References
1. UICC. In: Sobin LH, Wittekind CH, eds. TNM classification of malignant tumours, 6 ed. New York, NY: Wiley-Liss, 2002.
2. Krege S, Beyer J, Souchon R, et al. European consensus conference on diagnosis and treatment of germ cell cancer: a report of the second meeting of the European Germ Cell Cancer Consensus group (EGCCCG): part I. Eur Urol 2008;53:478.
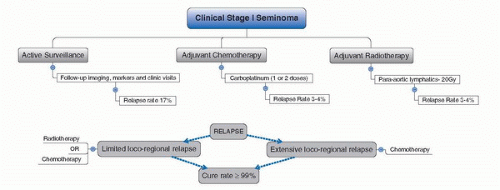
3. Warde P, Specht L, Horwich A, et al. Prognostic factors for relapse in stage I seminoma managed by surveillance: a pooled analysis. J Clin Oncol 2002;20:4448.
4. Loehrer PJ, Sr, Bosl GJ. Carboplatin for stage I seminoma and the sword of Damocles. J Clin Oncol 2005;23:8566.
5. Warde P, Gospodarowicz MK, Banerjee D, et al. Prognostic factors for relapse in stage I testicular seminoma treated with surveillance. J Urol 1997;157:1705.
6. Sava T, Consoli F, Santo A, et al. Adjuvant treatment in the management of testis-confined germ cell tumours after orchidectomy. BJU Int 2008;101:155.
7. Bosl GJ, Bajorin DF, Sheinfeld J, et al.: Cancer of the testis. In: Devita VT Jr, Hellman Rosenberg SA, eds.: Cancer: principles and practice of oncology. 7th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2005, pp 1269-1290.
8. Chung P, Parker C, Panzarella T, et al. Surveillance in stage I testicular seminoma – risk of late relapse. Can J Urol 2002;9:1637.
9. Gurkaynak M, Akyol F, Zorlu F, et al. Stage I testicular seminoma: para-aortic and iliac irradiation with reduced dose after orchiectomy. Urologia Internationalis 2003;71:385.
10. Garcia-Serra AM, Zlotecki RA, Morris CG, et al. Long-term results of radiotherapy for early-stage testicular seminoma. Cancer clinical trials. Am J Clin Oncol 2005;28:119.
11. Bokemeyer C, Schmoll HJ. Secondary neoplasms following treatment of malignant germ cell tumors. J Clin Oncol 1993;11:1703.
12. Travis LB, Curtis RE, Storm H, et al. Risk of second malignant neoplasms among long-term survivors of testicular cancer. J Natl Cancer Inst 1997;89:1429.
13. Travis LB, Fossã SD, Schonfeld SJ, et al. Second cancers among 40, 576 testicular cancer patients: Focus on long-term survivors. J Natl Cancer Inst 2005;97:1354.
14. Van Leeuwen FE, Stiggelbout AM, Van den Belt-Dusebout AW, et al. Second cancer risk following testicular cancer: a follow-up study of 1,909 patients. J Clin Oncol 1993;11:415.
15. Wanderas EH, Fossa SD, Tretli S. Risk of subsequent non-germ cell cancer after treatment of germ cell cancer in 2006 Norwegian male patients. Eur J Cancer Part A 1997;33:253.
16. Oliver RTD, Mason MD, Mead GM, et al. Radiotherapy versus single-dose carboplatin in adjuvant treatment of stage I seminoma: a randomised trial. Lancet 2005;366:293.
17. Loehrer PJ Sr, Bosl GJ. Carboplatin for stage I seminoma and the sword of damocles. J Clin Oncol 2005;23:8566.
18. Groll RJ, Warde P, Jewett MA. A comprehensive systematic review of testicular germ cell tumor surveillance. Crit Rev Oncol Hematol 2007;64:182.
19. National Comprehensive Cancer Network: The NCCN Testicular Cancer Clinical Practice Guidelines in Oncology http://www.nccn.org, vol. 1, Version 2, 2009.
20. Albers P, Albrecht W, Algaba F, et al. Guidelines on testicular cancer. Arnhem, The Netherlands: European Association of Urology (EAU), 2008:54.
21. Ernst DS, Brasher P, Venner PM, et al. Compliance and outcome of patients with stage 1 non-seminomatous germ cell tumors (NSGCT) managed with surveillance programs in seven Canadian centres. Can J Urol 2005;12:2575.
22. Moynihan C. Testicular cancer: the psychosocial problems of patients and their relatives. Cancer Surv 1987;6:477.
23. Koocher GP, O’Malley JE, Gogan JL, et al. Psychological adjustment among pediatric cancer survivors. J Child Psychol Psychiatry 1980;21:163.
24. Berkmen F, Hizli F. Stage I testicular seminoma: surveillance versus adjuvant radiotherapy. Turk J Cancer 2006;36:169.
25. Fleshner N, Warde P. Controversies in the management of testicular seminoma. Semin Urol Oncol 2002;20:227.
26. Rudberg L, Carlsson M, Nilsson S, et al. Selfperceived physical, psychologic, and general symptoms in survivors of testicular cancer 3 to 13 years after treatment. Cancer Nurs 2002;25:187.
27. Fossa SD, Abyholm T, Normann N, et al. Posttreatment fertility in patients with testicular cancer. III. Influence of radiotherapy in seminoma patients. Br J Urol 1986;58:315.
28. Fosså SD, Horwich A, Russell JM, et al. Optimal planning target volume for stage I testicular seminoma: a medical research council randomized trial. J Clin Oncol 1999;17:1146.
29. Joos H, Sedlmayer F, Gomahr A, et al. Endocrine profiles after radiotherapy in stage I seminoma: impact of two different radiation treatment modalities. Radiother Oncol 1997;43:159.
30. Huddart RA, Norman A, Shahidi M, et al. Cardiovascular disease as a long-term complication of treatment for testicular cancer. J Clin Oncol 2003;21:1513.
31. Zagars GK, Ballo MT, Lee AK, et al. Mortality after cure of testicular seminoma. J Clin Oncol 2004;22:640.
32. Van Den Belt-Dusebout AW, Nuver J, De Wit R, et al. Long-term risk of cardiovascular disease in 5-year survivors of testicular cancer. J Clin Oncol 2006;24:467.
33. Horwich A. Mortality and cancer incidence following radiotherapy for seminoma of the testis. Radiother Oncol 1994;30:193.
34. Jones WG, Fossa SD, Mead GM, et al. Randomized trial of 30 versus 20 Gy in the adjuvant treatment of stage I testicular seminoma: a report on Medical Research Council Trial TE18, European Organisation for the Research and Treatment of Cancer Trial 30942 (ISRCTN18525328). J Clin Oncol 2005;23:1200.
35. Jones RH, Vasey PA. Part I: testicular cancer-management of early disease. Lancet Oncol 2003;4:730.
36. Horwich A. Radiotherapy in stage I seminoma of the testis. J Clin Oncol 2004;22:585.
37. Fossa SD, Aass N, Kaalhus O. Radiotherapy for testicular seminoma stage I: treatment results and long-term post-irradiation morbidity in 365 patients. Int J Radiat Oncol Biol Phys 1989;16:383.
38. Niewald M, Freyd J, Fleckenstein J, et al. Low-dose radiotherapy for Stage I seminoma-long-term results. Int J Radiat Oncol Biol Phys 2006;66:1112.
39. Fatigante L, Ducci F, Campoccia S, et al. Longterm results in patients affected by testicular seminoma treated with radiotherapy: risk of second malignancies. Tumori 2005;91:144.
40. Classen J, Schmidberger H, Meisner C, et al. Para-aortic irradiation for stage I testicular seminoma: results of a prospective study in 675 patients. A trial of the German testicular cancer study group (GTCSG). Br J Cancer 2004;90:2305.
41. Raman JD, Nobert CF, Goldstein M. Increased incidence of testicular cancer in men presenting with infertility and abnormal semen analysis. J Urol 2005;174:1819.
42. Yeoh E, Razali M, O’Brien PC. Radiation therapy for early stage seminoma of the testis. Analysis of survival and gastrointestinal toxicity in patients treated with modern megavoltage techniques over 10 years. Aust Radiol 1993;37:367.
43. Schmoll HJ, Souchon R, Krege S, et al. European consensus on diagnosis and treatment of germ cell cancer: a report of the European Germ Cell Cancer Consensus Group (EGCCCG). Ann Oncol 2004;15:1377.
44. Schmoll HJ, Souchon R, Krege S, et al. European consensus on diagnosis and treatment of germ cell cancer: a report of the European Germ Cell Cancer Consensus Group (EGCCCG). Ann Oncol 2004;15:1377.
45. Feldman DR, Bosl GJ. Treatment of stage I seminoma: is it time to change your practice? J Hematol Oncol 2008;1:22.
46. Dieckmann KP, Bruggeboes B, Pichlmeier U, et al. Adjuvant treatment of clinical Stage I seminoma: is a single course of carboplatin sufficient? Urology 2000;55:102.
47. Oliver RTD, Edmonds PM, Ong JYH, et al. Pilot studies of 2 and 1 course carboplatin as adjuvant for stage I seminoma: should it be tested in a randomized trial against radiotherapy? Int J Radiat Oncol Biol Phys 1994;29:3.
48. Fosså SD, Gilbert E, Dores GM, et al. Noncancer causes of death in survivors of testicular cancer. J Natl Cancer Inst 2007;99:533.
49. Martin JM, Panzarella T, Zwahlen DB, et al. Evidence-based guidelines for following stage 1 seminoma. Cancer 2007;109:2248.
50. Meinardi MT, Gietema JA, Van Der Graaf WTA, et al. Cardiovascular morbidity in longterm survivors of metastatic testicular cancer. J Clin Oncol 2000;18:1725.
51. Travis LB, Andersson M, Gospodarowicz M, et al. Treatment-associated leukemia following testicular cancer. J Natl Cancer Inst 2000;92:1165.
52. Tarin TV, Sonn G, Shinghal R. Estimating the risk of cancer associated with imaging related radiation during surveillance for stage I testicular cancer using computerized tomography. J Urol 2009;181:627.
53. Rustin GJ, Mead GM, Stenning SP, et al. Randomized trial of two or five computed tomography scans in the surveillance of patients with stage I nonseminomatous germ cell tumors of the testis: medical research council trial TE08, ISRCTN56475197—The National Cancer Research Institute Testis Cancer Clinical Studies Group. J Clin Oncol 2007;25:1310.
54. Warde P, Gospodarowicz MK, Panzarella T, et al. Long term outcome and cost in the management of stage I testicular seminoma. Can J Urol 2000;7:967.
55. Lawrentschuk N, Fleshner N. Clinical stage I seminoma: the case for surveillance. World J Urol 2009;27:433.
56. Cheville JC. Classification and pathology of testicular germ cell and sex cord-stromal tumors. Urol Clin North Am 1999;26:595.
57. International Consultation on Urological Diseases (ICUD): Report of Consensus: Management of Stage I & Stage II Testicular Seminoma. Shanghai, Société Internationale d’Urologie 30th World Congress, Presented by Warde P, 2009.
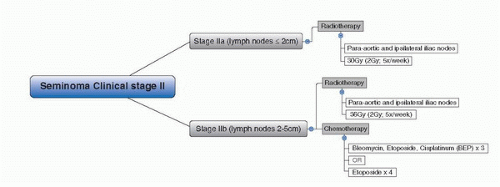
58. Zagars GK, Pollack A. Radiotherapy for stage II testicular seminoma. Int J Radiat Oncol Biol Phys 2001;51:643.
59. Chung PW, Warde PR, Panzarella T, et al. Appropriate radiation volume for stage IIA/B testicular seminoma. Int J Radiat Oncol Biol Phys 2003;56:746.
60. Classen J, Schmidberger H, Meisner C, et al. Radiotherapy for stages IIA/B testicular seminoma: final report of a prospective multicenter clinical trial. J Clin Oncol 2003;21:1101.
61. Zagars GK, Pollack A. Radiotherapy for stage II testicular seminoma. Int J Radiat Oncol Biol Phys 2001;51:643.
62. Classen J, Schmidberger H, Meisner C, et al. Radiotherapy for stages IIA/B testicular seminoma: final report of a prospective multicenter clinical trial. J Clin Oncol 2003;21:1101.
63. Detti B, Livi L, Scoccianti S, et al. Management of Stage II testicular seminoma over a period of 40 years. Urol Oncol 2009;27:534.
64. Chung PWM, Gospodarowicz MK, Panzarella T, et al. Stage II testicular seminoma: patterns of recurrence and outcome of treatment. Eur Urol 2004;45:754.
65. Patterson H, Norman AR, Mitra SS, et al. Combination carboplatin and radiotherapy in the management of stage II testicular seminoma: comparison with radiotherapy treatment alone. Radiother Oncol 2001;59:5.
66. Krege S, Beyer J, Souchon R, et al. European consensus conference on diagnosis and treatment of germ cell cancer: a report of the second meeting of the European Germ Cell Cancer Consensus Group (EGCCCG): part II. Eur Urol 2008;53:497.