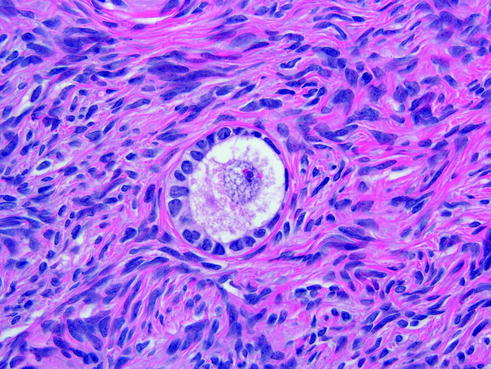
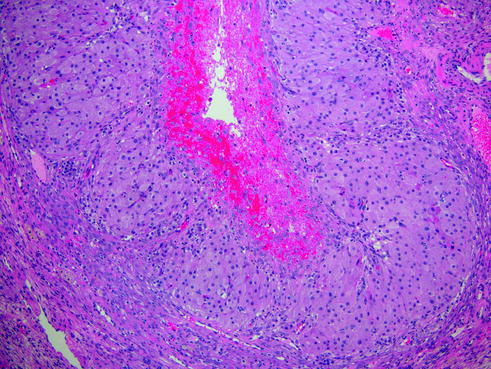

Fig. 14.15
Primordial follicles. They are composed of a primary oocyte surrounded by a single layer of flattened follicular cells

Fig. 14.16
Corpus luteum of pregnancy. Note the eosinophilic cytoplasm of the granulosa cells in response to progestin stimulation
14.4.2 Non-neoplastic Disorders of the Ovary
14.4.2.1 Follicle Cysts
If the luteal surge does not occur properly (which commonly occurs around menarche and menopause), follicle cysts lined by luteinized granulosa cells or theca cells may result [64]. They typically are small (less than 4–5 cm), thin-walled cysts with smooth surfaces, containing either blood or serous fluid.
14.4.2.2 Corpus Luteum Cysts
If a corpus luteum attains a diameter over 3 cm, it may be termed a corpus luteum cyst. These structures may rupture, leading to irritation of the peritoneum.
14.4.2.3 Polycystic Ovarian Syndrome (PCOS)
The hallmark finding is bilateral ovarian enlargement with numerous small cystic follicles surrounded by an eosinophilic collagenized stroma. The etiology of the disorder is not known and disease presentation may be variable with infertility (due to persistent anovulation), obesity, and hirsutism. Elevated levels of luteinizing hormone are seen which are thought to stimulate theca cells to synthesize excess androgens, some of which are converted to estrogens; thus, these patients may be susceptible to estrogen-driven endometrial carcinomas [65, 66].
14.4.2.4 Stromal Hyperthecosis (Cortical Stromal Hyperplasia)
There is significant overlap in clinical presentation between PCOS and stromal hyperthecosis, although the latter is more commonly seen in postmenopausal women. In this case, there is bilateral ovarian enlargement (up to 7–8 cm), but instead of numerous cysts, there is proliferation of luteinized stromal cells which are often arranged in nests [67]. Clinically, women with stromal hyperthecosis often display striking virilizing symptoms, along with obesity. Similar to PCOS, patients with stromal hyperthecosis are at increased risk of developing estrogen associated endometrial carcinomas.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree