Chapter Outline
Principles of flow cytometric immunophenotyping 330
Multicolour flow cytometric immunophenotyping 331
Methods 332
Detection of membrane antigens 333
Detection of surface immunoglobulin 334
Detection of intracellular antigens 335
Simultaneous detection of intracellular and membrane antigens 336
Acquisition and data analysis 336
Immunological markers in acute leukaemia 336
Immunological markers in chronic lymphoproliferative disorders 342
Minimal residual disease detection by flow cytometry 345
HIV monitoring 346
Antibody panels 346
Acknowledgement
The authors would like to acknowledge Kathryn Moss for her contribution to the HIV monitoring section.
Principles of flow cytometric immunophenotyping
The primary methods for immunophenotyping in the haematological setting are immunocytochemistry (described in previous editions of this book) and flow cytometry.
In this chapter we shall focus on flow cytometry and its application to the diagnosis and treatment response of haematological neoplasms.
Flow cytometry is a laser-based technology, which is able to identify and quantify cell populations. Cells in suspension are manipulated into a stream of fluid where single cells are interrogated by an electronic detection system. Flow cytometry has many applications and is now routinely used in the diagnosis of haematological malignancies. It can provide quantitative data on a number of cell parameters for a large number of cells, in the region of thousands of cells per second, making it a powerful diagnostic tool.
A flow cytometer has five main components:
- •
Lasers providing a monochromatic light
- •
A flow cell with a liquid stream (sheath fluid), which carries and aligns the cells so that they pass in single file through the laser beam
- •
Optical systems and filters regulating the light signals
- •
Photomultiplier detectors (PMTs) that generate data on forward light scatter (FSC) (which provides an approximation of cell size) and side light scatter (SSC) (which relates to cell complexity), as well as converting fluorescence signals from light into electrical signals that are processed by a computer
- •
A computer for analysis of the signals.
Flow cytometry relies on the application of fluorescent conjugates or fluorochromes attached to a monoclonal antibody (McAb) that has specific affinity for an antigen on the cell surface, in the cytoplasm or in the nucleus. Each fluorochrome has a characteristic peak excitation and emission wavelength, and the emission spectra often overlap, requiring compensation. Consequently, the combination of fluorescent labels that can be used depends on the wavelength of the laser used to excite the fluorochromes and on the detectors available.
Immunophenotyping of haematological neoplasms by flow cytometry involves the labelling of white blood cells or their precursors with fluorescent-labelled monoclonal antibodies directed against target cellular proteins or antigens. When appropriate antibodies – usually combined as ‘panels’ – are chosen, the cell lineage and stage of differentiation of leukaemic or lymphoma cells can be determined, allowing the leukaemia/lymphoma to be classified. The accurate classification of haematological neoplasms is critical for the clinician’s choice of treatment and for overall prognosis.
In summary, immunophenotyping by flow cytometry facilitates:
- •
The identification and quantification of cell populations within a sample
- •
The differentiation of normal from abnormal cells
- •
The differentiation of reactive from neoplastic cells
- •
The identification of the differentiation or maturation stage of a cell population
- •
The quantification of tumour infiltration.
However it is the interpretation of the data provided by the described techniques which poses the greatest challenge and the reliable diagnosis of leukaemia relies on the following:
- •
Knowledge of physical characteristics/antigen expression on normal cells
- •
The ability to distinguish between different patterns of expression of antigens
- •
The ability to identify aberrant antigen expression
- •
The identification of a robust leukaemia-associated immunophenotype (LAIP).
These will be discussed in more detail in the sections below.
Multicolour flow cytometric immunophenotyping
Flow cytometry is now a well-established technique for identifying immunophenotypic profiles in haematological neoplasms. Historically three- or four-colour antibody flow cytometry panels were used, and these panels remain of great utility. However, developments in instrumentation, namely an increase in both the number of lasers and in the availability of fluorochrome conjugates, have facilitated the design of eight- to ten-colour antibody panels. Such panels can provide data on up to 12 to 14 cellular parameters simultaneously, allowing the accurate identification and quantification of normal and abnormal cell populations in bone marrow and peripheral blood. The application of such extensive panels has provided a greater understanding of normal maturation stages in haemopoiesis and specifically the antigens expressed or down regulated at the different stages of this process. This provides a template to which abnormal cell populations can be compared. This new knowledge has increased the accuracy of diagnosis and allowed the identification of very small cell populations and subpopulations.
The advances in flow cytometry instrumentation and reagents mentioned above have facilitated the routine use of eight- to ten-colour antibody panels in the clinical laboratory setting. The use of such multicolour panels is generally what is referred to as multicolour or multiparameter flow cytometry (MFC). There are many advantages associated with the use of MFC but the corresponding pitfalls must also be considered.
Advantages of MFC:
- •
Increased accuracy. Using large numbers of fluorochromes is associated with an exponential increase in the information obtained from a single combination of antibodies in the same tube, permitting a more reliable identification.
- •
Smaller sample size. Increased number of antibodies per tube means fewer tubes and less sample needed but allows acquisition of more cellular events resulting in smaller coefficients of variation and increased data precision. This is of particular relevance to paucicellular samples such as cerebrospinal fluid (CSF) and fine needle aspirates (FNA) and also paediatric samples.
- •
Cost effectiveness. Less usage of repeating backbone or gating antibodies.
- •
Increased efficiency. Less time is required for sample processing and acquisition.
- •
Increased sensitivity for minimal residual disease monitoring.
Disadvantages:
- •
Increased complexity of compensation. Inaccurate compensation is probably the main source of erroneous data in MFC. This can be solved by applying compensation matrices but this requires expertise.
- •
Challenges of antibody panel validation. It is crucial to run fluorescence minus one controls for all new antibody combinations and to check for stearic hindrance between antibodies used to label antigens that are in close proximity on the cell.
- •
Tandem dye conjugate issues. Tandem dyes are conjugates of two fluorochromes, but this can lead to problems in resonance excitation transfer if exposed to light. Ideally a compensation matrix should be performed for each new tandem dye conjugate lot.
- •
Increased need for expertise in data analysis and interpretation.
- •
Human error associated with pipetting a high number of antibodies into a single tube. This can be overcome by preparing in-house McAb cocktails, which have been shown be stable for up to 4 weeks.
Many advances have been made in addressing the issues outlined above and groups such as the EuroFlow Consortium, the Multicolour Immunophenotyping Group UK (MIG UK), European Group for the Immunological Characterization of Leukemias (EGIL), Harmonemia and others have made great advances in standardising MFC protocols. Software tools such as Kaluza ( www.beckmancoulter.com ) and Infinicyt ( www.infinicyt.com ) have been developed to aid in data analysis and interpretation ( Fig. 16-1 ). Currently there is a move towards commercially available kits that include lyophilised or freeze-dried antibody ‘cocktails’ to overcome the issues associated with tandem conjugates. Some kits also include the use of standardised instrument set up facilitating the use of software analysis using libraries to compare normal with neoplastic cases .

Methods
Sample preparation
Flow cytometry can be performed on any sample where cells are available in suspension. Peripheral blood, bone marrow, CSF, ascitic fluid, pleural fluid and FNAs all provide such suspensions, requiring only red cell lysis and staining with appropriate antibody panels, as described below. Lymph node, spleen, liver and bone marrow trephine biopsy specimens (not in fixative) can also be processed for flow cytometry following tissue disaggregation to obtain a cell suspension.
Detection of membrane antigens
For all the methods described below the tube(s) should be clearly labelled with the name of the patient, type of specimen, laboratory number and the combination of fluorochrome-conjugated McAbs or multicolour cocktail used prior to any staining.
- 1.
Stain–Lyse–Wash method:
- •
Pipette 100 μl of the specimen into a round-bottom tube. Note : if the cell count of a specimen is known to be high, dilute this accordingly, aiming for a final cell concentration of 1 to 2 × 10 6 per tube.
- •
Add the appropriate volume of McAb combination or multicolour cocktail.
- •
Incubate in the dark at room temperature for 15 min.
- •
Add 1 ml of ammonium chloride-based lysing solution and incubate for 10 min at room temperature in the dark.
- •
Centrifuge for 5 min at 300 g and discard the supernatant. Repeat this step.
- •
Resuspend the cells in 0.2–0.5 ml of sheath fluid solution (e.g. Isoton, www.beckmancoulter.com ) and acquire data on the flow cytometer without delay.
- •
- 2.
Stain–Lyse–No Wash method:
This method utilises the same procedure as the previous methodology, but after incubation with the lysing solution, the sample data are acquired on the flow cytometer. This method is ideal for samples with few cells because it minimises cell loss during the centrifugation of the washing step.
- 3.
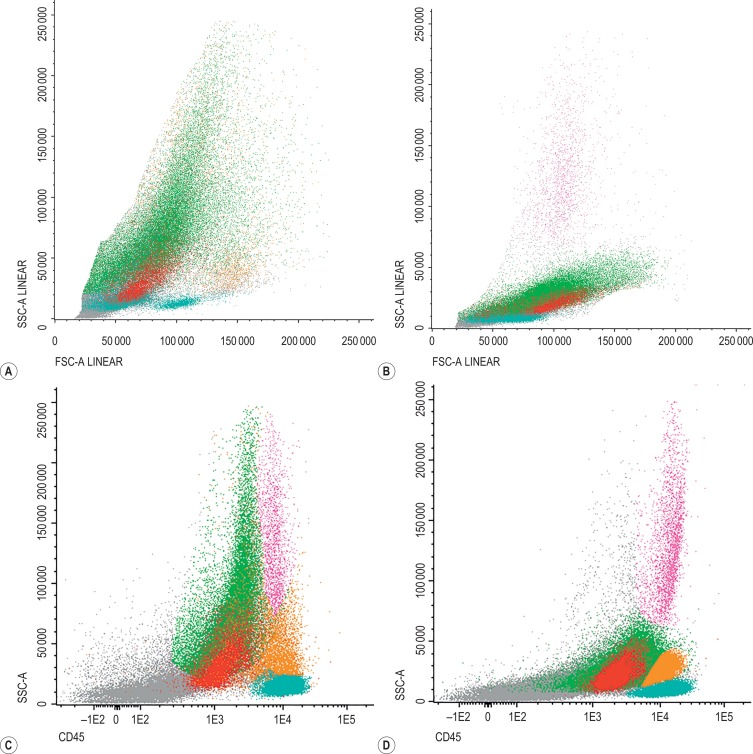
Lyse–Stain–Wash method ( Fig. 16-2 ):

Figure 16-2
Normal peripheral blood flow cytometry dot plots showing a mixture of polyclonal B cells and T lymphocytes.
A, Gate on FSC/SSC lymphocytes (orange). B, CD19-positive lymphocytes (blue) and CD3-positive lymphocytes (green). The orange population are not B or T lymphocytes, and are likely to be NK cells. C, Polyclonal B lymphocytes gated on CD19; some cells express kappa and some lambda. D, CD4-positive T cells (green, top cluster). E, CD8-positive T cells (grey).
This method of bulk specimen lysis is used for minimal residual disease (MRD) monitoring in order to facilitate and enrich the acquisition of leucocytes.
- •
Pipette 5–10 ml of peripheral blood or bone marrow into a tube and add the same volume of ammonium chloride lysing solution, mix gently and incubate for 10 min at room temperature.
- •
Centrifuge for 5 min at 300 g, discard supernatant after centrifugation and resuspend the cell pellet in 10 ml of phosphate-buffered saline (PBS) containing sodium azide and bovine serum albumin (PBS–Azide–BSA).
- •
Repeat this washing procedure. If the cell pellet still contains red cells the lysing step can be repeated.
- •
Finally resuspend the cell pellet in 10 ml of PBS–Azide–BSA and perform a white cell count.
- •
Aliquot a volume of cell suspension containing 10 × 10 6 cells/tube.
- •
Add appropriate volume of McAb, and incubate in the dark.
- •
Repeat washing procedure and resuspend in 0.2–0.5 ml of sheath fluid.
- •
Acquire data on the flow cytometer without delay.
- •
Detection of surface immunoglobulin
Lymphoproliferative disorders (LPD) of mature B cells are distinguished from their normal counterparts by identifying two main types of phenotypic abnormalities: surface immunoglobulin light chain restriction and aberrant B-cell antigen expression.
Staining for surface immunoglobulins requires some extra steps in the sample preparation. This is to avoid any nonspecific binding due either to cytophilic antibodies binding to Fc receptors (monocytes and some lymphocytes) or to the binding of antibodies to cell membranes of damaged or dying cells.
This nonspecific staining can be avoided by washing the sample with an isotonic solution prior to staining for surface immunoglobulins. Nonspecific staining can also be minimised by incubating the cells with serum prior to staining.
Finally, some B-cell LPDs such as chronic lymphocytic leukaemia (CLL) may express surface immunoglobulins very weakly and it is preferable to use polyclonal antibodies (PcAb) to detect light chain restriction in these cases ( Fig. 16-3 ).
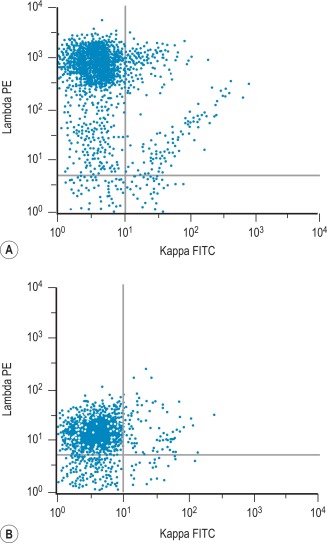
Two methods are suitable for detecting surface membrane immunoglobulin (SmIg) of blood and bone marrow cells, according to whether a PBS wash or a lysing procedure is used as the first step.
Method 1: wash–stain–lyse–wash
- •
Pipette 100 μl of the specimen into a round-bottom tube.
- •
Add 2 ml of PBS–Azide–BSA kept at 37 °C and centrifuge for 5 min at 300 g. Using a Pasteur pipette, carefully discard the supernatant.
- •
Repeat the procedure and resuspend the specimen in 50 μl of PBS–Azide–BSA.
- •
Add the appropriate McAb/PcAb combination, (e.g. anti-kappa and anti-lambda, CD19, CD45 or any multicolour cocktail).
- •
Incubate at room temperature in the dark for 15 min.
- •
Add 1 ml of ammonium chloride-based lysing solution and incubate for 10 min at room temperature.
- •
Add 1 ml of PBS–Azide–BSA, centrifuge for 5 min at 2000 revolutions per minute (rpm), and discard the supernatant. Repeat this step.
- •
Resuspend cells in 0.2–0.5 ml of sheath fluid solution (e.g. Isoton).
- •
Acquire data on a flow cytometer without delay.
Method 2: lyse–stain–wash
- •
Pipette 100 μl of the specimen into a round bottomed tube.
- •
Add 2 ml of ammonium chloride lysing solution, incubate for 10 min at room temperature, and wash twice in PBS–Azide–BSA as above.
- •
Add the appropriate volume of McAb/PcAb combination, according to the manufacturer’s instructions.
- •
Incubate in the dark for 15 min at room temperature. Add 2 ml of PBS–Azide–BSA, centrifuge for 5 min at 300 g and discard the supernatant. Repeat this step.
- •
Resuspend cells in 0.2–0.5 ml of sheath fluid (e.g. Isoton) and acquire data on a flow cytometer without delay.
Detection of intracellular antigens
This method is applied to identify antigens expressed within the cell (i.e. in the cytoplasm or nucleus); for example, intracellular immunoglobulin, myeloperoxidase (MPO), lysozyme, CD3, CD79a, BCL2, terminal deoxynucleotidyl transferase (TdT) and Ki67.
Several kits containing solutions to fix and permeabilise cells to detect cytoplasmic or nuclear antigens are commercially available. Their reliability and consistency for detecting particular nuclear and cytoplasmic (c) antigens may vary. These reagents can have some effect on the light scatter pattern as cells have a tendency to shrink after fixation ( Fig. 16-4 ).
The kits contain two solutions: solution A is the fixing agent based on a paraformaldehyde solution and solution B is a lysing agent based on a combination of a lysing solution and a detergent.
The methods below follow the manufacturer’s kit instructions for the “Fix and Perm” kit (Invitrogen) ( www.thermofisher.com ).
Method
- •
Pipette 100 μl of the specimen into a round-bottom tube.
- •
Add 100 μl of solution A (fixative) and incubate at room temperature for 15 min.
- •
Wash twice in PBS–Azide–BSA by centrifuging for 5 min at 300 g.
- •
Add 100 μl of solution B (lysing agent) and the appropriate amount of fluorochrome-conjugated McAb or antibody cocktail.
- •
Incubate at room temperature in the dark for 15 min.
- •
Wash twice in PBS–Azide–BSA, centrifuging for 5 min at 300 g.
- •
Resuspend in 0.2–0.5 ml of sheath fluid solution (e.g. Isoton).
- •
Acquire data on a flow cytometer without delay.
Simultaneous detection of intracellular and membrane antigens
The first step in simultaneous detection of intracellular and membrane antigens is to stain for membrane antigens followed by cytoplasmic or nuclear antigen staining ( Fig. 16-5 ).
- •
Pipette 100 μl of specimen into a round-bottom tube.
- •
Add the appropriate fluorochrome-conjugated McAbs or cocktail to detect the membrane antigen.
- •
Incubate at room temperature in the dark for 15 min.
- •
Without washing, add 100 μl of solution A (fixative).
- •
Incubate at room temperature in the dark for 15 min.
- •
Continue with the steps described previously for intracellular antigen detection.
Acquisition and data analysis
Data acquisition: According to the 2014 British Committee for Standards in Haematology (BSCH) guidelines all data files should have clear unique identification. The number of events to be acquired depends on the desired sensitivity, sample volume and cellularity.
For diagnostic LPD samples, acquisition of data representing 50 000 lymphocytes is recommended but acquisition of between 500 000 and 1 × 10 6 total events is required for MRD detection.
Fluidics stability should be monitored by the inclusion of a time versus fluorescence plot, and doublets should be discriminated on a FSC-H (height) versus FSC-A (area) analysis.
Data analysis: there are multiple software options available for the interpretation of FSC files. It is important to exclude debris and doublets at the beginning of the analysis. Analysis templates allow populations of interest to be identified by applying sequential gating i.e. light scatter and CD45 followed by lineage-specific antigens (see Fig. 16-1 ).
Immunological markers in acute leukaemia
Immunophenotyping by MFC is a well-established and widely used technique for identification and characterisation of leukemic cells in acute leukaemia. According to the World Health Organisation (WHO) classification of Tumours of Haematopoietic and Lymphoid Tissues, MFC should be integrated with morphology, cytogenetics and molecular studies at presentation and follow up. It is important to note that even if immunophenotyping is significantly more sensitive, specific and objective than morphological assessment, the latter remains the gold standard criterion for establishing the blast percentage both at presentation and post-treatment.
In the diagnosis of acute leukaemia by immunophenotyping, the most commonly used approach at least in Europe is a two-step process. First a screening panel including lineage markers and immature markers (CD34/TdT, HLA-DR) allows the classification of most cases into the broad categories of myeloid (MPO positive), B cell (CD19, CD79a, cCD22) or T cell (cCD3) and, less frequently, mixed phenotype acute leukaemia (MPAL). There are numerous panels described in the literature; recently the EuroFlow Consortium has published thoroughly validated eight-colour panels for the diagnosis of haematological malignancies. Their screening tube for the categorisation of acute leukaemia – acute leukaemia orientation tube (ALOT) – includes MPO, CD34, CD19 and CD79a, CD7, cCD3 and SmCD3.
Once the lineage of the blasts has been established, a secondary panel of antibodies is necessary to define the blast differentiation and maturation stage ( Table 16-1 ).
| Panel | Markers in Acute Leukaemia |
|---|---|
| First-line screening | CD34, TdT, HLA-DR, MPO, CD19, CD79a, CD22, CD3 |
| Second-line B ALL | CD10, cμ, CD20, CD58, CD38 |
| Second-line T ALL | CD1a, CD2, CD3, CD4, CD5, CD7, CD8, TCR |
| Second-line myeloid | CD13, CD33, CD117, CD16, CD11b |
| Second-line monocytic | CD14, CD64, CD36, CD4, CD300e |
| Second-line erythroid | CD105, CD36, CD71, CD235a |
| Second-line megakaryocyte | CD61, CD41, CD42 |
In acute myeloid leukaemia (AML) further staining will identify mainly granulocytic and monocytic commitment and more infrequently erythroid or megakaryocytic lineage. Whilst in acute lymphoblastic leukaemia (ALL) the extended phenotyping will further sub-classify the disease on the basis of B- and T-cell differentiation or maturation stage.
This subsequent phenotyping is crucial in defining the LAIP. More than 95% of cases of ALL and over 80% of AML show an aberrant antigen expression pattern or LAIP as defined by over/under expression of markers, asynchronous antigen expression and/or cross-lineage expression. These LAIPs are extremely useful for following response to treatment and MRD levels. In the case of ALL, they normally permit the unequivocal detection of abnormal blasts amongst normal lymphoid precursors. In AML, the LAIP’s sensitivity and specificity will depend on how frequently the aberrancy is found in the abnormal blasts and how much it deviates from a normal myeloid maturation pattern.
Multiparameter flow cytometry in acute lymphoblastic leukaemia
The vast majority of cases of ALL express TdT and/or CD34. For B-lineage assignment, strong expression of at least two B-cell associated markers is required (CD19, CD79a, cCD22, CD20) whereas cCD3 expression is sufficient as a T-cell lineage marker. Of note is that almost all T-ALL co-express CD7, but CD7 alone is not sufficient for T-cell lineage assignment (around 20% of AML co-express CD7).
From MFC studies performed in normal bone marrow we have learnt in detail the normal maturation pattern of the B-cell compartment.
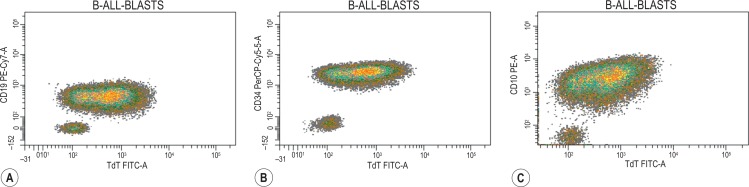
Thus a normal CD34 + early B-cell precursor acquires initially TdT, CD79a, cCD22, CD10 and CD19. Then as CD45 expression is gained, there is sequential loss of CD34/TdT, downregulation of CD10 and gain of CD20. At a later stage, cytoplasmic μ chains are expressed prior to the precursor becoming an immature B cell with SmIgM expression. The immunophenotypic classification of B-cell precursor ALL (B-ALL) is based on this physiological sequence: pro-B or early precursor ALL (blast positive for TdT, CD19, CD79a, cCD22); common ALL (positive for CD10 but not cμ); pre-B-ALL (blasts express cμ); very rarely B-ALL expresses SmIg. Abnormal precursors of B-ALLs will show a maturation arrest in one of the stages described above plus asynchronous expression of some antigens (for example co-expression of CD20 and TdT or CD22 positivity in CD10-negative blasts), over- or under-expression of certain antigens (CD58, CD38) and cross-lineage expression (usually expression of myeloid antigens such as CD66c, CD33, CD15). Over 95% of B-ALL have a phenotype that differs from that of normal B-cell precursors, and this difference allows the unequivocal identification of these blasts at relapse. Although it is recognised that, after treatment, leukaemias can exhibit an immunophenotype switch, it is still possible to discriminate between normal and abnormal B-cell precursors in most post-treatment bone marrows by identifying any deviation from normal B-cell development ( Fig. 16-6 ).
There is also a degree of correlation between these immunological subtypes and molecular genetics and prognosis. For example the majority of B-ALL with t(1;19)(q23;p13.3) display a pre-B phenotype (CD34 −, CD9 +, CD19 +, CD10 +, cμ +); the presence of t(4;11) is associated with a pro-B phenotype and usually with CD15 cross-reactivity. A Ph-positive B-ALL in general has a common ALL phenotype with expression of CD66c and CD25.
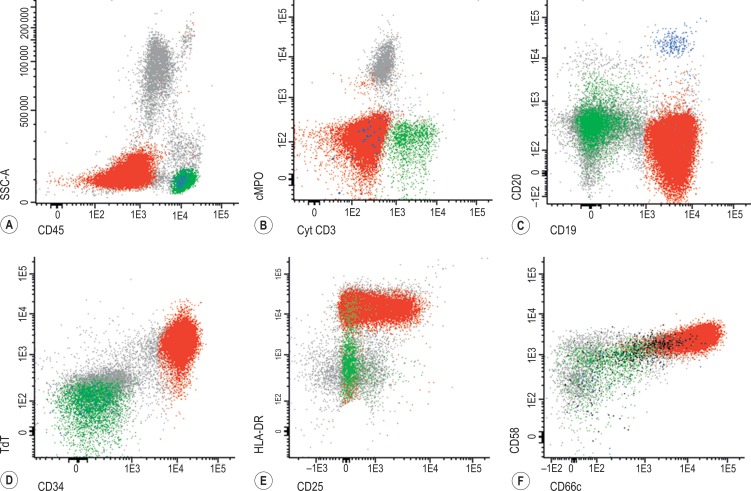
Similarly to B-ALL, precursor T-ALL has been further classified into different subgroups that mimic normal T-cell differentiation: pro-T (positive for cCD3 and CD7 with absence of other T-cell associated antigens), pre-T (additional expression of CD2 and/or CD5), cortical (CD1a positive) and mature T-ALL (sCD3 +, CD4 + or CD8 +). More recently a new subgroup of leukaemias derived from an early T-cell progenitor (ETP) and associated with dismal outcome with standard therapy has been described. These ETP-ALL characteristically lack CD1a and CD8 and show dim expression of CD5 together with expression of at least one myeloid or stem cell marker ( Fig. 16-7 ).
In contrast to B-ALL, the association between immunophenotypic profile and genotype is not so clear, the exception being overexpression of the following oncogenes: LYL1, TLX1 (HOX11) and TAL1 associated with a pro-T, early cortical and late cortical phenotype respectively.
Multiparameter flow cytometry in acute myeloid leukaemias
AML is a more complex and heterogeneous disease than ALL, which partly explains the less standardised approach to the immunophenotyping of this disease. In contrast to their lymphoid counterparts, myeloid leukaemias usually show more than one blastic population by MFC. Normal myeloid cells differentiate to neutrophils, monocytes, erythrocytes and megakaryocytes, and also, less often, to basophils, eosinophils, mast cells and dendritic cells. AML can show differentiation to any of these myeloid cell subsets. Most of the panels for identification of myeloid blasts include the following markers of immaturity: CD34, HLA-DR and CD117. The characteristic normal neutrophil maturation pattern of CD13 (biphasic expression), CD16 and CD11b expression is lost in AML. Acute promyelocytic leukaemia (APL) has a characteristic light scatter profile (high SCC due to the granularity) with negativity for CD34 and HLA-DR and positivity for CD117, CD33 (homogeneous) and CD13 (weak), which is similar to the promyelocyte stage except for abnormal lack of CD15 expression.
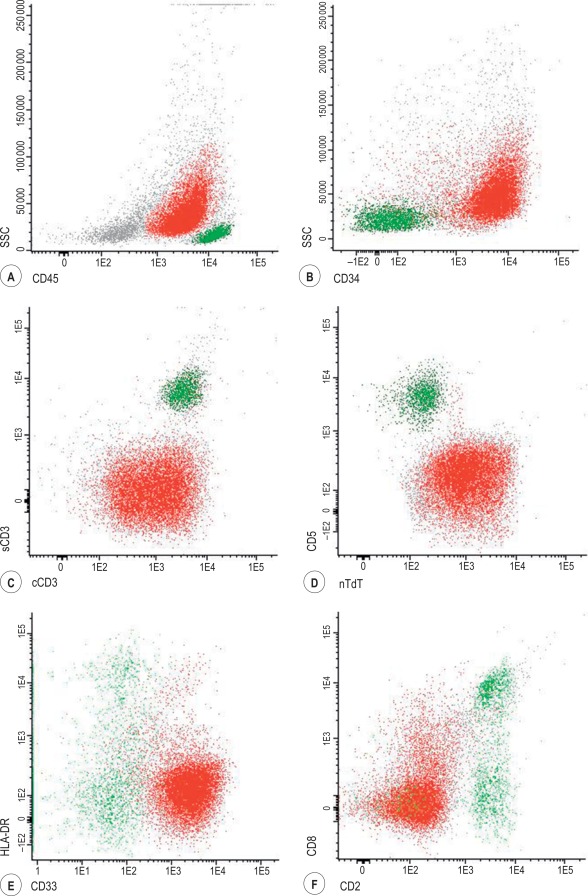
Normal monocytic differentiation includes sequential acquisition of CD4, CD64, CD36, CD14 and CD300e. AML with monocytic differentiation shows an increased monocytic compartment with a variable proportion of the cells displaying either an immature-monoblastic phenotype (CD64 + CD14 −), asynchronous antigen expression (CD64 + CD14 − CD300e +), abnormal loss of antigens normally expressed by monocytes (i.e. CD13, HLA-DR) and/or cross-antigen expression (CD56 in particular) ( Fig. 16-8 ).