Inflammatory breast cancer (IBC) is one of the rarest and most aggressive forms of breast cancer, having relatively distinct clinicopathological features and the lowest survival rates. Its presentation was first described in 1814 by Bell as “a purple color on the skin over the tumor accompanied by shooting pains,” which he identified as “a very unpropitious beginning.”1 In 1889, Bryant noted the role of cancer cells obstructing the lymphatics in the development of inflammatory signs.2 The term “inflammatory carcinoma of the breast” was coined by Lee and Tannebaum in 1924. Currently, based on the American Joint Committee on Cancer (AJCC) Staging System, IBC is classified as T4d and described as a “clinicopathological entity that is characterized by diffuse erythema and edema (peau d’orange), often without an underlying mass. These clinical findings should involve the majority of the breast . . “3 It should be stressed that IBC is primarily a clinical diagnosis and thus, in the absence of clinical findings, involvement of the dermal lymphatics alone does not indicate the diagnosis of IBC. In this chapter, we review the epidemiology, clinical presentation, diagnostic modalities, and treatment, and discuss the current controversies of IBC.
The incidence of IBC is 1% to 6% in the United States. However, results from the 2005 Surveillance, Epidemiology, and End Results program revealed that the IBC incidence rate increased from 2.0/100,000 woman-years for 1988 to 1990 to 2.5/100,000 woman-years for 1997 to 1999. This is a marked contrast with the incidence rates for other types of invasive breast cancers, which decreased during the same time.4
Before 1990, it was believed that IBC was particularly prevalent in North Africa, specifically in Tunisia, because pre-1990 reports revealed a higher than 50% incidence of IBC in those areas.5 However, a review of 419 cases diagnosed as T4d between 1975 and 1996 at a single Tunisian institution revealed the incidence of IBC in Tunisia to be approximately 7%.5 This decrease in IBC incidence was attributed to the fact that prior studies had included not only T4d breast cancers, but also ulcerated breast cancers (T4b), which clearly exemplifies the complexities in the diagnosis of IBC.
Studies done using breast biopsy tissue from Tunisian patients with IBC revealed an increased incidence in the detection of viral sequences and antigens that resemble mouse mammary tumor virus.6 This relationship is currently under investigation in several institutions in the United States to confirm the etiopathogenetic role of mouse mammary tumor virus in IBC. Currently, various molecular factors associated with IBC have been identified, but the causative agent remains to be isolated.
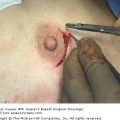
IBC has a characteristic clinical presentation of skin changes and breast enlargement that occurs rapidly, typically within 3 months (Fig. 90-1).7 Although the AJCC definition calls for clinical findings to involve the majority of the breast, some researchers are calling for the disease to be diagnosed when there is less skin involvement so that an earlier diagnosis and possibly a better outcome might be obtained.8 IBC is often considered to have 3 clinical variants: primary, secondary, and occult (Table 90-1).2 The occult variant has a better prognosis.
Figure 90-1
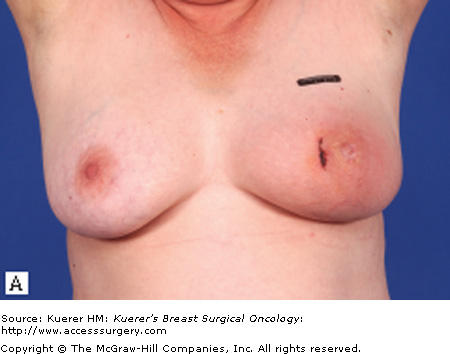
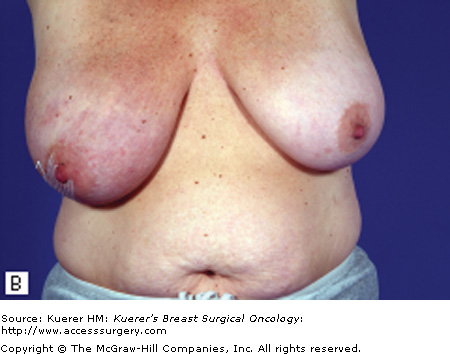
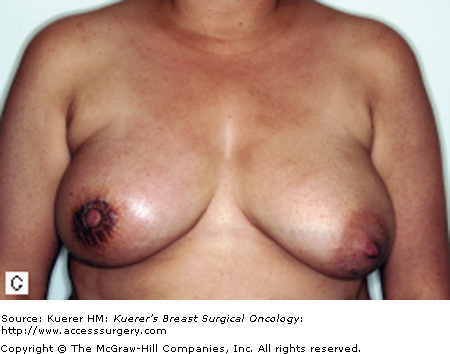
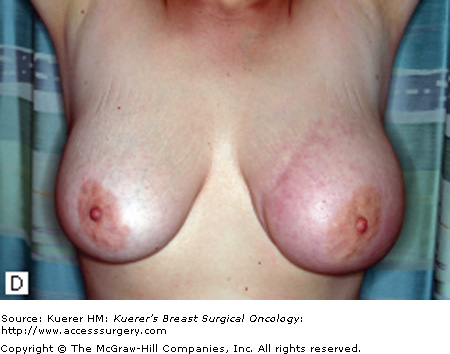
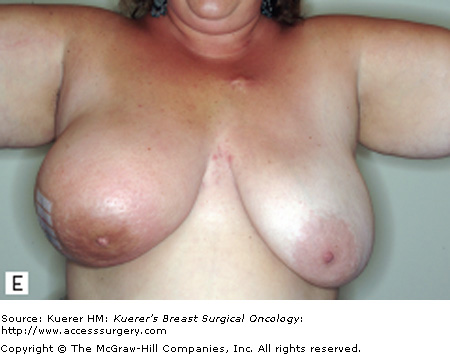
Characteristic clinical presentations of patients with IBC. A. Patient with left breast IBC; note erythema and peau de orange. B. Patient with right breast IBC; note erythema and marked breast edema. C. Patient with right breast IBC; note skin changes and edema. D. Patient with left breast IBC; note breast edema and erythema. E. Patient with right breast IBC; note erythema and peau de orange.
| Variant | Characteristics |
|---|---|
| Primary | Signs of inflammation |
| No palpable breast mass | |
| Enlarged ipsilateral axillary lymph node | |
| Presence of clinically, radiologically, and pathologically indistinct invasive carcinoma | |
| Secondary | Skin manifestation in breast that previously contained cancer |
| After mastectomy, skin manifestation in opposite breast or distant cutaneous recurrence | |
| Clinical course and behavior similar to primary IBC | |
| Occult | No clinical evidence of inflammation |
| Tumor emboli present in dermal lymphatics on histological exams | |
| Has a better prognosis |
Although IBC is most commonly associated with ductal carcinoma, lobular and medullary carcinomas may also be associated with an IBC diagnosis. The mean age at diagnosis is 58 years, and black women appear to be more affected with this disease than are other women.4 IBC is often estrogen receptor (ER) negative (up to 83% of cases), and it is associated with a higher nuclear grade tumor and a higher incidence of lymph node involvement compared with other types of breast cancer.9 As reported in the literature, the incidence of involved axillary lymph nodes can reach 72.3% (range, 60%-85%), with supraclavicular lymph node involvement seen in 11.6% of cases.10 Further, IBC is associated with a higher incidence (∼20%) of distant metastasis at the time of diagnosis than are other breast cancers. It has also been reported that a high body mass index in breast cancer patients is associated with a poor overall survival rate.11 However, Chang et al11 found no statistical survival difference between obese and nonobese patients with IBC. They also found that premenopausal women with IBC had a worse survival rate than did postmenopausal women with IBC.11
In general, the diagnosis of IBC is an independent predictor of a poor prognosis, with death from this disease occurring nearly twice as often as in other breast cancers.8,12,13 In their institutional review of 398 patients treated for IBC at the University of Texas MD Anderson Cancer Center between 1974 and 2005, Gonzalez-Angulo et al13 revealed a median overall survival duration of 4.2 years and a mean time to disease recurrence of 2.3 years. These findings demonstrate a marked contrast with those for stage I breast cancer, which has a 5-year overall survival rate of 90% or more.14 Finally, both “estrogen (ER)/ progesterone (PR)-negative receptor status” and high nuclear grade are associated with a poorer overall survival rate for patients diagnosed with IBC.
IBC has characteristic clinical findings, but may be confused with locally advanced breast cancer (LABC), which can present with the same physical findings. Thus, a thorough medical history is crucial to distinguish between the 2 as treatment and prognosis can differ significantly. LABC and IBC were previously thought to represent different points on the same disease spectrum, but it has since been well established that they are distinct entities, each with distinct clinical behaviors and outcomes (Table 90-2).12,15 For example, in IBC the physical signs are secondary to the characteristic dermal lymphatic invasion by tumor cells, whereas in LABC the physical signs typically are believed to represent tumor that has progressed as a result of delayed diagnosis or neglect.16 A critical difference between the 2 is the time frame during which the signs of disease develop. IBC arises quickly (<3 months), while LABC often develops slowly. IBC is usually detected by the patient because of the rapid progression of changes to the skin, and fewer than 50% of IBC patients present with a well-defined mass.7 In contrast, LABC is usually detected by the patient because of the presence of a mass over a long period of time, which is sometimes neglected.
| LABC | IBC |
|---|---|
| Arises over a longer period of time (>3 months) | Arises quickly (<3 months) |
| No dermal lymphatic involvement16 | Probable dermal lymphatic involvement16 |
| Palpable mass usually detected by patient | Rapid, progressive changes to skin usually detected by patient |
| Older patients | Younger patients |
| Positive ER/PR common*,12 | Negative ER/PR common*,12 |
| Positive node involvement common†,12 | |
| High tumor grade common12 | |
| No micrometastasis | Possible micrometastasis |
| Possible increase in angiogenesis and lymphangiogenesis | Probable increase in angiogenesis and lymphangiogenesis |
| Possible overexpression of HER-2/neu, RhoC GTPase, and NF-κB15 | Probable overexpression of HER-2/neu, RhoC GTPase, and NF-κB15 |
| Incidence increases with age‡,12 | Incidence plateaus after age 50 years12 |
| Overall survival rate of 44%-79% at 5 years7 | Overall survival rate of 41% at 5 years7 |
The treatment strategy for both IBC and LABC includes neoadjuvant chemotherapy, surgery, and radiotherapy. However, 2 of the most important reasons for distinguishing between them are the differences in the actual local treatment prescribed and the survival outcomes. The outcome for patients with IBC is less favorable than that for patients with LABC, with IBC patients being at twice the risk of death from disease as LABC patients.12 A series studied by Hortobagyi et al7 revealed 5-year overall survival rates for patients with stage IIIA or IIIB LABC to be 79% and 44%, respectively, while the overall 5-year survival rate for patients with IBC was 41%.
Mastitis is another condition that must be differentiated from IBC. Mastitis usually occurs during lactation, and the skin changes are accompanied by the flu-like symptoms of fatigue, fever, chills, and leukocytosis.2 An abscess may also be present. A mammogram may be normal or show increased tissue density, while a sonogram may show increased vasculature, skin thickening, or an abscess in the breast.17 Mastitis responds well to antibiotics; therefore, any suspected mastitis that persists for longer than 2 weeks or that does not respond to antibiotics warrants a biopsy to exclude IBC.
Radiation dermatitis must also be distinguished from IBC. It is usually limited to areas within the radiation field and often resembles a sun burn. Moist desquamation can occur, but typically these changes resolve within 2 to 3 weeks. Edema is not typically observed.2
Other conditions to be considered in the differential diagnosis of IBC include breast lymphoma, Paget disease, erysipelas, tuberculosis, metastatic gastric or ovarian cancer,18 and calciphylaxis.19 With so many conditions to be excluded, it is imperative to obtain a thorough medical history and to perform a complete physical exam, as well as radiological studies and biopsies, to distinguish all these conditions from IBC. A heightened awareness of IBC is extremely important to its early diagnosis because the longer it goes undiagnosed the worse it is for a patient who has an already poor survival outcome.
Although IBC is a clinical diagnosis, imaging studies play an important role in the complete assessment of patients because they provide characterization and delineation of the disease in the breast and enable the evaluation of the contralateral breast, regional lymph node basin, distant metastasis, and response to therapy. The standard imaging modalities used in IBC are mammography and ultrasonography, both of which provide better imaging than in previous years owing to technological advances. Similarly, technological innovations have enabled magnetic resonance imaging (MRI) and positron emission tomography by means of computed tomography (PET/CT) to be used in the early diagnosis and staging of IBC. Because technological advances in radiology have improved the imaging of breast tissue, the idea that IBC is not associated with a breast mass7 is being challenged by current findings.
Despite its standard use, mammography is the least sensitive imaging modality used in the differential diagnosis of IBC. The importance of mammography lies in the early detection of abnormality. The most common mammographic findings for IBC are skin thickening and trabecular distortion, which are seen in 83% and 73% of IBC patients, respectively.20 On mammograms, skin thickening indicative for IBC is first seen in the inferior areolar region, in contrast with other etiologies in which skin thickening is local or segmental.21
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree