Inflammatory and Lymphoid Lesions
ACUTE INFLAMMATION
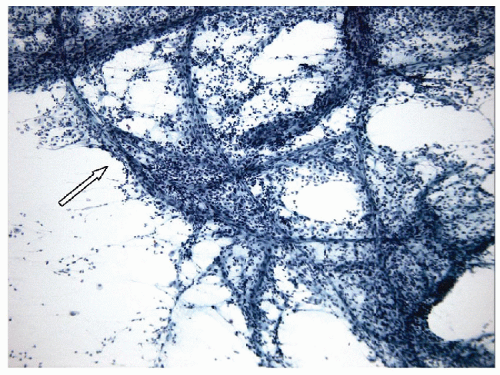
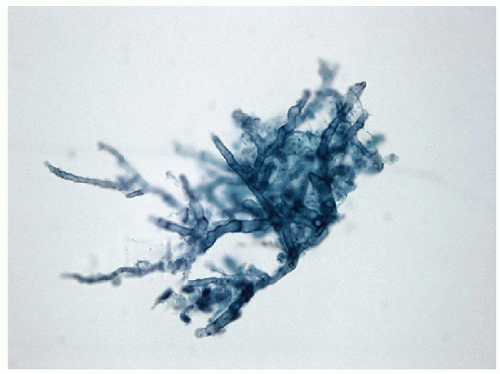
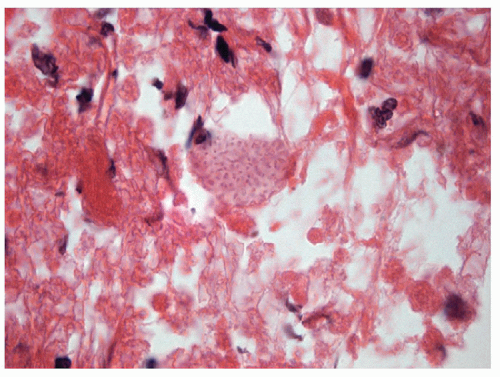
Acute inflammation of the thyroid occurs in the setting of infection and/or trauma, after radiation, or in association with tumor necrosis. Infection is rare in economically advantaged countries, and when it does occur, it is often associated with structural abnormalities including fistulas [1, 2, 3, 4]. It is more commonly seen in impoverished areas of the world [5, 6] or in immunocompromised hosts [7]. Acute inflammation often is attributed to direct extension from adjacent oropharyngeal structures, but there may also be hematogenous dissemination of systemic infection or there may be local infection associated with severe trauma. Fine needle aspiration (FNA) frequently shows acute inflammatory cells, macrophages, necrotic debris, granulation tissue-like vascular fragments, and rarely an identifiable infectious agent (Figs. 8.1, 8.2, 8.3, 8.4, e-Figs. 8.1, 8.2, 8.3). Histologic biopsy will identify acute inflammatory cells, often with microabscess formation, necrotic debris, and evidence of vascular damage. The degree of inflammation may vary, particularly in immunocompromised hosts. The material should be stained to identify infectious organisms that may be bacterial, fungal, or viral. Cultures for microbes can be helpful and may provide important information about sensitivity to specific antibacterial or antifungal therapies.
The important differential diagnosis is anaplastic thyroid carcinoma that can have large areas of tumor necrosis that can resemble acute inflammation but is generally sterile, yielding no organisms on culture.
GRANULOMATOUS (SUBACUTE) INFLAMMATION
Granulomatous inflammation of the thyroid may occur as a result of a variety of disorders:
2. Palpation thyroiditis [9]
4. Infectious processes (mycoses, mycobacterial infection, etc.)[1]
de Quervain’s thyroiditis is an idiopathic, possibly autoimmune, or viral disorder also known as subacute thyroiditis or granulomatous thyroiditis. This distinct clinicopathologic entity rarely results in thyroid biopsy because of its characteristic clinical manifestations. Patients usually develop painful thyroid enlargement, typically close to 10 days following a viral upper respiratory tract infection. There may be associated transient hyperthyroidism and associated biochemical disturbances that are attributed to rupture of thyroid follicles and release of stored thyroid hormone. This may be followed by a transient phase of hypothyroidism due to tissue damage. The disease is self-limited and resolves spontaneously.
Other forms of subacute and granulomatous inflammation include “painless” subacute thyroiditis and the focal type of granulomatous inflammation that results from palpation of the thyroid during clinical examination (“palpation thyroiditis”). The latter is a common finding focally in most thyroids that are removed surgically.
Sarcoidosis may involve the thyroid gland, and infections process may occur that are granulomatous in nature. Fungal infections are more common in immunocompromised patients, while tuberculosis is rare.
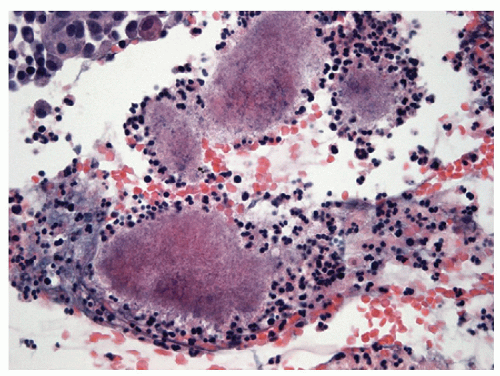
FNA reveals granulomatous inflammation admixed with follicular epithelium and a variable amount of colloid (Figs. 8.5 and 8.6, e-Figs. 8.4, 8.5, 8.6, 8.7, 8.8, 8.9, 8.10, 8.11). Multinucleated giant cells may be seen engulfing colloid in cases of de Quervain’s and palpation thyroiditis, both of which lack necrosis as does sarcoidosis. When necrosis is appreciated, the possibility of an infectious etiology should be considered and a careful search for organisms should be initiated. Unless an infectious agent is identified on routine or special staining, FNA is often not able to establish a specific definitive diagnosis and one is left with the rather generic diagnosis of “granulomatous thyroiditis.”
Histologic examination may provide a more specific diagnosis by revealing the folliculocentric nature of de Quervain’s thyroiditis with widespread granulomas that are centered on ruptured follicles. This contrasts with the more focal and haphazard arrangement of granulomas in infections and the interstitial positioning of granulomas in sarcoidosis. In contrast, palpation thyroiditis is also folliculocentric but focal and usually limited to single follicles with infiltration of the colloid by macrophages (Fig. 8.7, e-Figs. 8.12 and 8.13).
CHRONIC INFLAMMATION
Chronic lymphocytic thyroiditis is a very common disorder. The inflammation may be diffuse or focal. It may be primary, due to autoimmune disease, or it may be secondary to infection, medication, or radiation. It should be remembered that the mere presence of lymphoid tissue does not indicate an inflammatory lesion as lymphoid tissue may be found in association with branchial cleft-like cysts, ultimobranchial body remnants, papillary carcinomas, and other malignancies that have undergone
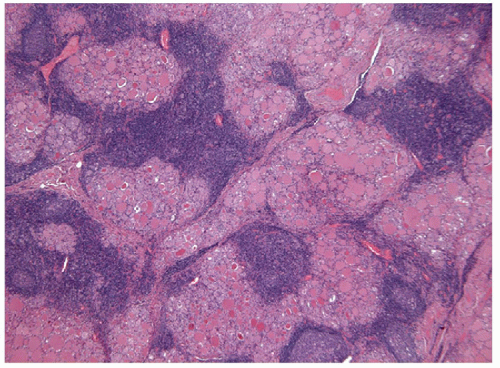
degeneration or necrosis. Thymic tissue within or around thyroid is a common source of difficulty but is usually distinguished by the presence of Hassall’s corpuscles and/or other epithelioid elements (Figs. 8.8 and 8.9). Therefore, the identification of chronic inflammation in a thyroid biopsy creates a complex diagnostic dilemma. The thyroid biopsy that contains chronic inflammatory cells represents one of the most difficult areas and must be carefully evaluated for the presence of associated pathologies. More importantly, the presence of inflammation in the thyroid results in reactive atypia and even dysplasia of adjacent follicular epithelium that can result in the erroneous diagnosis of papillary thyroid carcinoma [12].
degeneration or necrosis. Thymic tissue within or around thyroid is a common source of difficulty but is usually distinguished by the presence of Hassall’s corpuscles and/or other epithelioid elements (Figs. 8.8 and 8.9). Therefore, the identification of chronic inflammation in a thyroid biopsy creates a complex diagnostic dilemma. The thyroid biopsy that contains chronic inflammatory cells represents one of the most difficult areas and must be carefully evaluated for the presence of associated pathologies. More importantly, the presence of inflammation in the thyroid results in reactive atypia and even dysplasia of adjacent follicular epithelium that can result in the erroneous diagnosis of papillary thyroid carcinoma [12].
Autoimmune thyroiditis has been classified by many authors in various ways [9]. The commonest form is Hashimoto’s thyroiditis, a well-defined clinicopathologic syndrome consisting of goiter, hypothyroidism, and lymphocytic thyroiditis that is widely accepted to be of autoimmune etiology [13, 14]. Patients have antibodies to thyroglobulin and to thyroid peroxidase (formerly known as the “microsomal antigen”) [15]. Some patients also have antibodies to a colloid component other than thyroglobulin (“second colloid antigen”) and occasionally to thyroid hormones. Patients with this disorder are most often women with a female-male ratio of 10:1 and onset between 30 and 50 years of age. The thyroid typically becomes a diffuse, lobulated, asymmetrical, nontender goiter. Most patients with longstanding disease are hypothyroid. Occasionally, there is a transient episode
of hyperthyroidism known as “hashitoxicosis” early in the course of the disease. This has been attributed to release of stored hormone during tissue destruction or to stimulation by antibodies to the TSH receptor [15].
of hyperthyroidism known as “hashitoxicosis” early in the course of the disease. This has been attributed to release of stored hormone during tissue destruction or to stimulation by antibodies to the TSH receptor [15].
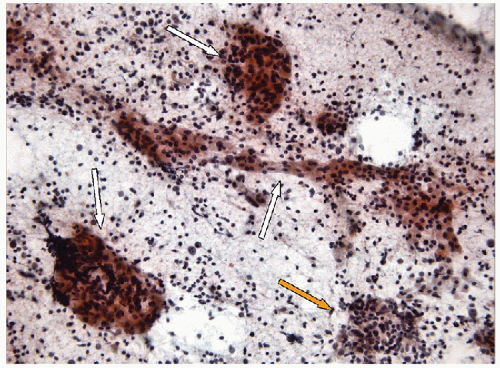
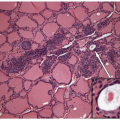
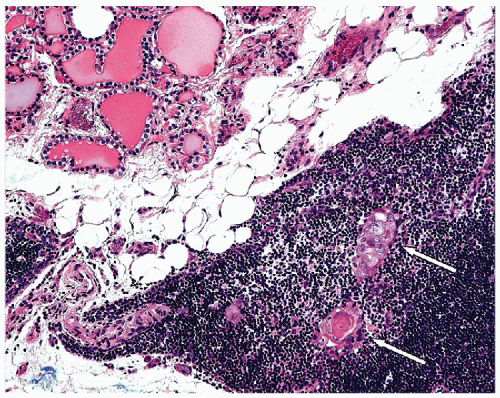 FIGURE 8.9 Intrathyroidal Thymus. The presence of the Hassall’s corpuscles (white arrows) in this intrathyroid lymphoid tissue confirms that it is ectopic thymic tissue (hematoxylin and eosin stain). |
Other forms of autoimmune chronic lymphocytic thyroiditis include a nongoitrous variant that may be associated with variable degrees of atrophy, the juvenile variant that has a tendency to be associated with hyperplasia, painless thyroiditis with hyperthyroidism, and postpartum thyroiditis. The distinction of these different forms of chronic thyroiditis is largely based on clinical and biochemical features. There are subtle morphological differences in some of the variants, but the main differences involve the presence and degree of oncocytic change and the presence and degree of fibrosis. In contrast to these autoimmune disorders, diffuse toxic goiter of autoimmune etiology, known as Graves’ disease, is usually associated with only mild focal inflammation and is therefore not discussed in this differential diagnosis, and the reader is referred to Chapter 9 (Papillary lesions) for a review of this subject.
Biopsy of the thyroid in patients with chronic lymphocytic thyroiditis usually results from the identification of a nodule. The presence of thyroid growth-stimulating immunoglobulins (TGI) in these patients and/or compensatory TSH excess due to tissue destruction, and hypothyroidism have been implicated in the development of hyperplastic nodules that present as discrete masses. A nodular appearance on ultrasound of cases of lymphocytic thyroiditis may be accentuated by the difference in echotexture between portions of the gland densely infiltrated with lymphoid cells
and the often fibrotic stroma, alternating with those portion of the gland that are relatively spared from the inflammation and epithelial rich.
and the often fibrotic stroma, alternating with those portion of the gland that are relatively spared from the inflammation and epithelial rich.
FNA of Lymphocytic Thyroiditis
In FNA samples, the hallmark of lymphocytic thyroiditis is the presence of a significant lymphoid population. Lymphoid cells may represent one of the following four conditions:
1. Aspiration of a chronic inflammatory infiltrate in the thyroid.
2. Hemodilution of the sample with peripheral blood.
3. Aspiration of a perithyroid lymph node.
4. Aspiration of a lymphoma.
In many cases, it is a simple task to recognize the lymphocytic infiltrate as a component of lymphocytic thyroiditis. However, when the lymphoid population is more scant, it may be difficult to determine if the lymphoid cells represent an inflammatory cell population or simply lymphocytes carried into a hemodiluted sample. This can be made more difficult in samples processed by liquid-based methods such as ThinPrep that employ erythrolysis, removing the red blood cells and leaving behind the leukocytes to generate a pseudo-inflammatory infiltrate. Lymphoid cells that normally circulate in the blood are essentially small lymphocytes; therefore, to confirm a true inflammatory infiltrate, one should look for centroblasts, plasma cells, tingible body macrophages, and germinal center fragments. The finding of mast cells or a significant number of eosinophils also suggests a true inflammatory infiltrate rather than contaminating blood.
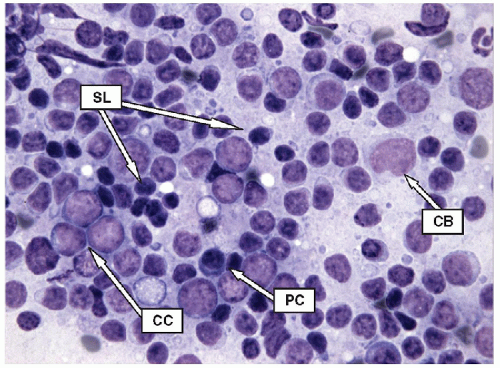
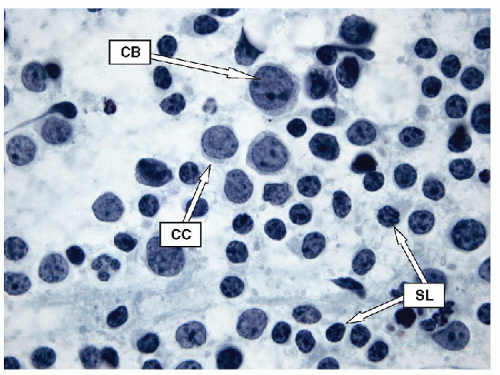
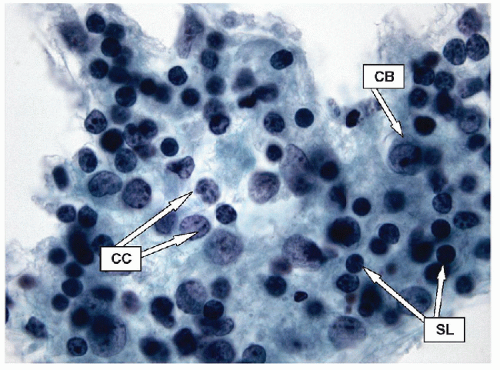
Lymphoid infiltrates are composed of very specific cells. The three lymphoid cells that are the foundation of most of inflammatory infiltrates are small lymphocytes, centrocytes, and centroblasts. The morphology of the lymphoid cells is often best appreciated on air-dried samples.
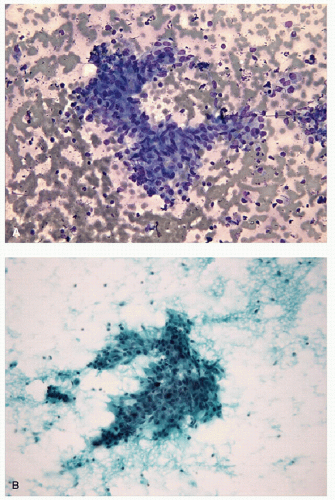
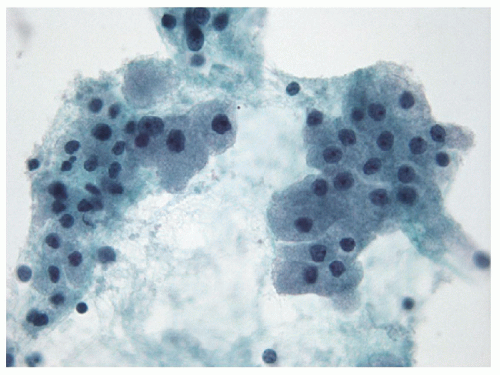
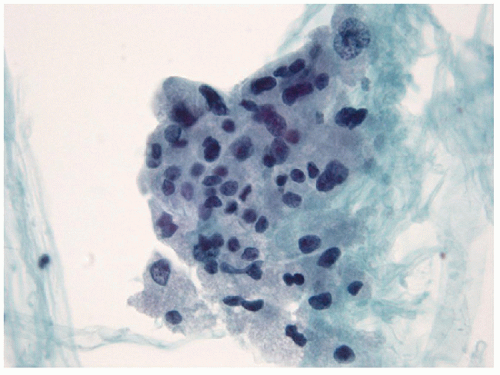
Small lymphocytes are smaller than a neutrophil and have scant cytoplasm with round, regular nuclei; dark chromatin; and lack nucleoli. Centrocytes, or small cleaved cells, are slightly larger than small lymphocytes and are characterized by cleaved nuclei containing clumped chromatin and inconspicuous nucleoli. Visualization of nuclear clefting is dependent on the orientation of the nucleus and will not be evident in all cells. Nuclear clefts are exaggerated by some degree in direct smears that are wet fixed, as the alcohol-induced shrinkage artifact tends to accentuate the nuclear irregularities. Centroblasts, or large noncleaved cells, are large lymphoid cells with a small quantity of cytoplasm and round nuclei with a fine chromatin pattern and one to three nucleoli that are often marginalized against the nuclear membrane (Figs. 8.10, 8.11, 8.12, e-Figs. 8.14 and 8.15).
The ratio of these lymphoid cells to one another is variable, and in some cases, small lymphocytes may dominate, but when secondary follicular structures develop, the proportion of centrocytes and centroblasts is increased and germinal center fragments may be appreciated in the aspirate (Fig. 8.13). The recognition of germinal center fragments is added by
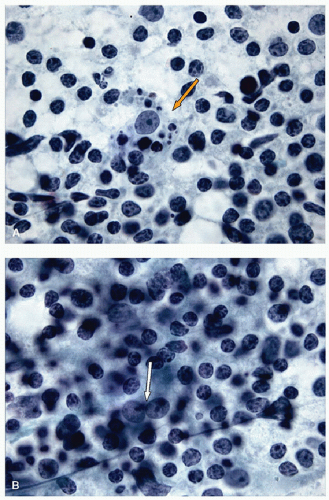
the identification of tingible body macrophages (Fig. 8.14A) and follicular dendritic cells (Fig. 8.14B), with enmeshed centroblasts and centrocytes. Plasma cells will be present in many cases, and other lymphoid cells such as immunoblasts may be seen.
the identification of tingible body macrophages (Fig. 8.14A) and follicular dendritic cells (Fig. 8.14B), with enmeshed centroblasts and centrocytes. Plasma cells will be present in many cases, and other lymphoid cells such as immunoblasts may be seen.
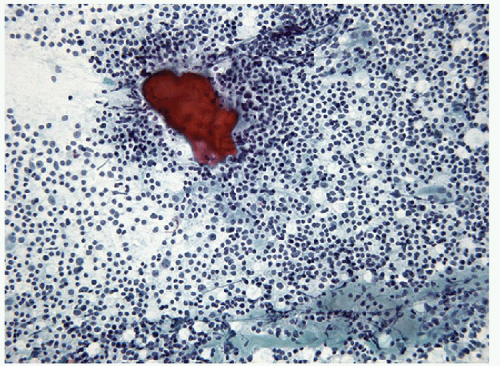
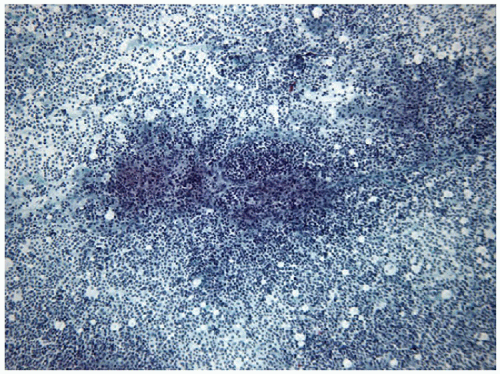 FIGURE 8.13 Lymphoid Germinal Center. When the lymphoid population shows florid follicular differentiation, numerous germinal center fragments may be found. Their cohesive nature makes them apparent at low-power examination but can also led to confusion with epithelial fragments, particularly when there is an abundance of centroblastic cells that may resemble oncocytic follicular cells. The clues to the correct diagnosis are the identification of the other lymphoid cells (small lymphocytes and centrocytes) in addition to finding of follicular dendritic cells and tingible body macrophages (Fig. 8.14) (direct smear, Papanicolaou stain). |
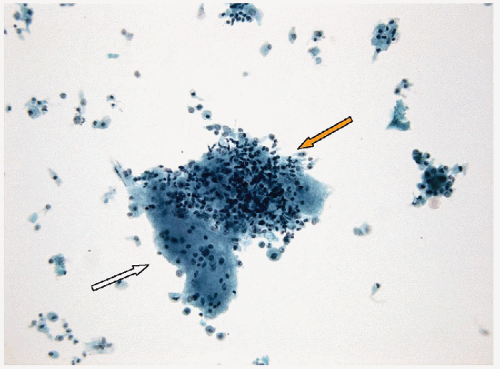
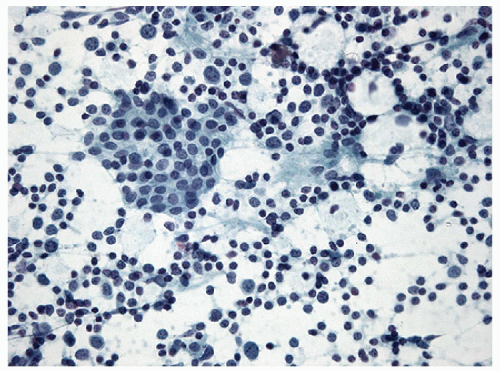 FIGURE 8.15 Bland follicular epithelium in lymphocytic thyroiditis. It may be impossible to distinguish lymphocytic thyroiditis from aspiration of a periglandular lymph node. Thus, to establish the diagnosis of lymphocytic thyroiditis, thyroid epithelium or colloid (Fig. 8.17) must be identified (direct smear, Papanicolaou stain). |
It can be very difficult or impossible to distinguish lymphoid tissue of a chronic inflammatory infiltrate from lymphoid tissue of a lymph node, as the component cells are the same in each. In histology, this is not a concern as the lymphoid infiltrate can be seen to be within the thyroid gland, or alternatively, the architecture of the nodal tissue (cortex, medulla, sinuses, and capsule) can be identified. However, in FNA samples, these anatomic findings are lacking, and therefore, an alternative approach must be applied to distinguish an inflammatory infiltrate within the thyroid gland from aspiration of a periglandular lymph node. In fact, the primary indicator is the presence of thyroid epithelium. Thus, to diagnose lymphocytic thyroiditis, the inflammatory infiltrate must be combined with follicular epithelium (Fig. 8.15). Care must be taken to ensure that germinal center fragments are not mistaken for epithelial groups (Fig. 8.16, e-Figs. 8.16, 8.17, 8.18, 8.19). Rarely, colloid may be present in the absence of follicular epithelium, and this too can serve as evidence of the needle entering the thyroid gland (Fig. 8.17). However, colloid depletion is common in lymphocytic thyroiditis, and infrequently colloid is present without epithelial cells.
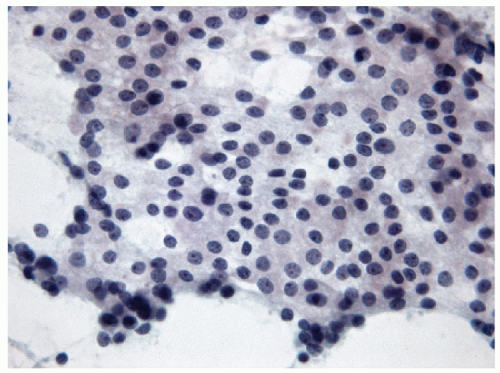
The follicular epithelium seen in lymphocytic thyroiditis is often bland; however, Hashimoto’s thyroiditis is one of the more commonly aspirated forms of lymphocytic thyroiditis, and oncocytic cells (also known as Hürthle cells) are frequently found. It is important to note that
while oncocytes are common in Hashimoto’s thyroiditis, the presence of oncocytes in an FNA does not indicate Hashimoto’s thyroiditis. Oncocytes are follicular epithelial cells that have undergone a metaplastic change characterized by an increase in cytoplasm that appears more vacuolated and granular with the granularity as a result of an increase in mitochondrial content. These mitochondria, due to their high protein content, take up more eosin and therefore not only is the cytoplasm granular but the granules are eosinophilic, a feature readily apparent on Pap stain material (Fig. 8.18). Oncocytes may also show significant cytoplasmic vacuolation and may begin to resemble macrophages (Fig. 8.19). The nuclei of oncocytes are significantly enlarged, round, and regular with a prominent, central eosinophilic nucleolus.
while oncocytes are common in Hashimoto’s thyroiditis, the presence of oncocytes in an FNA does not indicate Hashimoto’s thyroiditis. Oncocytes are follicular epithelial cells that have undergone a metaplastic change characterized by an increase in cytoplasm that appears more vacuolated and granular with the granularity as a result of an increase in mitochondrial content. These mitochondria, due to their high protein content, take up more eosin and therefore not only is the cytoplasm granular but the granules are eosinophilic, a feature readily apparent on Pap stain material (Fig. 8.18). Oncocytes may also show significant cytoplasmic vacuolation and may begin to resemble macrophages (Fig. 8.19). The nuclei of oncocytes are significantly enlarged, round, and regular with a prominent, central eosinophilic nucleolus.
The epithelium may be relatively unscathed in thyroiditis and appear as simple monolayered sheets. However, with colloid depletion, the epithelium may begin to take on the architecture of syncytial aggregates. Microfollicles should not be present in significant numbers, and transgressing vessel should be absent. Nuclear changes can occur in both the regular follicular epithelial cells and oncocytic cells of lymphocytic thyroiditis. There is often an irregular alteration of the chromatin that appears more pale and granular. Nuclear membrane irregularities up to and including nuclear grooving is a common finding. Nucleoli increase in prominence in regular follicular epithelium and are extremely prominent in oncocytic cells (Fig. 8.20). Clearly, these changes mimic to some degree
the changes of papillary carcinoma, and in the presence of lymphocytic thyroiditis, an extra degree of caution is warranted with a demand for all features of papillary carcinoma to be present before issuing a definitive diagnosis. Well-formed intranuclear inclusions are not a feature of lymphocytic thyroiditis and can be used to distinguish papillary carcinoma from thyroiditis.
the changes of papillary carcinoma, and in the presence of lymphocytic thyroiditis, an extra degree of caution is warranted with a demand for all features of papillary carcinoma to be present before issuing a definitive diagnosis. Well-formed intranuclear inclusions are not a feature of lymphocytic thyroiditis and can be used to distinguish papillary carcinoma from thyroiditis.
Histologic Biopsy of Lymphocytic Thyroiditis
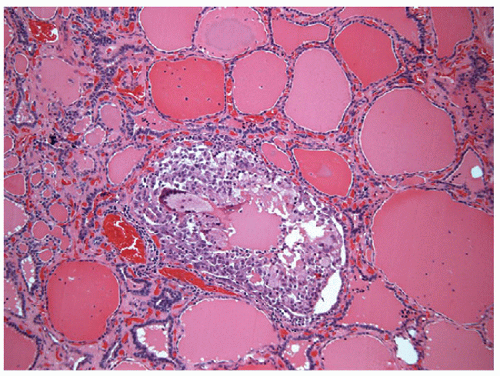
Similar to the FNA appearance of lymphocytic thyroiditis, in histologic sections, the tissue is diffusely infiltrated by a lymphoid population, including small lymphocytes, centrocytes, centroblasts, plasma cells, and occasionally immunoblasts. Lymphoid follicles may form and develop germinal centers (Figs. 8.21 and 8.22, e-Figs. 8.20 and 8.21).
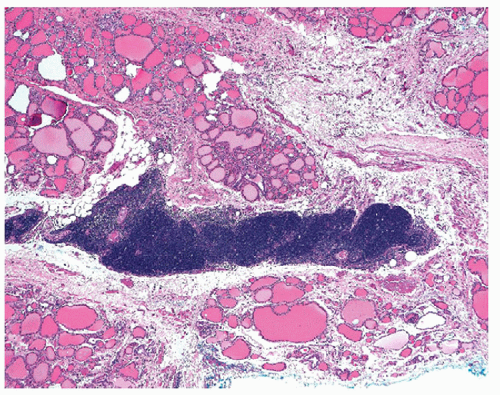
The glandular epithelium exhibits variable degrees of damage. Residual thyroid follicles either are atrophic, with sparse colloid and flattened epithelium, or exhibit oxyphilic metaplasia [16] (Figs. 8.23 and 8.24, e-Figs. 8.22 and 8.23). As in FNA specimens, the follicular epithelial cells may also exhibit marked cytologic atypia that can be characterized by irregular nuclear membranes, grooves, and even clearing of nucleoplasm (Fig. 8.25). These features that in the face of inflammation are considered reactive, may reflect dysplasia [12], and mimic papillary carcinoma [9, 17]. Areas of squamous metaplasia may be found [18].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree