Abstract
Pulsatile gonadotropin-releasing hormone (GnRH) release from the hypothalamus governs pituitary secretion of luteinizing hormone and follicle-stimulating hormone, which in turn regulate the production of gonadal sex steroids and gametes (ova and sperm). GnRH secretion is markedly influenced by a complex array of higher-level afferent inputs, such as neurons releasing kisspeptin, neurokinin B, and dynorphin. Feedback signals communicate the status of gonadal function and other facets of whole-body homeostasis to the neural systems regulating GnRH release and to pituitary gonadotropes, rendering a coordinated and tightly regulated feedback system that maintains appropriate gonadal function.
Keywords
Gonadotropin-releasing hormone, luteinizing hormone, follicle-stimulating hormone, kisspeptin, neurokinin B, dynorphin, KNDy neurons
Central Control of Reproduction
Successful reproduction is essential to the survival of a species. The reproductive system represents a highly complex functional organization of diverse tissues and signaling pathways that, when properly functioning, ensures a number of key endpoints, the most important of which are the adequate production of gametes (ova and sperm); successful delivery of gametes for fertilization; and, in women, physiologic preparation for possible pregnancy. Neuroendocrine systems are the principal drivers of reproductive function in both men and women. In particular, hypothalamic gonadotropin-releasing hormone (GnRH) is the primary, if not exclusive, feedforward signal to gonadotrope cells of the anterior pituitary, stimulating the synthesis and secretion of both luteinizing hormone (LH) and follicle-stimulating hormone (FSH). Together, these two gonadotropins direct the primary functions of the reproductive axis: gametogenesis and gonadal sex steroid synthesis.
Given its critical importance to a species, the reproductive system must be robust, continuing to function properly in the face of various physiologic perturbations. In contrast, in settings of marked physiologic stress (e.g., significantly reduced energy availability), mechanisms that temporarily limit fertility—the usual outcome of which is metabolically expensive in women—are biologically advantageous for the individual and, ultimately, the species. Appropriate function (or quiescence) of the reproductive system is governed by a number of intricate relationships. For example, feedback signals from the gonads (e.g., sex steroid concentrations) communicate the status of gonadal function to the hypothalamic-pituitary axis; these signals in turn influence GnRH and gonadotropin output, rendering a coordinated and tightly regulated feedback system that maintains gonadal function within narrow limits. The reproductive system also has extensive interactions with other neuroendocrine systems, such as those involved with energy balance and adaptations to stress. The reproductive neuroendocrine system integrates these myriad feedback signals, and the GnRH-secreting neuronal network represents the final common pathway for the central control of reproduction. Thus the regulation of GnRH secretion represents a major focus of reproductive neuroendocrinology.
Much of our understanding of reproductive neuroendocrinology has emerged from the study of rodents, sheep, and nonhuman primates, which largely reflects the ethical boundaries inherent to human research. Because many neurobiologic principles are similar among all mammals, these animal studies have been (and continue to be) indispensable. Nonetheless, certain aspects of reproductive neuroendocrinology may differ markedly among species. Thus, when available, human data will be prioritized throughout this chapter, but animal studies will also be discussed when appropriate, recognizing that specific findings may or may not be generalizable to humans. The reader is referred to Chapter 3 , Chapter 7 , Chapter 12 , Chapter 17 , Chapter 20 for additional treatment of neuroendocrine physiology and pathophysiology related to reproduction.
Neuroendocrinology: The Interface Between Neurobiology and Endocrinology
Endocrinology is the study of cell-to-cell signaling that occurs via specific chemicals (hormones) that travel through the bloodstream to influence remote targets. The term “neuroendocrinology” refers to the involvement of the central nervous system (CNS)—the hypothalamus in particular—in this process. This field of study has traditionally focused on hypothalamic neuron-derived factors that influence various target tissues, either directly, as with the hormones of the neurohypophysis, or indirectly, as with hypothalamic releasing factors that control anterior pituitary hormone secretion. Neuroendocrine systems direct a wide variety of critical biologic processes, such as growth and development, energy and fluid homeostasis, responses to stress, and reproduction.
Neurons are highly specialized and morphologically diverse cells that transmit information via electrical impulses called action potentials. Neurons have a cell body containing the cell nucleus, mitochondria, and synthetic organelles. Neurons also have cell processes that participate in the reception and delivery of electrical impulses ( Fig. 1.1 ). Dendrites are short processes—often extensively branched to increase surface area—that typically receive information (afferent electrical impulses). The axon is a single neuronal extension that generally transmits efferent electrical impulses away from the cell body in a process called neuronal firing. However, as described later, GnRH neuron fibers extending from the cell body to the median eminence (the location of GnRH release) in mice demonstrate characteristics of both axons and dendrites and, thus, have been called dendrons.
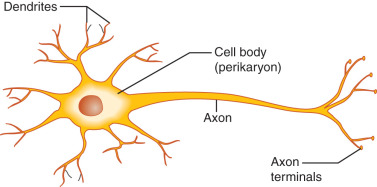
In unstimulated neurons, the inner portion of the neuronal membrane is negatively charged compared with the outer membrane surface; this resting membrane potential is typically between −50 and −75 mV in GnRH neurons. Such electrical polarization reflects transmembrane ionic differences, which are maintained by protein channels that govern transmembrane passage of specific ions (e.g., sodium, potassium, chloride). Regulated changes of transmembrane ion differences may cause the membrane potential to become more or less negative (hyperpolarization and depolarization, respectively). Depolarization to a certain threshold results in a rapid and temporary reversal of membrane potential (an action potential), which is propagated along the neuronal membrane. Notably, the amplitude of the action potential does not vary with the strength of stimulation; instead, once a threshold is reached, a full action potential occurs—the so-called all-or-none phenomenon . However, the degree of neuronal stimulation can alter the frequency of action potentials generated. In this way, neurons transmit information to other neurons and effector tissue cells.
Neuronal signals are transferred across neuron-to-neuron connections (synapses) via chemical neurotransmitters. This process begins with bursts of neuronal firing, which result in the opening of voltage-gated calcium channels at the axonal terminal. The influx of calcium promotes exocytosis of neurotransmitter-containing synaptic vesicles, releasing neurotransmitters into the synaptic cleft. Neurotransmitters then bind to specific ligand-dependent ion channels in the postsynaptic membrane, which can stimulate an action potential in the postsynaptic neuron. A wide variety of factors serve as neurotransmitters, including amino acids (e.g., acetylcholine, glutamate, γ-aminobutyric acid [GABA]), biogenic amines (e.g., norepinephrine, epinephrine, dopamine, serotonin), and neuropeptides (e.g., kisspeptin, neurokinin B [NKB], dynorphin, β-endorphin, somatostatin, proopiomelanocortin [POMC], neuropeptide Y [NPY]).
Bursts of neuronal firing can also elicit release of neuronal products into the bloodstream to influence remote targets (i.e., neurosecretion of neurohormones). Hypophysiotropic neurons are specialized hypothalamic neurons that secrete peptide-releasing factors—GnRH, corticotropin-releasing hormone (CRH), thyrotropin-releasing hormone (TRH), and growth hormone–releasing hormone (GHRH)—into the hypophyseal portal circulation. These releasing factors in turn stimulate specific anterior pituitary cell populations. In contrast, hypothalamic release of dopamine into the portal circulation provides tonic inhibition of pituitary prolactin secretion. Hypothalamic neurosecretion of vasopressin and oxytocin, which are released directly into the systemic circulation, alter the function of distant targets, such as the renal tubules and uterus, respectively.
Neuroglial cells (e.g., astrocytes, ependymal cells, oligodendrocytes, and microglia) represent approximately 90% of cells in the CNS. Neuroglia do not conduct action potentials, but they perform critical supportive functions. For example, astrocytes form the supportive framework of the CNS; help isolate synaptic junctions (to prevent nonspecific spread of neuronal impulses); facilitate nutrient delivery to neurons; and contribute to the blood-brain barrier. In addition, astrocytes have been implicated in the control of GnRH secretion and the mechanisms underlying pubertal onset. For example, astrocytes may impact neuronal activity via secretion of numerous growth factors, and astrocytes abundantly appose GnRH neurons; these contacts can influence synaptic input, and they may be influenced by estrogen in both rodents and nonhuman primates. Similarly, specialized ependymal cells (tanycytes) in the median eminence appear to modify access of GnRH neuron terminals to the hypophyseal portal blood.
Anatomy of the Reproductive Hypothalamic-Pituitary Axis
- ◆
GnRH neuronal cell bodies are located in the infundibular (arcuate) nucleus and the medial preoptic area of the hypothalamus.
- ◆
GnRH neurons extend processes (dendrons) to the median eminence, where GnRH gains access to the hypophyseal portal system.
- ◆
The hypophyseal portal circulation represents the functional connection between hypothalamic GnRH neurons and the gonadotropes of the anterior pituitary.
Portions of the hypothalamus and the anterior pituitary gland constitute the primary effector arm of the central reproductive axis. In particular, hypothalamic neural systems regulate GnRH release into the hypophyseal portal veins, with GnRH being the signal to gonadotropes (anterior pituitary) to secrete LH and FSH. In turn, these gonadotropins direct gonadal (ovarian and testicular) function.
Hypothalamus
The hypothalamus is located at the base of the brain ( Fig. 1.2 ). Although small (approximately 10 g, less than 1% of total brain weight), it performs critical functions for maintenance of whole-organism homeostasis, including regulation of hunger and body weight, growth, various aspects of metabolism, thirst and renal water handling, body temperature, autonomic function, sleep, circadian rhythms, and emotion. Importantly, the hypothalamus is also a primary control center for reproduction and influences sexual behavior.
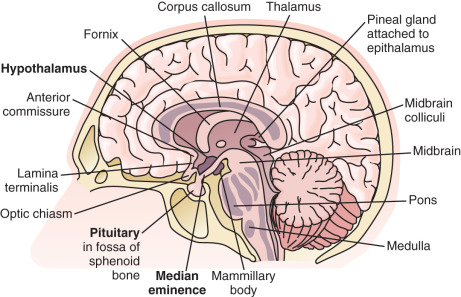
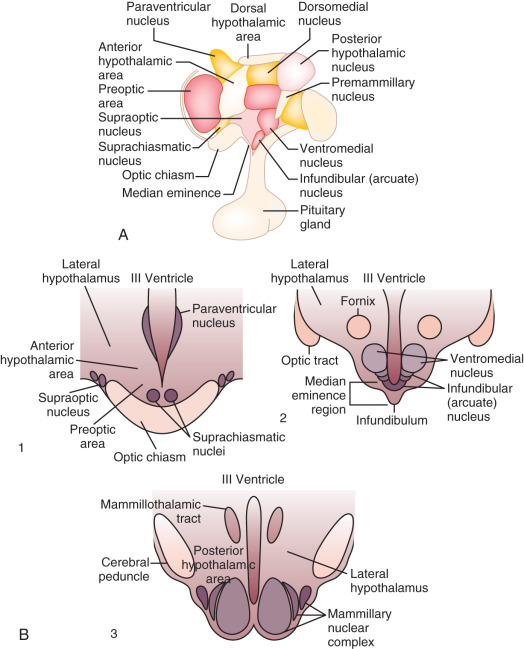
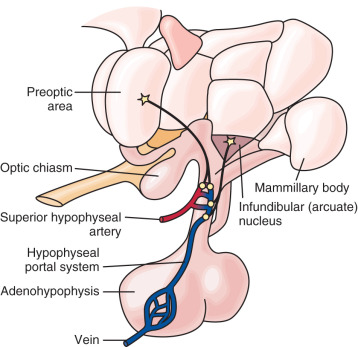
As an anatomic structure, the hypothalamus does not have discrete borders, but in general it forms the floor and inferior-lateral walls of the third ventricle ( Fig. 1.3 ). The medial portions of the hypothalamus are primarily made up of cell bodies, whereas the lateral portions are mostly composed of neuron fibers (axons), such as those connecting the medial hypothalamus to other areas of the brain. (The hypothalamus is extensively interconnected with other brain areas.) By convention, closely associated collections of neuron cell bodies are called nuclei; and the paraventricular, dorsomedial, ventromedial, and infundibular nuclei contain a majority of the neurons that secrete hypophysiotropic hormones into the portal circulation. (The human infundibular nucleus is the analogue to the arcuate nucleus in lower mammalian species.) GnRH cell bodies do not form discrete nuclei but are instead diffusely located throughout the preoptic area and the mediobasal hypothalamus ( Fig. 1.4 ); the latter is situated caudal to the preoptic area, extending from the retrochiasmatic area (i.e., the area situated behind the optic chiasm) to the mammillary bodies, and including both the infundibular (arcuate) nucleus and the median eminence.
Median Eminence
Positioned at the base of the third ventricle, the median eminence is part of the anatomic link between the hypothalamus and anterior pituitary. The internal zone of the median eminence is located along the ventral floor of the third ventricle and is largely composed of axonal fibers from both magnocellular neurons (larger neurons that secrete vasopressin and oxytocin) and hypophysiotropic neurons as they travel from hypothalamic nuclei/areas to their final destinations—the neurohypophysis (posterior pituitary) and the external zone of the median eminence, respectively ( Fig. 1.5 ). The external zone contains hypophysiotropic neuron terminals, which release hypophysiotropic hormones into an extensive capillary plexus—the proximal end of the hypophyseal portal system. Some nerve terminals in this zone act on other nerve terminals to influence hormone release (e.g., kisspeptin neurosecretion at GnRH neuron terminals influences GnRH release).
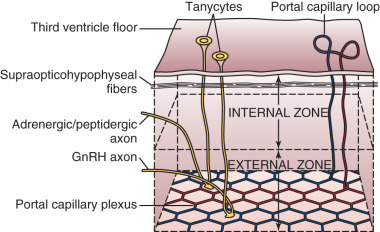
The ependymal layer lining the third ventricle includes a population of specialized ependymal cells called tanycytes, which have a short process extending toward the ventricular surface and a long process extending into the median eminence toward areas around portal capillaries. The latter tanycyte projections envelop or retract from GnRH nerve terminals during episodes of low and high GnRH neuronal activity, respectively. Thus tanycytes may influence GnRH secretion by physically isolating GnRH neuron terminals from portal capillaries—a regulated process. Tanycytes may also represent a link between cerebrospinal fluid and the external zone of the median eminence (e.g., by transporting substances from the third ventricle to portal blood).
The median eminence is among the so-called circumventricular organs, which lie adjacent to the ventricular system and represent openings in the blood-brain barrier. Although lipid-soluble molecules can diffuse in and out of the CNS relatively easily, and cellular transport mechanisms allow selective entry of ions, the blood-brain barrier functions to protect certain regions of the brain and hypothalamus from larger charged molecules, with physical protection provided by (1) tight junctions between endothelial cells and (2) neuron-capillary separation by both astrocyte foot processes and microglia. However, the CNS requires feedback signals—including hormonal, metabolic, and toxic cues—via macromolecules of peripheral origin that would otherwise be excluded by the blood-brain barrier; accordingly, capillaries of the circumventricular organs are fenestrated and permit transcapillary exchange of larger charged molecules (e.g., proteins, peptide hormones). Thus the median eminence represents a key access point for central sensing of peripheral cues. Similarly, fenestrated vessels readily allow entry of hypothalamic releasing factors into portal blood.
Hypophyseal Portal Circulation
No direct neuronal connections exist between the hypothalamus and the anterior pituitary. However, the hypophyseal portal circulation (hypothalamic-hypophyseal portal system, pituitary portal system) represents the functional connection between the median eminence and anterior pituitary (see Fig. 1.4 ). The superior hypophyseal artery—a branch of the internal carotid artery—subdivides to form an extensive capillary network in the external zone of the median eminence, with loops that reach into the inner zone. Capillary blood then drains into sinusoids that converge into the hypophyseal portal veins. Traversing the pituitary stalk, the hypophyseal portal system forms the primary blood supply of the anterior pituitary. The direction of blood flow is primarily, but not exclusively, from the hypothalamus to the anterior pituitary; some retrograde flow allows for short-loop hypothalamic feedback.
Pituitary Gland (Hypophysis)
The pituitary gland appears as an extension at the base of the hypothalamus and resides cradled within the sella turcica, a saddlelike structure of the sphenoid bone (see Fig. 1.2 ). The adenohypophysis (anterior pituitary) is of ectodermal origin, derived from an upward invagination of pharyngeal epithelium (Rathke pouch) during embryologic development. The adenohypophysis is composed of primarily the anterior lobe (pars distalis), which contains specialized cell populations that produce specific hormones: gonadotropes (the gonadotropins LH and FSH), mammotropes (prolactin), corticotropes (adrenocorticotropic hormone [ACTH]), thyrotropes (thyroid-stimulating hormone [TSH]), and somatotropes (growth hormone). The intermediate lobe is vestigial in adult humans but includes a small population of cells (e.g., POMC cells) in contact with the posterior lobe; the pars tuberalis is a slender layer of tissue (e.g., LH-producing cells and TSH-producing cells) surrounding the infundibulum (the funnel-shaped connection between the hypothalamus and the posterior pituitary) and pituitary stalk.
In contrast to the adenohypophysis, the neurohypophysis (posterior pituitary) is composed of neural tissue and forms as a downward extension of neuroectodermal tissue from the infundibulum during embryologic development. It is thus a direct extension of the hypothalamus. The neurohypophysis includes the infundibular stalk and the pars nervosa (posterior lobe of the pituitary). The supraoptic and paraventricular nuclei include magnocellular neurons that produce oxytocin and arginine vasopressin (AVP; also known as antidiuretic hormone [ADH]), respectively; these axons project to the posterior lobe of the pituitary, where oxytocin and AVP are secreted into a capillary network that drains into the hypophyseal veins (i.e., directly into the systemic circulation). The posterior lobe also includes specialized glial cells called pituicytes, which envelop or retract from magnocellular nerve terminals during episodes of low and high neuronal activity, respectively.
Gonadotropin-Releasing Hormone: The Final Common Pathway for the Central Control of Reproduction
- ◆
Pulsatile GnRH secretion is the proximate stimulus for LH and FSH synthesis and secretion by pituitary gonadotropes.
- ◆
Although numerous internal and external factors influence gonadotropin secretion via numerous neuronal pathways, GnRH is the final common pathway for the stimulation of LH and FSH release.
GnRH, previously called luteinizing hormone–releasing hormone (LHRH), is synthesized and released from a relatively small population of specialized hypothalamic neurons. GnRH was initially isolated from porcine hypothalami and shown to stimulate pituitary gonadotropin release. Although the primary function of GnRH is to regulate pituitary gonadotropin secretion, GnRH also appears to have autocrine and paracrine functions in diverse tissues (e.g., ovary, placenta).
The regulation of GnRH secretion is complex and involves overlapping pathways, which likely increases the robustness of central reproductive function. However, there are no known parallel or backup pathways for the stimulation of gonadotropin secretion. Thus natural fertility is absolutely dependent on appropriate GnRH secretion. For example, mice with mutations of the GnRH-1 gene are hypogonadal, but reproduction can be restored via GnRH-1 gene therapy or transplantation of fetal GnRH neurons. Similarly, a variety of human conditions associated with absent (or near-absent) GnRH secretion lead to pubertal failure, hypogonadotropic hypogonadism, and infertility, all of which can be fully reversed with exogenous GnRH therapy.
GnRH secretion is influenced by numerous factors, including sex steroids, energy availability, and stress. In some mammalian species, GnRH secretion is also affected by circadian rhythms, photoperiod (e.g., seasonal breeders such as sheep), social cues, and pheromones.
Gonadotropin-Releasing Hormone Structure
GnRH (GnRH-1 in particular) is a decapeptide, with the amino acid structure (pyro)Glu-His-Trp-Ser-Tyr-Gly-Leu-Arg-Pro-Gly-NH 2 . The amino acid structure of GnRH is identical in essentially all mammalian species; with the exception of the central Tyr-Gly-Leu-Arg segment, the amino acids of GnRH are highly conserved among vertebrate species. The GnRH-1 gene (GNRH1) is located on human chromosome 8 (8p11.2-p21) and produces a 92–amino acid precursor peptide called prepro-GnRH, which includes a signal sequence (23 amino acids), GnRH (10 amino acids), a proteolytic processing site (3 amino acids), and GnRH-associated peptide (56 amino acids) ( Fig. 1.6 ). The latter peptide can stimulate gonadotropin secretion and inhibit prolactin secretion, although its precise physiologic role, if any, remains unclear. The actions of GnRH are mediated through the GnRH type I receptor.
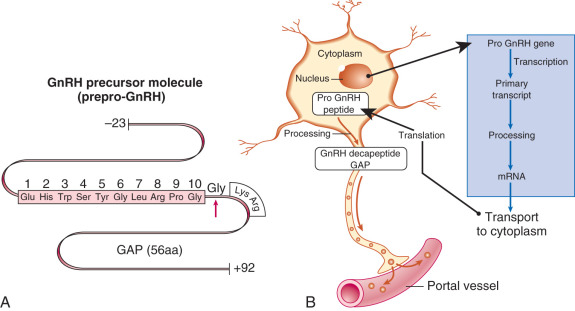
Another form of GnRH (GnRH-2) and its receptor have been identified in a variety of animal species, including humans. GnRH-2 is a decapeptide with similar structure to GnRH-1: (pyro)Glu-His-Trp-Ser- His -Gly- Trp – Tyr -Pro-Gly-NH2 (italicized amino acids denote differences compared with GnRH-1). However, the gene for GnRH-2 is located on human chromosome 20 (20p13). GnRH-2 is widely expressed in the CNS and extra-CNS tissues, and it may contribute to reproductive behavior regulation in some species. In lower animals, GnRH-2 can act via its own receptor, which is structurally and functionally distinct from the GnRH type I receptor. Although a homologue of the GnRH-2 receptor gene has been detected in humans, it includes a frameshift and premature stop codon. Thus the physiologic role of GnRH-2 in humans remains unclear.
Anatomy of Gonadotropin-Releasing Hormone-Secreting Neurons
GnRH neurons are a heterogeneous population of hypothalamic neurons. They are relatively few, numbering approximately 1500, and the majority of GnRH neuronal cell bodies are located in the infundibular (arcuate) nucleus—part of the mediobasal hypothalamus—and the medial preoptic area. Although GnRH neurons are rather loosely affiliated anatomically, they are functionally integrated and form a complex network with numerous interconnections, in addition to connections to other neuronal populations. Of interest in this regard, work in mice suggests that the GnRH neuron fibers extending from the cell body to the median eminence are morphologically atypical: although they do not exhibit many of the molecular markers classically associated with axons or dendrites, they demonstrate morphologic and functional characteristics of both axons and dendrites, including functional synaptic inputs throughout the length of the fiber. The term dendron has been proposed for such projections.
The GnRH neurons in the mediobasal hypothalamus—the infundibular (arcuate) nucleus in particular—appear to be requisite for gonadotropin secretion. For example, selective radiofrequency ablation of the arcuate nucleus in adult female monkeys obliterates gonadotropin secretion. GnRH neurons extend projections through the tuberoinfundibular tract to the median eminence, where neuron terminals gain access to the hypophyseal portal system.
The physiologic function of other GnRH neurons, which arise from the anterior and posterior hypothalamus and project to the limbic system and posterior pituitary, respectively, remains unclear, although some of these circuits may possibly be involved with various behavioral responses.
Embryologic Development of the Gonadotropin-Releasing Hormone Neuronal Network
The ontogeny of GnRH neurons in vertebrate species is unique among neuronal systems of the CNS: nascent GnRH neurons are initially identified outside of the CNS in the nasal placode (sometimes called the olfactory placode). However, GnRH cells migrate during embryologic development, as directly observed in embryonic nasal explant cultures and in embryonic head slices (mouse model). The specific migratory pathway of GnRH neurons was first demonstrated in mice by documenting the presence of GnRH-immunoreactive cells in different areas at different stages of embryonic development ( Fig. 1.7 ). Specifically, GnRH expression is first observed within the nasal placode circa embryonic day 10 or 11. By embryonic day 13, GnRH-expressing cells are primarily located around the cribriform plate, and GnRH-expressing cells begin to reach the hypothalamus by embryonic day 14, approaching their final positions around embryonic day 16. This migratory pathway has been confirmed in both nonhuman primates and humans.
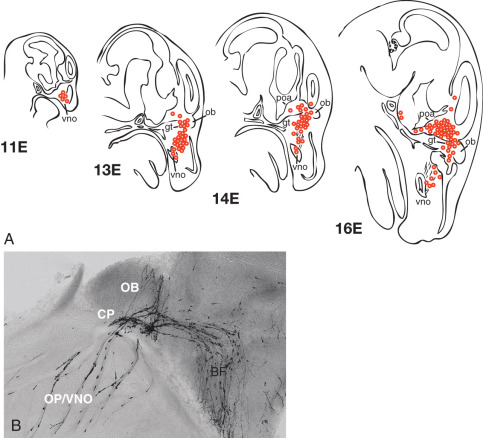
Successful migration of GnRH neurons is inextricably intertwined with olfactory system development, perhaps reflecting the close functional relationship between reproduction and the olfactory system (e.g., pheromones) in mammalian phylogeny. The nasal placode gives rise to nasal epithelium and olfactory sensory neurons, the latter of which extend axonal projections to the olfactory bulb. Vomeronasal neurons are a subset of olfactory neurons believed to be involved with pheromone detection; these axons originate in the vomeronasal organ and largely extend to the accessory olfactory bulb. At the level of the cribriform plate, some olfactory (vomeronasal) axons separate and form a branch that extends caudally into the forebrain. Of great importance, migrating GnRH neurons maintain adhesion to these axons; thus these olfactory neurons form a critical guidance track for GnRH neuronal migration across the nasal epithelium and through the forebrain toward the hypothalamus.
The dependence of GnRH neuronal migration on normal olfactory system development is exemplified by Kallmann syndrome, a form of congenital hypogonadotropic hypogonadism accompanied by absent sense of smell (anosmia). In this syndrome, faulty development of the olfactory system renders an inadequate guidance infrastructure for migrating GnRH neurons, leading to failure of GnRH neurons to reach the hypothalamus. The first identified cause of Kallmann syndrome was deletion of the Kallmann syndrome 1 sequence (KAL1) gene, which is located on the X chromosome (Xp22.3) and encodes anosmin-1, a secreted matrix glycoprotein expressed in the presumptive olfactory bulb. Although precise mechanisms are unclear, anosmin-1 is believed to be important for the formation of olfactory elements that provide migratory guidance to GnRH neurons as they move out of the nasal placode. Evaluation of a 19-week-old human fetus with X-linked Kallmann syndrome demonstrated GnRH-immunoreactive cells within a tangle of olfactory and vomeronasal nerves at the dorsal surface of the cribriform plate, along with the absence of olfactory tracts and bulbs. In a second human fetus (16 weeks) with X-linked Kallmann syndrome, GnRH was detected along terminal nerve fascicles in the nasal mucosa only. This syndrome illustrates that, without the guidance framework provided by the olfactory neuronal system, GnRH neurons fail to migrate into the hypothalamus and thus cannot release GnRH into the hypophyseal portal system.
A number of additional single-gene defects have been associated with Kallmann syndrome, including mutations in the genes for prokineticin 2 (PROK2) and its receptor (PROKR2) , fibroblast growth factor-8 (FGF8) and its receptor fibroblast growth factor receptor 1 (FGFR1) , NMDA receptor synaptonuclear signaling and neuronal migration factor ( NSMF ; formerly called nasal embryonic LH-releasing hormone factor [NELF]), and chromodomain helicase DNA binding protein 7 (CHD7) . The importance of these genes in GnRH neuronal development is corroborated by mouse studies. For example, in fetal mice lacking either Prok2 or Prokr2 , GnRH neurons are trapped in a tangled web of olfactory/vomeronasal axons, with few, if any, reaching the forebrain. Although these gene products are clearly important for GnRH neuron ontogeny, their precise roles remain uncertain.
Mouse studies suggest other important factors underlying GnRH neuron migration during prenatal development. For example, the chemokine (C-X-C motif) receptor 4 (CXCR4) is expressed on murine GnRH neurons and interacts with a secreted chemokine called stromal cell–derived factor-1 (SDF-1), which is present as a gradient in the nasal mesenchyme. This gradient, with highest concentrations at the cribriform plate, provides directional information as GnRH neurons migrate toward the cribriform plate; GnRH cell migration across the nasal compartment is markedly impaired in CXCR4 knockout mice. As another example, extension of the caudal branch of the vomeronasal nerve toward the ventral forebrain involves chemoattraction via interactions between netrin-1—a chemokine expressed as a gradient in the forebrain—and its receptor, deleted in colorectal cancer (DCC). In mice without either netrin-1 or DCC, the caudal branch of the vomeronasal nerve extends toward the cerebral cortex rather than the ventral forebrain; and GnRH neurons follow this path, ultimately residing in the cerebral cortex. Animal studies have suggested a number of such interactions in (1) guidance of olfactory neurons toward the forebrain and (2) the association between migrating GnRH neurons and axons of olfactory/vomeronasal nerves; but specific relevance to humans remains unclear.
After reaching the hypothalamus, GnRH neurons detach from olfactory nerve axons and may disperse further before resting. A critical next step is extension of GnRH neuronal projections (dendrons) to the median eminence, where GnRH gains access to the hypophyseal portal system.
Gonadotropin-Releasing Hormone Neuronal Firing and Gonadotropin-Releasing Hormone Secretion
GnRH neuronal activity is marked by bursts of action potentials (burst firing), the patterns and rates of which change across time; changes in GnRH neuron firing rates presumably relate to changes in GnRH secretion. Variable firing rate patterns (e.g., times of high and low firing rates) appear to be intrinsic to GnRH neurons, but they can also be altered by neurotransmitters and neuromodulators (e.g., glutamate, GABA, kisspeptin). Although sex steroids can markedly influence GnRH neuronal firing rates, GnRH neurons lack the primary receptors mediating sex steroid feedback (i.e., estrogen receptor alpha, progesterone receptor, androgen receptor); however, many studies suggest that sex steroid actions on GnRH neuronal activity are mediated primarily by afferent neurons (e.g., those secreting glutamate, GABA, kisspeptin).
GnRH neuron cell bodies are relatively scattered across the mediobasal hypothalamus and preoptic area, yet GnRH is secreted into the hypophyseal portal system in a coordinated, pulsatile fashion. Specifically, GnRH secretion is marked by episodic bursts of hormone release into the portal system, as demonstrated in rats, sheep, and monkeys. After being released into the portal vascular compartment, GnRH is rapidly degraded via enzymatic proteolysis, and the half-life of GnRH in the blood is very short—approximately 2 to 4 minutes. Thus GnRH presentation to gonadotrope cells is intermittent.
Pulsatile GnRH secretion is an absolute requirement for long-term stimulation of gonadotropin synthesis and secretion, and there is a relatively narrow window of GnRH pulse frequency and amplitude that will optimally stimulate gonadotropin secretion. Intermittent GnRH stimulation of gonadotrope cells can increase (or maintain) GnRH receptors on gonadotropes—the self-priming or autopriming effect. Thus intermittent GnRH stimulation facilitates or maintains gonadotrope responsiveness to GnRH. However, more frequent exposure to GnRH pulses can reduce gonadotropin responses to GnRH ; at one extreme, continuous GnRH receptor stimulation leads to marked desensitization of gonadotropin synthesis and secretion. In a classic experiment involving rhesus monkeys with hypothalamic lesions that abolished GnRH secretion, intermittent (once an hour) exogenous GnRH administration restored pituitary gonadotropin secretion. However, changing from intermittent to continuous GnRH administration resulted in marked desensitization of gonadotropin release ( Fig. 1.8 ). Although reduced GnRH receptor expression on gonadotropes (i.e., receptor downregulation) plays a role in desensitization, additional mechanisms contribute to the uncoupling of GnRH receptor agonism and gonadotropin synthesis.
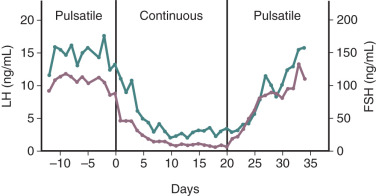
The foregoing phenomenon can be exploited therapeutically with the use of long-acting GnRH receptor agonists. Such agonists are peptides with structures very similar to that of GnRH but with amino acid substitutions that enhance receptor binding affinity, increase resistance to proteolytic degradation, or both ( Fig. 1.9 ), thus providing continuous GnRH receptor stimulation. Although initial GnRH receptor agonism temporarily (e.g., for 1 to 2 weeks) increases gonadotropin release (gonadotropin “flare”), continued agonism leads to desensitization of gonadotropin secretion with accompanying reductions of gonadal sex steroid concentrations to castrate levels (“medical oophorectomy,” “medical castration,” “pseudomenopause”), typically over several weeks. These agents are useful in the therapy of gonadotropin-dependent disorders such as central precocious puberty, endometriosis, and prostate cancer.
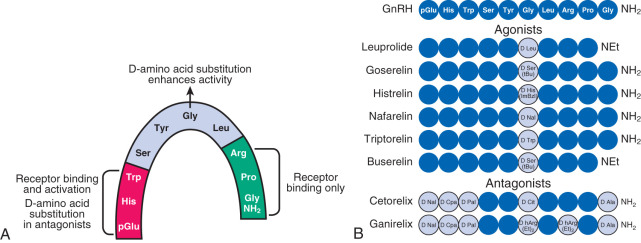
Peptide GnRH receptor antagonists are also available for clinical use. These antagonists reversibly bind to, but do not stimulate, the GnRH receptor (i.e., competitive antagonism). Thus these agents do not initially stimulate gonadotropin release, and they reduce gonadotropins more rapidly than GnRH agonists—usually within 24 to 72 hours.
Gonadotropin-Releasing Hormone Stimulation of Gonadotrope Cells
The specialized cells that synthesize and secrete gonadotropins (i.e., gonadotropes) are located mainly in the lateral portions of the anterior pituitary gland and constitute 7% to 10% of the adenohypophysis cell population. GnRH action at the pituitary gonadotrope begins with GnRH binding to the GnRH type I receptor on the plasma membrane. The GnRH type I receptor is a member of the seven-transmembrane receptor family, a G protein–coupled receptor, and encoded on chromosome 4. GnRH receptor density varies in different physiologic conditions and exhibits a positive correlation with gonadotrope responsiveness to GnRH (e.g., with both being high in rodents during preovulatory gonadotropin surges ). GnRH receptor density appears to be modulated primarily by GnRH, with intermittent GnRH stimulation leading to increased GnRH receptor expression; this is a central facet of the self-priming effect of GnRH and an important mechanism by which GnRH action is modulated in different physiologic states.
A majority of gonadotropes synthesize and secrete both LH and FSH. A detailed description of intracellular mechanisms of GnRH action on the gonadotrope is provided in Chapter 2 . Briefly, GnRH receptor binding activates the guanosine triphosphate (GTP)-binding protein G q/11 , leading to an increase in second messengers inositol 1,4,5-triphosphate (IP 3 ) and 1,2-diacylglycerol (DAG). Further intracellular signaling involves increased intracellular calcium and activation of various protein kinase C (PKC) isoforms, mitogen-activated protein kinases (e.g., extracellular signal–regulated kinase [ERK], c-Jun NH 2 -terminal kinase [JNK], and p38), calcium/calmodulin-dependent kinase II (Ca/CaMK II), and adenylate cyclase.
Each gonadotropin consists of two protein subunits, α and β. The 92–amino acid α-subunit is common to both LH and FSH—in addition to human chorionic gonadotropin (hCG) and TSH. β-Subunits for LH (LHβ) and FSH (FSHβ) are 121 and 117 amino acids in length, respectively, and account for the biologic specificity of these two hormones. GnRH stimulates gene expression of LHβ, FSHβ, and α-subunit, and the latter noncovalently dimerizes with either LHβ or FSHβ to form LH or FSH, respectively. Gonadotropins also undergo variable posttranslational modification, primarily glycosylation (addition of oligosaccharide moieties to specific amino acids), which influences bioactivity and elimination half-life. The gonadotropins are then packaged into secretory granules for eventual secretion.
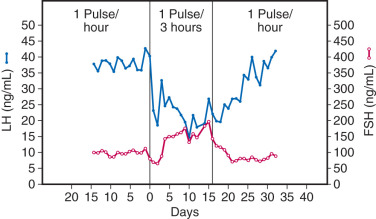
Although GnRH is the primary stimulus for LH and FSH synthesis and release from a common cell type, concentrations of these two gonadotropins vary differentially throughout ovulatory cycles, with FSH predominance in the early follicular phase and LH predominance in the late follicular phase. This sequential pattern of FSH and LH predominance is important for normal follicular maturation, ovarian steroid production, and subsequent ovulation. At least two mechanisms govern differential gonadotropin secretion throughout ovulatory cycles. First, both estradiol and inhibins selectively inhibit FSH release from gonadotropes during the mid- and late follicular phase and the luteal phase. Second, different patterns of pulsatile GnRH release differentially affect gonadotropin synthesis and secretion. Specifically, rapid (high frequency) GnRH pulses favor LH, whereas slower (low frequency) GnRH pulses favor FSH synthesis and secretion. For example, studies in ovariectomized, GnRH-deficient monkeys reveal that a decrease in the frequency of exogenously administered GnRH pulses from one pulse per hour to one pulse every 3 hours results in a 65% increase in plasma FSH, despite a 50% decrease in LH ( Fig. 1.10 ). Similar findings have been described in sheep and humans. Detailed studies in rats demonstrate that rapid GnRH pulse stimulation favors α-subunit and LHβ mRNA expression, whereas slow GnRH pulses favor FSHβ mRNA expression. The mechanisms effecting differential LH and FSH expression in response to changes in GnRH pulse frequency are complex but include variations of GnRH receptor number on the gonadotrope cell surface and alterations of gonadotrope activin β B and follistatin expression (discussed later in the chapter).