Islets of Langerhans: Morphology and Postnatal Growth
Susan Bonner-Weir
The islets of Langerhans are clusters of endocrine tissue scattered throughout the exocrine pancreas in all vertebrates higher in evolutionary development than the bony fish (teleosts). In the adult mammal, the islets are 1% to 2% of the pancreatic mass and thus comprise approximately 1 g of tissue in the adult human. Islets are complex structures of cells and function both separately as micro-organs and in concert as the endocrine pancreas. Although the direct secretion of insulin and glucagon from islets into the portal vein has obvious advantages with respect to influence on hepatic function, it is not clear why the endocrine pancreas is dispersed throughout the exocrine pancreas. One suggestion is that the local insular-acinar portal system helps regulate the exocrine function of the pancreas, with this function providing some evolutionary advantage (1).
The pancreas of the adult human contains approximately 200 U, or 8 mg, of insulin (2) and that of the adult rat contains about 100 μg of insulin. The size of an islet can range from only a few cells and less than 40 μm in diameter to approximately 10,000 cells and 400 μm in diameter. The average rat islet is 150 μm in diameter and contains approximately 45 ng of insulin. In the rat, and probably in other mammals, islets smaller than 160 μm in diameter represent 75% of the islets in number but only 15% of the islet volume, whereas islets larger than 250 μm in diameter represent only 15% of the islets in number but 60% of the islet volume (3).
Islet mass is dynamic, adjusting to meet the changing needs of the individual, whose size and level of activity vary at different stages of life. When islet mass cannot adjust to meet the demand, diabetes mellitus results.
Although studies of the islets of nonmammalian vertebrates have been useful in extending our knowledge, we have a far more detailed understanding of the structure, function, and changes in mass of mammalian islets. In a text on diabetes, the emphasis should be on the human islet, but our present understanding of islets is based mainly on rodent studies. Thus, the rodent islet will be used as the paradigm.
This chapter will first address the cellular components of the islet and their organization. We now know that islets are not all the same, and this heterogeneity will be discussed. The manner in which the structural organization of an islet defines its function will be addressed. Finally, issues of islet growth after birth, both normal and compensatory, will be discussed. Embryonic development of the endocrine pancreas has been discussed in the previous chapter (Chapter 2).
OVERALL PANCREATIC ANATOMY
The origin of the pancreas as separate primordia is thought to be the basis of the regional distribution of glucagon-producing and pancreatic polypeptide-producing cells (4). The dorsal pancreas, supplied with blood by the celiac trunk via the gastroduodenal and splenic arteries and drained by one main pancreatic duct, contains the glucagon-rich islets with few pancreatic polypeptide-containing cells. The opposite distribution is
found in the ventral pancreas, which is supplied with blood from the superior mesenteric artery via the inferior pancreatico-duodenal artery and is drained by a separate exocrine duct. Here the islets contain pancreatic polypeptide-producing cells and few, if any, glucagon-producing cells. The degree of fusion of these ducts differs among species.
found in the ventral pancreas, which is supplied with blood from the superior mesenteric artery via the inferior pancreatico-duodenal artery and is drained by a separate exocrine duct. Here the islets contain pancreatic polypeptide-producing cells and few, if any, glucagon-producing cells. The degree of fusion of these ducts differs among species.
The normal organization of the islet, with its central core of β-cells and its peripheral non-β-cells, is observed only after fetal day 18.5 (5). In adult mammals, 70% to 80% of the islet consists of insulin-producing β-cells, 5% is somatostatin-producing δ-cells, and 15% to 20% is either glucagon-producing α-cells or pancreatic polypeptide-producing PP cells. The proportions of the different cell types differ with age because the different islet cells do not have the same pattern of growth. For example, in both rats and humans, the percentage of islet cells that are δ-cells is considerably greater in perinatal than in adult individuals (6). At birth the β-cells are usually only 50% of the islet, and with postnatal replication of the β-cells and increase in cell volume (7), their proportion increases.
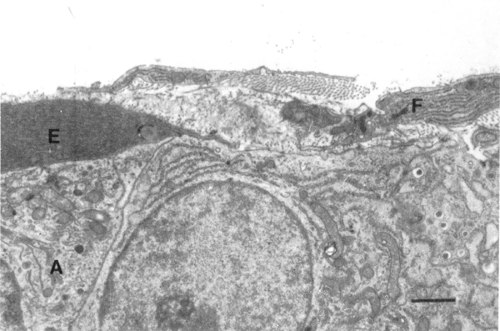
Islets differentiate from the pancreatic ductal epithelium as do exocrine cells, but the question of whether they are derived from the same or different precursor/stem cell populations remains unanswered. Embryonic development of the pancreas as ductules that proliferate, branch, and then differentiate was described by Pictet and Rutter (8). Islet hormone-containing cells are first seen as single cells among the exocrine cells of the terminal pancreatic tubules and then as clusters of cells within the exocrine basement membrane. These clusters then become separated from the exocrine tissue to form distinct islets (8). In the adult pancreas, sometimes the only separation between exocrine and islet cells are their respective basement membranes (Fig. 3.1). However, islets are usually surrounded by at least a partial capsule of fibroblasts and collagen fibers.
In the pancreas of adults of many species, islet cells of all types can be immunolocalized in the pancreatic ducts as occasional single cells or small budding islets. An increased occurrence of these cells has been observed under numerous experimental conditions, including dietary treatment with soybean trypsin inhibitor (9), overexpression of interferon-γ in the β-cells of transgenic mice (10), after partial pancreatectomy (11), and after cellophane wrapping of the head of the pancreas (12), as well as with some human diseases, including recent-onset type 1 diabetes mellitus (insulin-dependent) (13) and severe liver disease. Adult ductal epithelium can be stimulated to undergo morphogenic changes that result in a substantial formation of new islets. In the young adult rat, 3 days after a 90% pancrea-tectomy, 10% to 15% of the pancreatic remnant volume is composed of proliferating ductules, which differentiate into new islets and exocrine tissue within another 3 to 4 days and largely account for the doubling of remnant mass found within 1 week of surgery (11). When adult ductal epithelium was wrapped in fetal mesenchyme and implanted in nude mice, approximately 20% of the grafts were found to contain “islet-like cell clusters” with hormone immunostaining budding from the ducts (13,14). Adult human ductal tissue can be driven to differentiate into islets in vitro (15). The differentiation process can be reprogrammed, as seen in adult rats depleted of copper with a resultant destruction of acinar tissue but “normal islets and ducts.” After copper repletion, hepatocytes, rather than acinar cells, regenerate in the pancreas, this being an example of transdifferentiation (16).
PHYLOGENETIC CONSIDERATIONS
The comparative aspects of the endocrine pancreas was the focus of much study in the 1970s (17,18,19,20). The first step in the development of a separate islet organ is found in the vertebrate class Agnatha, the primitive jawless fish (represented today by hagfish and lampreys). In hagfish all of the β-cells and most of the δ-cells are no longer in the gut mucosa and are restricted to the bile duct and adjacent islet organ, whereas the glucagon/gastrin-producing cells remain in the gut mucosa (17). The first appearance of a pancreas in which β-, α-, δ-, and
PP-cells are all represented is in the cartilaginous fish (class Chondrichthyes), in which the islet cells are found in the parenchyma, in the pancreatic ducts, or disseminated in the exocrine pancreas (17,21). The bony fish (class Osteichthyes) have large accumulations of endocrine tissue, sometimes called the Brockmann bodies, near the spleen and pylorus and, in addition, have small islets scattered throughout the exocrine tissue, often in association with small ducts. The descriptions of islets in amphibians and reptiles are widely variant, but there seems to be a pattern of three types of islets: large splenic islets with the α-cell population greater than the β-cell population; islets of intermediate size with more β-cells but still a majority of α-cells; and small islets with mostly β-cells. This heterogeneity of islets has been better defined for the birds (class Aves): Dark, or α, islets are composed of α- and δ-cells; and light, or β, islets are composed of β- and δ-cells (PP-cells are often extrainsular). In this class, as in mammals, the regional distribution of islet types has an embryologic origin.
PP-cells are all represented is in the cartilaginous fish (class Chondrichthyes), in which the islet cells are found in the parenchyma, in the pancreatic ducts, or disseminated in the exocrine pancreas (17,21). The bony fish (class Osteichthyes) have large accumulations of endocrine tissue, sometimes called the Brockmann bodies, near the spleen and pylorus and, in addition, have small islets scattered throughout the exocrine tissue, often in association with small ducts. The descriptions of islets in amphibians and reptiles are widely variant, but there seems to be a pattern of three types of islets: large splenic islets with the α-cell population greater than the β-cell population; islets of intermediate size with more β-cells but still a majority of α-cells; and small islets with mostly β-cells. This heterogeneity of islets has been better defined for the birds (class Aves): Dark, or α, islets are composed of α- and δ-cells; and light, or β, islets are composed of β- and δ-cells (PP-cells are often extrainsular). In this class, as in mammals, the regional distribution of islet types has an embryologic origin.
COMPONENTS OF THE ISLETS OF LANGERHANS
Endocrine Cells
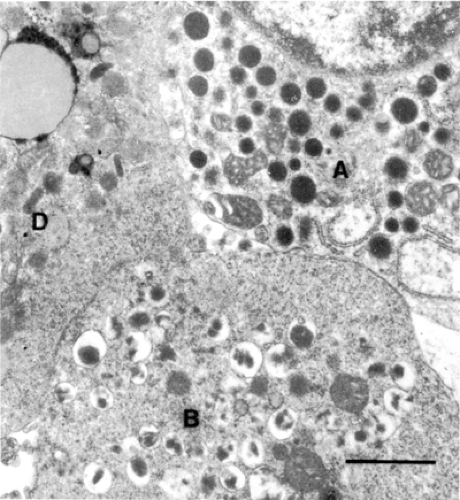
There are four major endocrine cell types in mammalian islets: the insulin-producing β-cell, the glucagon-producing α-cell, the somatostatin-producing δ-cell, and the pancreatic polypeptide-producing PP-cell (the latter three will be referred to collectively as the non-β-cells) (Fig. 3.2). Ultrastructural and immunocytochemical techniques are used to distinguish these cell types and have identified other minor cell types (Table 3.1). Numerous other peptides and hormones have been localized to the islet cells with the use of sensitive immunostaining techniques (Table 3.1). Localization of several of these peptides is confusing because the type of cells that are immunostained varies with species. For example, calcitonin gene-related peptide (CGRP) co-localizes with somatostatin in the rat δ-cells but with insulin in mouse β-cells (22). Similarly, an antibody to pancreastatin stains α- and δ-cells in humans but β- and δ-cells in pigs (23,24). An additional level of complexity is introduced by evidence that the hormones thyrotropin-releasing hormone and gastrin are expressed in the islets only during the perinatal period (25,26). It is presently unclear how any of these other hormones/peptides function in the islet. The sensitivity of the techniques and the overlap of antibody recognition may be responsible for some of these confusing data. Another explanation for the overlap may be the sequential differentiation of the islet cell types.
TABLE 3.1. Characteristics of the Endocrine Cells of the Islets of Langerhansa | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||
The β-cells are polyhedral, being truncated pyramids, and are usually well granulated, with secretory granules 250 to 300 nm in diameter (Fig. 3.3). It has been estimated that each mouse β-cell contains approximately 10,000 granules (27). There are
two forms of insulin granules: mature granules that have an electron-dense core and a loosely fitting granule-limiting membrane with the appearance of a spacious halo; and immature granules with little or no halo, moderately electron-dense contents, and a clathrin coat. Immature granules have been shown to be the major, if not the only, site of conversion from proinsulin to insulin (28). Other changes, such as the shedding of the clathrin coat, acidification of the granule contents, and crystallization of insulin, occur as the granule matures from a proinsulin-rich granule to an insulin-rich granule (28). In some species the electron-dense core of the mature granule is visibly crystalline. Insulin is not the only peptide in the granules; there are at least 100 other peptides (29).
two forms of insulin granules: mature granules that have an electron-dense core and a loosely fitting granule-limiting membrane with the appearance of a spacious halo; and immature granules with little or no halo, moderately electron-dense contents, and a clathrin coat. Immature granules have been shown to be the major, if not the only, site of conversion from proinsulin to insulin (28). Other changes, such as the shedding of the clathrin coat, acidification of the granule contents, and crystallization of insulin, occur as the granule matures from a proinsulin-rich granule to an insulin-rich granule (28). In some species the electron-dense core of the mature granule is visibly crystalline. Insulin is not the only peptide in the granules; there are at least 100 other peptides (29).
The α-cells are usually smaller and more columnar than the β-cells and well granulated, with granules 200 to 250 nm in diameter (Fig. 3.2). The granules are electron-dense with a narrow halo of less-dense material and a tightly fitting granule-limiting membrane; there is little species variation.
The δ-cells are usually smaller than either α- or β-cells, are well granulated, and are often dendritic in shape. The electron density of granules within a δ-cell varies greatly (Fig. 3.2). Each granule, 200 to 250 nm in diameter, contains material of homogeneous moderate density that fills the granule-limiting membrane.
PP-cells are the most variable among species. In humans the granules are elongated, very electron dense, and 120 to 160 nm in diameter. In dogs and cats, the granules are spherical, variable in electron density, and approximately 300 nm in diameter (30).
Capsulem
The existence of an islet capsule has been a controversial issue, perhaps because the capsule is only a single layer of fibroblasts and the collagen fibers laid down by these fibroblasts (Fig. 3.1). Furthermore, frequently the capsule between islet and exocrine cells is absent. The extant capsule does overlay the efferent blood vessels of the islet and seems to define a subcapsular interstitial space.
Microvasculature
The islet is highly vascularized and has a direct arteriolar blood supply (31). Islet capillaries are fenestrated, whereas fenestration is decreased or absent in capillaries of the surrounding exocrine tissue (32). The fenestrae render these capillaries highly permeable. The passage of horseradish peroxidase into the pericapillary interstitial space of the islet takes only 45 seconds, whereas it takes 5 minutes in cardiac muscle (33). The blood flow to the islets has been found to be disproportionately large (10% to 20% of the pancreatic blood flow) for the 1% to 2% of pancreatic volume (34,35,36,37). Factors regulating islet blood flow may affect islet hormone secretion. High concentrations of glucose have been shown to enhance pancreatic blood flow and to preferentially increase islet blood flow (38). In addition, several of the peptides immunochemically localized in islet nerves and/or endocrine cells, for example, CGRP, are vasoactive (39). The relation of the microvasculature to islet endocrine cells is discussed below. Lymphatic vessels, while common in the pancreas, have not been found within the islets (40).
Nerves
The pancreas is innervated by sympathetic fibers from the celiac and superior mesenteric ganglia and by parasympathetic fibers from the vagus nerve (41,42). These parasympathetic fibers synapse in small ganglia dispersed in the pancreas. They may act as pacemakers for the oscillations in hormone secretion (43) that occur without extrinsic nervous connections, as in the isolated perfused canine pancreas (44).
Within the pancreas, nerve fibers terminate in perivascular, periacinar, and peri-insular areas. Within the islets, the nerves follow the blood vessels and terminate within the pericapillary space, within the capillary basement membrane, or closely apposed to the endocrine cells. Because no specialized synapses are found, it has been suggested that neurotransmitters released into the interstitial space may affect a number of neighboring islet cells. In some species, nerve cell bodies are close to or even within the islets (45).
The distribution and number of the different types of nerve fibers differ among species (41,42,45). Cholinergic nerves, identified histochemically by cholinesterase activity or ultrastructurally by the presence of electron-lucent vesicles, are common in rat, cat, and rabbit islets. Adrenergic nerves, as identified immunochemically by antibodies to catecholamine-forming enzymes (including tyrosine hydroxylase) (45) and ultrastructurally by the presence of dense cored vesicles, are common in the hamster, dog, and cat. Peptidergic nerves are less well defined, being identified either immunochemically as containing substance P, enkephalin, vasoactive intestinal polypeptide (VIP), galanin, gastrin-releasing peptide, CGRP, or cholecystokinin, or ultrastructurally by the presence of large granules. A further complication is introduced by the finding of neuropeptide Y and galanin in adrenergic nerves and the identification of VIP in cholinergic nerves (46). Both substance P and CGRP seem to be localized in the sensory fibers. The presence of peptidergic nerve cell bodies in pancreatic ganglia suggests that some of these intrinsic nerves may be involved in the synchronization of islet secretion (45).
The autonomic nervous system modulates islet hormone secretion (41,45,47). Cholinergic stimulation elicits increased secretion of insulin, glucagon, and pancreatic polypeptide. Its effects on somatostatin are less clear but appear to be inhibitory. β-Adrenergic stimulation similarly elicits increased secretion of insulin, glucagon, pancreatic polypeptide, and somatostatin (48). β-Adrenergic stimulation decreases secretion
of insulin and somatostatin, but its effect on glucagon secretion varies with species. The effects of stimulation of peptidergic nerves have not been studied as closely, but VIP, cholecystokinin, and galanin can affect islet-cell secretion. VIP causes an increase in insulin secretion that is direct (49). VIP neurons form a peri-insular network in rats (50), but the peptide does not change the islet blood flow as it increases the total pancreatic blood flow (49).
of insulin and somatostatin, but its effect on glucagon secretion varies with species. The effects of stimulation of peptidergic nerves have not been studied as closely, but VIP, cholecystokinin, and galanin can affect islet-cell secretion. VIP causes an increase in insulin secretion that is direct (49). VIP neurons form a peri-insular network in rats (50), but the peptide does not change the islet blood flow as it increases the total pancreatic blood flow (49).
ORGANIZATION OF THE COMPONENTS OF THE ISLETS OF LANGERHANS
The distribution of the endocrine cells is nonrandom, with a core of β-cells surrounded by a discontinuous mantle of non-β-cells one to three cells thick (51,52). The islets of most mammalian species have this pattern, but those of humans and other primates have a somewhat more complex arrangement (Fig. 3.4). Sections of human pancreas show many different islet profiles, including oval and cloverleaf patterns, differences that have resulted in controversy about whether they actually have a mantle-core arrangement (52,53,54). Nonetheless, in three dimensions, human islets can be considered as composites of several mantle-core subunits or as lobulated with mantle-core lobules (52,55). In sectioned tissue, incomplete fusion of such subunits and penetrations of islet vasculature can appear as invaginations of the islet periphery. Most of the non-β-cells are found along these invaginations and the periphery (52,53), thus maintaining a mantle-core arrangement. Confocal microscopy allows a better appreciation of the three-dimensional organization of islet cells (56). With confocal microscopy, α-cells are seen as a sheet or a peel at the islet periphery and δ-cells are seen as more peripheral to the α-cells in the human but less peripheral in the rat. This pattern of organization is based on intrinsic qualities of the cell surfaces of the different cell types, as shown by reaggregation studies. When single dispersed rat islet cells were allowed to reaggregate, the aggregates showed the same nonrandom organization as native islets, even when the proportions of β-cells and non-β-cells were reversed (57). Both β-cells and non-β-cells have high levels of calcium-dependent cell-adhesion molecules (cadherins), but non-β-cells also contain calcium-independent cell-adhesion molecules (58). It is thought that this differential expression of cell-adhesion molecules may be responsible for the segregation of β-cells and non-β-cells.
The microvasculature forms the infrastructure of an islet. The rat islet has been studied most extensively and will serve as the prototype in this discussion, but this generalization needs to be verified. Afferent vessels to an islet number one to three arterioles, depending on islet size. Each arteriole penetrates the islet through discontinuities of the non-β-cell mantle and enters directly into the β-cell core, where it branches into a number of fenestrated capillaries (3). These capillaries follow a tortuous path, passing first through the β-cell core and then through the non-β-cell mantle. Often the vessels pass along the inside of the mantle before penetrating it. The efferent vessels are collecting or postcapillary venules. It is unclear where the transition from capillary to collecting venule occurs, but collecting venules can often be seen in indentations of the islet. There appears to be a single continuous circulation through the islet. Although each capillary should have functionally different arterial and venous portions, no morphologic differences have yet been found.
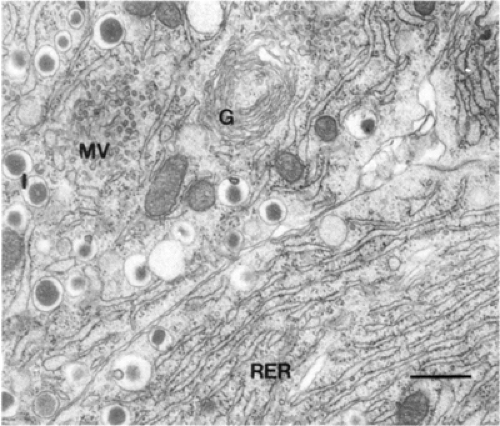
The pattern of microvasculature varies, depending on islet size (Fig. 3.5). In large islets, the efferent vessels coalesce into collecting venules within the subcapsular space. In small islets, the efferent capillaries extend into the exocrine tissue for 50 to 100 μm before coalescing into collecting venules. Within the islet, endocrine cells are organized around the microvasculature (Fig. 3.6). In the rat, the β-cells have been found to have two faces on capillaries (59). In cross-section, eight to ten β-cells are seen to form a rosette around one of the capillaries. In three dimensions this pattern would be tubelike. The outer face of each β-cell would be against a capillary, which is probably arterial and orthogonal to the central capillary. In β-cells experimentally degranulated with glyburide, the remaining granules are polarized toward the central “venous” capillary, suggesting an in situ polarity of the β-cells. Along the interface of three or more β-cells and extending from capillary face to capillary face are elaborate canaliculi (59). Desmosomes that act as mechanical attachments are preferentially found near these canaliculi. These canaliculi contain large numbers of microvilli (59,60) that have been found to be enriched in glucose transporter 2 (GLUT-2) (61). The presence of these elaborate structures suggests a bulk flow of interstitial fluid through these canaliculi in an arterial-to-venous direction and an uptake of glucose by the β-cells that is carried out mainly by the transporters on these
microvilli. Thus, these canaliculi may serve as the initial interface for glucose sensing by the β-cell. Insulin secretory granules appear to be released from the lateral and apical surfaces to enter the venous (central) capillaries.
microvilli. Thus, these canaliculi may serve as the initial interface for glucose sensing by the β-cell. Insulin secretory granules appear to be released from the lateral and apical surfaces to enter the venous (central) capillaries.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree