Organ
Surgery
Radiation therapy
Chemotherapy
Kidney
Late proteinuria possible with single kidney
Renal failure if bilateral
A single normal kidney usually provides good function
Bilateral injury can lead to renal dysfunction
Glomerular and tubular injury (cisplatin, ifosfamide)
Ureter
Urinary diversion may be necessary
Fibrosis rare: may occur after high-dose or intraoperative irradiation
Bladder
Dysfunction due to partial or total organ loss may occur
Fibrosis, focal ulceration possible
Loss of capacity if large fraction of kidney irradiated
Contracture and functional loss possible after hemorrhagic cystitis (cyclophosphamide, ifosfamide)
Secondary bladder cancer (cyclophosphamide, ifosfamide)
Urethra
Stricture requiring dilation possible
Vagina
Dysfunction with partial or total organ loss
Fibrosis, ulceration, fistula, and maldevelopment possible
Uterus
Dysfunction due to partial or total organ loss may occur
Maldevelopment and fibrosis possible
Prostate
Dysfunction due to partial or total organ loss may occur
Loss of glandular function is possible
15.2 Pathophysiology
15.2.1 Normal Organ Development
Normal fetal development of the GU structures begins with successive development of pronephric, mesonephric, and metanephric tubules around the third, sixth, and twelfth weeks of gestation in the embryo, respectively. After 12 weeks, the urinary bladder has developed and separated from the rectum. The prostate and testes in boys, and the ovaries and uterus in girls, are also formed at approximately 12 weeks gestation. The vagina develops somewhat later. After birth, prostatic and vaginal-uterine growth proceeds very slowly until adolescence, when the organs enlarge during pubertal growth.
15.2.2 Organ Damage and Developmental Problems
The multimodal treatment of cancers with surgery, RT, and chemotherapy may cause structural or functional impairment of the GU organs and tissues. Table 15.1 summarizes late organ damage to the GU system.
15.2.3 Surgery
Removal of a paired structure, such as a kidney, is not usually associated with subsequent functional impairment, unless the remaining organ has been damaged from either therapy or the tumor. (In fact, the remaining kidney may undergo compensatory hypertrophy [1].) Conversely, the removal of a nonpaired structure such as the bladder, prostate, or uterus can produce severe and life-long impairment, such as urinary incontinence or infertility. Urinary diversion after total cystectomy for bladder sarcoma in childhood can be associated with infection and eventual renal impairment from pyelonephritis, ureteral, or stomal obstruction or both [2, 3]. In addition, ureterocolic diversion and bladder augmentation have occasionally been associated with early development of colon cancer [4]. It is also seen with reconstruction of the neurogenic bladder. The hypothesis is that the irritation of urine on bowel mucosa can be carcinogenic. An interesting canine study showed hyperplasia at the anastomoses [5]. Continent diversion techniques, using repeated catheterization of an indwelling ileal or colonic bladder, may provide better results [6]. Continent diversion is accomplished by creating a reservoir usually of bowel that the patient empties periodically via catheterization throughout the day.
15.2.4 Radiation Therapy
Organ injury following RT is generally classified as acute (occurring during or soon after therapy) and late (occurring months to years following therapy). Whereas the acute effects are usually transient, late effects are usually progressive. Acutely, RT frequently causes irritation of the mucosa of the bladder and urethra (causing cystitis and urethritis) or of the vagina and vulva (causing pruritus, discomfort, and candidal overgrowth). These symptoms usually occur after approximately 20 Gy of radiation. Since almost all children receiving GU tract irradiation are also receiving chemotherapy, normal acute tissue toxicities are seen earlier than they would be seen without concurrent chemotherapy. Typically, cystitis occurs after 3–4 weeks of radiation, but it can occur after 2 weeks with concurrent therapy. Occasionally, some morbidity is seen after doses as low as 8–10 Gy. Acute injury of the kidney, prostate, and uterus is generally not clinically apparent. The later effects of RT are dose dependent and due to progressive vascular and parenchymal cell damage, generally leading to scarring, fibrosis, and sometimes necrosis. Malignant tumors can be seen following irradiation, generally occurring a minimum of 4–5 years following completion of radiation [7]. A discussion of the late effects for each organ follows.
15.2.4.1 Kidney
Irradiation appears to cause renal dysfunction secondary to tubular damage. Nephropathy generally occurs when doses in excess of 20–25 Gy are delivered to both kidneys [8]. When chemotherapeutic agents are used as well, lower doses (10–15 Gy) can cause significant injury. In general, if only a portion (less than one-half to one-third) of the kidney is irradiated, then higher doses may be tolerated without demonstrable functional deficits. The sequelae may be more prominent and occur at lower doses in infants. Hyper-renin hypertension can also occur secondary to radiation-induced renal artery narrowing. This phenomenon has been noted most often in children (especially infants) and should be distinguished from other types of renal radiation-induced hypertension. Irradiation to the remaining kidney following nephrectomy may hinder the normal hypertrophic response.
15.2.4.2 Bladder, Ureter, and Urethra
Radiation can induce inflammation and fibrosis and cause dysfunction due to a reduction in bladder capacity and contractility. Although it is not certain, the underlying etiology seems to be radiation-induced vascular ischemia of the muscular wall [9–11]. The risk of developing bladder dysfunction is related to both the radiation dose and the percentage of the bladder wall irradiated [9, 12]. In data compiled in adults, it is clear that a small volume of the bladder can tolerate fairly high doses of radiation [9, 12]. (Radiation for prostate or bladder cancer in adults routinely results in irradiation of portions of the bladder with 60–70 Gy.) However, high doses may cause focal injury to part of the bladder wall, resulting in bleeding and stone formation [13–17]. It is believed that stone formation is associated with bacteriuria, which can occur after damage to the bladder. When the entire bladder is irradiated, doses of >50 Gy may result in severe contraction and secondary whole organ dysfunction. Consequently, both the radiation dose and volume of organ irradiated must be considered when assessing the risk of injury. Similarly, scarring and fibrosis can occur in the urethra and ureter, causing dysfunction of these structures [17–19]. Doses less than 50 Gy may slow or hamper the full development of the bladder, due to lesser degrees of fibrosis (Fig. 15.1).
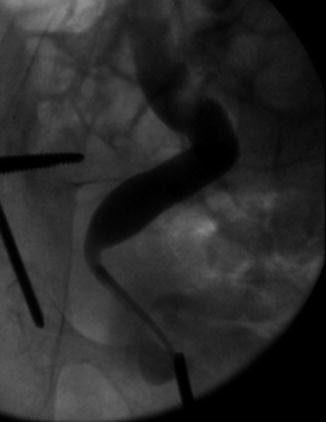

Fig. 15.1
Stenosis (arrow) in ureter in a 13-year-old who received 41.4 Gy pelvic radiotherapy at age 2 for rhabdomyosarcoma
15.2.4.3 Prostate, Uterus, and Vagina
The exact pathophysiology of radiation-induced late effects is less well defined than for the kidneys and bladder. When irradiated to high doses in an adult, the vagina undergoes loss of the epithelium and slow reepithelialization over a 2-year period. It is likely that the prominent late effects in the uterus and vagina are related to progressive fibrosis, leading to loss of function [20]. The prostate may lose its secretory capacity, resulting in ejaculatory dysfunction.
15.2.5 Chemotherapy
The major chemotherapeutic agents that cause damage to the GU tract are the platinum compounds (cisplatin and carboplatin) and alkylating agents (cyclophosphamide and ifosfamide). The toxicity of the antimetabolite, methotrexate, is largely preventable and reversible [21].
15.2.5.1 Kidney
Cisplatin causes both glomerular and renal tubular damage, with wasting of divalent and monovalent cations (magnesium, calcium, and potassium). Cumulative doses as low as 450 mg/m2 are associated with some renal toxicity. Proximal tubular damage predominates, especially in a low chloride environment [22]. Elevated serum concentration of creatinine and decreased glomerular filtration rate (GFR) with azotemia also occur and are dose and age related. These effects vary both in severity and chronicity [22, 23]. Prior cisplatin administration may delay the renal clearance of methotrexate [24]. Carboplatin has a better renal toxicity profile than cisplatin. The replacement of cisplatin with carboplatin in standard regimens is being tested in adult studies, but in some instances, the tumor efficacy is not equivalent. Carboplatin at doses used in stem cell transplantation has been associated with renal dysfunction [25]. At present, the routine use of amifostine to protect renal integrity and function is not indicated [26, 27]. Its routine use as a renal protector is being investigated in clinical trials; a COG study did not show this approach to be effective in germ cell tumors [28].
The acute effects of ifosfamide, seen most commonly in young (<3-year-old) children or those with prior renal dysfunction or nephrectomy, include renal tubular damage with hyperphosphaturia, glycosuria, and aminoaciduria, followed by the inability to acidify the urine – the so-called Fanconi syndrome [29–31]. Hypophosphatemia and acidosis can lead to inhibition of statural growth, as well as to bone deformity (renal rickets) in prepubertal and pubertal children. Glomerular damage may accompany the tubular damage, leading to diminished GFR, with increased serum creatinine and azotemia. Median doses of 54 g/m2 have been reported to cause progressive glomerular toxicity [32], and chronic glomerular and tubular toxicity has been reported [32, 33]. Risk factors include total ifosfamide dose [33], prior cisplatin administration [30, 31], and age. Recovery of renal function is possible over time [34].
Methotrexate toxicity is usually acute and reversible. The drug and its metabolites precipitate in the renal tubules. Adequate hydration and leucovorin administration will prevent most renal damage. (Doses up to 12 g/m2 can be given safely if the appropriate precautions are taken [35].)
15.2.5.2 Bladder
Bladder damage, including hemorrhagic cystitis, fibrosis, and occasional bladder shrinkage, can occur following chronic administration of alkylating agents such as cyclophosphamide [36] and ifosfamide [37]. The metabolic by-products of these drugs include acrolein (of the same chemical class as the aniline dyes), which is excreted in the urine and irritates the bladder mucosa. This leads to exposure of submucosal blood vessels and subsequent bleeding [38]. Fortunately, drug-induced hemorrhagic cystitis and related fibrosis can nearly always be prevented by increased hydration during drug administration and the concomitant administration of intravenous or oral mercaptoethane sulfonate (MESNA). MESNA serves as a chemical sponge that binds the metabolites, thereby inactivating them and preventing their toxic action on the urothelium. Cyclophosphamide has also been associated with the induction of bladder tumors [39]. The interaction between RT and chemotherapy and their effects on hemorrhagic cystitis are discussed in Sect. 15.3.
Radiation may interact with a number of chemotherapeutic agents in an additive or synergistic fashion. The most notable example for the organs of the GU tract, particularly the kidneys, is the interaction between radiation and the antibiotics, actinomycin-D and doxorubicin [40]. There is a significant enhancement of the radiation effects when the agents are given concurrently, but this may also occur when the modalities are used sequentially. Radiation may also interact with cyclophosphamide, increasing the severity and chronicity of hemorrhagic cystitis. Therefore, great care is necessary when evaluating patients who have received or will receive RT to fields that include the kidney or bladder, if those patients also have received or will receive chemotherapy. This is of particular importance in patients who have nephrectomy or a fused or ectopic kidney, where the functional renal tissue may have been purposefully or inadvertently irradiated. It is critical in these cases to have precise information on the definition of the radiation portals.
Conditioning regimens for bone marrow transplantation (BMT) often include chemotherapy and total body irradiation. Data is emerging on late renal toxicity, such as hematuria and renal insufficiency [41, 42]. Renal biopsy reveals both parenchymal and vascular glomerular changes [43]. This data is from two published sources of the effects on ALL (n = 44) and neuroblastoma (n = 15) patients. Most patients received twice daily radiation (interfraction time of 4–6 h) to total doses equaling 12–14 Gy. Another group has shown that many patients with hematologic malignancies already come into the BMT process with a decreased (but normal) GFR when compared to those undergoing the transplant for nonmalignant diseases [43]. This is likely related to the intensive systemic therapy that they have already received. Approximately 1 year after transplant, there is a significant decrease in GFR which stabilizes or slightly improves at over the ensuing 5 years. TBI-containing regimens show a greater decrease in the GFR. Hemorrhagic cystitis after bone marrow transplantation may also be associated with BK polyomavirus [44].
15.3 Clinical Manifestations
Table 15.2 summarizes the available data for the late genitourinary effects in childhood survivors of cancer. Each organ system is discussed below.
Table 15.2
Incidence of late genitourinary effects following treatment for childhood cancer
Author | Tumor | Therapy | N | Follow-up (yrs) | Endpoint studied | Result |
|---|---|---|---|---|---|---|
Barrera | Wilms’ | Nephrectomy | 16 | >13 | Mild proteinuria tubular function DBP >90 | 2/16 |
25 % | ||||||
Ritchey | Wilms’: unilateral | Nephrectomy | 5,368 | >10 | Renal failure | 0.28 % |
Makipernaa | Wilms’ | Nephrectomy + ipsilateral RT (20–40 Gy) | 30 | 19 | HTN BUN/creatinine | 17 % normal |
Paulino | Wilms’ | Nephrectomy + RT (12–40 Gy) + CT | 42 | 15 | Serum BUN and creatinine HTN | Elevated in 1 patient 7 % |
Thomas | Wilms’ | Nephrectomy + RT (15–44 Gy) | 24 | 13 | Low-grade renal failure and UTIs | 4 % |
Wikstad | Wilms | Nephrectomy + ipsilateral RT + contralateral kidney RT | 22 | 13 | GFR | 82 % compared to normal controls; stable over time |
(5–15 Gy) | BP | Normal | ||||
Raney | Bladder or prostate sarcoma | Surgery + CT+/− RT (25–55 Gy) | 109 | 8 | Bladder dysfunction | 25 % |
Urinary diversion | 50 % | |||||
HTN | 1 % | |||||
Elevated BUN/Cr | 6 % | |||||
Hematuria (intact bladder vs. diversion) | 20 % vs. 39 % | |||||
Bacteriuria (intact bladder vs. diversion) | 8 % vs. 35 % | |||||
Abnormal renal imaging (intact bladder vs. diversion) | 20 % vs. 37 % | |||||
Heyn | Paratesticular rhabdomyosarcoma | Surgery + RT (16–58 Gy) + CT | 86 | >4 | Ejaculatory dysfunction | 7 % |
Normal blood pressure | 96 % | |||||
Normal BUN and creatinine | 100 % | |||||
Ureteral obstruction | 3 % | |||||
Hemorrhagic cystitis | 34 % | |||||
Normal bladder function | 100 % | |||||
Hale | Germ cell tumors | Surgery +/− RT (20–40 Gy) +/− CT | 73 | 11 | Neurogenic bladder | 15 % |
Hemorrhagic cystitis | 13 % | |||||
Recurrent UTI | 9 % | |||||
Ureteral/urethral stenosis | 3 % | |||||
Bladder atrophy | 75 % | |||||
Hydronephrosis | 100 % | |||||
Ritchey | Retroperitoneal tumors | Surgery + EBRT (18–50 Gy) + IORT (10–25 Gy) | 4 | 2 | Bilateral hydronephrosis | 50 % |
Renal artery stenosis | 4 % | |||||
Renal atrophy | 25 % | |||||
Stea | Pelvic sarcomas | CT + RT (55–60 Gy) +/−IORT +/− BMT (with TBI) | 23 | 2 | Vaginal stenosis | 4 % |
Cystitis | 4 % | |||||
Fistulas (IORT) | 100 % | |||||
Tarbell | ALL | BMT (with TBI) | 28 | 2 | Renal dysfunction | 32 % |
Guinan | Leukemia | BMT (with TBI) | 115 | 3 | GFR | 41 % |
Esiashvili | Leukemia | BMT (with TBI) | 60 | Elevated Cr | Acute: 45 % | |
Delayed:25 % | ||||||
Guinan | Neuroblastoma | BMT | 11 | Renal dysfunction | 64 % |
15.3.1 Kidney
15.3.1.1 Surgery
Unilateral nephrectomy in childhood results in contralateral hyperplasia [45, 46]. Normal kidney function is usually seen following resection of one of the two kidneys [47, 48]. Normal function can continue with as little as one-third of one kidney remaining. Radiation in moderate doses (14–15 Gy) to the remaining kidney may decrease the amount of hyperplasia that otherwise would have taken place [49, 50].
15.3.1.2 Radiation
Acute radiation nephropathy is an extremely uncommon occurrence, requiring greater than 30–40 Gy to the kidney. Subacute radiation nephropathy, characterized by hypertension and a decreased GFR, may occur 6–8 weeks to several months after doses equal to or greater than 15 Gy of radiation to both kidneys.
Significant late renal dysfunction occurs following radiation doses >20 Gy [51]. In children, even lower doses (5–20 Gy) can cause renal dysfunction. If a significant volume of the renal tissue is left unirradiated, the damage may not be clinically significant, although regional dysfunction within the irradiated portions of the kidney can be demonstrated. However, if all or the majority of the patient’s renal tissue is irradiated, clinical renal dysfunction will result.
The more frequent use of three-dimensional and four-dimensional RT planning is prompting investigations into the volume-based analysis of clinical damage. The first of these studies suggests that over 2 years low-grade nephrotoxicity may be associated with the volume of kidney receiving 20 Gy or more [52]. The volume of kidney within the high-dose RT envelope may also be reduced by a better understanding of the individual motion of the organ using sophisticated computed tomography during the treatment planning and frequent (sometimes daily) verification of the set up during the treatment course [53, 54].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree