Abstract
Cancer patients are surviving their disease in increasing numbers, and options to preserve their reproductive health exist for both males and females. Coupling the need of cancer patients with the existing or emerging options from the laboratory is critical to the overall quality of life for survivors. These same fertility interventions also may be applied to men and women facing fertility-threatening nononcologic disease or treatments. Guidance to clinicians is provided by Society guidelines and professional organizations. While not all investigational technologies are available at all locations, and treatment strategies for each disease are constantly changing, the guidance in this chapter provides the most comprehensive series of tools enabling actionable clinician knowledge. The goal of any fertility intervention is to ensure that the primary disease burden is eliminated and that reproductive health is maintained or preserved. These goals are largely possible.
Keywords
Oncofertility, radiation, chemotherapy, iatrogenic, ovary, testis, sperm, oocyte
Introduction
Since the 7th edition of this textbook, there have been major advances in fertility preservation, including the following:
- ◆
Fertility preservation has seen a large expansion into nononcologic indications.
- ◆
Egg cryopreservation is now considered the standard of care.
- ◆
Approximately 95 live births have been reported from ovarian tissue cryopreservation and transplantation.
- ◆
Formalized male fertility preservation programs are emerging globally, which offer testicular tissue cryopreservation.
- ◆
Multiple international professional societies have issued guidelines focused on fertility preservation.
Oncofertility—a discipline that merges oncology with fertility—has moved rapidly from the realm of individual champions, who provide high-level but autonomous care for addressing the fertility needs of cancer patients, to an integrated program that has become the standard of care in many institutions. The necessity of oncofertility, and its progress, has accelerated in parallel to the many advances in oncologic care, including earlier diagnostics and the emergence of targeted biologic therapeutics, methods to reduce radiation dose and field, and localized surgical procedures. With these advances, cancer patients are surviving their disease in increasing numbers and converting what was once a mortal diagnosis to a chronic illness, and, in some cases, a curable disease. The data are particularly compelling for pediatric cancer survivors; a patient treated for cancer between the years 1975 and 1979 had a 55% likelihood of surviving 5 years. In children diagnosed between 2005 and 2011 (the most recent interval for which we have data) that number has risen to 83%. By 2020, it is estimated that 1 in 750 adults will be a survivor of childhood cancer. Although these statistics are encouraging, they now also raise tangible concerns about the health of cancer survivors as they age. These concerns are largely related to the off-target effects of the cancer treatments themselves. Cancer survivors are at significant increased risk of second neoplasias, cardiovascular disease, and death at an early age. A major complication of cancer treatment is compromised reproductive function, which ranges from the destruction of gametes to the loss of pituitary and gonadal hormone production. It is in this setting that oncofertility is working to provide information about the iatrogenic effects of drugs on reproductive organs and to develop strategies that will preserve and restore biological function for cancer survivors.
Advances in the oncology arena are being paralleled by those in reproductive medicine and science; together, the oncofertility field and the broader field of fertility preservation are continuously improving and expanding the fertility preservation repertoire for men, women, and children in need. Since the topic of fertility preservation was first included in the 7th edition of this textbook, we have witnessed major advances in this area. New populations and communities that can benefit from fertility preservation have been identified and engaged. These include younger and younger individuals with cancer; those with nononcologic conditions whose disease or treatment may also threaten fertility; individuals with differences of sex determination; and transgender individuals.
From a clinical perspective, techniques that were once perhaps considered an impossibility are now a reality. For example, in the United States, the American Society for Reproductive Medicine (ASRM) removed the “experimental” label from mature oocyte cryopreservation in 2012, and now females can freeze eggs rather than embryos as standard of care. This change greatly reduces anxiety for patients who needed to find a sperm donor to create embryos; this issue was of significant concern for many newly diagnosed cancer patients. In addition to advances in traditional assisted reproductive technologies (ART), the success of investigational clinical technologies is also increasing. Approximately 95 live births have been reported in the literature following ovarian tissue cryopreservation ( ) and transplantation, and this is likely an underestimation. Remarkably, one ovarian tissue transplant was performed in an adolescent who had not yet transitioned through puberty, and the transplant was able to restore endocrine function. In another case, ovarian tissue that was harvested in a young child (13 years, 11 months) was fully functional and able to produce a live, healthy offspring when transplanted into the same individual more than a decade later. We have also witnessed the success of the first series of human uterine transplants, which creates tangible hope that cancer survivors who require a surrogate to support a pregnancy will now be able to carry their own offspring to term. For males, fertility preservation options are often considered simpler than for females because postpubertal males can bank semen relatively easily, and the navigation of young male patients to sperm banks is relatively straightforward. However, the options for prepubertal males are significantly lacking. The recent emergence of formalized male oncofertility programs around the world is facilitating the development of investigational techniques, such as testicular tissue cryopreservation.
From a basic science perspective, progress is being made on a daily basis to bridge experimental and standard fertility preservation technologies. Protocols continue to emerge from basic research laboratories that isolate follicles for growth ex vivo with the goal of creating high-quality in vitro-derived eggs. Human eggs and nonhuman primate embryos have been created from these culture systems, but more research is necessary to improve the efficiency and to understand the impact that long-term culture has on egg quality. Tissue-specific bioprosthetics, including male and female reproductive gonads and tracts, as well as patient-derived induced pluripotent stem (iPS) cell derivatives to populate these tissues, represent the cutting-edge of bench research, with early proof-of-principle evidence that will continue to emerge over the coming years.
The field of fertility preservation is mobilized now more than ever. Professional and clinical societies have moved with alacrity to issue guidance to healthcare teams about the emerging fertility preservation options for their patients. In addition, comprehensive registries are being established to track long-term fertility and reproductive health outcomes in cancer survivors. It is clear that oncofertility is now a global mission. Hundreds of sites worldwide are invested in sharing resources, methodologies, and other experiences in the field. Such global collaborations not only increase knowledge of fertility-threatening conditions and treatments in other countries, but also provide critical insights concerning broad cultural issues related to reproduction and fertility, as well as their impact on fertility preservation and oncofertility. By reducing duplicative efforts, proactively addressing barriers and challenges, and raising awareness worldwide, the field of fertility preservation has accelerated the pace of research and rapidly translated it to clinical care.
The goal of this chapter is to provide a comprehensive overview of the need, the current clinical methods, and the emerging future techniques of fertility preservation.
Fertility Preservation in Both Nononcologic and Oncologic Conditions
- ◆
In some cases, cancer itself may negatively impact fertility.
- ◆
A growing list of nononcologic conditions are associated with compromised fertility, including autoimmune diseases, hematologic conditions, metabolic disorders, gender and sex diversity, and postsurgical etiologies.
- ◆
Cancer treatments, such as surgery, radiation, and chemotherapy can negatively impact reproductive function in both males and females.
- ◆
In addition to infertility or sterility, cancer treatments may result in other adverse outcomes including depression, psychological disorders, and sexual dysfunction.
- ◆
Patient-specific variables (i.e., age, genetics, medical history, baseline fertility, and treatment regimen) will dictate how an individual’s reproductive potential will be affected by cancer treatment.
Reproductive Dysfunction As a Result of Nononcologic Conditions or Their Treatment
Although the predominant indication for fertility preservation is to avoid the iatrogenic effect of cancer treatments (chemotherapy, radiotherapy, surgery) on fertility, there is a growing list of nononcologic conditions where fertility preservation should be considered due to the underlying pathology of the disease itself or the effect of specific treatment regimens ( Table 33.1 ). In fact, approximately 15% of ovarian tissue cryopreservation cases performed through the Oncofertility Consortium’s National Physician Cooperative were done for nononcologic indications (unpublished data). For example, rheumatologic and autoimmune diseases, such as systemic lupus erythematosus, are treated with highly gonadotoxic agents. Nononcologic hematologic disorders, such as thalassemia major, sickle cell anemia, aplastic anemia, Fanconi anemia, and myeloproliferative diseases, do not inherently compromise reproductive function, but they are treated with bone marrow transplantation or hematopoietic stem cell transplant. These treatments require exposure to alkylating chemotherapy and radiation, which can significantly damage the ovarian reserve, or the number of primordial follicles in the ovary, which dictates a female’s reproductive lifespan.
| Autoimmune Diseases | Transplantation and Transfusion-Related Disorders | Genetic Conditions | Metabolic Conditions | Gender and Sex Diversity | Postsurgical Etiologies/Other |
|---|---|---|---|---|---|
|
|
|
|
|
|
Several genetic conditions can also impact fertility. For example, females with Turner syndrome—who have either partial or complete absence of one X chromosome—are at risk of premature ovarian failure. In mosaic Turner syndrome, an X chromosome is lost during embryonic development so that some proportion of the body’s cells contains two normal X chromosomes. Rare cases of pregnancy have been reported in women with mosaic Turner syndrome, and recent work suggests that women with mosaic Turner syndrome may be candidates for fertility preservation by either tissue cryopreservation (in prepubertal girls) or hormone stimulation followed by oocyte cryopreservation (in postpubertal girls and women). Spontaneous puberty and normal gonadotropin and estrogen levels were found to be predictive for the presence of mature follicles in girls with Turner mosaicism, and several case reports have described fertility preservation efforts in this population. Men with Klinefelter syndrome (XXY) have normal testes development up to puberty, but then develop hypergonadotrophic hypogonadism with degeneration of the seminiferous tubules and almost total loss of sperm production. Small areas of the tubules may still produce viable sperm that are recoverable by surgical extraction procedures. Finally, females with BRCA mutations that confer an increased risk for ovarian cancer may undergo prophylactic oophorectomy and opt for fertility preservation at that time.
Surgical complications may also threaten fertility due to the disruption of tissue, the formation of adhesions, and parenchymal damage. In boys, cryptorchidism, even after orchiopexy, can significantly decrease fertility later in life. Testicular tissue biopsy cryopreservation has been performed in boys with cryptorchidism, with spermatogonial survival and normal hormone production achieved after thawing. In males, genitourinary trauma is a serious nononcological condition that can threaten fertility. In fact, in 2016, the Secretary of Defense outlined a series of reforms to ensure fertility preservation for wounded service members.
Gender and sex diverse individuals, including transgender individuals or those with differences in sex development (DSD) may be at risk of infertility, but the reasons differ depending on the population. For example, transgender individuals possess inherent normal reproductive function, but infertility may occur as a result of medical interventions required to facilitate phenotypic transition to the affirmed gender. On the other hand, infertility in individuals with DSD may be due to abnormal gonadal development, altered endocrine function, or premature gonadal failure. Working groups for individuals with gender and sex diversity have assembled to develop unified fertility preservation protocols for these populations.
Reproductive Dysfunction As a Result of Oncologic Conditions or Their Treatment
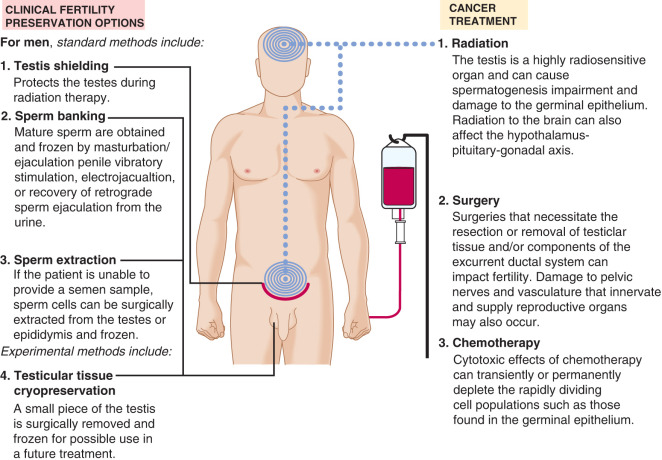
With greater numbers of patients winning the battle against cancer, there is an increased demand that quality of life for survivors be as close to normal as possible. For many young men and women, a high quality of life includes regaining reproductive function and preserving the option to have a family. Yet an unfortunate consequence of the cancer treatments that will preserve these patients’ lives is that nearly all aspects of both the male and female reproductive axis can be compromised. In the female, the hypothalamic-pituitary-gonadal (HPG) axis, the ovaries and ovarian follicles, and the uterus are directly affected by cancer treatments; in the male, the testes are at risk, specifically the germinal epithelium and Leydig cells ( Figs. 33.1 and 33.2 ).
Cancer, Cancer Treatment, and Female Reproductive Function
A fertile woman must have: (1) a functioning neuroendocrine system that regulates the menstrual cycle and can maintain a pregnancy; (2) a healthy pool of follicles that will grow in response to hormonal cues and produce mature and fertilizable gametes; and (3) a receptive uterus that will support embryo implantation and fetal development to term. It is unclear whether cancer itself affects female reproductive function. In one recent study, the success of ovarian stimulation was compared in women with malignancies prior to cancer treatment to a similar group of women seeking assisted reproduction for male-factor infertility. No differences were found between these two groups of patients in terms of the number of dominant follicles and retrieved oocytes, proportion of mature eggs, or fertilization rates. However, a small meta-analysis that examined all types of malignancies found that there were fewer numbers of oocytes retrieved from women with breast and hormone-dependent cancers.
Cancer treatment can affect all aspects of the female reproductive axis (see Fig. 33.1 ). For example, radiotherapy can damage growing follicles, which triggers either repair or elimination pathways. In general, actively dividing cells are more susceptible to radiation-induced death, and because oocytes in the young adult are arrested in prophase of meiosis I, they are more resistant to radiation than cells in mitosis. Primordial follicles, which are quiescent, also appear to be more resistant to radiation compared with growing follicles. Nevertheless, the human oocyte is sensitive to radiation therapy. The LD50, or the radiation dose required to destroy 50% of immature human oocytes, is less than 2 Gy. Mathematical modeling predicts that the effective sterilizing dose of radiation is inversely correlated with age: it is 20.3 Gy at birth and 16.5 Gy at 20 years of age.
In addition to being radiosensitive, the ovary is also chemosensitive. Alkylating agents, especially cyclophosphamide and busulfan, are more gonadotoxic compared to other chemotherapeutic agents, including platinum-based drugs, plant alkaloids, and antimetabolites ( Table 33.2 ). Alkylating agents, which cause DNA breaks irrespective of cell cycle stage, are associated with a high risk of primordial follicle death and compromised stromal cell function. Follicle destruction, whether by radiation- or chemotherapy-induced mechanisms, not only leads to gamete loss, but also results in impaired ovarian hormone production and uterine dysfunction. Although follicles may resist cancer therapies, the ovarian reserve may be compromised and depleted early, resulting in premature menopause. Moreover, both the negative effects of radiation and chemotherapy on estrogen-producing growing follicles can severely impair the menstrual cycle. It is also becoming clear that not only are the germ cells potential targets of iatrogenic insults, but so are the somatic components of the ovary including stromal cells and granulosa cells.
| High Risk | Intermediate Risk | Low Risk | Very Low/No Risk | Unknown Risk |
|---|---|---|---|---|
| Female | ||||
|
|
|
|
|
| Male | ||||
|
|
|
|
|
| Pediatric | ||||
|
|
|
|
|
The female neuroendocrine axis (HPG axis) is also vulnerable to off-target effects of cancer therapies. The HPG axis controls the menstrual cycle and pregnancy by regulating the secretion of hormones, including gonadotropin-releasing hormone (GnRH), follicle-stimulating hormone (FSH), luteinizing hormone (LH), estradiol, progesterone, and prolactin. Radiation therapy, particularly targeting the cranium, can cause altered hypothalamic and pituitary function. There is also evidence that hypothalamic dysfunction can occur after chemotherapy in the absence of cranial irradiation.
The uterus functions primarily to support embryo implantation as well as fetal growth and development. Although chemotherapy alone does not appear to adversely impact the uterus, radiotherapy can have negative long-term consequences on the ability of the uterus to support a future pregnancy. Radiotherapy can reduce uterine volume and elasticity, damage the uterine musculature and endometrium, and decrease the vasculature. If women are able to conceive following radiotherapy, they may have an increased risk of adverse pregnancy outcomes, including miscarriage, placental abnormalities, preterm birth, and delivery of infants with low birth weights.
Cancer, Cancer Treatment, and Male Reproductive Function
Normal male reproduction is also dependent on a tightly regulated HPG axis. In males, the pulsatile release of GnRH from the hypothalamus acts on cells within the anterior pituitary gland to stimulate the production and release of FSH. FSH is secreted into the bloodstream and binds to FSH receptors on the basolateral aspect of Sertoli cells within the testicle, which stimulates the production of androgen-binding protein by Sertoli cells and is responsible for the initiation of spermatogenesis. FSH production is modulated by inhibin B, a glycoprotein produced by the Sertoli cells that feeds back to the hypothalamus and pituitary to suppress FSH production and hypothalamic secretion of GnRH. LH is also produced by gonadotroph cells within the anterior pituitary gland; like FSH, the secretion of LH is regulated by the pulsatile release of GnRH. LH is secreted and stimulates Leydig cells to produce testosterone, and testosterone and FSH act synergistically on the Sertoli cells to initiate and support spermatogenesis. The precise endocrine relationship shared by the hypothalamus, pituitary gland, and testicles is essential to the normal production of sperm. Disturbance of this balance can have detrimental effects on spermatogenesis, resulting in decreased sperm number and quality.
Evidence suggests that cancer itself can affect male reproductive function. For example, at the time of cancer diagnosis, patients often present with impairment of the normal HPG axis. These findings are commonly noted in men with testicular tumors that produce alpha-fetoprotein (AFP) and beta-human chorionic gonadotropin (β-hCG). In 1987, Carroll and coworkers reported that two-thirds of men with testicular cancer had abnormal FSH, LH, and/or β-hCG levels. Specifically, decreased FSH levels and elevated LH and β-hCG levels were observed in men with poor semen parameters. The study authors asserted that β-hCG might have an inhibitory effect on FSH in some patients, resulting in impaired spermatogenesis. Other etiologic mechanisms have been proposed for the endocrine disruption that is sometimes seen in patients with cancer, including central cytokine effects. While this pathway has been suggested to play a role in oncologic anorexia-cachexia syndrome, it has not been specifically studied in terms of hormonal reproductive regulation. More work is needed to specifically elucidate the mechanisms by which oncologic disease processes disrupt the normal HPG axis. In addition, semen quality parameters tend to be poor prior to treatment in men with testicular cancer, leukemia, or lymphoma.
The testis, like the ovary, is both radiosensitive and chemosensitive (see Fig. 33.2 ). Chemotherapy is widely used in the treatment of cancers affecting males, but its cytotoxic effects on normal tissue can transiently or permanently deplete rapidly dividing cell populations, such as the testicular germinal epithelium. Chemotherapeutic regimens that include alkylating agents are among the most gonadotoxic and often lead to permanent oligospermia or azoospermia. These changes in spermatogenesis are typically seen within 90 to 120 days after administration of the alkylating agent. Platinum-based chemotherapy, such as cisplatin and carboplatin, predominately affect spermatogonia and spermatocytes. While most men treated with platinum regimens will experience an associated decline in semen parameters, the majority (80%) will have a return of sperm in the ejaculate within 5 years of completion of chemotherapy. Antimetabolites, such as 5-fluorouracil, methotrexate, gemcitabine, and 6-mercaptopurine, can also impair spermatogenesis, although the effects are commonly transient. Clinicians should be mindful of the fact that, while patients might start on a “fertility friendly” chemotherapeutic regimen, transition to other regimens is not uncommon in the course of cancer care. The best time point for sperm cryopreservation to help ensure optimal fertility preservation is prior to the initiation of chemotherapy.
The testis is a highly radiosensitive organ, and radiation-associated declines in semen parameters are commonly seen within 60 to 70 days after radiation treatment. Doses as small as 0.1 Gy can impair spermatogenesis, while doses of just 1.2 Gy have the potential to cause permanent azoospermia. Sperm concentration is typically observed 4 to 6 months after completion of radiation treatment, and recovery of spermatogenesis is often noted within 10 to 24 months. Fractionated radiation regimens result in more pronounced spermatogenesis impairment than nonfractionated regimens; the belief is that repeated injury to the germ cells impairs recovery and further diminishes spermatogonial stem cell and progenitor pools, thus leading to more severe and lasting damage than that caused by single, larger doses. In contrast to germ cells, Leydig cells are relatively resistant to radiotherapy. While germinal epithelial damage is observed at low doses of radiation, Leydig cell injury is generally not evident until radiation exposure is 20 Gy or more, at which point serum LH elevation and serum testosterone decline are commonly seen.
In addition to the effects of chemotherapy and radiation therapy, surgical procedures can also lead to iatrogenic impairment of male reproductive potential (see Fig. 33.2 ). Orchiectomy leads to loss of testicular mass and thus germ cell mass, with diminished sperm production capability. Patients undergoing radical prostatectomy for prostate cancer, the most common solid cancer in males, sustain disruption of their excurrent ductal system via removal of their prostate gland and seminal vesicles. Men being treated with cystoprostatectomy for bladder cancer are subjected to similar excurrent ductal disruption. Retroperitoneal surgery can often result in disruption of the sympathetic nerves that control seminal emission and ejaculation. Retroperitoneal lymph node dissection (RPLND) for men with testicular cancer is one example of this kind of surgery. Template-driven, nerve-sparing approaches are now widely used in the course of RPLND, and these methods represent a technical approach to fertility preservation in males. Finally, pelvic and abdominal exenterative procedures (removal of all or most organs within the pelvic cavity) can necessitate resection of gonadal tissue and/or the components of the excurrent ductal system. If the possibility of such a resection arises during surgical planning, preoperative sperm cryopreservation should be strongly considered.
Additional Adverse Reproductive Outcomes Following Cancer
As described previously, cancer and its treatments may lead to infertility or sterility due to premature ovarian failure, oligomenorrhea, uterine dysfunction, or altered endocrine function in females; and testicular failure, disruption of the excurrent ductal system, and impaired endocrine function in males. However, there are also a number of other side effects of cancer or its treatment that can alter reproductive function. These effects are often overlooked or dismissed, but pose a significant challenge to the reestablishment of a high quality of life in cancer survivors. These include increased depression, psychological disorders, and sexual function issues. Furthermore, patients often report a loss of self-identity or feelings of attractiveness.
It is quite difficult to predict precisely how a given individual’s reproductive potential will be affected by cancer or cancer treatments, as there are many case-dependent variables that contribute to reproductive risk. These factors include, but are not limited to, a patient’s age, genetic background, medical history, and fertility potential prior to diagnosis and treatment. In addition, the radiation and/or chemotherapy treatment regimen used—including dose, duration, frequency, field of treatment, and combination—has a significant bearing on future reproductive function.
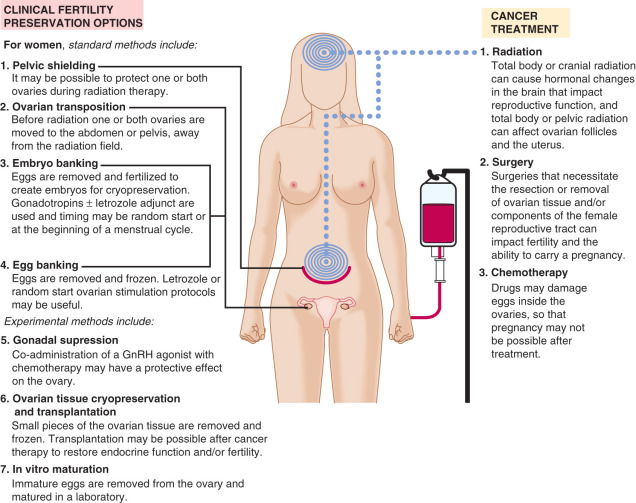
Oncofertility Medical Management
- ◆
Current clinical fertility preservation options for females include ovarian shielding, ovarian transposition, egg or embryo freezing, GnRH agonists, ovarian tissue cryopreservation and transplantation, and oocyte in vitro maturation.
- ◆
Current clinical male fertility preservation options for males include testicular shielding, sperm freezing, surgical sperm extraction, and testicular tissue cryopreservation.
- ◆
Additional considerations in the setting of fertility preservation include surrogacy, uterine transplantation, adoption, and contraception.
- ◆
Complex scenarios in oncofertility medical management include pediatric and obstetrical oncology cases.
- ◆
Assessment of an individual’s reproductive potential across the treatment continuum can inform optimal fertility preservation options.
Relevant Sex Differences in Reproduction
Innate biological differences in gametogenesis between males and females have largely dictated the types of fertility preservation methods that are currently available or are being developed. In females, it is commonly accepted that oocyte production ceases during midgestation, resulting in a fixed number of primordial follicle-enclosed oocytes (the ovarian reserve)—approximately 1 million—at birth that set an individual’s reproductive life span. The fate of these follicles is to remain quiescent, to activate, or to die. Of those primordial follicles that are activated, only a small fraction will develop to the terminal Graafian follicle stage and produce oocytes that will be ovulated. Once destroyed, there is no mechanism to replace these follicles, as replenishment by ovarian germline stem cells does not appear to be physiologically relevant. By puberty, the primordial follicle number decreases to approximately 400,000, and reproductive senescence occurs at the time of menopause, when approximately 1000 primordial follicles remain in the follicle pool. In addition to having a finite supply, relatively invasive techniques are required to access female oocytes.
In contrast to oocytes, sperm are continuously generated in the adult testis. The process begins when type A pale spermatogonia differentiate into type B spermatogonia. The type B spermatogonia then differentiate into spermatocytes, which undergo meiosis to form spermatids and ultimately mature haploid spermatozoa. This process results in the production of millions of mature sperm daily. Because germline stem cells are very active in the male, there is an increased potential for slow recovery of fertility posttreatment if the spermatogonial stem cells are not irreversibly damaged, and the mitotically quiescent type A dark spermatogonia are able to transform into actively dividing and differentiating type A pale spermatogonia. Compared with oocytes, sperm are numerous and can be obtained using relatively simple and noninvasive methods.
Unlike adults, prepubertal males and females have far fewer fertility preservation options because their gonads are not fully developed. Testes of prepubertal males do not contain mature haploid gametes but instead are populated with spermatogonial stem cells that are poised to initiate spermatogenesis and generate sperm at the onset of puberty. Thus, current fertility preservation treatments in prepubertal boys relies primarily on removing and cryopreserving testicular tissue for potential future use with emerging oncofertility techniques (see sections on “ Clinical Fertility Preservation Options for Male Patients ” and “ Emerging Fertility Preservation Techniques ”). Prepubertal females have follicles that develop in waves until the secondary stage, but are then eliminated by atresia. It is not until puberty, when cyclic production of gonadotropins promotes the development of follicles to mature stages, that fertilization-competent eggs are ovulated. Thus, prepubertal females cannot make use of most standard fertility preserving technologies (e.g., hormone stimulation and in vitro fertilization [IVF]) that involve the collection of mature gametes. Instead, current fertility preservation treatments for prepubertal females include ovarian tissue cryopreservation and potentially in vitro maturation (IVM) (see sections on “ Clinical Fertility Preservation Options for Female Patients ” and “ Emerging Fertility Preservation Techniques ”).
Clinical Fertility Preservation Options for Female Patients
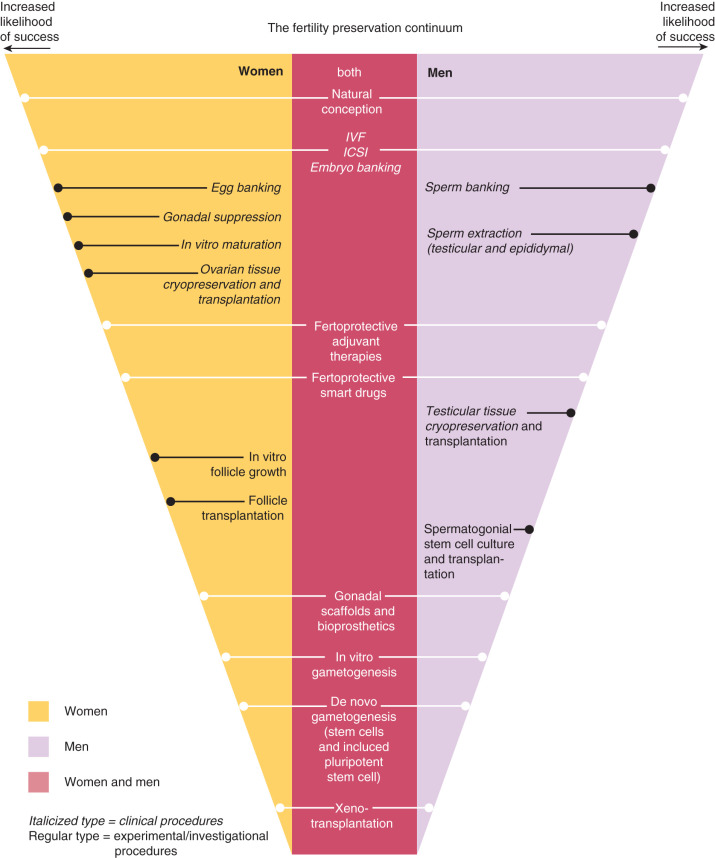
For females, the menu of fertility preservation options is constantly expanding as research breakthroughs are being translated into clinical use ( Table 33.3 and Figs. 33.1 and 33.3 ). Although there are many approaches to fertility preservation, ranging from standard to experimental to theoretical, the unique set of patient- and case-based factors dictate the best course of action. These factors include a patient’s age, ovarian reserve prior to the start of treatment, and type of cancer, as well as the dose, duration, and timing of cancer therapy. It should also be emphasized that female cancer patients who seek fertility preservation are typically young. Thus the well-documented decline in egg quality and fertility with advanced maternal age is unlikely to be a compounding factor for these young women. An overview of female fertility preservation options that are currently used clinically follows (see Table 33.3 and Figs. 33.1 and 33.3 ).
| Fertility Preservation Option | Male | Female | Suitable for Prepubertal Patients | Potentially Delays Cancer Treatment >2 Weeks | Potentially Delays Cancer Treatment <2 Weeks | Requires Hyperstimulation | Requires Sperm Donor at Time of Procedure | Potentially Preserves or Restores Natural Reproductive Function Hormonal | Requires Additional ART Procedures to Attempt Pregnancy | Additional Considerations |
|---|---|---|---|---|---|---|---|---|---|---|
| Standard | ||||||||||
| Ovarian transposition/ Oophoropexy | ● | ● | ● | ● |
| |||||
| Gonadal shielding | ● | ● | ● | ● |
| |||||
| Embryo cryopreservation | ● | ● | ● | ● | ● | |||||
| Sperm cryopreservation | ● | ● | ● |
| ||||||
| Egg cryopreservation | ● | ● | ● | ● |
| |||||
| Investigational | ||||||||||
| Immature oocyte cryopreservation | ● | ● | ● |
| ||||||
| Isolation of an oocyte or an egg from a natural cycle | ● | ● | ● |
| ||||||
| Isolation of oocytes from an ovarian biopsy | ● | ● | ● |
| ||||||
| Ovarian tissue cryopreservation followed by transplantation | ● | ● | ● | ● |
| |||||
| Ovarian hormonal suppression | ● | ● | ● |
| ||||||
| Uterine transplantation | ● | ● | ||||||||
| Testicular sperm extraction (TESE) | ● | ● | ● | ● |
| |||||
| Testicular fine needle aspiration (TFNA) | ● | ● | ● | ● |
| |||||
| Microsurgical epididymal sperm aspiration (MESA) | ● | ● | ● | ● |
| |||||
| Percutaneous epididymal sperm aspiration | ● | ● | ● | ● |
| |||||
| Pre-clinical | ||||||||||
| Ovarian tissue cryopreservation followed by in vitro follicle growth | ● | ● | ● | ● |
| |||||
| Follicle isolation and cryopreservation | ● | ● | ● | ● |
| |||||
| Transplantation of isolated follicles | ● | ● | ● | ● |
| |||||
| Xenotransplantation of ovarian tissue or follicles | ● | ● | ● | ● |
| |||||
| Testicular tissue cryopreservation followed by transplantation | ● | ● | ● | ● | ● |
| ||||
| Spermatogonial stem cell cryopreservation followed by transplantation | ● | ● | ● | ● |
| |||||
| Xenotransplantation of testicular tissue or spermatogonial stem cells | ● | ● | ● | ● |
| |||||
| In vitro spermatogenesis | ● | ● | ● | ● |
| |||||
| Use of fertoprotective drugs | ● | ● | ● | |||||||
| Derivation of oocytes and sperm from stem cells | ● | ● | ||||||||
| Nonbiological/third party | ||||||||||
| Sperm, egg, or embryo donor | ● | ● | ● |
| ||||||
| Surrogate | ● | ● | ● |
| ||||||
| Adoption | ● | ● | ● |
| ||||||
Shielding and Ovarian Transposition
Ovarian shielding, through the use of a lead shield, has been used clinically for decades to protect the gonads in female patients undergoing abdominal or pelvic radiation. However, shielding is not possible in cases requiring total body irradiation or where the ovaries overlap with the radiation field. In such cases, ovarian transposition, or oophoropexy, is a potential fertility sparing option function in which the ovaries are surgically moved outside of the pelvic area and radiation field. Where the ovaries are placed depends on the type of cancer. Normal endocrine function and fertility occur following ovarian transposition, but this technique is contraindicated if there is a risk of spreading cancer cells.
Embryo Freezing
Controlled ovarian stimulation (COS) for egg or embryo cryopreservation has had the most proven success and should be offered as the first-line therapy for fertility preservation in postpubertal females. In the normal menstrual cycle, a tightly controlled sequence of hormones results in the development and ovulation of a single egg. Briefly, in the follicular phase of the menstrual cycle, the selection of the single dominant follicle from the pool of available follicles is determined by FSH responsiveness. The follicle that is most sensitive to FSH will upregulate FSH receptors and increase granulosa cell numbers. Granulosa cells produce estrogen and the increased estrogen levels have a negative feedback effect on FSH production by the pituitary. The dominant follicle continues to grow because of its newly increased responsiveness due to increased FSH receptor density. However, the other follicles suffer from FSH withdrawal and undergo atresia. LH receptors appear on the dominant follicle as it approaches ovulation and a pituitary-mediated LH surge signals to resume meiosis and ovulation. By this mechanism, a single follicle is recruited and a single egg is released in a natural menstrual cycle. The duration of the rise in FSH above a critical threshold determines the number of dominant follicles that are selected in a menstrual cycle.
In COS cycles, exogenous FSH is given in higher doses and over a longer period of time to enable recruitment of multiple follicles and capture of more than one egg per cycle. GnRH agonists or antagonists can be used to block the release of GnRH, and therefore the LH surge, to prevent premature ovulation and allow more time to recruit multiple follicles. hCG is used in COS cycles to mimic the endogenous LH surge due to its longer half-life and its ability to promote ovulation similar to LH. Newer protocols have been developed that use GnRH agonists (GnRH-a) to cause an endogenous LH surge to trigger final oocyte maturation. The choice of COS protocol, either downregulation with GnRH agonists or antagonists and ovulation triggered with hCG or GnRH-a, is dependent on the patient’s age, ovarian reserve, and physician preference. Following COS, mature eggs are aspirated and either frozen or fertilized using ART procedures, such as IVF or intracytoplasmic sperm injection (ISCI). Embryos are then cryopreserved by slow-freezing or vitrification and stored for the patient’s future use. Following cancer treatment, these embryos can be thawed and transferred back to the patient’s (or a surrogate’s) uterus.
Cryopreservation of embryos has been highly successful, and millions of children worldwide have been born using cryopreserved and thawed embryos. The obstetrical outcomes of children born after implantation of cryopreserved embryos are comparable to those born after implantation of embryos produced in fresh IVF and ICSI cycles, although long-term follow-up child health studies are still needed. Importantly, although the number of high-quality embryos is lower in the setting of fertility preservation relative to the general population, the cumulative live birth rate was similar to that achieved with fresh embryo in noncancer patients. Despite this success, there are several potential drawbacks of this method in the context of oncofertility. This procedure requires time. Conventional downregulation protocols begin in the luteal phase of the preceding cycle and can take 4 to 6 weeks from initiation to oocyte retrieval. Conventional antagonist cycles similarly can take 1 to 4 weeks from presentation depending on where a woman is in her menstrual cycle. This potential delay in cancer treatment may be detrimental for women with aggressive or advanced cancers. In addition, COS is contraindicated for women with hormone-sensitive cancers. The use of random start protocols and adjuvant letrozole treatment for ovarian stimulation may overcome these challenges, as described in more detail later. Embryo banking also presupposes that a patient is willing to use a sperm donor or has an existing partner who has agreed to create embryos, which is an issue that is an obstacle for adolescent and young adult patients. Moreover, COS is not applicable to prepubertal patients. Finally, a myriad of ethical and potential legal concerns exist surrounding the generation and cryopreservation of embryos that may or may not be used.
Egg Freezing
For women who want to maintain their reproductive autonomy, but do not want to create embryos at the time of cancer diagnosis, egg freezing is a potential option. In this technique, a woman undergoes COS to recruit multiple follicles to grow, and mature eggs are aspirated and cryopreserved for later fertilization. Random start protocols and adjuvant letrozole treatment for ovarian stimulation as described later are also applicable to egg freezing. Accumulating data suggest that egg freezing is a viable option, especially in an oncofertility setting. When performed at experienced fertility centers, the live birth rate of IVF using cryopreserved eggs is comparable to that of conventional IVF using fresh eggs, making this a viable option for biological preservation of gametes without the need to have a known or selected sperm donor. In the United States, the ASRM removed the experimental label of egg freezing in 2012, and this technique is now considered the standard of care. Interestingly, the number of females over the age of 18 undergoing ovarian tissue cryopreservation has decreased since this decision, based on data from the Oncofertility Consortium.
Random Start Protocols
For patients with a new diagnosis of cancer where time is critical, the need to initiate immediate COS due to time constraints has led to a flurry of research into various clinical protocols to optimize success while minimizing wait time. Random start stimulation protocols, which allow patients to undergo COS on the day of presentation, regardless of the menstrual cycle phase, have similar outcomes to conventional follicular phase start protocols. Studies comparing late follicular phase and luteal phase stimulation start protocols were as effective as conventional early follicular phase start protocols with regard to the number of oocytes and mature oocytes retrieved, the fertilization rates, and the embryo development rates. For patients initiating gonadotropins in the late follicular phase with no dominant follicle present, the ovaries are stimulated and the LH surge is ignored. After ovulation, a secondary cohort of follicles is then recruited, and a GnRH antagonist is initiated to capture this secondary cohort and to prevent ovulation. Alternatively, if the patient is in the late follicular phase and a dominant follicle is present (>15 mm), ovulation is triggered with hCG, and gonadotropins are initiated after ovulation to capture the secondary follicle cohort. If the patient presents in the luteal phase, gonadotropins and GnRH antagonists are administered in a similar fashion. Administration of the final oocyte maturation trigger can occur either with hCG or the GnRH agonist, depending on the clinical scenario.
Letrozole Protocols
COS result in estrogen levels that are 10 to 20 times higher than physiologic levels during a natural menstrual cycle. It has been hypothesized that high levels of estrogen cause accelerated tumor growth in patients with estrogen-sensitive tumors, such as certain breast cancers. One way to avoid the risk posed by supraphysiologic estrogen levels is to cryopreserve oocytes or embryos on a natural menstrual cycle without COS. However, the results of natural-cycle IVF are poor; one study showed an average of only 0.6 embryos cryopreserved per patient. Success rates increase with increasing numbers of retrieved oocytes and embryos, making COS advantageous.
Adjuvant letrozole, an aromatase inhibitor, has been used in combination with FSH to decrease estrogen levels during COS. Aromatase converts androgens to estrogens in many tissues, including the granulosa cells of ovarian follicles. Letrozole can be used at a daily dose of 2.5 to 7.5 mg in anovulatory patients for ovulation induction while maintaining low estrogen levels. Letrozole also can be used as an adjunct to gonadotropin stimulation in the setting of COS for oocyte or embryo cryopreservation in estrogen-sensitive cancer patients. Two prospective studies were done comparing letrozole to other agents in the setting of fertility preservation for cancer patients. The first study compared 5 mg of daily letrozole plus FSH, 60 mg daily tamoxifen plus FSH, and 60 mg of tamoxifen alone. The authors found that patients who used either letrozole plus FSH or tamoxifen plus FSH had significantly more oocytes retrieved than those who used tamoxifen alone. Peak estrogen levels were lowest in the letrozole plus FSH group. In a later study, the same authors compared letrozole to anastrozole in a similar population of breast cancer patients undergoing IVF for fertility preservation. In this study, letrozole was shown to be superior to anastrozole in the suppression of estradiol levels, although there was no statistically significant difference in the number of oocytes retrieved. Other studies have found that titrating the dose of letrozole up to 10 mg/day can keep estradiol levels suppressed below 500 pg/mL. Long-term follow-up studies have not shown an increased risk for cancer recurrence in patients who underwent fertility preservation protocols using letrozole as adjunct therapy. Subsequent clinical pregnancy rates have been shown to be similar to the United States national mean among infertile patients undergoing IVF.
Gonadotropin-Releasing Hormone Agonists
The success of using GnRH analogues (agonists, GnRH-a) as adjuvant treatment for fertility preservation remains controversial. The mechanism of action by which GnRH agonists protect fertility is unclear, but it may involve a decrease in ovarian perfusion and therefore a decrease in the amount of cytotoxic chemotherapy delivered to the ovary. Another potential mechanism is hypothesized to be an upregulation of intragonadal antiapoptotic molecules, such as sphingosine-1-phosphate. While COS for egg or embryo cryopreservation should be offered as first-line therapy for fertility preservation, the use of a GnRH agonist may be considered in addition to COS or as an alternative to COS when time or other barriers exist.
Several randomized studies have failed to show a statistically significant benefit of GnRH agonists in ovarian protection, but these were small trials with relatively short follow-up periods. However, data from two larger randomized studies have provided stronger evidence in support of GnRH agonists as adjuvant therapy for ovarian protection during cytotoxic chemotherapy. In 2011, the PROMISE-GIM6 trial demonstrated a reduction in ovarian failure rates when a GnRH agonist was given concurrently with chemotherapy in patients with breast cancer. The PROMISE-GIM6 trial included women with early-stage breast cancer who were randomly allocated to receive chemotherapy alone (anthracycline-based, anthracycline plus taxane-based, or cyclophosphamide-based regimens) or chemotherapy plus triptorelin. Triptorelin was given as an intramuscular dose of 3.75 mg at least 1 week prior to the onset of chemotherapy and continued every 4 weeks for the duration of treatment. The primary endpoint of the study was the incidence of chemotherapy-induced early menopause. Early menopause was defined as no resumption of menstrual activity with postmenopausal levels of both FSH and estradiol for 1 year after the end of chemotherapy. A total of 281 patients were randomized. Treatment with triptorelin was associated with a significant risk reduction of developing early menopause (odds ratio [OR], 0.28; 95% confidence interval [CI], 0.14 to 0.59, P < .001). Premature menopause occurred in 25.9% of patients treated with chemotherapy alone compared to 8.9% treated with chemotherapy and triptorelin.
A second larger randomized controlled trial, POEMS/S0230, showed similar results with the GnRH agonist goserelin. In this study, 218 patients with stage I to IIIa estrogen receptor (ER)-negative and progesterone receptor (PR)-negative cancers who were going to receive cyclophosphamide-containing chemotherapy were eligible to participate, and 135 patients were included in the final analysis. Patients were randomized to adjuvant treatment with goserelin or no adjuvant treatment. Goserelin 3.6 mg was administered 1 week prior to the initial dose of chemotherapy and then every 4 weeks during chemotherapy, with the final dose given 2 weeks after the last cycle of chemotherapy. The primary objective was to compare the rate of ovarian failure between the two treatment groups. Ovarian failure was defined as amenorrhea for the preceding 6 months and FSH levels in the postmenopausal range at 2 years. Of 69 patients, 15 (22%) in the no adjuvant group experienced ovarian failure compared to 5 of 66 (8%) in the goserelin group (OR, 0.30; 95% CI, 0.09 to 0.97).
Ovarian Tissue Cryopreservation and Transplantation
Removal of ovarian tissue prior to cancer treatment may be the only option for fertility preservation for prepubertal females, those who do not have time to undergo ART prior to starting cancer treatment, or those who cannot tolerate the supraphysiological hormone exposure required for most ART procedures. Following surgical removal, ovarian tissue is cryopreserved and can be used later for transplantation once the patient has been deemed cancer free. Although the current approach is to cryopreserve ovarian cortical strips, methods are being developed to freeze whole ovaries for later transplantation through vascular anastomosis. The goals of ovarian tissue transplantation are to restore both fertility and endocrine function. Human ovarian tissue transplantation was first tested in a series of heterologous transplants, using either fresh cortical tissue or whole ovaries, between nine pairs of monozygotic twins that were discordant for premature ovarian failure. Heterologous transplantation of fresh ovarian tissue resulted in 14 pregnancies and 11 live births in 7 of the 9 recipients. In an oncofertility setting, ovarian tissue is cryopreserved and stored for an indeterminate amount of time prior to transplant. To date, approximately 95 live births have been documented worldwide in the literature following ovarian tissue transplantation.
All human live births to date have occurred following orthotopic ovarian transplantation, in which human ovarian tissue is transplanted back to its original location either on the remaining ovary or near the infundibulopelvic ligament or ovarian fossa. An active area of research is heterotopic transplantation, in which the ovarian tissue is grafted subcutaneously at various sites, including the brachioradialis fascia of the forearm or under the rectus sheet in the lower abdomen. Heterotopic transplantation is an attractive option because it can be performed in patients who have had pelvic irradiation and may have ovarian scarring; also it allows for simpler monitoring of follicle development. Thus far, heterotopic transplantation in women has had varying degrees of reproductive success, resulting in restored endocrine function, follicle development, gametes capable of fertilization, and preimplantation embryo development. Although biochemical pregnancies in humans have been reported from gametes derived from heterotopic transplantation, no live births have been reported. Another area of active research is in vitro activation (IVA), which is designed to harness the activity of residual follicles in patients with primary ovarian insufficiency. IVA exposes ovarian tissue to stimulators of the Akt pathway prior to transplantation, which has resulted in several live births. However, the efficacy, safety, and mechanisms of IVA warrant further investigation.
There are several risks and concerns associated with ovarian tissue transplantation. It is difficult to estimate the true efficacy of this technique because, although live births have been reported worldwide following this procedure, it is not known how many total transplants have been attempted that may not have been successful. In addition, the number, quality, and distribution of follicles within the cortical tissue from each patient cannot be predicted, and it is not known how long the transplants will remain functional. Finally, there is a lack of robust methods to detect residual cancer cells in the ovarian tissue, and thus there is an inherent risk of reintroducing cancer cells upon transplantation. Obtaining definitive data about ovarian tissue transplantation outcomes is expected to be a slow process, as it could be decades before children who have frozen their tissue return to use it.
In Vitro Maturation
IVM is a procedure that involves the collection of immature oocytes from growing antral follicles from minimally stimulated or unstimulated ovaries. These oocytes are within cumulus-oocyte-complexes (COCs) that are then matured in a laboratory setting prior to being cryopreserved or fertilized. Immature oocytes can be aspirated in vivo from unstimulated or mildly stimulated ovaries, and live birth incidences using IVM-derived eggs have been reported upwards of approximately 40% per procedure. IVM can also be performed ex vivo in combination with ovarian tissue cryopreservation. In this case, preparation of the harvested ovarian tissue into cortical strips disrupts small antral follicles in the medulla, releasing COCs that can be used for IVM. This approach has been used to generate additional mature eggs from females (prepubertal and adult), which otherwise would have been discarded as part of standard ovarian tissue cryopreservation, and an ongoing pregnancy has been reported. Although the initial reports of IVM are promising, our overall understanding of the quality and reproductive potential of gametes that did not reach full maturity in vivo is lacking. This lack of knowledge is even more pronounced for prepubertal females and warrants further study.
Clinical Fertility Preservation Options for Male Patients
Fertility preservation techniques for males are generally more feasible and less invasive than those available to females. This dichotomy arises because most males are able to provide ejaculated semen samples with viable sperm for cryopreservation. Very small quantities of sperm can be frozen and used in IVF or ICSI after cancer to achieve biological paternity. However, some patients may experience anejaculation (the inability to ejaculate), aspermia (the lack of an antegrade ejaculate), retrograde ejaculation, necrospermia (only dead sperm present in the ejaculate), or azoospermia. Each of these scenarios can hamper standard male fertility preservation measures, and therefore require more invasive and sometimes investigational measures for fertility preservation. Fortunately, the armamentarium of male reproductive therapeutics that is commonly used to treat such conditions can be applied in the setting of fertility preservation (see Table 33.3 and Figs. 33.2 and 33.3 ). An overview of male fertility preservation options that are currently used clinically follows (see Table 33.3 and Figs. 33.2 and 33.3 ).
Testicular Shielding
Gonadal shielding is also a means to preserve fertility in males who must undergo radiation therapy. Similar to females, gonadal shielding is a particularly important consideration when pelvic or lower abdominal fields are being treated, as these can result in significant gonadal exposure through radiation scatter.
Masturbation/Ejaculation
The ejaculate is the preferred source of sperm for cryopreservation. Men engaging in semen collection are first asked to provide an ejaculate sample themselves through masturbation. For this, men are typically asked to abstain from ejaculation for 2 to 3 days prior to the collection of each sample. Patients are discouraged from use of lubricants, jellies, and saliva during the collection process, as these all have spermatotoxic properties. Semen collection can be performed on an inpatient or outpatient basis. In the setting of cancer care and management, coordination of semen collection is essential. Narcotics and sedatives are commonly used in diagnostic and therapeutic procedures for males recently diagnosed with cancer, and these agents can hamper semen collection efforts. Assuring the patient that his privacy will be maintained during semen collection is also critical, as anxiety can impair efforts to produce a semen sample by masturbation.
Penile Vibratory Stimulation
Some men experience difficulty reaching climax and are thus unable to produce an ejaculated semen sample by masturbation. Patients at risk for this problem include men on narcotics and sedatives, with peripheral neuropathy, or with significant pelvic, perineal, or genital edema. Men with a prior history of pelvic surgery and/or trauma or arterial disease also have a heightened risk for difficulties with ejaculation. For these individuals, penile vibratory stimulation (PVS) can help optimize genital tactile stimulatory input and trigger the ejaculatory reflex. To facilitate PVS, a vibratory stimulation device is applied to the ventral surface of the penis, just proximal to the corona. Commercially available vibratory devices commonly feature variable amplitude and frequency settings, allowing patients to modulate these parameters to optimize their stimulatory input. Care must be taken in patients with edema, peripheral neuropathy, or other conditions that reduce tactile perception of the penis, as penile skin damage or breakdown can occur in these individuals without their knowledge. Patients are usually able to use these devices successfully alone or in private with their partner. If these efforts are not successful, a health care provider can perform the PVS procedure.
Electroejaculation
In some instances, vibratory stimulation does not result in ejaculation. For such cases, another option is electroejaculation (EEJ). EEJ triggers ejaculation through the use of a probe, which is inserted transrectally to deliver an electrical current to the pelvic structures in a rhythmic fashion. The resultant electrical stimulation of the seminal vesicles, prostate gland, and pelvic nerves usually leads to antegrade seminal expulsion via the urethra. In some cases, the semen may travel in a retrograde fashion back into the bladder. To cover this possibility, sperm wash media should be instilled into the bladder prior to EEJ, and the bladder should be drained after the procedure via catheter to insure the collection of all viable sperm. EEJ requires the use of an anesthetic, except in patients with a history of a complete spinal cord injury. Care must be taken during the EEJ procedure to monitor rectal temperature to avoid rectal mucosal burns. Anoscopy is usually performed immediately prior to and after EEJ procedures. Monitoring of the rectal mucosa in this fashion is highly recommended. As noted in the previous section on PVS, this technique is recommended for patients demonstrating anejaculation, who may include those taking narcotics and sedatives, males with peripheral neuropathy or past pelvic surgery/trauma, and men with significant pelvic, perineal, or genital edema.
Recovery of Retrograde Sperm Ejaculation from the Urine
As briefly described earlier, some patients experience the retrograde passage of their ejaculate back into the bladder instead of antegrade through the urethra and out the tip of the penis. Patients at increased risk for retrograde ejaculation include men with autonomic neuropathy, men who have sustained sympathetic nerve injury due to prior surgical procedures, such as RPLND or pelvic exenteration, and men with bladder neck incompetence, including those with a history of bladder neck surgery. In general, the bladder is a hostile environment for sperm, as urine is highly spermatotoxic. In some cases, sympathomimetic agents such as pseudoephedrine can facilitate conversion from retrograde to antegrade ejaculation. However, in cases where medical therapy is unsuccessful, recovery of viable sperm from the bladder can be achieved by urethral catheterization to drain urine and then instilling sperm wash media into the bladder prior to ejaculation. After ejaculation, the patient can either urinate out the media or the urethral catheter can be replaced to allow collection of media from the bladder. The media is then centrifuged, and the sperm are isolated in the resultant pellet. These sperm can then be resuspended in media and cryopreserved.
Surgical Sperm Extraction Procedures
A number of surgical sperm extraction procedures are available to permit sperm retrieval from the testicular tissue or the epididymis. Surgical sperm extraction is generally reserved for those patients with obstruction of their excurrent ductal system, men with ejaculatory dysfunction nonresponsive to PVS and/or EEJ, and men with necrospermia or nonobstructive azoospermia. A number of centers are now also collecting sperm from peripubertal patients, and patients with DSD (e.g., Klinefelter syndrome). A number of sperm extraction procedures are available, and surgeon preference often dictates which approach is used.
For men with obstructive azoospermia, a blockage in the sperm delivery system (i.e., epididymis, vas deferens, or ejaculatory duct) prevents passage of sperm in the semen. This may include men with a history of vasectomy who want to preserve future fertility potential after receiving a cancer diagnosis. For these men, the ejaculate contains no sperm, so sperm extraction from either the epididymis (percutaneous epididymal sperm aspiration [PESA] or microsurgical epididymal sperm aspiration [MESA]) or from the testicle (microsurgical testicular sperm extraction [micro-TESE], testicular sperm extraction [TESE], or testicular fine needle aspiration [TFNA]) are all options. Sperm retrieval rates are generally high in patients with obstruction, but the underlying systemic response to cancer in oncologic patients can markedly inhibit sperm production in some cases, as was previously detailed.
For men with nonobstructive azoospermia, the underlying problem is sperm production. Nonobstructive azoospermia can arise due to endocrine abnormalities, such as hypogonadism, which is often associated with oncologic diagnoses. As was described earlier in this chapter, low testosterone is frequently seen in cancer patients even before the initiation of cancer therapy. Hypogonadism can also result from narcotic use, which leads to suppression of LH secretion and decreased production of testosterone by Leydig cells. Another common cause of nonobstructive azoospermia is germ cell sloughing. This can occur in men after a febrile systemic response to cancer, as is sometimes seen in lymphoma. These men will often be found to have either nonobstructive azoosperma or necrospermia at the time of attempted sperm cryopreservation. Finally, men with testicular cancer are sometimes found to have nonobstructive azoospermia at the time of semen collection. In some men, impaired spermatogenesis may be associated with a local inflammatory response to the testicular neoplasia; in others, germ cell absence may have preceded the development of cancer.
For all men with nonobstructive azoospermia, the aim is to isolate and extract sperm from the testicular tissue. Onco-TESE, described by Schrader and coworkers, involves surgical extraction of sperm from the testicle in male cancer patients with azoospermia. Most clinicians prefer the micro-TESE technique in such cases, as this microsurgical approach enables the systematic inspection of the seminiferous tubules in search of dilated, opaque tubules that often bear sperm. Clinicians should realize that spermatogenesis is often “patchy” and heterogeneous, and so careful, methodical inspection of the tissue is technically important. As reported by Schrader and coworkers, published sperm retrieval rates for onco-TESE are approximately 50%.
Testicular Tissue Cryopreservation
While mature testis tissue has been used for fertility preservation, namely through recovery of germ cells that can be used for IVF, to our knowledge, cryopreserved immature (prepubertal) testis tissue has yet to be used to restore fertility due to a need for poststorage maturation. However, testicular tissue cryopreservation is the only fertility preservation option for prepubertal oncologic patients who cannot produce a semen sample and whose testes do not yet possess mature sperm for surgical collection and cryopreservation. To this end, cryopreservation of immature testis tissue is being further developed as an investigational fertility preservation option and is becoming increasingly performed both abroad and in the United States.
Role of Gamete and Reproductive Tissue Transport, Cryopreservation, and Storage in Fertility Preservation
Cryopreservation is a central component of fertility preservation, as reproductive tissues must be harvested before fertility-threatening treatment is initiated and must be stored until treatment is completed. As the array of fertility preservation options expand for both males and females, so does the need to develop long-term storage methods for diverse cell types (oocytes, eggs, spermatogonia, spermatids, spermatozoa, and embryos) and complex tissues containing these cell types (testicular tissue, ovarian tissue, and follicles). Although there has been some success with preserving sperm through freeze-drying, most reproductive tissues are stored using cryotechniques. In general, these methods require that a tissue is cooled from its physiological temperature to subzero temperatures, is maintained at that temperature for an extended time, and is able to regain cellular viability upon thawing. Because reproductive cells and tissues differ in their size, number, composition, and structure, there is no standard protocol for cryopreservation. The success of this technology is highly site-dependent, and patients should be referred to specialized centers with trained staff who can address their specific needs.
In the field of ART, egg, sperm, and embryo cryopreservation are considered standard technologies. The two main types of cryopreservation techniques that are routinely used in ART are slow freezing and vitrification. With slow freezing, cells or tissues are exposed to low concentrations of cryoprotectants, which results in a lowering of the intracellular water content during a slow cooling process. The main risk of slow freezing is ice crystal formation during both freezing and thawing. Vitrification was developed to avoid ice crystal formation; in this method, the cells or tissues are equilibrated in high concentrations of cryoprotectants, and then plunged directly in liquid nitrogen. Vitrification is simple, rapid, and economical; however, its main drawback is that it is performed in an open system in which the cells or tissues are in direct contact with liquid nitrogen. Current efforts are focused on developing adequate closed systems to ensure safety. Whether slow freezing or vitrification methods are employed, the greatest risk of cryodamage occurs during either the cooling or the thawing process. Little damage to cells and tissues is thought to occur during extended storage.
Recently, the investigational procedure of ovarian and testicular tissue cryopreservation has become more widespread in the field of fertility preservation, especially in cases involving prepubertal males and females whose gametes are not yet mature. Freezing and thawing tissues versus individual cells is complex as it must be done in such a way that maintains cell-to-cell interactions in a characteristic architecture. This is essential if both endocrine and reproductive functions are to be restored when thawed tissues are re-introduced into the patient. Both somatic cells (male: Leydig and Sertoli; female: granulosa, cumulus, and theca) and germ cells (male: spermatogonia and spermatids; female: oocytes) and their arrangement within the tissue must be protected. Gonadal tissue can be frozen as cell suspensions, tissue pieces, or whole organs. Ovarian and testicular tissue cryopreservation can be performed at the clinical site where the tissue was removed, or if the relevant expertise is not present at that location, the tissue can be transported at cold temperatures for period of up to 20 to 24 hours. Most tissues are frozen using slow-freezing protocols, although promising results—including live births—have also been obtained with vitrification of ovarian tissue. Coupled with the freezing of gonadal tissue is the need to develop and optimize methods for transplantation or isolation of immature gametes from the thawed tissue followed by in vitro growth to maturity. As already mentioned, there have been approximately 95 live births reported following transplantation of frozen and thawed cortical ovarian tissue. In contrast, we are unaware of any attempts of testicular tissue transplantation in humans. However, in the male, the advent of microinjection technologies has allowed germ cells at different stages of development to be isolated from testicular tissues and used for fertilization. For example, pregnancies and live births can be achieved with ejaculated, seminal, and testicular spermatozoa, even in cases of low sperm number or quality. In addition, injection of immature round spermatids can be performed clinically. For additional potential fertility preservation applications of ovarian and testicular tissue, see the “ Emerging Fertility Preservation Techniques ” section .
Assessing Reproductive Potential Before, During, and After Cancer Treatment
Assessing a patient’s reproductive function in an oncofertility setting is best performed before cancer treatment begins because doing so can help predict how vulnerable a patient may be to cancer therapies and how the patient is likely to recover. This understanding will ultimately help with the selection of the most beneficial fertility preservation option. Measuring reproductive function after cancer therapy may indicate whether a potential for restored natural fertility exists or whether the patient must rely on previously performed fertility preservation methods.
In females, ovarian reserve is defined as the functional potential of the ovary and reflects the number and quality of oocytes it contains. Although there is no single predictive marker of ovarian reserve, several tests have been developed ( Table 33.4 ). Currently, serum anti-müllerian hormone (AMH) is one of the most robust markers for evaluating ovarian reserve. AMH is produced by the granulosa cells in developing ovarian follicles, and AMH levels do not vary with the menstrual cycle day, oral contraceptive use, or pregnancy. Serum AMH levels decrease rapidly after the age of 37, and this drop precedes and is related to the onset of menopause. A study performed in regularly menstruating women assessed several hormonal markers of aging and found that serum AMH was the most accurate in predicting the occurrence of the menopausal transition within 4 years. Serum AMH also correlates with the number of antral follicles (the antral follicle count [AFC]), which can be determined by counting the number of 2 to 10 mm diameter follicles by transvaginal ultrasound. A strong correlation between AMH levels and AFC has been well established in clinical studies. The number of small antral follicles is one of the best markers of ovarian age related to the ovarian reserve. It has also been shown that serum AMH levels and AFC, when analyzed together, are indicative of primordial follicle number independent of age. Thus, in combination, serum AMH levels and AFC are strong correlates of ovarian age and the remaining ovarian reserve.