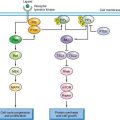
FIGURE 30.1 Model for the generation of genetic lesions during lymphomagenesis. B-NHL–associated genetic lesions appear to be due to mistakes occurring during the physiologic processes of somatic hypermutation and class-switch recombination in the highly proliferative environment of the germinal center (top). These include chromosomal translocations, which in most cases juxtapose the Ig genes to one of several protooncogenes (e.g., BCL6 or MYC), and aberrant somatic hypermutation of multiple target genes AID, activation-induced cytidine deaminase; SHM, somatic hypermutation; CSR, class-switch recombination.
Normal T-Cell Development
T-cell development proceeds through sequential stages defined according to the expression of the molecules CD4 and CD8. Committed lymphoid progenitors exit the bone marrow and migrate to the thymus as early T-cell progenitors or double-negative 1 (DN1) cells, which lack expression of CD4 and CD8 as well as of the T-cell receptor (TCR).31 In the thymic cortex, T cells advance through the double-negative stages DN2, DN3, and DN4, while undergoing specific rearrangements at the TCRγ locus in order to acquire expression of the pre-TCR.31 Those thymocytes that have successfully recombined the pre-TCR will be selected to further differentiate into double-positive cells (DP; CD4+CD8+), which express a complete surface TCR and can then enter a process of positive and negative selection in the medulla, before exiting the thymus as single positive T cells.31 The end result of this process is a pool of mature T cells that exhibit coordinated TCR and coreceptor specificities as required for effective immune responses to foreign antigens. Most mature T-NHLs arise from postthymic T cells in the lymphoid organs.
GENERAL MECHANISMS OF GENETIC LESION IN LYMPHOMA
Chromosomal Translocations
Chromosomal translocations are the genetic hallmark of malignancies derived from the hematopoietic system. Lymphoma-associated translocations represent reciprocal and balanced recombination events that occur between two specific chromosomes, are clonally represented in each tumor case, and are often recurrently associated with a given tumor type.
Although the precise molecular mechanisms that are responsible for the generation of translocations remain partially obscure, significant advances have been obtained during the past decade in our understanding of the events that are required for their initiation.32 It has now been documented that chromosomal translocations occur at least in part as a consequence of mistakes during Ig and TCR gene rearrangements in B and T cells, respectively. Based on the characteristics of the chromosomal breakpoint, three distinct scenarios can be distinguished: (1) translocations derived from mistakes of the RAG-mediated V(D)J recombination process, as is the case for translocations involving IGH and CCND1 in MCL or IGH and BCL2 in follicular lymphoma (FL)32–34; (2) translocations mediated by errors in the AID-dependent CSR process, such as those involving the Ig genes and MYC in sporadic Burkitt lymphoma (BL)32; and (3) translocations occurring as by-products of the AID-mediated SHM mechanism, which also generates DNA breaks, such as those joining the Ig and MYC loci in endemic BL.32 Conclusive experimental evidence for the involvement of antibody-associated remodeling events has been recently provided through in vivo studies performed in lymphoma-prone mouse models, where the removal of the AID enzyme was sufficient to abrogate the generation of MYC-IGH translocations in normal B cells undergoing CSR35,36 and to prevent the development of GC-derived B-NHL.37,38
The common feature of all NHL-associated chromosomal translocations is the presence of a protooncogene in proximity to the chromosomal recombination sites. In most lymphoma types, and in contrast with acute leukemias, the coding domain of the oncogene is not affected by the translocation, but its pattern of expression is altered as a consequence of the juxtaposition of heterologous regulatory sequences derived from the partner chromosome (protooncogene deregulation) (Fig. 30.2). Two distinct types of protooncogene deregulation (i.e., homotopic and heterotopic) can be distinguished. Homotopic deregulation occurs when the protooncogene becomes constitutively expressed in the lymphoma cell, while its expression is tightly regulated in normal lymphoid cells. Conversely, heterotopic deregulation occurs when the protooncogene is not expressed in the normal tumor counterpart and undergoes ectopic expression in the lymphoma. In most types of NHL-associated translocations, the heterologous regulatory sequences responsible for protooncogene deregulation are derived from antigen receptor loci that are expressed at high levels in the target tissue.32 However, in certain translocations, such as the ones involving BCL6 in diffuse large B-cell lymphoma (DLBCL), different promoter regions from distinct chromosomal sites can be juxtaposed to the protooncogene in individual tumor cases, a concept known as promiscuous translocations.39–46
Less commonly, B-NHL–associated chromosomal translocations juxtapose the coding regions of the two involved genes to form a chimeric unit that encodes for a novel fusion protein, an outcome typically observed in chromosomal translocation associated with acute leukemia (Fig. 30.2). Examples are the t(11;18) of mucosa-associated lymphoid tissue (MALT) lymphoma and the t(2;5) of anaplastic large cell lymphoma (ALCL). The molecular cloning of the genetic loci involved in most recurrent translocations has led to the identification of a number of protooncogenes involved in lymphomagenesis (Table 30.1).
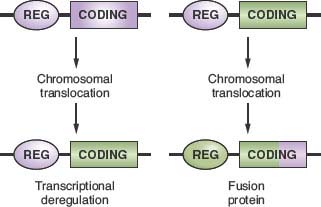
FIGURE 30.2 Molecular consequences of chromosomal translocations. Top panel: schematic representation of the two protooncogenes involved in prototypic chromosomal translocations, with their regulatory (REG) and coding sequences. Only one side of the balanced, reciprocal translocations is indicated. Bottom panel: two distinct outcomes of chromosomal translocations. In the case of transcriptional deregulation (left scheme), the normal regulatory sequences of the protooncogene are substituted with regulatory sequences derived from the partner chromosome, leading to deregulated expression of the protooncogene. In most cases of B-NHL, the heterologous regulatory regions derive from the Ig loci. In the case of fusion proteins (right scheme), the coding sequences of the two involved genes are joined in frame into a chimeric transcriptional unit that encodes for a fusion protein, characterized by novel biochemical and functional properties.
Aberrant Somatic Hypermutation
The term aberrant somatic hypermutation (ASHM) defines a recently identified mechanism of genetic lesion that is uniquely associated with B-NHL, particularly DLBCL, leading to the mutation of multiple non-Ig genes.47 ASHM has been proposed to derive from a malfunction in the physiologic SHM process, although the mechanism involved in this malfunction has not been identified.
In GC B cells, SHM is tightly regulated both spatially and temporally to introduce mutations only in the rearranged IgV genes8 as well as in the 5’ region of a few other genes, including BCL6 and the CD79 components of the B-cell receptor,48–52 although the functional role of the mutations found in these other genes remains obscure. On the contrary, multiple mutational events were found to affect numerous loci in over 50% of DLBCL cases, as well as in a few other lymphoma types, including, among others, AIDS-associated B-NHL, primary central nervous system lymphomas, and posttransplant lymphoproliferative disorders.53–57 The identified target loci comprise more than 10% of the genes transcribed in B cells and include several well-known protooncogenes such as PIM1 and MYC, one of the most frequently altered human oncogenes.47 These mutations are typically distributed within ∼2 Kb from the transcription initiation site (i.e., the hypermutable domain in the Ig locus)58 and, depending on the genomic configuration of the target gene, may affect nontranslated as well as coding regions, thus altering the response to factors that normally regulate their expression, or changing key structural and functional properties.47 This is the case of MYC, where a significant number of events lead to amino acid changes with proven functional consequences in activating its oncogenic potential. However, a comprehensive characterization of the potentially extensive genetic damage caused by ASHM is still lacking.
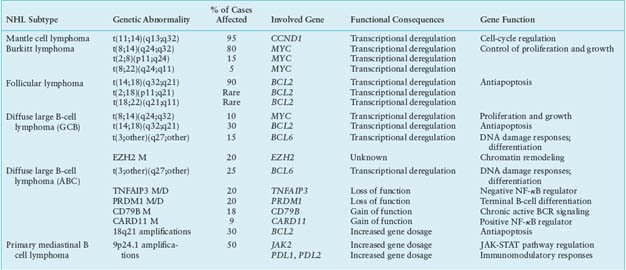
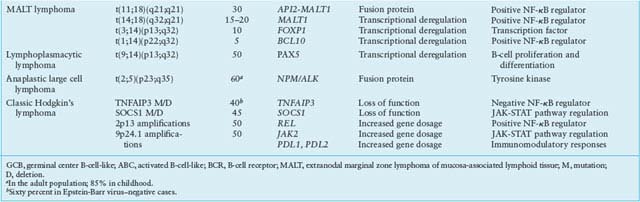
TABLE 30.1
MOST COMMON GENETIC LESIONS ASSOCIATED WITH NON-HODGKIN’S LYMPHOMA (NHL)
Other Mechanisms of Protooncogene Alteration
In addition to chromosomal translocations and ASHM, the structure of protooncogenes and/or their pattern of expression can be altered by gene copy number amplifications and somatic point mutations. Compared with epithelial cancer, only a few genes have been identified so far as specific targets of chromosomal amplification in B-NHL, as exemplified by REL and BCL2 in DLBCL,59–62 and PD-1 ligands in primary mediastinal B-cell lymphoma (PMBCL).63 However, the recent introduction of advanced cytogenetic and high-resolution, genome-wide single nucleotide polymorphism array technologies is likely to reveal a more complex scenario, leading to the identification of additional chromosomal sites of amplification. Somatic point mutations may alter the coding sequence of the target protooncogene and thus the biological properties of its protein product, as observed in MYC and BCL2.25,64–66 More recently, a number of genes involved in the activation of the NF-kB transcription complex have also been found to harbor oncogenic point mutations in DLBCL, leading to constitutive activation of NF-kB.67,68 Mutations of the RAS genes, a very frequent protooncogene alteration in human neoplasia, are virtually absent in lymphomas.69
Inactivation of Tumor Suppressor Genes
Until recently, the TP53 gene, possibly the most common target of genetic alteration in human cancer,70 remained one of few bona fide tumor suppressor genes involved in the pathogenesis of NHL, although at generally low frequencies and restricted to specific disease subtypes, such as BL and DLBCL derived from the transformation of FL or CLL.71–73 The mechanism of TP53 inactivation in NHL is similar to that detected in human neoplasia in general, entailing point mutation of one allele and chromosomal deletion or mutation of the second allele.70 In recent years, additional genes have been identified as targets of biallelic inactivation in B-NHL through specific chromosomal deletions and/or mutations. Two such genes lie on the long arm of chromosome 6 (6q), a region long known to be deleted in a large percentage of aggressive lymphomas, and associated with poor prognosis74,75: the PRDM1/BLIMP1 gene on 6q21, which is biallelically inactivated in ∼25% of activated B-cell-like (ABC)-DLBCL cases,76–78 and the gene encoding for the negative NF-kB regulator INFAIP3 on chromosome 6q23, which is commonly lost in ABC-DLBCL, PMBCL, and subtypes of marginal-zone lymphoma and HL.67,79–81
Deletions of chromosome 13q14.3 represent the most frequent lesions in CLL (>50% of cases)82,83 and encompass three noncoding elements, namely the DLEU2/mir-15a/16-1 cluster,84–87 whose deletion in mice promotes the development of CLL,88 documenting its pathogenetic role. Tumor suppressor inactivation via epigenetic transcriptional silencing was described for CDKN2A (p16/INK4a) as an infrequent event in various B-NHL.89,90 More recently, monoallelic inactivating mutations and deletions were found to affect the acetyltransferase genes CREBBP and EP300 in a significant proportion of DLBCL and FL, suggesting a role as haploinsufficient tumor suppressors.91 Major efforts are currently ongoing to identify the total complement of genetic lesions that are associated with the development of various lymphoma types by taking advantage of recently developed genome-wide technologies.
Infectious Agents
Viral and bacterial infections have both been implicated in the pathogenesis of lymphoma. At least three viruses are associated with specific NHL subtypes: the Epstein-Barr virus (EBV), the human herpesvirus-8 (HHV-8/KSHV), and the human T-cell leukemia virus type 1 (HTLV-1). Other infectious agents, such as human immunodeficiency virus (HIV), hepatitis C virus (HCV), Helicobacter pylori, and Chlamydophila psittaci, have an indirect role in NHL pathogenesis by either impairing the immune system and/or providing chronic antigenic stimulation.
EBV was initially identified in cases of endemic African BL92,93 and subsequently detected also in a fraction of sporadic BL, HIV-related lymphomas and primary effusion lymphomas (PELs).71,94–100 On infection of a B lymphocyte, the EBV genome is transported into the nucleus, where it exists predominantly as an extrachromosomal circular molecule (episome).101 The formation of circular episomes is mediated by the cohesive terminal repeats, which are represented by a variable number of tandem repeats sequence.101,102 Because of this termini heterogeneity, the number of tandem repeats sequences enclosed in newly formed episomes may differ considerably, thus representing a clonal marker of a single infected cell.102 Evidence for a pathogenetic role of the virus in NHL infected by EBV is at least twofold. First, it is well recognized that EBV is able to significantly alter the growth of B cells.101 Second, EBV-infected lymphomas usually display a single form of fused EBV termini, suggesting that the lymphoma cell population represents the clonally expanded progeny of a single infected cell.71,94 Nonetheless, the role of EBV in lymphomagenesis is still unclear as the virus infects virtually all humans during their lifetime and its transforming genes are commonly not expressed in the tumor cells of BL.
HHV-8 is a gammaherpesvirus initially identified in tissues of HIV-related Kaposi sarcoma103 and subsequently found to infect PEL cells as well as a substantial fraction of multicentric Castleman disease.104–107 Phylogenetic analysis has shown that the closest relative of HHV-8 is herpesvirus saimiri, a gamma-2 herpesvirus of primates associated with T-cell lymphoproliferative disorders.108 Like other gammaherpesviruses, HHV-8 is also lymphotropic and can be found in lymphocytes both in vitro and in vivo.103,106,107 Lymphoma cells naturally infected by HHV-8 harbor the viral genome in its episomal configuration and display a marked restriction of viral gene expression, suggesting a pattern of latent infection.108
HTLV-1 is a member of the lentivirus group that can immortalize normal T cells in vitro and can cause adult T-cell leukemia/lymphoma (ATLL).109–112 Unlike acutely transforming retroviruses, the HTLV-1 genome does not encode a viral oncogene. Moreover, this retrovirus does not transform T cells by cis-activation of an adjacent cellular protooncogene because the provirus appears to integrate randomly within the host genome.110–112 Rather, the pathogenetic effect of HTLV-1 seems to be due to viral production of a transregulatory protein (HTLV-1 tax) that activates the transcription of several host genes.113–119
An association between B-NHL and infection by HCV, a single-stranded RNA virus of the Flaviviridae family, has been proposed because of the increased risk of developing lymphoproliferative disorders among HCV-positive patients.120 Although the underlying mechanisms remain unclear, current models suggest that chronic B-cell stimulation by antigens associated with HCV infection may induce nonmalignant B-cell expansion, which subsequently evolves into B-NHL by accumulating additional genetic lesions.
A causal link between antigen stimulation by H. pylori and MALT lymphoma originating in the stomach is documented by the observation that H. pylori can be found in the vast majority of the lymphoma specimens,121–123 and long-term complete regression of the disease is achieved in 70% of cases on eradication of infection with antibiotics.124 However, cases with t(11;18)(q21;21) respond poorly to antibiotic eradication.125
C. psittaci, an obligate intracellular bacterium, was recently linked to the development of ocular adnexal marginal zone B-cell lymphoma, although variations in prevalence among different geographic areas remain a major investigational issue.4,126,127 In this indolent lymphoma, C. psittaci causes both local and systemic persistent infection, presumably contributing to lymphomagenesis via its mitogenic activity as well as through its ability to promote polyclonal cell proliferation and to induce resistance to apoptosis in the infected cells in vivo. Notably, bacterial eradication with antibiotic therapy is often followed by lymphoma regression.128
MOLECULAR PATHOGENESIS OF B-CELL NON-HODGKIN’S LYMPHOMA
The following section will focus on well-characterized genetic lesions that are associated with the most common types of B-NHL, classified according to the World Health Organization classification of lymphoid neoplasia.2 The molecular pathogenesis of HIV-related NHL will also be addressed, while the pathogenesis of other B-cell NHL types remains far less understood.
Mantle Cell Lymphoma
Cell of Origin
MCL is an aggressive disease representing ∼5% of all NHL diagnoses and generally regarded as incurable.2,89 Based on immunophenotype, gene expression profile, and molecular features, such as the presence of unmutated IgV genes in most cases, MCL is thought to derive from naïve, pre-GC peripheral B cells located in the inner mantle zone of secondary follicles (Fig. 30.3).
Genetic Lesions
MCL is typically associated with the t(11;14)(q13;q32) translocation that juxtaposes the IGH gene on chromosome 14q32 to a region containing the CCND1 gene (also known as BCL1) on chromosome 11q13 (Table 30.1).129–131 The translocation consistently leads to homotopic deregulation and overexpression of cyclin D1, a member of the D-type G1 cyclins that regulates the early phases of the cell cycle and is normally not expressed in resting B cells.132–134 By deregulating cyclin D1, t(11;14) is thought to contribute to malignant transformation by perturbing the G1-S phase transition of the cell cycle.89 In addition to t(11;14), up to 10% of MCLs overexpress aberrant or shorter cyclin D1 transcripts, as a consequence of secondary rearrangements, microdeletions, or point mutations in the gene 3’ untranslated region.135–137 These alterations may lead to cyclin D1 overexpression through the removal of destabilizing sequences and the consequent increase in mRNA half-life. Typically, this subset of cases is characterized by high proliferative activity and a more aggressive clinical course.138 The pathogenetic role of cyclin D1 deregulation in human neoplasia is suggested by the ability of the overexpressed protein to transform cells in vitro and to promote B-cell lymphomagenesis in transgenic mice,139–141 although a specific animal model that faithfully recapitulates the features of the human MCL is still lacking. Importantly, the frequency and specificity of this genetic lesion, together with the expression of CCND1 in the tumor cells, provide an excellent marker for MCL diagnosis.2
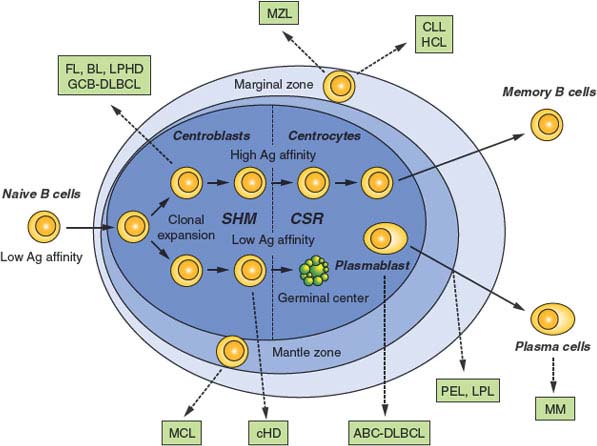
FIGURE 30.3 Normal B-cell development and lymphomagenesis. Schematic representation of a lymphoid follicle, constituted by the germinal center (GC) and the mantle zone, along with the surrounding marginal zone. B cells that have successfully rearranged their Ig genes in the bone marrow move to peripheral lymphoid organs as naïve B cells. On encounter with a T-cell–dependent antigen, B cells become proliferating centroblasts in the GC and eventually mature into centrocytes. These events are associated with the activation of somatic hypermutation and class-switch recombination. Only GC B cells with high affinity for the antigen will be positively selected to exit the GC and further differentiate into plasma cells or memory B cells, while low-affinity clones are eliminated by apoptosis. Dotted arrows indicate the putative normal counterpart of various lymphoma subtypes, as identified based on the presence of somatically mutated IgV genes, as well as on distinctive phenotypic features. MCL, mantle cell lymphoma; FL, follicular lymphoma; BL, Burkitt lymphoma; DLBCL, diffuse large B-cell lymphoma (GCB, germinal center B-cell-like; ABC, activated B-cell-like); CLL, chronic lymphocytic leukemia/ HCL, hairy cell leukemia; MM, multiple myeloma; LPHD, lymphocyte predominance Hodgkin disease; LPL, lympho plasma cytic lymphoma; MZL, marginal zone lymphoma; PEL, primary effusion lymphoma.
Other genetic alterations involved in MCL include frequent biallelic inactivation of the ATM gene by genomic deletions and mutations,142 loss of the TP53 gene in 20% of patients, where it represents a marker of poor prognosis,143 and inactivation of the CDKN2A gene by genomic deletion, mutation, or hypermethylation in approximately half of the cases belonging to the MCL variant characterized by a blastoid cell morphology.144 In a small number of cases, BMI1 is amplified and/or overexpressed, possibly as an alternative mechanism to the loss of CDKN2A.138,145
Burkitt Lymphoma
Cell of Origin
BL is an aggressive lymphoma comprising three clinical variants, namely sporadic Burkitt lymphoma (sBL), endemic Burkitt lymphoma (eBL), and HIV-associated BL, often diagnosed as the initial manifestation of AIDS.2 The presence of highly mutated IgV sequences that carry the hallmark of SHM,146–149 together with the expression of a distinct gene expression signature,150,151 indicates the derivation from a GC B cell.
Genetic Lesions
All BL cases, including the leukemic variants, share a virtually obligatory genetic lesion, that is, chromosomal translocations involving the MYC gene on region 8q24 and one of the Ig loci on the partner chromosome.152,153 In ∼80% of cases, this is represented by the IGH locus, leading to t(8;14)(q24;q32), while in the remaining 20% of cases either IGk (2p12) or IGλ (22q11) are involved.152–155 Although fairly homogeneous at the microscopic level, these translocations display a high degree of molecular heterogeneity. The t(8;14) breakpoints are located 5’ and centromeric to MYC, whereas they map 3’ to MYC in t(2;8) and t(8;22).152–156 Further molecular heterogeneity derives from the exact breakpoint sites on chromosomes 8 and 14 in t(8;14). Translocations of eBL tend to involve sequences on chromosome 8 at an undefined distance 5’ to MYC (>1,000 Kb) and sequences on chromosome 14 within or in proximity to the Ig IH region.157,158 In sBL, t(8;14) preferentially involves sequences within or immediately 5’ to MYC (<3 Kb) on chromosome 8, and sequences within the Ig switch regions on chromosome 14.157,158
The common consequence of t(8;14), t(2;8), and t(8;22) is the ectopic and constitutive overexpression of the MYC protooncogene,159–161 which is normally not detected in most proliferating GC B cells.11 At least two distinct mechanisms are responsible for MYC deregulation, including juxtaposition of the MYC coding sequences to heterologous enhancers derived from Ig loci,159–161 and structural alterations of the gene 5’ regulatory sequences, which alter the responsiveness to cellular factors controlling its expression.162 In fact, the MYC exon 1/intron 1 junction, encompassing critical regulatory elements, is either decapitated by the translocation or mutated in the translocated alleles. Oncogenic activation of MYC can also be due to amino acid substitutions within the gene exon 2, encoding for the protein transactivation domain.64,65 These mutations can abolish the ability of p107, a nuclear protein related to RB1, to suppress MYC activity,163 or can increase protein stability.164,165
MYC is a nuclear phosphoprotein that functions as a sequence specific DNA-binding transcriptional regulator controlling proliferation, differentiation, and apoptosis, all of which are implicated in carcinogenesis.166,167 In addition, MYC controls DNA replication independent of its transcriptional activity, a property that may promote genomic instability by inducing replication stress.168 Consistent with its involvement in multiple cellular processes, the MYC target gene network is estimated to include ∼15% of all protein-coding genes as well as noncoding RNAs.167,169 In vivo, MYC is found mainly in heterodimeric complexes with the related protein MAX, and such interaction is required for MYC-induced stimulation of transcription and cell proliferation.170–176 In B-NHL carrying MYC translocations, constitutive expression of MYC induces transcription of a subset of target genes that have diverse roles in regulating cell growth by affecting DNA replication, energy metabolism, protein synthesis, and telomere elongation.167,176,177 In addition, deregulated MYC expression is thought to cause genomic instability, thus contributing to tumor progression by facilitating the occurrence of additional genetic lesions.178 Dysregulation of MYC expression in a number of transgenic mouse models leads to the development of aggressive B-cell lymphomas with high penetrance and short latency.165,179,180 These mouse models confirm the pathogenetic role of deregulated MYC in B cells, although the resulting tumors tend to be more immature than the human BL, most likely because of the early activation of the promoter sequences used for expression of the MYC transgene.
Cooperating oncogenic events in BL include loss of TP53 by mutation and/or deletion (30% of both sBL and eBL cases),72 inactivation of CDKN2B by hypermethylation,181 and deletions of 6q, detected in ∼30% of cases, independent of the clinical variant.74 Additionally, one contributing factor to the development of BL is monoclonal EBV infection, present in virtually all cases of eBL and in ∼30% of sBL.92,94,182,183 The consistent expression of EBER, a class of small RNA molecules, has been proposed to mediate the transforming potential of EBV in BL.184 However, because EBV infection in BL displays a peculiar latent infection phenotype characterized by negativity of both EBV-transforming antigens LMP1 and EBNA2, the precise pathogenetic role of the virus has remained elusive.185
Follicular Lymphoma
FL represents the second most common type of B-NHL, accounting for ∼20% of all diagnoses, and the most common low-grade B-NHL.2 Over time, FL tends to transform into an aggressive lymphoma with a diffuse large cell architecture (Fig. 30.3).2
Cell of Origin
FL arises from a GC-derived B cell, as documented by the presence of somatically mutated Ig genes that show evidence of ongoing SHM activity, and by the expression of specific GC B-cell markers such as BCL6 and CD10.1
Genetic Lesions
The genetic hallmark of FL is represented by chromosomal translocations affecting the BCL2 gene on chromosome band 18q21, which are detected in 80% to 90% of cases independent of cytologic subtype, although less frequent in grade 3 FL130,186–189 (Table 30.1). In t(14;18), the rearrangement joins the 3’ untranslated region of BCL2 to an Ig JH segment, resulting in the ectopic expression of BCL2 in GC B cells,186,187,190–194 where its transcription is normally repressed by BCL6.16,25 Approximately 70% of the breakpoints on chromosome 18 cluster within the major breakpoint region, while the remaining 5% to 25% map to the more distant minor cluster region, located ∼20 kb downstream of the BCL2 gene.186,187,190,191 Rearrangements involving the 5’ flanking region of BCL2 have been described in a minority of cases.195 The BCL2 gene encodes a 26-kD integral membrane protein that controls the cell apoptotic threshold by preventing programmed cell death196–199; BCL2 may thus contribute to lymphomagenesis by inducing resistance of tumor cells to apoptosis independent of antigen selection. Nevertheless, additional genetic aberrations are likely required for malignant transformation, a major role being played by chronic antigen stimulation.200–202
More recently, somatic mutations of the polycomb-group oncogene EZH2, which encodes a histone methyltransferase responsible for the trimethylation of Lys27 of histone H3 (H3K27), were found in 7% of FL patients.203 These mutations result in the replacement of a single tyrosine (Tyr641) in the SET domain of the EZH2 protein, and were associated with increased levels of H3K27me3 through a mechanism that involves altered substrate catalytic specifity.203 However, the precise mechanism by which this amino acid change contributes to tumorigenesis remains to be clarified.
Chromosomal translocations of the BCL6 gene are detected in 6% to 14% of all FL cases, and were shown to have a significantly higher prevalence in the group of patients known to eventually transform into aggressive DLBCL.204–207 Other genetic lesions are also predominantly observed in FL cases that have undergone histologic progression to a high-grade NHL, and include deletions of chromosome 6 (20% of the cases),74 TP53 mutations (25% to 30%),73,208–210 inactivation of CDKN2A through deletion, mutation, and hypermethylation (one-third of patients),144,211 rearrangements of MYC in rare cases,212 and a variety of copy number aberrations.213 Overall, the molecular events that lead to the clinical progression of FL remain poorly characterized.
Diffuse Large B-Cell Lymphoma
DLBCL is the most common type of B-NHL, accounting for ∼40% of all new diagnoses in adulthood, and includes cases arising de novo, as well as cases that derive from the clinical evolution of various, less aggressive B-NHL types (i.e., FL and CLL).2,214
Cell of Origin
Over the past decade, the advent of genome-wide gene expression profile technologies has allowed the identification of multiple phenotypic DLBCL subgroups that reflect the derivation from B cells at various differentiation stages. These include at least three well-characterized subtypes: a GC B-cell-like (GCB) DLBCL, which appears to derive from proliferating GC centroblasts; an ABC DLBCL, which shows a transcriptional signature related to plasmablastic B cells presumably blocked during post-GC differentiation; and PMBCL, which is postulated to arise from thymic B cells; the remaining 15% to 30% of cases remain unclassified.215–218 Stratification according to gene expression profiles has prognostic value, as patients diagnosed with a GCB-DLBCL display a better overall survival compared with ABC-DLBCL,62 but does not direct differential therapy, and is imperfectly replicated by immunophenotyping or morphology219,220; thus, it is not officially incorporated into the World Health Organization classification. A separate classification scheme identified three subsets defined by the expression of genes involved in oxidative phosphorylation, B-cell receptor/proliferation, and tumor microenvironment/host inflammatory response.221
Genetic Lesions
The heterogeneity of DLBCL is reflected in the catalogue of genetic lesions that are associated with its pathogenesis. These include balanced reciprocal translocations deregulating the expression of protooncogenes, gene amplifications, chromosomal deletions, single-point mutations, and aberrant somatic hypermutation.222,223 Notably, most of these abnormalities are preferentially or exclusively associated with individual DLBCL phenotypic subtypes, indicating that GCB- and ABC-DLBCL use distinct oncogenic pathways.224
GCB-DLBCL. Genetic lesions that are specific to GCB-DLBCL include the t(14;18) and t(8;14) translocations, which deregulate the BCL2 and MYC oncogenes in 34% and 10% of cases, respectively25,62,225–227; mutations affecting an autoregulatory domain within the BCL6 5’untranslated exon 1228–230; mutations of the EZH2 gene203; and deletions of the tumor suppressor PTEN.224 In addition, recent studies have identified frequent monoallelic mutations and deletions inactivating the acetyltransferase genes CREBBP and EP300 predominantly in this subtype of DLBCL, where they affect nearly 40% of cases.91
Somatic mutations of the BCL6 5’ regulatory sequences are detected in up to 75% of DLBCL cases,48,231,232 and reflect the activity of the physiologic SHM mechanism that operates in normal GC B cells.48,49,52 However, functional analysis of numerous mutated BCL6 alleles revealed that a subset of mutations are specifically associated with DLBCL while being absent in normal GC cells or in other B-cell malignancies.229 These mutations deregulate BCL6 transcription by disrupting an autoregulatory circuit through which the BCL6 protein controls its own expression levels via binding to the promoter region of the gene229,230 or by preventing CD40-induced BCL6 down-regulation in post-GC B cells.233 Because the full extent of mutations deregulating BCL6 expression has not been characterized, the fraction of DLBCL cases carrying abnormalities in BCL6 cannot be determined.
Approximately 50% of all DLBCL are also associated with ASHM.47 The number and identity of the genes that accumulate mutations in their coding and noncoding regions due to this mechanism varies in different cases and is still largely undefined. However, preferential targeting of individual genes has been observed in the two main COO-defined DLBCL subtypes, with mutations of MYC and BCL2 being found at significantly higher frequencies in GCB-DLBCL, and mutations of PIM1 almost exclusively observed in ABC-DLBCL. ASHM may therefore contribute to the heterogeneity of DLBCL via the alteration of different cellular pathways in different cases. Mutations and deletions of the TP53 tumor suppressor gene are mostly detectable in cases originating from the transformation of FL, and are therefore more often associated with chromosomal translocations involving BCL2 and with a GCB-DLBCL phenotype.73
ABC-DLBCL. Several genetic abnormalities are observed almost exclusively in ABC-DLBCL, including amplifications of the BCL2 locus on 18q24234,235; mutations within the NF-kB (CARD11, TNFAIP3/A20)67,68 and B-cell receptor signaling (CD79B)236 pathways; inactivating mutations and deletions of BLIMP176–78; chromosomal translocations deregulating the BCL6 oncogene; deletion or lack of expression of the p16 tumor suppressor gene and, rarely, mutations of the ATM gene.237,238
Chromosomal translocations affecting band 3q27 cause rearrangements of the BCL6 gene in up to 35% of all DLBCL cases,75,205,239 with a twofold higher frequency in the ABC-DLBCL subtype228 (Table 30.1). These rearrangements juxtapose the intact coding domain of BCL6 downstream and in the same transcriptional orientation to heterologous sequences derived from the partner chromosome, including IGH (14q23), IGk (2p12), IGλ (22q11), and at least 20 other chromosomal sites unrelated to the Ig loci.39–46 The majority of these translocations result in a fusion transcript in which the promoter region and the first noncoding exon of Bcl6 are replaced by sequences derived from the partner gene.40,240 Because the common denominator of these promoters is a broader spectrum of activity throughout B-cell development, including expression in the post-GC differentiation stage, the translocation prevents the down-regulation of BCL6 expression that is normally associated with differentiation into post-GC cells. Deregulated expression of a normal BCL6 gene product may play a critical role by enforcing the proliferative phenotype typical of GC cells while blocking terminal differentiation, as confirmed by a mouse model in which deregulated BCL6 expression causes DLBCL.241
In up to 25% of ABC-DLBCL, the PRDM1 gene is inactivated by a variety of genetic lesions, including truncating or missense mutations and/or genomic deletions, as well as by transcriptional repression through constitutively active, translocated BCL6 alleles.76–78 The PRDM1 gene encodes for a zinc finger transcriptional repressor that is expressed in a subset of GC B cells undergoing plasma cell differentiation and in all plasma cells,242,243 and is an essential requirement for terminal B-cell differentiation.244 Thus, BLIMP1 inactivation may contribute to lymphomagenesis by blocking post-GC B-cell differentiation. Notably, translocations deregulating the BCL6 gene are virtually never found in BLIMP1 mutated DLBCLs, suggesting that BCL6 deregulation and BLIMP1 inactivation represent alternative oncogenic mechanisms converging on the same pathway (Fig. 30.4).
A predominant feature of ABC-DLBCL is the constitutive activation of the NF-kB signaling pathway, initially evidenced by the selective expression of a signature enriched in NF-kB target genes, and by the requirement of NF-kB for proliferation and survival in ABC-DLBCL cell lines. A number of recent studies have led to the identification of multiple oncogenic alterations affecting positive and negative regulators of NF-kB, specifically in this disease subtype, providing genetic evidence for this phenotypic characteristic. In up to 30% of cases, the TNFAIP3 gene encoding for the negative regulator A20 is biallelically inactivated by mutations and/or deletions, thus preventing termination of NF-kB responses.67,79 The tumor suppressor role of A20 was documented by the observation that reconstitution of A20 knockout cell lines with wild type protein induces apoptosis and blocks proliferation, in part due to suppression of NF-kB activity.67,79 In an additional ∼10% of ABC-DLBCL, the CARD11 gene is targeted by oncogenic mutations clustering in the coiled-coil domain and enhancing its ability to transactivate NF-kB target genes.67,68 Less commonly, mutations were found in a variety of other genes encoding for NF-kB components, overall accounting for over half of all ABC-DLBCL67 and suggesting that yet unidentified lesions may be responsible for the NF-kB activity in the remaining fraction of cases. In addition to constitutive NF-kB activity, ABC-DLBCLs display evidence of chronic active BCR signaling, which is associated with somatic mutations affecting the immunoreceptor tyrosine-based activation motif signaling modules of CD79B and CD79A in 10% of ABC-DLBCL biopsy samples but rarely in other DLBCLs.236
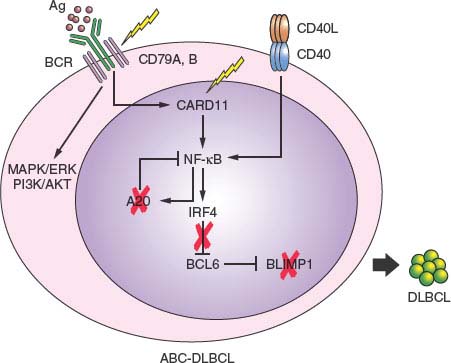
FIGURE 30.4 Pathway lesions in activated B-cell-like diffuse large B-cell lymphoma (ABC-DLBCL). Schematic representation of a germinal center centrocyte, expressing a functional surface B-cell receptor (BCR) and CD40 receptor. In normal B cells, engagement of the BCR by the antigen (spheres) or interaction of the CD40 receptor with the CD40L presented by T cells induce activation of the NF-kB pathway, including its targets IRF4 and A20. IRF4, in turn, down-regulates BCL6 expression, allowing the release of BLIMP1 expression, a master plasma cell regulator required for plasma cell differentiation. In ABC-DLBCL, multiple genetic lesions converge on this pathway and disrupt it at multiple levels in different cases (percentages as indicated), presumably contributing to lymphomagenesis by favoring the antiapoptotic and proproliferative function of NF-kB while blocking terminal B-cell differentiation through mutually exclusive deregulation of BCL6 and inactivation of BLIMP1.
PMBCL. PMBCL is a tumor observed most commonly in young female adults, which involves the mediastinum and displays a distinct gene expression profile, largely similar to a particular type of HL.217,218 A genetic hallmark of both PMBCL and HL is the amplification of chromosomal region 9q24, detected in nearly 50% of patients.224,245 This relatively large interval encompasses multiple genes of possible pathogenetic significance, including the gene encoding for the JAK2 tyrosine kinase or the PDL1 and PDL2 genes, which encode for inhibitors of T-cell responses.63,224,245 Besides contributing to lymphomagenesis, elevated expression levels of these genes may partly explain the unique features of these lymphoma types, which are characterized by a significant inflammatory infiltrate. PMBCL also shares with HL the presence of genetic lesions affecting the NF-kB pathway and the deregulated expression of receptor tyrosine kinases.81,246–248
Extranodal Marginal Zone Lymphoma of Mucosa-Associated Lymphoid Tissue
Cell of Origin
MALT lymphoma represents the third most common form of B-NHL,2 and has steadily risen in incidence over the past 2 decades.249 The presence of rearranged and somatically mutated IgV genes,30,250 together with the architectural relationship with mucosa-associated lymphoid tissue,2 indicate the post-GC origin of these tumors, possibly from a marginal zone, memory B cell (Fig. 30.3). A critical role for antigen stimulation, particularly in the pathogenesis of gastric MALT lymphoma, is supported by the observation that (1) this disease is associated with chronic infection of the gastric mucosa by H. pylori in virtually all cases,121–123 (2) eradication of H. pylori by antibiotic treatment can lead to tumor regression in ∼70% of cases,124,251 and (3) MALT lymphoma cells express autoreactive BCR, in particular to rheumatoid factors.252,253
Whether the development of MALT lymphoma arising in body sites other than the stomach is also dependent on antigen stimulation remains an open question. In this respect, it is remarkable that salivary gland and thyroid MALT lymphoma are generally a sequela of autoimmune processes, namely Sjögren syndrome and Hashimoto thyroiditis, respectively.
Genetic Lesions
Of several structural aberrations that are selectively and recurrently associated with MALT lymphoma, most target the NF-kB signaling pathway, suggesting a critical role in the pathogenesis of the disease. The most common one is the t(11;18)(21;21) translocation, which involves the API2 (BIRC3) gene on 11q21 and the MALT1 gene on 18q21,254,255 and is observed in 25% to 40% of gastric and pulmonary MALT lymphomas.256–258 API2, a member of the family of Inhibitor of Apoptosis Proteins, plays an evolutionary conserved role in regulating programmed cell death in diverse species. MALT1, together with BCL10 and CARD11, is a component of the CBM ternary complex and plays a central role in BCR and NF-kB signaling activation.90 Notably, the wild type proteins encoded by these two genes are incapable of activating NF-kB, as opposed to the API/MALT1 fusion protein, suggesting that the translocation may confer a survival advantage to the tumor by leading to inhibition of apoptosis and constitutive NF-kB activation without the need for upstream signaling.254,255,259 In an additional 15% to 20% of cases, the MALT1 gene is translocated to the IGH locus in t(14;18)(q32;q21).260,261
Recurrent abnormalities of chromosomal band 1p22, generally represented by t(1;14)(p22;q32), occur in ∼5% of MALT lymphomas and cause deregulated expression of BCL10, a cellular homologue of the equine herpesvirus-2 E10 gene, which encodes an amino-terminal caspase recruitment domain (CARD) homologous to that found in several apoptotic molecules.262,263 BCL10, however, does not have proapoptotic activity in vivo, where it functions as a positive regulator of antigen-induced activation of NF-kB.90,264,265 Thus, the translocation may provide both antiapoptotic and proliferative signals mediated via NF-kB transcriptional targets. Trisomy 3 represents a recurrent numerical abnormality in MALT lymphomas; however, the genes involved remain presently unknown.266,267
A more recently identified translocation associated with, although not restricted to, MALT lymphoma is t(3;14)(p13;q32),268,269 which leads to deregulated expression of FOXP1, a member of the Forkhead box family of winged-helix transcription factors involved in regulation of Rag1 and Rag2, and essential for B-cell development.270 Finally, homozygous or hemizygous loss of TNFAIP3 due to mutations and/or deletions has been reported in 20% of MALT lymphoma patients, where it is mutually exclusive with other genetic lesions, leading to NF-kB activation.80 Genetic lesions also involved in other lymphoma types include BCL6 alterations and TP53 mutations.271–273
Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma
Cell of Origin
CLL is a malignancy of mature, resting B lymphocytes, which originates from the oncogenic transformation of a common cellular precursor resembling an antigen-experienced B cell.274,275 This notion was conclusively demonstrated when gene expression profile studies revealed that, although CLL can express somatically mutated or unmutated IgV genes at approximately equal percentages,276,277 all cases share a homogeneous signature more related to that of CD27+ memory and marginal zone B cells.278,279 Moreover, analysis of the Ig gene repertoire in these patients indicates very similar, at times almost identical, antigen receptors among different individuals.280–285 This finding, known as stereotypy, strongly supports a critical role for the antigen in CLL pathogenesis. The histogenetic heterogeneity of CLL carry prognostic relevance, as cases with mutated Ig genes associate with a significantly longer survival.286,287 Intriguingly, 6% of the normal elderly population develops a monoclonal B-cell lymphocytosis (MBL) that seems to be the precursor to CLL in 1% to 2% of cases.288
Genetic Lesions
Different from most mature B-NHLs, and consistent with the derivation from a post-GC or GC-independent B cells, CLL cases are largely devoid of balanced, reciprocal chromosomal translocations.82,83 On the contrary, CLL is recurrently associated with several numerical abnormalities, including trisomy 12 and monoallelic or biallelic deletion/inactivation of chromosomal regions 17p, 11q, and 13q14 (Table 30.1).82,83 Of these, deletion of 13q14 represents the most frequent chromosomal aberration, being observed in up to 76% of cases as a monoallelic event, and in 24% of cases as a biallelic event. Interestingly, this same deletion is also found in those with MBL.288 In all affected cases, the minimal deleted region encompasses a long noncoding RNA (DLEU2) and two microRNAs expressed as a cluster, namely miR-15a and miR-16-1.84,85,87 The causal involvement of 13q14-minimal deleted region–encoded tumor suppressor genes in CLL pathogenesis was recently demonstrated in vivo in two animal models that developed clonal lymphoproliferative diseases including MBL, CLL, and DLBCL, at 25% to 40% penetrance.88 Trisomy 12 is found in approximately 16% of patients evaluated by interphase fluorescent in situ hybridization and correlates with a poor survival, but no specific genes have been identified.289–291 Deletions of chromosomal region 11q22-23 (18% of cases) almost invariably encompass the ATM gene and may thus promote genomic instability.292–294 Because these mutations may occur in the patient germ line, it is thought that they may account, at least in part, for the familial form of the disease. A similar pathogenetic mechanism may be involved in cases with 17p13 genomic deletions (∼7%), which include the TP53 tumor suppressor and are frequently accompanied by mutation of the second allele.72,295 A higher frequency of TP53 alterations is observed after transformation of CLL/SLL to Richter syndrome, a highly aggressive lymphoma with a poor clinical outcome.72
HIV-Related NHL
The association between an immunodeficiency state and the development of lymphoma has been recognized in several clinical conditions, including congenital (e.g., Wiskott-Aldrich syndrome), iatrogenic (e.g., treatment with immunosuppressor agents), and viral-induced (e.g., AIDS) immunodeficiencies. HIV infection has emerged as a major risk factor for lymphomagenesis, prompting detailed investigations of the molecular pathophysiology of HIV-related NHL, which are primarily classified into three clinicopathologic categories: HIV-related BL, DLBCL, and PEL.2,296–298 Based on the site of origin, HIV-related NHLs are generally grouped into systemic HIV-related NHL (i.e., DLBCL and BL) and HIV-related PCNSL, which is characterized by a uniform morphology consistent with a diffuse architecture of large cells.2,296–298
Cell of Origin
HIV-related NHLs invariably derive from B cells that have experienced the GC reaction, as indicated by the presence of somatically mutated Ig and BCL6 genes, as well as by several phenotypic and genome-wide transcriptional features.296–298 Based on the presence or absence of immunoblastic features, and on the expression pattern of the BCL6 protein, the EBV-encoded LMP1, and the CD138/syndecan-1 antigen, a proteoglycan associated with the terminal phases of B-cell differentiation, both HIV-related DLBCL and PCNSL can be segregated into two distinct histogenetic categories: cases displaying the BCL6+/CD138−/LMP1− phenotype closely resemble the phenotype of GC B cells; conversely, BCL6−/CD138+/LMP1+ cases are morphologically consistent with immunoblastic lymphoma, plasmacytoid, and reflect a post-GC stage of B-cell differentiation.296–298 PEL consistently derives from B cells, reflecting a preterminal stage of B-cell differentiation.105,106,299,300
Genetic Lesions
The different categories of HIV-related NHL associate with distinctive molecular pathways. Cases of HIV-related BL consistently display activation of MYC by chromosomal translocations that are structurally similar to those found in sporadic BL, while rearrangements of BCL6 are always absent.296–298,301 HIV-related BLs also frequently harbor mutations of TP53 (60%), mutations of the BCL6 5’ noncoding regions (60%), and, in 30% of cases, infection of the tumor clone by EBV, although the EBV-encoded antigens LMP1 and EBNA2 are not expressed.302,303 Stimulation and selection by antigens, frequently represented by autoantigens, appear to be a prominent feature of HIV-related BL.148,304
Different from BL, the most frequent genetic alteration detected in HIV-related DLBCL is infection by EBV, which occurs in approximately 60% to 70% of the cases and associates frequently, although not always, with expression of LMP1.71,98,303 Also different from HIV-BL, HIV-related DLBCL displays rearrangements of BCL6 in 20% of cases.301 Mutations of BCL6 5’ noncoding regions occur in 70% of cases.305
All HIV-related PCNSLs harbor EBV infection.306 However, only the subset of cases with immunoblastic plasmacytoid morphology expresses the LMP1-transforming protein of EBV.100 HIV-related PCNSLs display evidence of ASHM54 and harbor oncogenic mutations in the CARD11 gene (16% of cases),307 which may explain in part the presence of constitutive NF-kB activity previously recognized in this lymphoma subtype. Although some reports have suggested that HHV-8 may be related to PCNSL pathogenesis in immunocompromised patients, extensive analysis of HIV-related PCNSL has unequivocally ruled out this hypothesis.308,309
The last type of HIV-related NHL that has been characterized at the molecular level is represented by PEL, also known as body cavity–based lymphoma.105,106,299 This lymphoma entity is associated with HHV-8 infection in 100% of cases and clinically presents as effusions in the serosal cavities of the body (pleura, pericardium, and peritoneum) in the absence of solid tumor masses.105,106,299 In addition to HHV-8, cases of PEL frequently carry coinfection of the tumor clone by EBV.97,99,105,106,299
MOLECULAR PATHOGENESIS OF T-CELL NHL
Peripheral T-cell lymphomas (PTCLs) encompass a highly heterogeneous and relatively uncommon group of diseases representing 5% to 10% of all cases of NHL worldwide, with significant geographic variation in both incidence and relative prevalence.2 PTCLs arise from mature postthymic T cells and, according to the clinical presentation of the disease, are listed as leukemic or disseminated, predominantly extranodal, cutaneous, and predominantly nodal.2 Although the study of T-cell neoplasms is hampered by the rarity of these diseases and the difficulty of collecting homogeneous sample series, significant advances have been made over the past decades in our understanding of their biology, classification, and prognosis.
Adult T-Cell Lymphoma/Leukemia (HTLV-1–Positive)
Cell of Origin
The term ATLL encompasses a spectrum of lymphoproliferative diseases associated with HTLV-1 infection and mainly restricted to southwestern Japan and the Caribbean basin.2,310 The United States and Europe are considered low-risk areas as less than 1% of the population are HTLV-1 carriers,311 and only 2% to 4% of seropositive individuals eventually develop ATLL.2,110,111 Clonal rearrangement of the TCR is evident in all cases, and clonal integration of the virus has been observed.312,313
Genetic Lesions
Compared with other mature T-cell tumors, the molecular pathogenesis of ATLL has been elucidated to a wider extent. Particularly, the role of HTLV-1 has been linked to the production of a transregulatory protein (HTLV-1 tax), which markedly increases expression of all viral gene products and transcriptionally activates the expression of certain host genes, including IL2, CD25 (the α chain of the IL-2 receptor), c-sis, c-fos, and GM-CSF.113–117,242 Indeed, a property of ATLL cells is the constitutive high level expression of IL-2 receptors. The central role of these genes in normal T-cell activation and growth, together with the results of in vitro studies, support the notion that tax-mediated activation of these host genes represents an important mechanism by which HTLV-1 initiates T-cell transformation.113 In addition, tax interferes at multiple sites with DNA damage repair functions and with mitotic checkpoints,118,119,314 consistent with the fact that ATLL cells harbor a high frequency of karyotypic abnormalities.
The long period of clinical latency that precedes the development of ATLL (usually 10–30 years), the small percentage of infected patients who develop this malignancy, and the observation that leukemic cells from ATLL are monoclonal suggest that HTLV-1 is not sufficient to cause the full malignant phenotype.110–112 A model for ATLL therefore implies an early period of tax-induced polyclonal T-cell proliferation which, in turn, facilitates the occurrence of additional genetic events, leading to the monoclonal outgrowth of a fully transformed cell. In this respect, a recurrent genetic lesion in ATLL is represented by mutations of the TP53 tumor suppressor gene, which is inactivated in 40% of cases.315,316
Peripheral T-Cell Lymphoma, Not Otherwise Specified
This category represents the largest and most heterogeneous group of PTCLs, and includes all cases that lack specific features allowing classification within another entity. The majority of these cases derive from αβ CD4+ T cells and show aberrant defective expression of one or several T-cell–associated antigens.317 Based on gene expression profile analysis, PTCL not otherwise specified (NOS) as a group appears to be most closely related to activated T cells than to resting T cells, and can be segregated according to similarities with the transcriptional signature of CD4+ and CD8+ T cells. However, no correlation can be observed between gene expression profile and immunophenotype, likely reflecting the variable detection of T-cell antigens in the disease.
Genetic Lesions
Clonal numerical and structural aberrations are found in most PTCL NOS by conventional cytogenetics and in all cases by more sensitive approaches such as array-based methods. For a few loci, correlation between gene copy number and expression has been confirmed, suggesting a pathogenetic role. Candidate genes targeted by copy number gains include CDK6 on chromosome 7q, MYC on chromosome 8, and the NF-kB regulator CARD11 at 7p22, while losses of 9p21 are associated with reduced expression of CDKN2A/B.318 Chromosomal translocations involving the TCR loci have been reported in rare cases and remain poorly understood because the identity of the translocation partner has not been identified, with few exceptions: the BCL3 gene, the poliovirus receptor-related 2 (PVRL2) gene, found in the t(14;19)(q11;q13) translocation, and the IRF4 gene, cloned in two cases.319–321
Angioimmunoblastic T-Cell Lymphoma
Cell of Origin
Angioimmunoblastic T-cell lymphoma (AITL) is an aggressive disease of the elderly and accounts for about one-third of all PTCL in Western countries.322 The tumor cells display a mature CD4+CD8–T-cell phenotype, with frequent aberrant loss of one or several T-cell markers, and coexpression of BCL6 and CD10 in at least a fraction of cells. Recently, gene expression profile studies allowed the conclusive establishment of the cellular derivation of AITL from follicular T-helper cells,323 as initially suspected based on expression of single markers.324
Genetic Lesions
Clonal aberrations have been reported in up to 90% of AITL patients and are mostly represented by chromosomal imbalances, while chromosomal translocations affecting the TCR loci are extremely rare.318 However, the scarce number of genetic studies performed so far has failed to provide any significant clues regarding the oncogenic pathways involved in AITL.
Cutaneous T-Cell Lymphoma
Genetic lesions are involved in a limited but significant fraction of primary CTCLs showing molecular markers of clonality.325 Rearrangements of the NFKB2 gene at 10q24 encode for a protein that lacks the ankyrin regulatory domain required for regulating the physiological NFKB2 nuclear/cytoplasmic distribution, but retains the rel effector domain and can bind kappa B sequences in vitro.326 The translocation may thus contribute to lymphoma development by causing constitutive activation of the NF-kB pathway.
Anaplastic Large Cell Lymphoma
Cell of Origin
ALCL is a distinct subset of T-NHL (∼12% of cases) whose normal cellular counterpart has not yet been established.2,310 The tumor is composed of large pleomorphic cells that exhibit a unique phenotype characterized by positivity for the CD30 antigen and loss of most T-cell markers.2,327 Based on the expression of a chimeric protein containing the cytoplasmic portion of anaplastic lymphoma kinase (ALK) as a consequence of translocations involving the ALK gene, ALCL may be subdivided into two groups, which display distinct transcriptional signatures328: the most common and curable ALK-positive ALCL and the more aggressive ALK-negative ALCL.2,329–331 However, the identification of a common 30-genes predictor that can discriminate ALCL from other T-NHL, independent of ALK status, suggests that these two subgroups are closely related and may derive from a common precursor.332
Genetic Lesions
The genetic hallmark of ALK+ ALCL is chromosomal translocations involving band 2p23 and a variety of chromosomal partners, with t(2;5)(p23;q35) accounting for 70% to 80% of the cases.2,333 Cloning of the translocation breakpoint in t(2;5) demonstrated the involvement of the ALK gene on 2p23 and the nucleophosmin (NPM1) gene on 5q35.334 As a consequence, the aminoterminus of NPM is linked in frame to the catalytic domain of ALK, driving transformation through multiple molecular mechanisms334: (1) the ALK gene, which is not expressed in normal T lymphocytes, becomes inappropriately expressed in lymphoma cells, conceivably because of its juxtaposition to the promoter sequences of NPM1, which are physiologically expressed in T cells; and (2) all translocations involving ALK produce proteins with constitutive tyrosine activity, due in most cases to spontaneous dimerization induced by the various fusion partners.333 Constitutive ALK activity, in turn, results in the activation of several downstream signaling cascades, with the JAK-STAT and PI3K-AKT pathways playing central roles.335–338 The transforming ability of the chimeric NPM/ALK protein has been proven both in vitro and in vivo in transgenic mouse model.339–341
In a minority of cases, fusions other than NPM-ALK cause the abnormal subcellular localization of the corresponding chimeric ALK proteins and the constitutive activation of ALK. Among these alternative rearrangements, the most frequent involve TPM3 or TPM4, TRK-fused genes,342 ATIC,343,344 CLTCL1, and MSN. No recurrent cytogenetic abnormality has been described in ALK-negative ALCL, and the molecular events responsible for this disease subtype remain largely unknown.
MOLECULAR PATHOGENESIS OF HL
HL is a B-lymphoid malignancy characterized by the presence of scattered large atypical cells—the mononucleated Hodgkin cells and the multinucleated Reed-Sternberg cells (HRS)—residing in a complex admixture of inflammatory cells.2,345 Based on the morphology and phenotype of the neoplastic cells, as well as on the composition of the infiltrate, HL is segregated into two major subgroups: nodular lymphocyte-predominant HL (NLPHL; ∼5% of cases) and classic HL (cHL), comprising the nodular sclerosis, mixed cellularity, and lymphocyte-depletion and lymphocyte-rich variants. Until recently, molecular studies of HL have been hampered by the paucity of the tumor cells in the biopsy (typically <1%, although occasional cases can present >10% HRS cells). However, the introduction of sophisticated laboratory techniques allowing the isolation and enrichment of neoplastic cells has markedly improved our understanding of HL histogenesis.
Cell of Origin
Despite the HRS of cHL cells having lost expression of nearly all B-cell–specific genes,346–348 both HL types represent clonal expansions of B cells, as revealed by the presence of clonally rearranged and somatically mutated Ig genes.349,350 In about 25% of cHL cases, nonsense mutations disrupt originally in-frame VH gene rearrangements (crippling mutations), thereby preventing antigen selection and suggesting that HRS cells of cHL have escaped apoptosis through a mechanism not linked to antigen stimulation.350
Genetic Lesions
A number of structural alterations lead to the constitutive activation of NF-kB in cHL. Nearly half of the cases display amplification of REL, associated with increased protein expression levels351,352; gains or translocations of the positive regulator BCL3 were also reported.319 More recently, a number of inactivating mutations were found in genes coding for negative regulators of NF-kB, including NFKBIA (20% of cases), NFKBIE (15%), and TNFAIP3 (40%).81,353–355 Notably, TNFAIP3-mutated cases are invariably EBV-negative, suggesting that EBV infection may substitute in part for the pathogenetic function of A20 in causing NF-kB constitutive activation.81,355 Amplification of JAK2 and inactivating mutations of SOCS1, a negative regulator of the JAK-STAT signaling pathway, are often found in NLPHL245,248; in an additional large number of cases, constitutive JAK-STAT activity is sustained by autocrine and paracrine signals.345 BCL6 translocations have been reported in the lymphocytic and histiocytic cells of NLPHD, but only rarely in cHL,356,357 and translocations of BCL2 or mutations in positive or negative regulators of apoptosis (e.g., TP53, FAS, BAD, and ATM) are virtually absent.345 Finally, an important pathogenetic cofactor in cHL, but not NLPHL, is represented by monoclonal EBV infection, which occurs in approximately 40% of cHL and up to 90% of HIV-related HL, suggesting that infection precedes clonal expansion.345 Of the viral proteins encoded by the EBV genome, infected HRS cells most commonly express LMP1, LMP2, and EBNA1 but not EBNA2.345
Selected References
The full list of references for this chapter appears in the online version.
2. Swerdlow SH, Campo E, Harris NL, et al. WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues. Lyon, France: IARC, 2008.
5. Rajewsky K. Clonal selection and learning in the antibody system. Nature 1996;381:751–758.
6. Klein U, Dalla-Favera R. Germinal centres: role in B-cell physiology and malignancy. Nat Rev Immunol 2008;8:22–33.
17. Phan RT, Dalla-Favera R. The BCL6 proto-oncogene suppresses p53 expression in germinal-centre B cells. Nature 2004;432:635–639.
28. Muramatsu M, Kinoshita K, Fagarasan S, et al. Class switch recombination and hypermutation require activation-induced cytidine deaminase (AID), a potential RNA editing enzyme. Cell 2000;102:553–563.
29. Revy P, Muto T, Levy Y, et al. Activation-induced cytidine deaminase (AID) deficiency causes the autosomal recessive form of the Hyper-IgM syndrome (HIGM2). Cell 2000; 102:565–575.
35. Ramiro AR, Jankovic M, Eisenreich T, et al. AID is required for c-myc/IgH chromosome translocations in vivo. Cell 2004;118:431–438.
37. Pasqualucci L, Bhagat G, Jankovic M, et al. AID is required for germinal center-derived lymphomagenesis. Nat Genet 2008;40:108–112.
41. Ye BH, Lista F, Lo Coco F, et al. Alterations of a zinc finger-encoding gene, BCL-6, in diffuse large-cell lymphoma. Science 1993;262:747–750.
47. Pasqualucci L, Neumeister P, Goossens T, et al. Hypermutation of multiple proto-oncogenes in B-cell diffuse large-cell lymphomas. Nature 2001;412:341–346.
48. Pasqualucci L, Migliazza A, Fracchiolla N, et al. BCL-6 mutations in normal germinal center B cells: evidence of somatic hypermutation acting outside Ig loci. Proc Natl Acad Sci U S A 1998;95:11816–11821.
49. Shen HM, Peters A, Baron B, Zhu X, Storb U. Mutation of BCL-6 gene in normal B cells by the process of somatic hypermutation of Ig genes. Science 1998;280:1750–1752.
67. Compagno M, Lim WK, Grunn A, et al. Mutations of multiple genes cause deregulation of NF-kappaB in diffuse large B-cell lymphoma. Nature 2009;459:717–721.
68. Lenz G, Davis RE, Ngo VN, et al. Oncogenic CARD11 mutations in human diffuse large B cell lymphoma. Science 2008;319:1676–1679.
69. Neri A, Knowles DM, Greco A, McCormick F, Dalla-Favera R. Analysis of RAS oncogene mutations in human lymphoid malignancies. Proc Natl Acad Sci U S A 1988;85:9268–9272.
72. Gaidano G, Ballerini P, Gong JZ, et al. p53 mutations in human lymphoid malignancies: association with Burkitt lymphoma and chronic lymphocytic leukemia. Proc Natl Acad Sci U S A 1991;88:5413–5417.
76. Mandelbaum J, Bhagat G, Tang H, et al. BLIMP1 is a tumor suppressor gene frequently disrupted in activated B-cell like diffuse large B-cell lymphoma. Cancer Cell 2010;18(6):568.
79. Kato M, Sanada M, Kato I, et al. Frequent inactivation of A20 in B-cell lymphomas. Nature 2009;459:712–716.
81. Schmitz R, Hansmann ML, Bohle V, et al. TNFAIP3 (A20) is a tumor suppressor gene in Hodgkin lymphoma and primary mediastinal B cell lymphoma. J Exp Med 2009; 206:981.
82. Dohner H, Stilgenbauer S, Benner A, et al. Genomic aberrations and survival in chronic lymphocytic leukemia. N Engl J Med 2000;343:1910–1916.
84. Calin GA, Dumitru CD, Shimizu M, et al. Frequent deletions and down-regulation of micro-RNA genes miR15 and miR16 at 13q14 in chronic lymphocytic leukemia. Proc Natl Acad Sci U S A 2002;99:15524–15529.
88. Klein U, Lia M, Crespo M, et al. The DLEU2/miR-15a/16-1 cluster controls B cell proliferation and its deletion leads to chronic lymphocytic leukemia. Cancer Cell 2010; 17:28–40.
94. Neri A, Barriga F, Inghirami G, et al. Epstein-Barr virus infection precedes clonal expansion in Burkitt’s and acquired immunodeficiency syndrome-associated lymphoma. Blood 1991;77:1092–1095.
103. Chang Y, Cesarman E, Pessin MS, et al. Identification of herpesvirus-like DNA sequences in AIDS-associated Kaposi’s sarcoma. Science 1994;266:1865–1869.
130. Tsujimoto Y, Yunis J, Onorato-Showe L, et al. Molecular cloning of the chromosomal breakpoint of B-cell lymphomas and leukemias with the t(11;14) chromosome translocation. Science 1984;224:1403–1406.
132. Motokura T, Bloom T, Kim HG, et al. A novel cyclin encoded by a bcl1-linked candidate oncogene. Nature 1991;350:512–515.
152. Dalla-Favera R, Bregni M, Erikson J, et al. Human c-myc onc gene is located on the region of chromosome 8 that is translocated in Burkitt lymphoma cells. Proc Natl Acad Sci U S A 1982;79:7824–7827.
153. Dalla-Favera R, Martinotti S, Gallo RC, Erikson J, Croce CM. Translocation and rearrangements of the c-myc oncogene locus in human undifferentiated B-cell lymphomas. Science 1983;219:963–967.
167. Meyer N, Penn LZ. Reflecting on 25 years with MYC. Nat Rev Cancer 2008;8:976–990.
168. Dominguez-Sola D, Ying CY, Grandori C, et al. Non-transcriptional control of DNA replication by c-Myc. Nature 2007;448:445–451.
169. Eilers M, Eisenman RN. Myc’s broad reach. Genes Dev 2008;22:2755–2766.
179. Adams JM, Harris AW, Pinkert CA, et al. The c-myc oncogene driven by immunoglobulin enhancers induces lymphoid malignancy in transgenic mice. Nature 1985;318:533–538.
188. Cleary ML, Smith SD, Sklar J. Cloning and structural analysis of cDNAs for bcl-2 and a hybrid bcl-2/immunoglobulin transcript resulting from the t(14;18) translocation. Cell 1986;47:19–28.
196. Hockenbery D, Nunez G, Milliman C, Schreiber RD, Korsmeyer SJ. Bcl-2 is an inner mitochondrial membrane protein that blocks programmed cell death. Nature 1990; 348:334–336.
215. Alizadeh AA, Eisen MB, Davis RE, et al. Distinct types of diffuse large B-cell lymphoma identified by gene expression profiling. Nature 2000;403:503–511.
217. Savage KJ, Monti S, Kutok JL, et al. The molecular signature of mediastinal large B-cell lymphoma differs from that of other diffuse large B-cell lymphomas and shares features with classical Hodgkin lymphoma. Blood 2003;102:3871–3879.
218. Rosenwald A, Wright G, Leroy K, et al. Molecular diagnosis of primary mediastinal B cell lymphoma identifies a clinically favorable subgroup of diffuse large B cell lymphoma related to Hodgkin lymphoma. J Exp Med 2003; 198:851–862.
224. Lenz G, Wright GW, Emre NC, et al. Molecular subtypes of diffuse large B-cell lymphoma arise by distinct genetic pathways. Proc Natl Acad Sci U S A 2008;105:13520–13525.
229. Pasqualucci L, Migliazza A, Fracchiolla N, et al. Mutations of the BCL6 proto-oncogene disrupt its negative autoregulation in diffuse large B-cell lymphoma. Blood 2003;101:2914–2923.
232. Migliazza A, Martinotti S, Chen W, et al. Frequent somatic hypermutation of the 5’ noncoding region of the BCL6 gene in B-cell lymphoma. Proc Natl Acad Sci U S A 1995;92:12520–12524.
233. Saito M, Gao J, Basso K, et al. A signaling pathway mediating downregulation of BCL6 in germinal center B cells is blocked by BCL6 gene alterations in B cell lymphoma. Cancer Cell 2007;12:280–292.
236. Davis RE, Ngo VN, Lenz G, et al. Chronic active B-cell-receptor signalling in diffuse large B-cell lymphoma. Nature 2010;463:88–92.
251. Wotherspoon AC. Gastric lymphoma of mucosa-associated lymphoid tissue and Helicobacter pylori. Annu Rev Med 1998;49:289.
274. Caligaris-Cappio F, Hamblin TJ. B-cell chronic lymphocytic leukemia: a bird of a different feather. J Clin Oncol 1999;17:399–408.
278. Klein U, Tu Y, Stolovitzky GA, et al. Gene expression profiling of B cell chronic lymphocytic leukemia reveals a homogeneous phenotype related to memory B cells. J Exp Med 2001;194:1625–1638.
279. Rosenwald A, Alizadeh AA, Widhopf G, et al. Relation of gene expression phenotype to immunoglobulin mutation genotype in B cell chronic lymphocytic leukemia. J Exp Med 2001;194:1639–1647.
288. Rawstron AC, Bennett FL, O’Connor SJ, et al. Monoclonal B-cell lymphocytosis and chronic lymphocytic leukemia. N Engl J Med 2008;359:575.
310. de Leval L, Bisig B, Thielen C, Boniver J, Gaulard P. Molecular classification of T-cell lymphomas. Crit Rev Oncol Hematol 2009;72:125–143.
333. Chiarle R, Voena C, Ambrogio C, Piva R, Inghirami G. The anaplastic lymphoma kinase in the pathogenesis of cancer. Nat Rev Cancer 2008;8:11–23.
334. Morris SW, Kirstein MN, Valentine MB, et al. Fusion of a kinase gene, ALK, to a nucleolar protein gene, NPM, in non-Hodgkin’s lymphoma. Science 1994;263:1281–1284.
349. Küppers R, Rajewsky K, Zhao M, et al. Hodgkin disease: Hodgkin and Reed-Sternberg cells picked from histological sections show clonal immunoglobulin gene rearrangements and appear to be derived from B cells at various stages of development. Proc Natl Acad Sci U S A 1994;91:10962–10966.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree