The reconstructive approach to breast defects is challenging, as the resultant glandular tissue deficiency not only needs to be replaced or reconstructed following the “tissue-like” principle but also necessitates a contour deformity restoration. Conservative breast surgery with postoperative irradiation has replaced modified radical mastectomy as the preferred treatment for early invasive breast cancer. Breast-conservation surgery with quadrantectomy or lumpectomy procedures creates primary or delayed secondary (postradiation) defects of the conical breast shape with or without involvement, and subsequent repositioning, of the nipple–areola complex (NAC).
The objective in breast-conservation surgery is to establish disease-free surgical margins while the reconstructive part will tend to preserve as much glandular tissue as possible allowing primary, tension-free closure with an acceptable aesthetic appearance and satisfying breast symmetry. When primary closure without tissue distortion is not feasible, mobilization of adjacent or locoregional tissue is warranted.
Up to 30% of patients are dissatisfied with the aesthetic result after partial mastectomy with irradiation.1 Distortion, retraction, and mammary volume changes together with NAC repositioning extenuating asymmetry all have a profound impact on the aesthetic appearance of the breast. Therefore, we prefer to perform immediate reconstruction whenever it is indicated and feasible, as operating on irradiated breasts has high complication rates with frequently poor esthetic results. During immediate reconstruction, the breast can be manipulated prior to radiation. This potentially decreases complications and improves the outcome. As for any conservative breast therapy, tumors up to 3 cm in diameter are generally considered safe for quadrantectomy associated with postoperative radiotherapy. On the other hand, any immediate partial reconstruction should be delayed if the surgeon is uncertain about the margins or tumor extension despite the preoperative radiologic assessment. A delayed immediate reconstruction can still be performed within a few days after the definitive margins become known.2
The combination of wide glandular breast tissue excision with subsequent immediate reconstruction has been considered a decisive stage in the evolution of breast cancer surgery. This approach allows not only wider tumor resection to obtain safer margins but is also advantageous in handling the healthy resultant glandular tissue to achieve better aesthetic outcomes. However, if there is any doubt about the margins of resection or tumor extension, the procedure should be delayed and the delayed stage can still be done a few days later when the definitive margins are known. Many surgeons have suggested incorporating a reduction mammaplasty procedure during the tumor resection in hypertrophic breasts.3-7
The size and location of the breast defect and the ratio of breast volume to resection volume are fundamental determinants to choose the methods of reconstruction. One of the relative contraindications for rearranging the breast parenchyma is a large tumor to breast ratio, with smaller breasts requiring different, more challenging reconstruction approaches. An extensive resection in a smaller breast necessitates the recruitment of nonglandular tissue, and different flaps are available to fill the tissue deficiency.
If the defect involves less than 30% volume deficiency in a relatively moderate to large breast, rearrangement surgery using dermoglandular or glandular flaps based on breast tissue can be performed as long as a tension-free closure can be obtained and the final outcome will be aesthetically acceptable.1,3,8-10 Skin rotation flaps or lateral thoracic axial skin flaps can be used for small lateral defects, but most of those flaps become unavailable when axillary lymph node dissection is part of the oncologic procedure.
The versatility of the pedicled perforator flaps available around the mammary gland is definitely an advantage to obtain acceptable results. Defects involving up to 30% of the breast volume are usually considered for reconstruction with pedicled flaps as well are defects involving an unacceptable aesthetic outcome with breast asymmetry. Our algorithm to recruit distant tissue for partial breast reconstruction is to turn first to pedicled flaps and then to free flaps with completion mastectomy if pedicled flaps are insufficient, unavailable, or potentially compromised due to previous surgery.
Free tissue transfer for immediate partial breast reconstruction is not recommended because it eliminates the use of the flap for a local recurrence or a new tumor in the contralateral breast. It is, as mentioned before, better to complete the mastectomy followed by the autologous reconstruction.
The thoracodorsal, intercostal, or superior epigastric pedicle axes can be used to elevate perforator flaps based on the desired location within the breast. The flaps can be raised in the axillary and back region as well as from the anterior thoracic and upper abdominal areas.
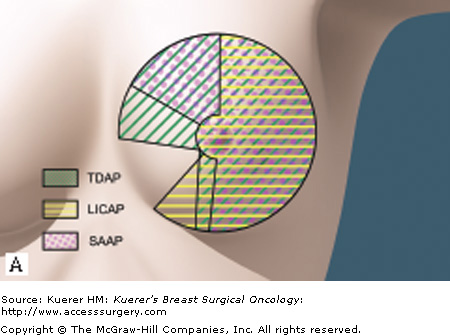
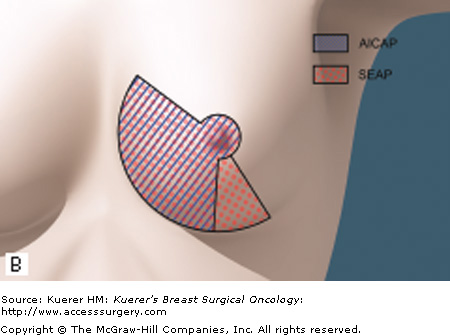
Depending on the tumor location and the extent of the resultant defect, a variety of pedicled perforator flaps are available (Fig. 77-1). The most commonly used pedicled perforator flaps used to reconstruct considerable breast defects are described next.
The thoracodorsal artery perforator (TDAP) flap, suitable for superior defects, is based on the perforators from the descending or the horizontal branches of the thoracodorsal vessels (Fig. 77-1).1,11
Intercostal artery perforator (ICAP) flaps are nourished through the perforators arising from the costal groove. The lateral intercostal artery perforator (LICAP) flap is suitable to restore defects in the lateral quadrant or small defects in the medial quadrant.12 The flap provides a short pedicle, but the flap can be rotated 180 degrees without torsion of the perforator. The LICAP flap is a good alternative to the TDAP flap for lateral and inferior breast defects. The anterior intercostal artery perforator (AICAP) flap is supplied by perforators originating from the muscular segment. The AICAP, on the other hand, is an option to reconstruct defects in the medial quadrant.
The serratus anterior artery perforator (SAAP) flap is based on the connection between the serratus branch of the thoracodorsal artery and the intercostal perforators. However, these perforators can be found in only 21% of the cases, as shown by our anatomic study.12
The superior epigastric artery perforator (SEAP) flap is based on perforators originating from the superficial or the deep branch of the superior epigastric artery. This flap can be used to reconstruct tissue deficiencies in the medial quadrant.13
The latissimus dorsi (LD) myocutaneous flap has an excellent blood supply and provides enough bulk tissue for filling glandular defects and pliable skin to substitute for skin deficiencies. However, the donor-site morbidity consists of seroma formation and the presence of conspicuous scars on the back with loss of some back muscular function.14 Avoiding a scar on the back can be achieved by harvesting the LD without a skin paddle through the lateral breast incision used for the oncologic excision or approaching the defect using a mini-LD flap.15
The perforator will determine the flap viability, and careful preoperative mapping of those perforators will influence the flap design and its location. This mapping reduces the operating time and complication rate significantly while giving the surgeon a reference point of how to design the flap to address the defect. Perforators are tracked using a Doppler ultrasound device. Perforators in the thoracodorsal region are sought out in a region 8 to 10 cm below the axillary crease and within 5 cm of the anterior border of the LD muscle.16,17 To avoid the background signal from the main thoracodorsal pedicle, the patient is positioned for perforator mapping in the same position as for the surgical procedure, which is a lateral position with 90 degrees of shoulder abduction and 90 degrees of elbow flexion.
When the signal is heard in front of the anterior border of the LD muscle, a direct perforator (a septal perforator) of the thoracodorsal pedicle can be expected. This direct perforator can be found in 55% of the cases, which makes the dissection easier and quicker, but the disadvantage is a shorter pedicle and the flap may not be able to reach more distant defects.16,17 This septal perforator arises from the thoracodorsal vessels at a high level and it may be damaged by axillary lymph node surgery. Therefore, it is mandatory to check the vascular course of this perforator before committing to raise the flap based on it.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree